introduction
A hallmark of advancing age is a progressive decline in maximal O2 consumption during whole-body exercise that appears to be concomitant to decrements in various central and peripheral components of O2 transport to skeletal muscle [1–3]. However, to date, owing to the confounding effects of physical activity and the technical challenges associated with assessing both peripheral O2 delivery and mitochondrial function in skeletal muscle in vivo, it is still equivocal whether the age-related decrease in O2 transport capacity is accompanied by a proportional decline in mitochondrial ATP synthesis capacity [4–10].
The acute manipulation of O2 availability combined with the in vivo measurement of peak mitochondrial ATP synthesis rate by 31P-MRS offers an opportunity to assess the matching of O2 supply and demand. Using this approach, it has recently been documented that enhanced O2 availability to the muscle, induced by higher fractions of inspired O2 (FiO2 = 1.0), resulted in unaltered mitochondrial function in the calf muscle of elderly individuals [11]. Somewhat in contrast to this finding, Wray et al. [12] reported that enhanced tissue perfusion induced by acute antioxidant administration in elderly subjects improved mitochondrial function, both assessed by MR imaging and spectroscopy techniques, suggesting that a relative hypoperfusion potentially restrains metabolic capacity under normal conditions of oxidative stress. While antioxidant administration in this population likely induced an increase in muscle blood flow by enhanced scavenging of free radicals and a subsequent increase in nitric oxide (NO) bioavailability [13], it may have also directly improved mitochondrial function [14], thus complicating the interpretation of the findings. Given these apparent conflicting results regarding O2 supply and demand in the elderly, circulatory occlusion and the subsequent reactive hyperemia (RH) upon cuff release at the offset of exercise appears to be an interesting alternative experimental model that can, rather simplistically and substantially, locally increase O2 supply [15]. Lacking of the limitations of the previous studies, the application of this approach in the elderly would thus allow the assessment of the putative mismatch between peripheral O2 supply capacity and intrinsic mitochondrial capacity for ATP synthesis with advancing age.
Therefore, the purpose of this study was to utilize an integrative approach combining vascular and metabolic measurements (31P-MRS, NIRS, and Doppler ultrasound) to determine whether, in the elderly, mitochondrial function is limited by O2 supply or the intrinsic mitochondrial capacity to synthesize ATP. To this aim, we examined whether a period of brief ischemia during exercise followed by RH would affect limb and capillary blood flow, tissue re-oxygenation and mitochondrial function in old untrained subjects. Based upon the work of Wray et al. [12] in the young and old, utilizing a somewhat different approach, we hypothesized that improved convective O2 delivery and capillary blood flow induced by superimposing RH at the offset of exercise would increase tissue O2 availability and ultimately result in a higher peak mitochondrial ATP synthesis rate in older subjects.
Results
Baseline
Table 1 summarizes tissue oxygenation indices, intracellular metabolite concentrations and pH at rest and at the end of the exercise. Immediately before each exercise bout (i.e. baseline), microvascular oxygenation (tissue oxygenation and deoxyhemoglobin), pH, and phosphorylated compounds ([PCr], [Pi] and [ADP]) were not significantly different between conditions (P > 0.05). Baseline mean arterial pressure was not significantly different between conditions (FF: 80 ± 30 mmHg; RH: 82 ± 32 mmHg, P>0.05)
Table 1. Metabolic and oxygenation indices at rest and the end of plantar flexion exercise in free flow and reactive hyperemia conditions.
| Free Flow | Reactive Hyperemia | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metabolism | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Resting concentrations | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PCr (mM) | 29 | ± | 7 | 29 | 5 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pi (mM) | 2.2 | ± | 0.6 | 1.6 | 0.6 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ADP (μM) | 15 | ± | 10 | 15 | 8 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| pH | 6.99 | ± | 0.02 | 6.96 | 0.03 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PDE (mM) | 1.5 | ± | 1.3 | 1.5 | 1.3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| End Exercise concentrations | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PCr (mM) | 21 | ± | 6 | 15 | ± | 4 | * | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pi (mM) | 8 | ± | 3 | 13 | ± | 3 | * | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ADP (μM) | 45 | ± | 20 | 67 | ± | 29 | * | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| pH | 7.00 | ± | 0.06 | 6.94 | ± | 0.06 | * | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Microvascular oxygenation | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Resting | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tissue oxygenation (%) | 58 | ± | 11 | 59 | 10 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Deoxyhemoglobin (µM) | 23 | ± | 8 | 23 | 8 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| End Exercise | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tissue oxygenation index (%) | 50 | ± | 25 | 39 | ± | 36 | * | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Deoxyhemoglobin (µM) | 25 | ± | 10 | 34 | ± | 12 | * | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Data expressed as mean ± SD. Phosphocreatine, PCr; Inorganic phosphate, Pi; ADP; Adenosine diphosphate; Phosphodiester, PDE. * reactive hyperemia significantly different from free flow (P < 0.05). | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Exercise
By the end of exercise, both microvascular oxygenation and the metabolic response differed significantly between free-flow and with circulatory occlusion (P < 0.05, Table 2). Mean arterial pressure was significantly augmented at the end of the exercise with circulatory occlusion (FF: 98 ± 14 mmHg; RH: 109 ± 24 mmHg, P<0.05).
Table 2. Phosphocreatine and tissue oxygenation dynamics during the recovery from plantar flexion exercise in free flow and reactive hyperemia conditions.
| Free Flow | Reactive Hyperemia | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| PCr offset kinetics and mitochondrial function | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Amplitude (mM) | 9 | ± | 3 | 15 | ± | 4 | * | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Time constant (s) | 46 | ± | 19 | 39 | ± | 9 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Vmax (mM.min-1) | 21 | ± | 10 | 29 | ± | 8 | * | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Deoxyhemoglobin offset kinetics | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Time Delay (s) | 34 | ± | 46 | 4 | ± | 5 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Amplitude (%) | 3 | ± | 2 | 11 | ± | 5 | * | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tau (s) | 43 | ± | 40 | 22 | ± | 18 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mean Response Time (s) | 77 | ± | 70 | 26 | ± | 20 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tissue oxygenation offset kinetics | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Time Delay (s) | 14 | ± | 15 | 4 | ± | 6 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Amplitude (%) | 9 | ± | 14 | 24 | ± | 31 | * | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tau (s) | 67 | ± | 89 | 19 | ± | 10 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mean Response Time (s) | 81 | ± | 90 | 22 | ± | 11 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| All values are expressed as mean ± SD. Peak rate of oxidative ATP synthesis, Vmax. * reactive hyperemia significantly different from free flow (P < 0.05) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Recovery period
Blood Flow and tissue oxygenation
Mean changes in blood flow, leg vascular conductance and convective O2 delivery dynamics during the recovery period in FF and RH conditions are displayed in Figure 1. Blood flow (FF: 1545 ± 549 ml; RH: 3262 ± 1165 ml; P < 0.01), leg vascular conductance (FF: 21 ± 9 ml.mmHg-1; RH: 38 ± 13 ml.mmHg-1; P < 0.01) and convective O2 delivery (FF: 0.30 ± 0.13 L.mmHg-1; RH: 0.64 ± 0.29 L.mmHg-1; P < 0.01) AUCs were all significantly greater in RH compared to FF. This augmented blood flow with RH resulted in an increased capillary blood flow (Figure 2) as indicated by the higher AUC in RH compared to FF (P < 0.05).
