Introduction
Skin aging is a continuous process which can be caused by both internal and external factors [1,2]. It is thought that intrinsic (chronological) and extrinsic (photo-induced) aging are realized through different pathways and can even cause different visual alterations of the skin [3,4]. These pathways are generally viewed to be connected with modifications of the connective tissue. This is featured in models for classical skin aging that implicate a progressive accumulation of senescent fibrocytes, leading to a subsequent reduction of collagen production and content in the aging skin [5]. Additional scenarios include the excessive oxidative phosphorylation leading to the accumulation of superoxides [6], the local overproduction of matrix metalloproteinases as a result of inflammation or UV irradiation (UVR) [7,8] and the reduced production of heat shock proteins [9].
Existing theories of skin aging exclude almost completely the involvement of any adipose tissue components. Whereas intrinsic aging is known in general to correlate with a continuous reduction of subcutaneous white adipose tissue (sWAT) and concurrent accumulation of visceral fat [10], extrinsic aging was till now considered to be fully independent of the state of adipose tissue. The reason for omitting adipose tissue from the equation is related to the low penetration depth of UVR into the human skin. The penetration of light waves into the skin is inversely related to its wavelength [11]. UVR is traditionally subdivided into three classes – UVA (320-400 nm), UVB (280-320 nm) and UVC (100-280 nm). UVA has the highest and the UVC the lowest penetration depth. The light energy transmission through a 70 µm thick epidermis was assessed to be about 0.27% at 290 nm and 9.5% at 313 nm [12]; penetration depths in the forearm skin were measured to be 20 µm at 290 nm and up to 60 µm at 320 nm [13]. Taking into account the characteristic thickness of the facial dermis layer of approximately 1-1.5 mm [14], it is clear that only a fractional part of UVR applied to the skin surface can reach the interface dermis/sWAT and thus directly influence the subcutaneous adipocytes.
These properties of UVR should exclude the adipose tissue from the list of global players in extrinsic skin aging. However, based on the results obtained during the recent past, we have reason to believe that adipose tissue should be considered as an important component of the process, in light of the presence and the properties of dermal adipocytes.
Dermal white adipose tissue – from little-known structure to a global player in skin physiology?
Dermal white adipose tissue (dWAT) appears in murine skin as a layer of adipocytes separated from sWAT by the panniculus carnosus [15]; in human and porcine skin, it appears in the form of so called “dermal cones” [16,17], giving raise to the superficial layer of sWAT (Fig. 1). This new adipose tissue depot [15,18] received much recent attention. It was shown that dWAT is involved in physiologically distinct processes, such as the cycling of hair follicles (HF) [19, 20] wound healing [21], homeostatic temperature regulation of the skin [22,23], skin protection against infection [24] and cutaneous fibrosis [25]. Moreover, it was hypothesized that dermal adipocytes can be involved in the long-term effects of soft tissue fillers [26], skin hyperpigmentation, development of hypertrophic scars and at least some skin efflorescences [27, 28]. Important reasons for such unusually multi-functional properties of dermal adipocytes are their high plasticity and the ability to change their phenotype in a very short time [27].
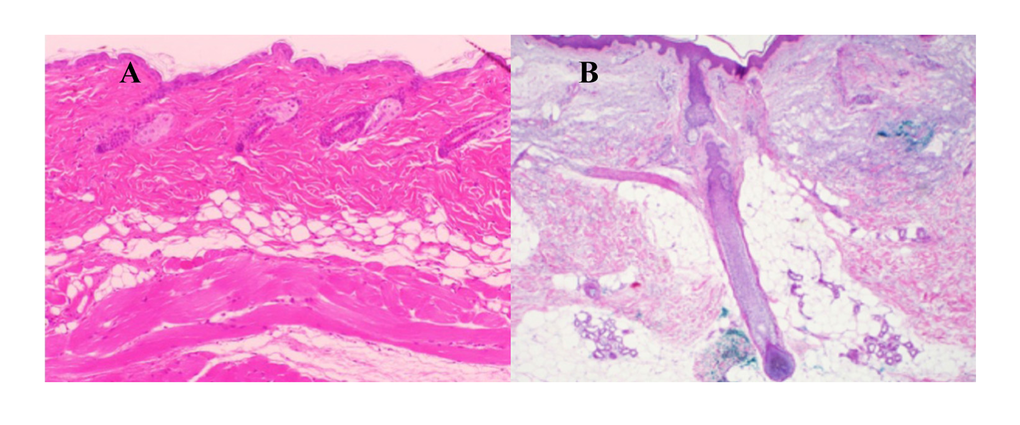
Figure 1. Typical layered dWAT structures in rodents and humans. (A) Section of the dWAT from the C57/BI6 mouse. This dWAT depot has the layered form placed parallel to the panniculus carnosus. (B) Human dermal adipocytes in the form of “dermal cones” around the pilosebaceous units. Single “dermal cones” can protrude into the upper dermis. These dermal cones are connected on the other end with sWAT. Pictures courtesy of Drs. Min Kim (A) and Travis Vandergriff (B), UT Southwestern Medical Center and published in [32].
Dermal adipocytes can protrude up to the upper dermis and produce the spatial “fat bridges” between the skin surface and the sWAT (Fig. 1B), thus connecting the regions which can be directly affected by UVR with much deeper fat layers. Paracrine signaling activity of dermal adipocytes discussed in [27] may be responsible for the transduction of the direct response to UVR induced in dWAT to the sWAT layer, providing the long-range, deep effects of UVR. At the same time, dWAT has the property to modulate its structure with rates that are much higher than the turnover rates characteristic for adipocytes in sWAT [27]. This can prompt quick structural and functional responses to different external physical impacts. These properties provide dWAT the ability to be one of the first-line responders to UVR in the skin.
Evolution of dWAT during intrinsic aging
Experimental systems to investigate the chronological evolution of damage to the human skin in vivo are very limited. For example, confocal microscopy allows the evaluation of the epidermis and upper dermis; however, alterations in the reticular dermis and in the superficial layer of sWAT cannot be assessed properly with this method, since penetration depth of the laser in the skin is very limited [29]. For this reason we will restrict ourselves mainly on the discussion of aging processes in murine skin where dWAT can be clearly visualized and quantified. To analyze the experimental results concerning the temporal modulation of dWAT, we have to take into account that dWAT demonstrates significant sexual dimorphism [30]. Thus, only animals of the same gender can be compared with each other.
Different experiments revealed an age-dependent evolution of the dWAT thickness in murine skin. One of the most accurate studies of this parameter was provided in [31], where the dWAT thickness was determined in BDF1 female mice for young (6-10 weeks), intermediate (13-19 weeks) and adult (26-34 weeks) animals. Additionally, these animals were divided into subgroups according to the stage of the hair follicle cycle, since dWAT thickness is known to be dependent on this parameter [19]. This is the reason for the spatially heterogeneous structure of dWAT, even in the same body area [32]. The results reported demonstrate the periodic evolution of dWAT: dWAT is present in 6-week-old mice (anagen phase of HFs), depleted in 9-week-old mice (telogen phase), significantly increased in 12-week-old mice (anagen phase) over its value in 6-week-old animals, reduced in 18-week-old mice (telogen phase) and then strongly increased in adult 31-week-old mice. Interestingly, this dWAT evolution is significantly correlated with skin stiffness, thereby demonstrating the inverse dependence of dWAT on skin thickness, being the lowest in 31-week-old mice. Similar behavior of dWAT was observed in C57BL/6 mice where the dWAT thickness is significantly increased in 12-month-old animals compared to the 2-month-old ones, and this property was found to be sex independent [33]. We also have to consider that the life-span of the C57BL/6 mice, which are often used in these experiments, is on average 29 months in males and 26.5 months in females [34]; thus the evolution between 2 and 12 months can be considered as “maturing”, but not as a “true” aging.
At the same time, age-dependent modifications of dWAT can easily be observed in different knock-out mouse models demonstrating accelerated aging phenotypes. 12-month-old mice carrying a deletion in the cannabinoid 1 receptor gene (Cnr1-/-) demonstrate strongly accelerated aging and have significantly reduced dWAT layers. Such a reduction was observed both in relation to the 2-month-old Cnr1-/- mice and the 12-month-old wild type mice. Another model which is useful in chronological aging studies is the PASG (proliferation associated SNF-2-like gene) null mouse, which also displays distinct signs of premature aging. PASG-/- skin exhibits almost complete depletion of dWAT comparing to the PASG+/+ skin [35]. In addition, p53 mutant mice displaying an early aging-associated phenotype demonstrate depleted dWAT structures in 24-month-old animals (which can be considered as very old), whereas the wild type mice of the same age show significantly reduced but still present dWAT layers [36].
Recapitulating, the chronological evolution of dWAT in intact mice seems to be associated with a periodic modulation of the volume of this depot till mid-age and its subsequent continuous strong involution in old animals. The last feature could be interpreted as a sign of a “true” aging.