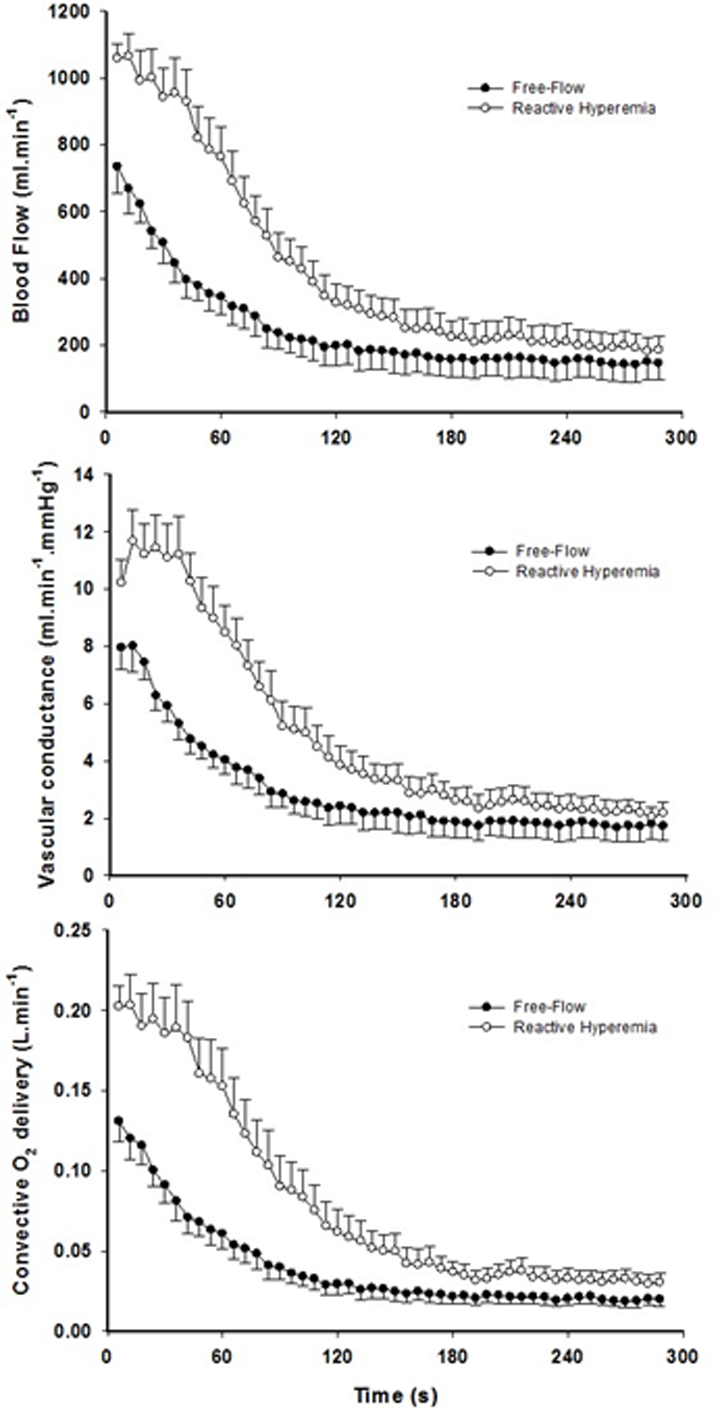
Figure 1. The effect of superimposing reactive hyperemia on the recovery from plantar flexion exercise on the peripheral arterial vasculature and O2 delivery. Blood flow (upper panel), vascular conductance (middle panel) and convective O2 delivery (lower panel) kinetics in older subjects. Area under the curve for blood flow (P < 0.01), vascular conductance (P < 0.01), and convective O2 delivery (P < 0.01) were all significantly greater in reactive hyperemia compared to free-flow conditions. Data are presented as mean ± SEM.
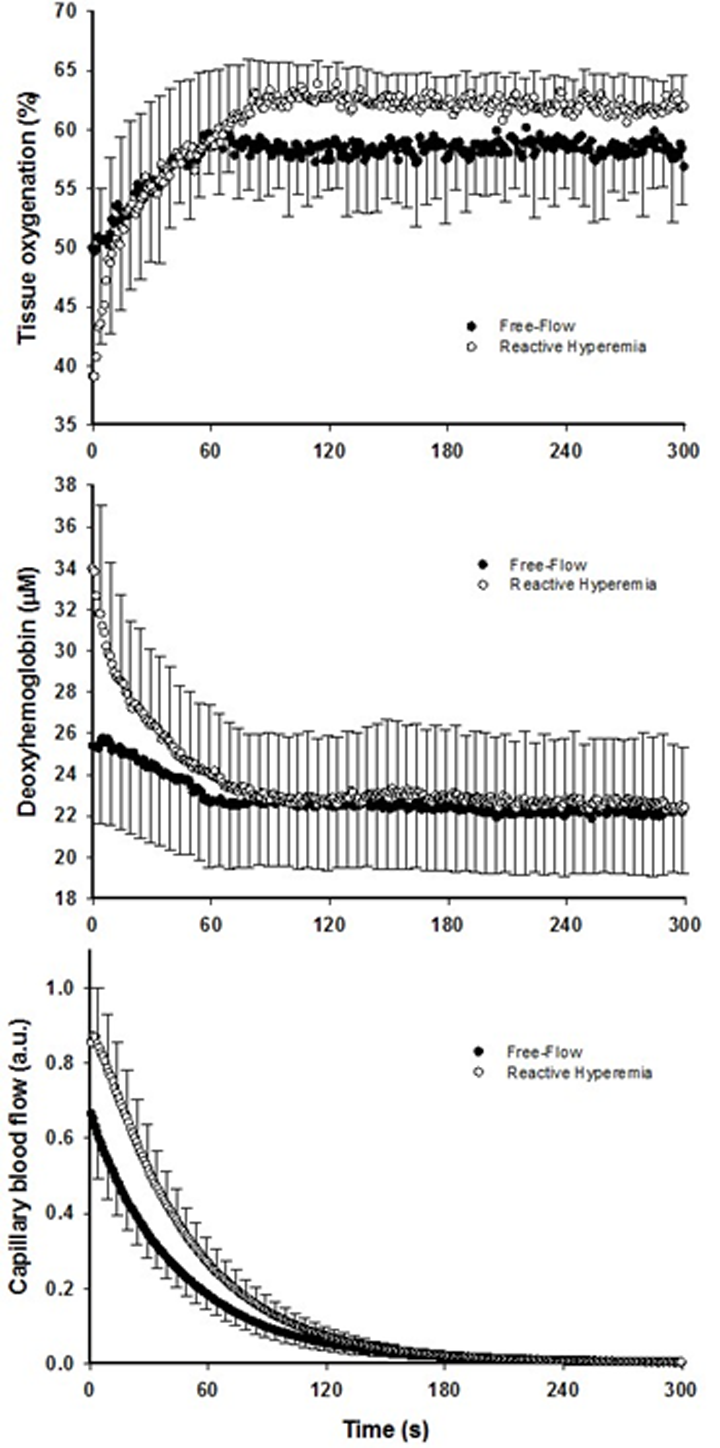
Figure 2. The effect of superimposing reactive hyperemia on the recovery from plantar flexion exercise on the peripheral microcirculation. Tissue oxygenation (upper panel), deoxyhemoglobin (middle panel), and capillary blood flow (bottom panel) kinetics in older subjects. Data are presented as mean ± SEM. Capillary blood flow and tissue oxygenation AUC were significantly greater in reactive hyperemia compared to free flow conditions (P < 0.05 and P < 0.01, respectively).
Although faster, the re-oxygenation kinetics were not significantly different owing to heterogeneous dynamics following the exercise-induced hyperemia (Figure 2, Table 2). However, a greater response in terms of amplitude during RH for both TOI and HHb was detected (P < 0.05). As a consequence, TOI (P < 0.01) AUC was significantly greater in RH compared to FF. Although not reaching significance (P = 0.07), Hbtot was greater upon release of the circulatory occlusion compared to FF (FF: 57 ± 28 µM; RH 64 ± 34 µM).
Metabolic offset kinetics assessed with 31P-MRS
Although shorter, the PCr resynthesis time constant did not reach significance in RH in comparison to FF (P = 0.15, Table 3), whereas the estimated mitochondrial ATP synthesis rate (Vmax) was significantly improved in RH (P < 0.05, Figure 3). The change in Vmax from RH to FF was neither correlated to changes in pH (r = -0.19, P = 0.56) nor [PCr] (r = -0.35, P = 0.26).
Table 3. Subject characteristics.
| Normal Range | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sample size | 12 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age (years) | 68 | ± | 8 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Anthropometric characteristics | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Height (cm) | 173 | ± | 9 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Weight (kg) | 74 | ± | 12 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BMI (kg/m2) | 25 | ± | 3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lower leg muscle volume (dL) | 21 | ± | 4 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lower leg adipose tissue thickness (mm) | 54 | ± | 18 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Functional characteristics | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Steps per day | 7047 | ± | 2573 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Moderate to vigorous activity (min/day) | 36 | ± | 20 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Blood characteristics | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Glucose (mg/dl) | 90 | ± | 11 | (74-106) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cholesterol (mg/dl) | 199 | ± | 42 | (118-210) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Triglycerides (mg/dl) | 132 | ± | 67 | (30-150) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HDL (mg/dl) | 49 | ± | 11 | (35-72) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| LDL (mg/dl) | 132 | ± | 67 | (0-100) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| WBC (K/ul) | 5.2 | ± | 1.3 | (3.7-9.9) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| RBC (M/ul) | 5.0 | ± | 0.5 | (4.0-5.6) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Haemoglobin (g/dl) | 15.2 | ± | 1.7 | (12.0-16.1) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hematocrit (%) | 45 | ± | 4 | (37.0-47.1) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Neutrophil (K/ul) | 3 | ± | 1 | (1.9-8.0) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lymphocyte (K/ul) | 1.5 | ± | 0.6 | (0.9-5.2) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Monocyte (K/ul) | 0.5 | ± | 0.1 | (0.16-1.50) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Data expressed as mean ± SD. Body mass index, BMI; high density lipoprotein, HDL; low density lipoprotein, LDL; white blood cells, WBC; red blood cells, RBC; normal range, (). | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
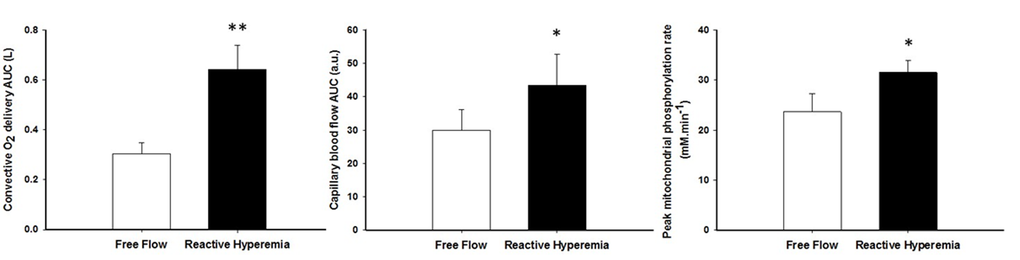
Figure 3. The effect of superimposing reactive hyperemia on the recovery from plantar flexion exercise on convective oxygen delivery (left panel), capillary blood flow (middle panel), and peak mitochondrial phosphorylation rate (Vmax)(right panel). Data are presented as mean ± SEM. * Reactive hyperemia significantly different from free flow condition (P < 0.05), ** (P < 0.01).
Discussion
Utilizing an integrative approach, this study sought to determine whether superimposing reactive hyperemia on the recovery from plantar flexion exercise would positively affect capillary blood flow, tissue re-oxygenation, and ultimately peak mitochondrial respiration rate in old untrained individuals. The three main findings of this study were that the release of brief circulatory occlusion at the offset of plantar flexion exercise in the elderly 1) substantially increased convective O2 delivery and muscle capillary blood flow, 2) this resulted in greater tissue re-oxygenation and microcirculatory O2 extraction, and 3) substantially improved peak mitochondrial ATP synthesis rate. Together, these findings indicate that an increase in O2 availability can enhance mitochondrial function in the plantar flexor muscles of the elderly, thereby revealing a metabolic reserve in skeletal muscle despite advancing age and low level of physical activity.
Increased convective O2 delivery and diffusional conductance for O2 after circulatory occlusion
The brief period of ischemia during exercise and the subsequent superimposing of the RH on the typically mono-exponential decay of blood flow post-exercise (FF) enhanced convective O2 delivery by 111% (Figures 1 and 3). Likewise, both limb blood flow and leg vascular conductance AUC were substantially augmented (+111% and +77%, respectively, Figure 1). As a result of these changes in bulk limb blood flow, capillary blood flow was significantly enhanced (+45%, Figure 2) as was microvascular re-oxygenation AUC (Figure 2), implying an improvement in local O2 availability.
Several mechanisms have been previously suggested to account for this increased muscle O2 delivery post-occlusion [15]. Among them, the accumulation of vasoactive metabolites and the subsequent reduction in vascular tone during the circulatory occlusion appear to be the most likely explanations. Accordingly, we observed a greater accumulation of [Pi] and a more pronounced acidosis at the end of exercise in RH in comparison to FF (Table 2). In addition, end-exercise microvascular oxygenation was significantly lower during the ischemic exercise, thus potentially eliciting the release of nitric oxide from the endothelium [16] or erythrocytes in the form of S-nitrosohemoglobin [17], or by augmenting the O2 dependent ATP-induced vasodilation in the microcirculation [18,19]. In addition to increasing bulk blood flow, these local adaptations would also promote an improvement in the regional match between microvascular perfusion and O2 utilisation post cuff occlusion thus further increasing the O2 availability in the active fibers.
In parallel with the large increase in convective O2 delivery in RH, it could be postulated that O2 diffusional conductance was also improved. Interestingly, the theoretical framework for the interaction between convective and diffusive O2 transport, developed by Wagner [20,21], offers to shed some light on this question. Using a modified version of this approach (Figure 4), it is readily apparent that, in addition to the RH-induced augmentation of convective O2 transport, diffusional O2 conductance was increased by ~90% in RH compared to FF. In fact, the increase O2 diffusional conductance in RH was similar in magnitude to the increase in convective O2 delivery (+111%), indicating that both components of O2 transport contributed, almost equally, to the improvement in muscle O2 utilization. In the present experimental conditions several mechanisms may have concurrently led to an enhanced capillary surface area available for O2 exchange, and thus greater diffusional O2 conductance in RH: 1) increased blood flow in originally poorly perfused capillaries (better perfusion-metabolism matching), 2) greater capillary hematocrit, as indicated by an 8% increase in total hemoglobin measured by NIRS, and 3) increased red blood cell velocity and fractional O2 extraction, increasing the length of individual capillaries over which blood–tissue O2 flux occurs (longitudinal recruitment) [26], as evidenced by both the greater capillary blood flow and deoxyhemoglobin signal (Figure 2).
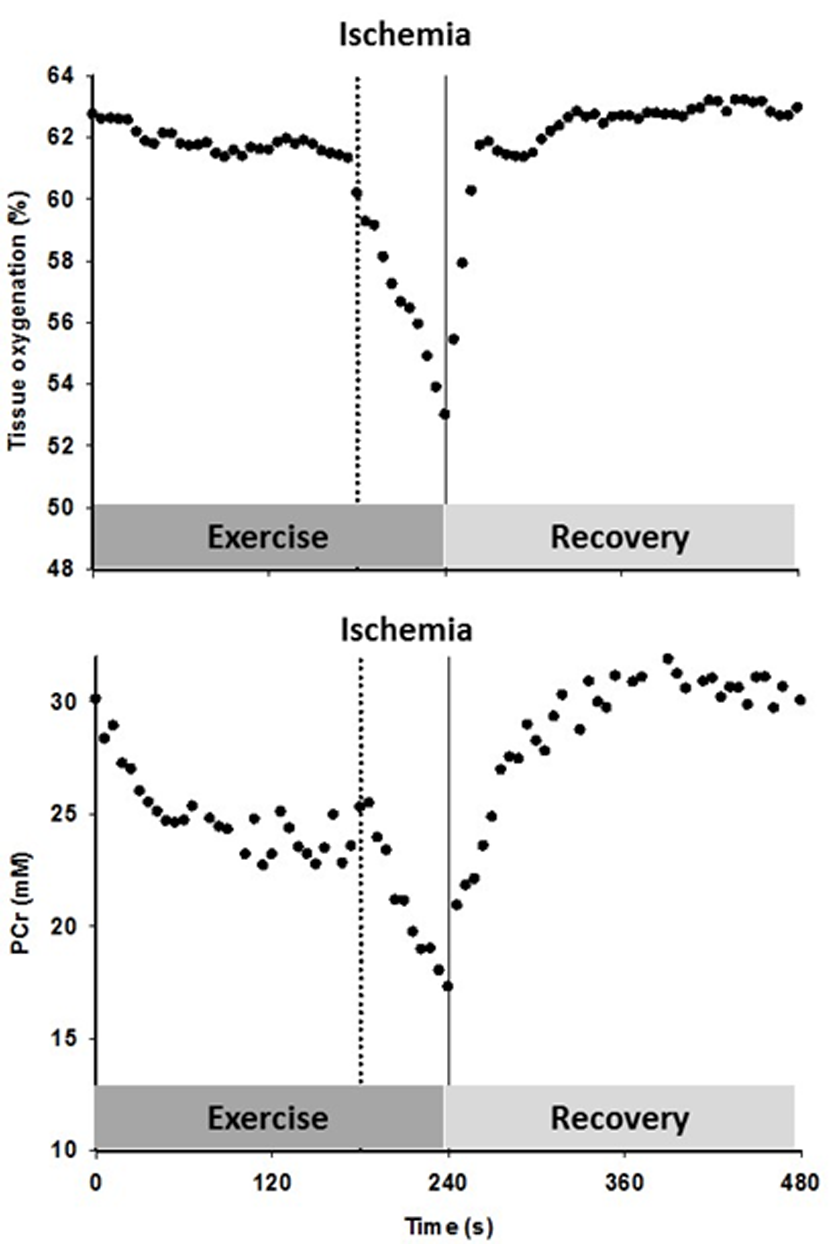
Figure 4. The effect of superimposing reactive hyperemia on the recovery from plantar flexion exercise on the relationship between microvascular partial pressure of O2 (PO2) and initial post exercise PCr resynthesis rate, an index of O2 utilization, in the context of diffusive and convective O2 transport. Muscle O2 utilization was inferred from the initial post exercise PCr resynthesis rate as this process is derived almost exclusively from oxidative phosphorylation [22]. Microvascular PO2 was derived from the tissue oxygen index [23], assuming that the near infrared spectroscopy signal mainly originates from hemoglobin [24], and then computed from the O2-hemoglobin dissociation curve [25]. Oxygen consumption, VO2; blood flow, Q; arterial oxygen content, CaO2; venous oxygen content, CvO2; diffusional conductance, DO2; Mean Capillary PO2, PCapO2.
Evidence of a metabolic reserve in aging skeletal muscle
According to our hypothesis, enhanced O2 availability increased peak mitochondrial phosphorylation rate by 36% (Figure 3). Although not significant, the PCr recovery time constant also tended to be faster in RH (P=0.15). This difference is likely related to the contrasting end-exercise muscle milieu in the RH and FF conditions (Table 2), which is well recognized to modulate the exercise PCr offset time constant [44,45] and confound the interpretation of this index. This inconsistency between the two indices can also be explained by the fact that our sample size was estimated to detect a large effect size on the peak mitochondrial phosphorylation rate. Given the greater variability of the PCr recovery time constant in the present experimental conditions, identifying a significant effect on this index would require to double the sample size to confirm our conclusion with the more robust Vmax parameter.
Overall, our results are consistent with the previously reported faster post-exercise PCr recovery kinetics induced by enhanced tissue perfusion following acute antioxidant administration in elderly subjects [12]. It should, however, be noted that, unlike the present experimental paradigm, antioxidant administration may exert both direct and indirect effects on muscle metabolism as enhanced scavenging of free radicals can induce a subsequent increase in NO bioavailability, which has been documented to improve both muscle blood flow [46,47] and mitochondrial function [14,48]. Based upon these considerations, our results provide the first unambiguous evidence of an excess capacity of mitochondrial phosphorylation relative to peripheral O2 transport in the calf muscle of untrained elderly subjects.
Interestingly, a prior study from our group using hyperoxia to increase the O2 gradient from blood to muscle did not observe such an improvement in mitochondrial function in sedentary older individuals [11]. These findings were interpreted as evidence that mitochondrial function in this population was likely limited by intrinsic mitochondrial capacity rather than O2 availability, which appears to be in conflict with the current findings. However, in this prior study, O2 availability was altered predominantly by an increase in blood PO2 augmenting the O2 gradient from blood to mitochondria. Owing to the sigmoidal shape of the O2-hemoglobin dissociation curve at high PO2 and the already almost fully saturated hemoglobin in normoxia, gains in arterial O2 concentration in hyperoxia are therefore rather small (<10% [20],). In contrast, in the present study, convective O2 delivery was dramatically enhanced in RH (+ 111%, Figure 3) and, interestingly, this also resulted in an improvement in diffusional O2 conductance of a similar magnitude (+ 90%, Figure 5). This difference may explain the discrepancy between studies.
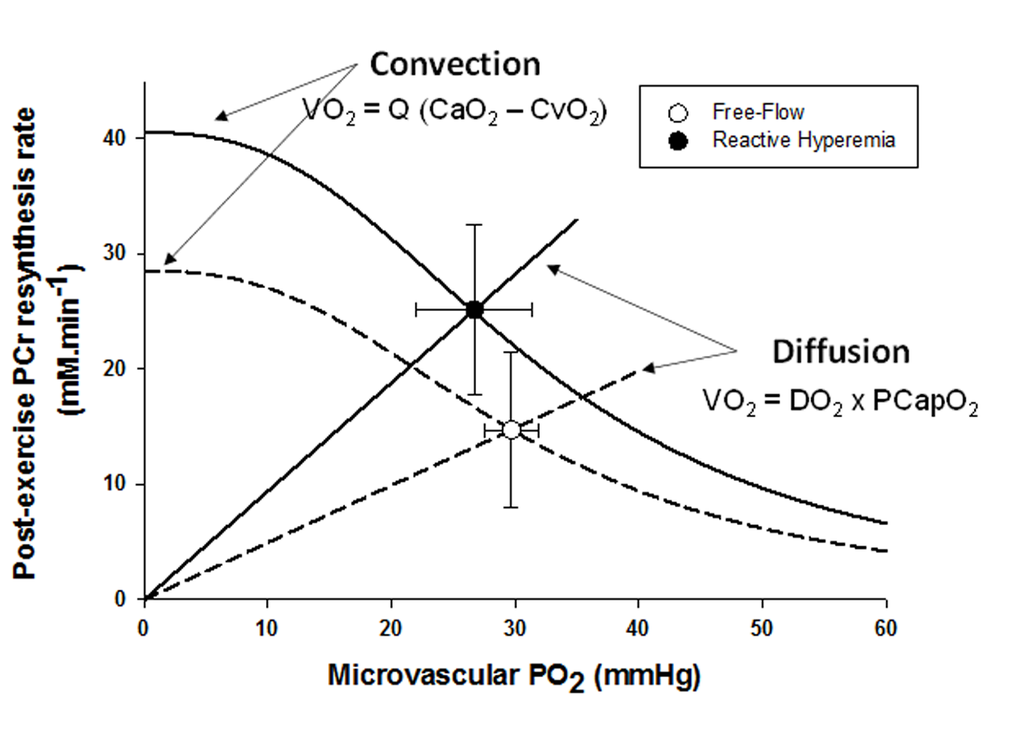
Figure 5. The timeline for the constant-load sub-maximal plantar flexion exercise and recovery, illustrated with an example of the tissue oxygenation and phosphocreatine (PCr) response to the protocol including cuff occlusion during the last min of exercise and the subsequent reoxygenation in an old subject.
One could question whether the increase in peak mitochondrial ATP synthesis rate might be the consequence of the confounding effects of differing end-exercise metabolic states on the calculation of Vmax or a direct effect on mitochondrial enzymatic activities. As for the first point, unlike the PCr recovery time constant, Vmax has been repeatedly demonstrated to be independent from the end-exercise metabolic state [49–51]. In agreement with this literature, Vmax was neither correlated to changes in pH nor [PCr] thus ruling out differing end-metabolic states as a confounding factor. As for the second point, several in vivo and in vitro studies suggest that, in conditions of high respiratory rate (ADP-stimulated respiration in isolated mitochondria or during the transition from exercise to rest), intrinsic mitochondrial function is not influenced by differences in metabolic state. For instance, lowering the pH from 7.4 to 6.38 did not affect the maximal ADP-stimulated respiration in isolated mitochondria from the skeletal muscle [52]. Consistent with this in vitro finding and our correlation analyses, the maximum rate of mitochondrial ATP synthesis measured by 31P-MRS has been documented to be similar following plantar-flexion exercise at two different durations, despite vastly different pH and metabolite concentrations [50]. Combined with the finding that the maximum rate of mitochondrial ATP synthesis is independent of pH and [PCr] across a wide range of metabolic states (pH ranging from 7.1 to 6.0, and [PCr] from 35 mM to 2 mM [49,51], these data suggest that it is very unlikely that the greater Vmax in RH was the result of an intrinsic improvement in mitochondrial function.
Perspective and significance
Despite the current evidence of a mitochondrial reserve in the elderly, one should not completely dismiss the potential role of factors downstream from the convective delivery of O2 in terms of contributing to the decline in muscle aerobic function with advancing age. Indeed, using an animal preparation, Hepple et al. [53] elegantly illustrated the complex interaction between muscle oxidative capacity and O2 delivery which determine maximal O2 uptake (VO2max) in skeletal muscle. Specifically, a combined reduction in O2 delivery and mitochondrial oxidative capacity resulted in a greater impairment in muscle VO2max than either intervention performed independently in young rats. The implication of this finding for the present results is that, even though intrinsic mitochondrial capacity appears to be in relative excess of convective O2 transport in the elderly, mitochondrial capacity may still influence muscle VO2max [53]. Therefore, the decline in mitochondrial function [54,55] and/or density [55] previously suggested by some in vitro reports may still play some role in the impaired ability of aged muscles to use O2 during high-intensity exercise.
From a theoretical standpoint, the findings from the present study challenge the classic concept of symmorphosis, which postulates that each component of the O2 transport system (heart, blood, capillaries and mitochondria) is matched to demand such that the functional capacity of each step does not limit the maximal overall capacity of the system [56]. Indeed, according to this theory, the peak rate of mitochondrial phosphorylation measured by 31P-MRS should not be sensitive to an acute increase in capillary blood flow and O2 availability within the skeletal muscle. In addition, despite the well-established impairments in the convective component of O2 transport to skeletal muscle with age [1,2], this does not appear to have resulted in a proportional decrease in mitochondrial function in the elderly, as demonstrated in the present study.
Methodological consideration
It should be noted that the volume of muscle interrogated by the NIRS was small and localized to the medial gastrocnemius, while, in contrast, the 31P-MR signal was detected with a relatively large surface coil, sampling a much greater muscle volume. However, considering that the 31P-MR reception sensitivity is greatest close to the coil, the gastrocnemius (lateral and medial) and the soleus contributed to the MR signal in a weighted fashion, with the soleus contributing to a much smaller extent. In addition, this study employed a within-subject comparison, such that 31P-MRS and NIRS signals originated, for each method, from the same muscle volume in both conditions. Therefore, while acknowledging the potential influence of these differing measurement approaches, it is reasonable to contend that our qualitative estimate of capillary blood flow was not greatly confounded by sampling volume differences between techniques.
Conclusion
In summary, using an integrative approach, this study has revealed a hemodynamic reserve in the contracting calf muscle accessible under conditions of superimposed reactive hyperemia, which appears, to some extent, to be blunted in the microcirculation with advancing age. In addition, this study provides the first direct evidence of an excess capacity of mitochondrial respiration relative to peripheral O2 transport capacity in the calf muscle of untrained elderly
Materials and Methods
Subjects
Following the attainment of informed consent, 12 healthy sedentary elderly subjects (10 men and 2 women), participated in this study (Table 3). The subjects were recruited based upon no evidence of regular physical activity above that required for activities of daily living (assessed by both questionnaire and accelerometry), and being greater than 60 years of age. All subjects were non-smokers, free of diabetes, and known cardiovascular, peripheral vascular, neuromuscular, or pulmonary disease. Additionally, none of the subjects were taking statins or other medications recognized to affect muscle or vascular function. Women taking hormone replacement therapy were excluded from the study. The study was approved by the Human Research Protection Programs of both the University of Utah and the Salt Lake City VA Medical Center.
Exercise protocol
After familiarization with the equipment, individual maximum work rate (WRmax) was determined by performing incremental dynamic plantar flexion exercise until exhaustion. On a separate day, subjects performed constant-load sub-maximal plantar flexion at ~40% of WRmax (frequency of 1 Hz) in the whole body MRI system (TimTrio, 2.9T Siemens Medical Systems, Erlangen, Germany) under conditions of FF and then RH, as PCr recovery kinetics are unaffected by prior exercise [50]. Specifically, after 2 min of rest, subjects exercised for 4 min followed by 5 min of recovery. In RH, min 3-4 was performed under ischemic conditions induced by a cuff occlusion (Figure 5). A blood pressure cuff, placed distal to the knee, was rapidly inflated to 250 mmHg to occlude the popliteal artery. In a subset of 9 subjects, this protocol was repeated on a separate day in order to measure limb and capillary blood flow as well as tissue oxygenation using Doppler ultrasound imaging and NIRS, respectively. Prior to initiation of this protocol, blood samples were collected to perform a complete blood cell count analysis.
31P MRS
31P-MRS was performed using a clinical 2.9T MRI system (Tim-Trio, Siemens Medical Solutions, Erlangen, Germany) operating at 49.9 MHz for 31P resonance. 31P MRS data were acquired with a dual tuned 31P-1H surface coil with linear polarization (Rapid biomedical GmbH, Rimpar, Germany) positioned under the calf at its maximum diameter. The 31P single-loop coil diameter was 125 mm surrounding a 110 mm 1H coil loop. The centering of the coil around the leg was confirmed by T1 weighted 1H localizing images and the coil was repositioned if the majority of the gastrocnemius muscle was not within this range. For all subjects a similar ratio between the volumes of gastrocnemius / soleus muscles was maintained within the coil. After a three-plane scout proton image, advanced localized volume shimming was performed. Before each experiment, two fully relaxed spectra were acquired at rest with 3 averages per spectrum and a repetition time of 30 s. Then, MRS data acquisition was performed throughout the rest-exercise-recovery protocol using a FID (free-induction-decay) pulse sequence with a 2.56 ms adiabatic-half-passage excitation radiofrequency pulse and the following parameters: repetition time = 2 s; receiver bandwidth = 5 kHz; 1024 data points; and 3 averages per spectrum). Saturation factors were quantified by the comparison between fully relaxed (TR = 30 s) and partially relaxed spectra (TR = 2s).
As previously described [57], relative concentrations of PCr, inorganic phosphate Pi, and ATP were obtained by a time-domain fitting routine using the Advanced Method for Accurate, Robust and Efficient Spectral (AMARES) fitting algorithm [58] incorporated into the CSIAPO software [49]. Intracellular pH was calculated from the chemical shift difference between the Pi and PCr signals. The free cytosolic [ADP] was calculated from [PCr] and pH using the creatine kinase equilibrium constant (KCK = 1.66 × 109 M-1) and assuming that phosphocreatine represents 85% of total creatine content [59]. The resting concentrations were calculated from the average peak areas of the three relaxed spectra (TR =30 s; N = 3) recorded at rest, assuming an 8.2 mM ATP concentration. When Pi splitting was evident, the pH corresponding to each Pi pool was calculated separately as pH1 and pH2 on the basis of the chemical shift of each peak relative to PCr. The overall muscle pH was then calculated as pH = pH1 (area Pi1/total Pi area) + pH2 (area Pi2/total Pi area).
Popliteal blood flow
Measurements of popliteal artery blood velocity and vessel diameter were performed in the popliteal fossa of the exercising leg proximal to the branching of the medial inferior genicular artery with a Logic 7 Doppler ultrasound system (General Electric Medical Systems, Milwaukee, WI). The ultrasound system was equipped with a linear transducer operating at an imaging frequency of 10 MHz. Vessel diameter was determined at a perpendicular angle along the central axis of the scanned area. Blood velocity was measured using the same transducer with a frequency of 5 MHz. All blood velocity measurements were obtained with the probes appropriately positioned to maintain an insonation angle of 60° or less. The sample volume was maximized according to vessel size and was centered within the vessel. Arterial diameter was measured off-line every 12 s using automated edge-detection software (Medical Imaging Applications, Coralville, IA), and mean velocity (Vmean) (angle corrected, and intensity-weighted area under the curve) was automatically calculated beat by beat (Logic 7). Using arterial diameter and Vmean, blood flow in the popliteal artery was calculated as blood flow = Vmean ∙ π (vessel diameter/2)2 ∙ 60, where blood flow is in milliliters per minute. Mean arterial pressure (MAP), heart rate, stroke volume, and cardiac output were determined with a Finometer (Finapres Medical Systems, Amsterdam, The Netherlands). Leg vascular conductance was then calculated as popliteal artery blood flow divided by MAP. Arterial O2 content (CaO2) was calculated as the sum of bound O2 (0.0134 ∙ Hb ∙ SaO2) and dissolved O2 (0.000031 ∙ PO2) assuming a constant SaO2 = 94% and PO2 = 70.8 mmHg, based upon typical values and a normal Hb association curve [60]. O2 delivery was then calculated as the product of CaO2 and popliteal artery blood flow.
Microvascular oxygenation and capillary blood flow
Microvascular oxygenation was assessed using the NIRS technique, which provides continuous, non-invasive measurements of oxygenated (HbO2), deoxygenated (HHb) and total (Hbtot) haemoglobin levels as well as a tissue oxygenation index (TOI, i.e. HbO2/Hbtot). Due to identical spectral characteristics, haemoglobin and myoglobin are not separated using NIRS. However, the signal is usually considered as being derived mainly from Hb [24]. In the present study, changes in microvascular oxygenation of the right gastrocnemius muscle were continuously monitored at 2 Hz using a near infrared frequency resolved spectroscopy oximeter (Oxiplex TS, ISS Inc., Illinois USA). The probe was positioned at the level of the largest circumference of the medial gastrocnemius and secured with Velcro straps and bi-adhesive tape. NIRS uses intensity-modulated light and the probe consisted of 8 infrared light sources (4 emitting at 690 nm and 4 emitting at 830 nm) and one detection channel (interoptode distance = 1.5 to 4.5 cm) including a selected light detector (photomultiplier tube), thus providing a measurement of absorption and the scattering coefficient of the tissues. Measurement of adipose tissue thickness under the NIRS sample site was performed with a Logic 7 Doppler ultrasound system (General Electric Medical Systems, Milwaukee, WI).
As previously described [15], the estimated capillary blood flow response following the offset of exercise was calculated from a modified version of the method proposed by Ferreira et al. [61,62] using the kinetics of muscle O2 consumption and the HHb data. Specifically, the PCr resynthesis rate, measured by 31P-MRS, which is derived almost exclusively from oxidative phosphorylation [22], was used as an index of muscle O2 consumption. Then, as the HHb response determined by NIRS is considered to reflect muscle capillary O2 extraction (i.e., CaO2-CvO2) [24], and based upon the Fick equation, the temporal characteristics of capillary blood flow were estimated using the PCr resynthesis rate to HHb ratio.
Data analysis
The PCr, HHb, and TOI recovery kinetics were determined by fitting the time-dependent changes during the recovery period to a mono-exponential curve described by the following equation:
Y (t) =Yend + Yres (1-e-(t-TD/τ))
where Yend is the level of [PCr], HHb, or TOI measured at end-of-exercise and Yres refers to the amount of PCr resynthesized or the resaturation during the recovery. Unlike TOI or HHb, there is no time delay (TD) in the resynthesis of PCr and therefore TD was fixed to 0 for PCr kinetics. Then, the initial rate of PCr resynthesis from 31P-MRS (ViPCr) was calculated from the derivative of equation (1) at time zero:
ViPCr = k · ∆[PCr]
in which ∆ [PCr], represents the amount of PCr resynthesized during the recovery and the rate constant k = 1/τ [63].
Then, peak rate of oxidative ATP synthesis from 31P-MRS (Vmax in mM.min-1) was calculated using the initial rate of PCr synthesis (ViPCr) during the recovery period and [ADP] obtained at the end of exercise as previously described [64]:
Vmax = ViPCr (1 + (Km/[ADP]end2.2))
in which Km (the [ADP] at half the highest oxidation rate) is 30 µM in skeletal muscle [63].
Model variables were determined with an iterative process by minimizing the sum of squared residuals (RSS) between the fitted function and the observed values. Goodness of fit was assessed by visual inspection of the residual plot and the frequency plot distribution of the residuals, Chi square values, and the coefficient of determination (r2), which was calculated as follows [65]:
r2 = 1- (SSreg/SStot)
with SSreg, the sum of squares of the residuals from the fit and SStot, and the sum of squares of the residuals from the mean. In a subset of subjects demonstrating the greatest drop in pH (n=4), we determined that a single exponential function rather than a double exponential function [66] provided the best fit of PCr recovery kinetics.
Statistical Analysis
The assessment of differences between FF and RH was performed with either paired t-tests or nonparametric Wilcoxon tests, where appropriate (Statsoft, version 5.5; Statistica, Tulsa, Oklahoma). Popliteal blood flow, leg vascular conductance, convective O2 delivery, capillary blood flow and TOI cumulative area under the curve (AUC) was calculated as the summed sec-by-sec response during the first 180 s of recovery and used to identify how differences over time were affected by the treatment. Statistical significance was accepted at P < 0.05. Results are presented as mean ± SD in tables and mean ± SEM in figures for clarity.
Acknowledgements
The Authors wish to thank all the subjects in this study for their committed participation in this research.
Funding
This work was funded in part by grants from the Flight Attendant Medical Research Institute (FAMRI), NIH National Heart, Lung, and Blood Institute (PO1 HL 091830, K99HL125756) and VA Merit grant E6910R.
Conflicts of Interest
No conflicts of interest, financial or otherwise, are declared by the author(s).
References
- 1. Poole D, Behnke B, Musch T. Capillary hemodynamics and oxygen pressures in the aging microcirculation. Microcirculation. 2006; 13:289–99. https://doi.org/10.1080/10739680600618793 [PubMed]
- 2. Poole JG, Lawrenson L, Kim J, Brown C, Richardson RS. Vascular and metabolic response to cycle exercise in sedentary humans: effect of age. Am J Physiol Heart Circ Physiol. 2003; 284:H1251–59. https://doi.org/10.1152/ajpheart.00790.2002 [PubMed]
- 3. Venturelli M, Schena F, Scarsini R, Muti E, Richardson RS. Limitations to exercise in female centenarians: evidence that muscular efficiency tempers the impact of failing lungs. Age (Dordr). 2013; 35:861–70. https://doi.org/10.1007/s11357-011-9379-1 [PubMed]
- 4. Taylor DJ, Kemp GJ, Thompson CH, Radda GK. Ageing: effects on oxidative function of skeletal muscle in vivo. Mol Cell Biochem. 1997; 174:321–24. https://doi.org/10.1023/A:1006802602497 [PubMed]
- 5. McCully KK, Fielding RA, Evans WJ, Leigh JS
Jr , Posner JD. Relationships between in vivo and in vitro measurements of metabolism in young and old human calf muscles. J Appl Physiol (1985). 1993; 75:813–19. [PubMed] - 6. Conley KE, Jubrias SA, Esselman PC. Oxidative capacity and ageing in human muscle. J Physiol. 2000; 526:203–10. https://doi.org/10.1111/j.1469-7793.2000.t01-1-00203.x [PubMed]
- 7. Wray DW, Nishiyama SK, Monnet A, Wary C, Duteil S, Carlier PG, Richardson RS. Multiparametric NMR-based assessment of skeletal muscle perfusion and metabolism during exercise in elderly persons: preliminary findings. J Gerontol A Biol Sci Med Sci. 2009; 64:968–74. https://doi.org/10.1093/gerona/glp044 [PubMed]
- 8. Lanza IR, Befroy DE, Kent-Braun JA. Age-related changes in ATP-producing pathways in human skeletal muscle in vivo. J Appl Physiol (1985). 2005; 99:1736–44. https://doi.org/10.1152/japplphysiol.00566.2005 [PubMed]
- 9. Kent-Braun JA, Ng AV. Skeletal muscle oxidative capacity in young and older women and men. J Appl Physiol (1985). 2000; 89:1072–78. [PubMed]
- 10. Lawrenson L, Poole JG, Kim J, Brown C, Patel P, Richardson RS. Vascular and metabolic response to isolated small muscle mass exercise: effect of age. Am J Physiol Heart Circ Physiol. 2003; 285:H1023–31. https://doi.org/10.1152/ajpheart.00135.2003 [PubMed]
- 11. Layec G, Haseler LJ, Richardson RS. Reduced muscle oxidative capacity is independent of O2 availability in elderly people. Age (Dordr). 2013; 35:1183–92. https://doi.org/10.1007/s11357-012-9442-6 [PubMed]
- 12. Wray DW, Nishiyama SK, Monnet A, Wary C, Duteil SS, Carlier PG, Richardson RS. Antioxidants and aging: NMR-based evidence of improved skeletal muscle perfusion and energetics. Am J Physiol Heart Circ Physiol. 2009; 297:H1870–75. https://doi.org/10.1152/ajpheart.00709.2009 [PubMed]
- 13. Wray DW, Nishiyama SK, Donato AJ, Carlier P, Bailey DM, Uberoi A, Richardson RS. The paradox of oxidative stress and exercise with advancing age. Exerc Sport Sci Rev. 2011; 39:68–76. https://doi.org/10.1097/JES.0b013e31820d7657 [PubMed]
- 14. Larsen FJ, Schiffer TA, Borniquel S, Sahlin K, Ekblom B, Lundberg JO, Weitzberg E. Dietary inorganic nitrate improves mitochondrial efficiency in humans. Cell Metab. 2011; 13:149–59. https://doi.org/10.1016/j.cmet.2011.01.004 [PubMed]
- 15. Layec G, Haseler LJ, Trinity JD, Hart CR, Liu X, Le Fur Y, Jeong EK, Richardson RS. Mitochondrial function and increased convective O2 transport: implications for the assessment of mitochondrial respiration in vivo. J Appl Physiol (1985). 2013; 115:803–11. https://doi.org/10.1152/japplphysiol.00257.2013 [PubMed]
- 16. Pohl U, Busse R. Hypoxia stimulates release of endothelium-derived relaxant factor. Am J Physiol. 1989; 256:H1595–600. [PubMed]
- 17. Stamler JS, Jia L, Eu JP, McMahon TJ, Demchenko IT, Bonaventura J, Gernert K, Piantadosi CA. Blood flow regulation by S-nitrosohemoglobin in the physiological oxygen gradient. Science. 1997; 276:2034–37. https://doi.org/10.1126/science.276.5321.2034 [PubMed]
- 18. Kirby BS, Crecelius AR, Richards JC, Dinenno FA. Sources of intravascular ATP during exercise in humans: critical role for skeletal muscle perfusion. Exp Physiol. 2013; 98:988–98. https://doi.org/10.1113/expphysiol.2012.071555 [PubMed]
- 19. Ellsworth ML, Sprague RS. Regulation of blood flow distribution in skeletal muscle: role of erythrocyte-released ATP. J Physiol. 2012; 590:4985–91. https://doi.org/10.1113/jphysiol.2012.233106 [PubMed]
- 20. Wagner PD. Determinants of maximal oxygen transport and utilization. Annu Rev Physiol. 1996; 58:21–50. https://doi.org/10.1146/annurev.ph.58.030196.000321 [PubMed]
- 21. Wagner PD. Systemic oxygen transport and utilization. J Breath Res. 2008; 2:024001. https://doi.org/10.1088/1752-7155/2/2/024001 [PubMed]
- 22. Quistorff B, Johansen L, Sahlin K. Absence of phosphocreatine resynthesis in human calf muscle during ischaemic recovery. Biochem J. 1993; 291:681–86. https://doi.org/10.1042/bj2910681 [PubMed]
- 23. Broxterman RM, Ade CJ, Wilcox SL, Schlup SJ, Craig JC, Barstow TJ. Influence of duty cycle on the power-duration relationship: observations and potential mechanisms. Respir Physiol Neurobiol. 2014; 192:102–11. https://doi.org/10.1016/j.resp.2013.11.010 [PubMed]
- 24. Koga S, Kano Y, Barstow TJ, Ferreira LF, Ohmae E, Sudo M, Poole DC. Kinetics of muscle deoxygenation and microvascular PO(2) during contractions in rat: comparison of optical spectroscopy and phosphorescence-quenching techniques. J Appl Physiol (1985). 2012; 112:26–32. https://doi.org/10.1152/japplphysiol.00925.2011 [PubMed]
- 25. Severinghaus JW. Simple, accurate equations for human blood O2 dissociation computations. J Appl Physiol. 1979; 46:599–602. [PubMed]
- 26. Poole DC, Copp SW, Hirai DM, Musch TI. Dynamics of muscle microcirculatory and blood-myocyte O(2) flux during contractions. Acta Physiol (Oxf). 2011; 202:293–310. https://doi.org/10.1111/j.1748-1716.2010.02246.x [PubMed]
- 27. Proctor DN, Le KU, Ridout SJ. Age and regional specificity of peak limb vascular conductance in men. J Appl Physiol (1985). 2005; 98:193–202. https://doi.org/10.1152/japplphysiol.00704.2004 [PubMed]
- 28. Chilibeck PD, Paterson DH, Cunningham DA, Taylor AW, Noble EG. Muscle capillarization O2 diffusion distance, and VO2 kinetics in old and young individuals. J Appl Physiol (1985). 1997; 82:63–69. [PubMed]
- 29. Coggan AR, Spina RJ, King DS, Rogers MA, Brown M, Nemeth PM, Holloszy JO. Histochemical and enzymatic comparison of the gastrocnemius muscle of young and elderly men and women. J Gerontol. 1992; 47:B71–76. https://doi.org/10.1093/geronj/47.3.B71 [PubMed]
- 30. Tanaka H, Reiling MJ, Seals DR. Regular walking increases peak limb vasodilatory capacity of older hypertensive humans: implications for arterial structure. J Hypertens. 1998; 16:423–28. https://doi.org/10.1097/00004872-199816040-00003 [PubMed]
- 31. Parker BA, Smithmyer SL, Jarvis SS, Ridout SJ, Pawelczyk JA, Proctor DN. Evidence for reduced sympatholysis in leg resistance vasculature of healthy older women. Am J Physiol Heart Circ Physiol. 2007; 292:H1148–56. https://doi.org/10.1152/ajpheart.00729.2006 [PubMed]
- 32. Heinonen I, Nesterov SV, Kemppainen J, Nuutila P, Knuuti J, Laitio R, Kjaer M, Boushel R, Kalliokoski KK. Role of adenosine in regulating the heterogeneity of skeletal muscle blood flow during exercise in humans. J Appl Physiol (1985). 2007; 103:2042–48. https://doi.org/10.1152/japplphysiol.00567.2007 [PubMed]
- 33. Heinonen I, Wendelin-Saarenhovi M, Kaskinoro K, Knuuti J, Scheinin M, Kalliokoski KK. Inhibition of α-adrenergic tone disturbs the distribution of blood flow in the exercising human limb. Am J Physiol Heart Circ Physiol. 2013; 305:H163–72. https://doi.org/10.1152/ajpheart.00925.2012 [PubMed]
- 34. Musch TI, Eklund KE, Hageman KS, Poole DC. Altered regional blood flow responses to submaximal exercise in older rats. J Appl Physiol (1985). 2004; 96:81–88. https://doi.org/10.1152/japplphysiol.00729.2003 [PubMed]
- 35. Trinity JD, Groot HJ, Layec G, Rossman MJ, Ives SJ, Morgan DE, Gmelch BS, Bledsoe A, Richardson RS. Passive leg movement and nitric oxide-mediated vascular function: the impact of age. Am J Physiol Heart Circ Physiol. 2015; 308:H672–79. https://doi.org/10.1152/ajpheart.00806.2014 [PubMed]
- 36. McDaniel J, Hayman MA, Ives S, Fjeldstad AS, Trinity JD, Wray DW, Richardson RS. Attenuated exercise induced hyperaemia with age: mechanistic insight from passive limb movement. J Physiol. 2010; 588:4507–17. https://doi.org/10.1113/jphysiol.2010.198770 [PubMed]
- 37. Groot HJ, Trinity JD, Layec G, Rossman MJ, Ives SJ, Richardson RS. Perfusion pressure and movement-induced hyperemia: evidence of limited vascular function and vasodilatory reserve with age. Am J Physiol Heart Circ Physiol. 2013; 304:H610–19. https://doi.org/10.1152/ajpheart.00656.2012 [PubMed]
- 38. Dinenno FA, Jones PP, Seals DR, Tanaka H. Limb blood flow and vascular conductance are reduced with age in healthy humans: relation to elevations in sympathetic nerve activity and declines in oxygen demand. Circulation. 1999; 100:164–70. https://doi.org/10.1161/01.CIR.100.2.164 [PubMed]
- 39. Taylor JA, Hand GA, Johnson DG, Seals DR. Augmented forearm vasoconstriction during dynamic exercise in healthy older men. Circulation. 1992; 86:1789–99. https://doi.org/10.1161/01.CIR.86.6.1789 [PubMed]
- 40. Thijssen DH, Rongen GA, van Dijk A, Smits P, Hopman MT. Enhanced endothelin-1-mediated leg vascular tone in healthy older subjects. J Appl Physiol (1985). 2007; 103:852–57. https://doi.org/10.1152/japplphysiol.00357.2007 [PubMed]
- 41. Wray DW, Nishiyama SK, Harris RA, Richardson RS. Angiotensin II in the elderly: impact of angiotensin II type 1 receptor sensitivity on peripheral hemodynamics. Hypertension. 2008; 51:1611–16. https://doi.org/10.1161/HYPERTENSIONAHA.108.111294 [PubMed]
- 42. Wray DW, Nishiyama SK, Richardson RS. Role of alpha1-adrenergic vasoconstriction in the regulation of skeletal muscle blood flow with advancing age. Am J Physiol Heart Circ Physiol. 2009; 296:H497–504. https://doi.org/10.1152/ajpheart.01016.2008 [PubMed]
- 43. Koch DW, Leuenberger UA, Proctor DN. Augmented leg vasoconstriction in dynamically exercising older men during acute sympathetic stimulation. J Physiol. 2003; 551:337–44. https://doi.org/10.1113/jphysiol.2003.042747 [PubMed]
- 44. Iotti S, Lodi R, Frassineti C, Zaniol P, Barbiroli B. In vivo assessment of mitochondrial functionality in human gastrocnemius muscle by 31P MRS. The role of pH in the evaluation of phosphocreatine and inorganic phosphate recoveries from exercise. NMR Biomed. 1993; 6:248–53. https://doi.org/10.1002/nbm.1940060404 [PubMed]
- 45. Roussel M, Bendahan D, Mattei JP, Le Fur Y, Cozzone PJ. 31P magnetic resonance spectroscopy study of phosphocreatine recovery kinetics in skeletal muscle: the issue of intersubject variability. Biochim Biophys Acta. 2000; 1457:18–26. https://doi.org/10.1016/S0005-2728(99)00111-5 [PubMed]
- 46. Donato AJ, Uberoi A, Wray DW, Nishiyama S, Lawrenson L, Richardson RS. Differential effects of aging on limb blood flow in humans. Am J Physiol Heart Circ Physiol. 2006; 290:H272–78. https://doi.org/10.1152/ajpheart.00405.2005 [PubMed]
- 47. Kirby BS, Voyles WF, Simpson CB, Carlson RE, Schrage WG, Dinenno FA. Endothelium-dependent vasodilatation and exercise hyperaemia in ageing humans: impact of acute ascorbic acid administration. J Physiol. 2009; 587:1989–2003. https://doi.org/10.1113/jphysiol.2008.167320 [PubMed]
- 48. Larsen FJ, Weitzberg E, Lundberg JO, Ekblom B. Dietary nitrate reduces maximal oxygen consumption while maintaining work performance in maximal exercise. Free Radic Biol Med. 2010; 48:342–47. https://doi.org/10.1016/j.freeradbiomed.2009.11.006 [PubMed]
- 49. Le Fur Y, Nicoli F, Guye M, Confort-Gouny S, Cozzone PJ, Kober F. Grid-free interactive and automated data processing for MR chemical shift imaging data. MAGMA. 2010; 23:23–30. https://doi.org/10.1007/s10334-009-0186-y [PubMed]
- 50. Walter G, Vandenborne K, McCully KK, Leigh JS. Noninvasive measurement of phosphocreatine recovery kinetics in single human muscles. Am J Physiol. 1997; 272:C525–34. [PubMed]
- 51. Lodi R, Kemp GJ, Iotti S, Radda GK, Barbiroli B. Influence of cytosolic pH on in vivo assessment of human muscle mitochondrial respiration by phosphorus magnetic resonance spectroscopy. MAGMA. 1997; 5:165–71. https://doi.org/10.1007/BF02592248 [PubMed]
- 52. Tonkonogi M, Sahlin K. Actively phosphorylating mitochondria are more resistant to lactic acidosis than inactive mitochondria. Am J Physiol. 1999; 277:C288–93. [PubMed]
- 53. Hepple RT, Hagen JL, Krause DJ. Oxidative capacity interacts with oxygen delivery to determine maximal O(2) uptake in rat skeletal muscles in situ. J Physiol. 2002; 541:1003–12. https://doi.org/10.1113/jphysiol.2001.013649 [PubMed]
- 54. Larsen RG, Callahan DM, Foulis SA, Kent-Braun JA. Age-related changes in oxidative capacity differ between locomotory muscles and are associated with physical activity behavior. Appl Physiol Nutr Metab. 2012; 37:88–99. https://doi.org/10.1139/h11-135 [PubMed]
- 55. Short KR, Bigelow ML, Kahl J, Singh R, Coenen-Schimke J, Raghavakaimal S, Nair KS. Decline in skeletal muscle mitochondrial function with aging in humans. Proc Natl Acad Sci USA. 2005; 102:5618–23. https://doi.org/10.1073/pnas.0501559102 [PubMed]
- 56. Weibel ER, Taylor CR, Weber JM, Vock R, Roberts TJ, Hoppeler H. Design of the oxygen and substrate pathways. VII. Different structural limits for oxygen and substrate supply to muscle mitochondria. J Exp Biol. 1996; 199:1699–709. [PubMed]
- 57. Layec G, Bringard A, Vilmen C, Micallef JP, Fur YL, Perrey S, Cozzone PJ, Bendahan D. Accurate work-rate measurements during in vivo MRS studies of exercising human quadriceps. MAGMA. 2008; 21:227–35. https://doi.org/10.1007/s10334-008-0117-3 [PubMed]
- 58. Vanhamme L, van den Boogaart A, Van Huffel S, van den Boogaart A, Van Huffel S, and van den Boogaart A, and Van Huffel S. Improved method for accurate and efficient quantification of MRS data with use of prior knowledge. J Magn Reson. 1997; 129:35–43. https://doi.org/10.1006/jmre.1997.1244 [PubMed]
- 59. Kemp GJ, Roussel M, Bendahan D, Le Fur Y, Cozzone PJ. Interrelations of ATP synthesis and proton handling in ischaemically exercising human forearm muscle studied by 31P magnetic resonance spectroscopy. J Physiol. 2001; 535:901–28. https://doi.org/10.1111/j.1469-7793.2001.00901.x [PubMed]
- 60. Crapo RO, Jensen RL, Hegewald M, Tashkin DP. Arterial blood gas reference values for sea level and an altitude of 1,400 meters. Am J Respir Crit Care Med. 1999; 160:1525–31. https://doi.org/10.1164/ajrccm.160.5.9806006 [PubMed]
- 61. Ferreira LF, Townsend DK, Lutjemeier BJ, Barstow TJ. Muscle capillary blood flow kinetics estimated from pulmonary O2 uptake and near-infrared spectroscopy. J Appl Physiol (1985). 2005; 98:1820–28. https://doi.org/10.1152/japplphysiol.00907.2004 [PubMed]
- 62. Harper AJ, Ferreira LF, Lutjemeier BJ, Townsend DK, Barstow TJ. Matching of blood flow to metabolic rate during recovery from moderate exercise in humans. Exp Physiol. 2008; 93:1118–25. https://doi.org/10.1113/expphysiol.2008.042895 [PubMed]
- 63. Kemp GJ, Radda GK. Quantitative interpretation of bioenergetic data from 31P and 1H magnetic resonance spectroscopic studies of skeletal muscle: an analytical review. Magn Reson Q. 1994; 10:43–63. [PubMed]
- 64. Trenell MI, Sue CM, Kemp GJ, Sachinwalla T, Thompson CH. Aerobic exercise and muscle metabolism in patients with mitochondrial myopathy. Muscle Nerve. 2006; 33:524–31. https://doi.org/10.1002/mus.20484 [PubMed]
- 65. Motulsky HJ, Christopoulos A. (2003). Fitting models to biological data using linear and nonlinear regression. A practical guide to curve fitting. (San Diego.
- 66. Iotti S, Gottardi G, Clementi V, Barbiroli B. The mono-exponential pattern of phosphocreatine recovery after muscle exercise is a particular case of a more complex behaviour. Biochim Biophys Acta. 2004; 1608:131–39. https://doi.org/10.1016/j.bbabio.2003.11.003 [PubMed]