UVR can modulate the metabolism of sWAT
UVR can significantly modulate sWAT metabolism. This effect is observable not only in chronically sun-damaged human skin, but even after a single UV exposure of a non-damaged skin [37]. These authors have shown that the free fatty acid and triglyceride content in sWAT of sun-exposed skin (forearm) is significantly lower than in the buttocks (sun-protected area) of the same subjects. At the same time, young subjects did not demonstrate such differences, which points to the UV-induced effect and not just to the regional variations in fat metabolism. Additionally, both chronic and single UVR exposure significantly reduces master adipogenic factors such as peroxisome proliferator-activated receptor γ (PPARγ); this reduction was rapid and remained stable for at least 72 h after acute UVR exposure. To explain these results, the authors assumed that some soluble factors (such as IL-6, IL-8, MCP-3 and PIGF) produced in upper dermis during UV exposure diffuse into the sWAT and trigger modification of sWAT metabolism. This idea was supported by the fact that the treatment of mature adipocytes from sWAT with these cytokines provides a reduction of triglyceride content. The list of cytokines which could be involved in signal transduction from the skin to the sWAT was further extended in [38], where it was shown that the exposure of preadipocytes to conditioned medium from solar irradiated epidermal-dermal equivalents, containing such inflammatory cytokines as IL-1α, IL-6, IL-11 and TNF-α, inhibited the differentiation of these cells into mature adipocytes. At the same time, application of antibodies neutralizing these cytokines was able to reduce the failure to differentiate significantly. This lead to the conclusion, that inflammatory cytokines are involved in the loss of sWAT during extrinsic aging.
A single low-dose (1.6 J/cm2) of UVB irradiation to Hos:HR-1 hairless mice decreases adiponectin levels in serum and even in ovarian adipose tissue within 24 h after irradiation [39]. This reduction correlated with the observed decreased levels of PPARγ in ovarial adipose tissue. Since the level of serum amyloid A in this study was shown to be significantly increased, the effect of UVB on remote adipose tissue depots was explained through endocrine responses mediated by amyloid A. In addition, reduced obesity was recently demonstrated after chronic exposure of C57BL/6 male mice to UVR independent of vitamin D production [40].
In all experimental studies mentioned above, it was assumed that the targets for UVR are in the epidermis and/or dermis and that the observed reaction of adipose tissue is of an indirect nature. However, since human dWAT structures can spatially reach the upper dermis (Fig. 1B), a direct effect of UVR on the dermal adipocytes cannot be fully excluded. While the lower parts of “dermal cones” (also known as “fat domes”) transverse the dermis and penetrate into the sWAT, the whole reaction chain leading to sWAT modifications can be theoretically realized through adipose tissue directly.
If this model is correct, the rate of extrinsic aging in humans should demonstrate spatial correlations with dWAT structures. Correlations of different pathological processes in the skin with its dWAT content are not unusual and were described, for example, in hypertrophic scarring [16], which predominantly appears in the areas having a high content of “dermal cones”. Analyzing the effects of UVR, these areas must additionally be separated into the sun-exposed and non-exposed areas. The most prominent sun-exposed body areas containing the “dermal cone” structures are the cheek, neck, dorsal hand and forearm; corresponding non-exposed areas containing “dermal cones” are the chest, abdomen, buttock and thigh. Body areas without dermal adipocytes are the palm and scalp [16].
From this point of view dWAT content correlates with a much more pronounced extrinsic aging process in the dorsal hand comparing to the palm area. Chronological skin aging demonstrates similar but not as pronounced differences in aging processes in palmar and dorsal regions of the hand. This can be an indication that UVR accelerates the processes of skin aging, whereas their basic components are determined by some other factors, one of which could be the local dWAT content. This can make skin aging not only body area dependent, but also spatially heterogeneous in the same body area, since dWAT can have a spatially heterogeneous structure [32]. This model allows the appearance of the “mosaic” structure of the aging skin.
Effects of UVR on the adipocytes in vitro
Irradiation of human adipose-derived stem cells (ADSCs) with UVA in vitro demonstrated suppression of adipogenic differentiation potential of these cells. Such suppression could be observed already by very low fluence of 0.05 J/cm2 and was gradually more severe as fluence increased up to 5.0 J/cm2 [41]. This effect was connected with an observed significant down-regulation of PPARγ expression caused by UVA and demonstrated strong dose- dependent effects. Accumulation of triglycerides in UVA-irradiated cells in this study was also significantly reduced in a dose-dependent manner. Taking into account that UVA of such low fluence as 0.05 J/cm2 was able to induce the pronounced effect on the ADSCs, we can assume that a direct effect of UVR on dermal adipocytes is possible, since such fluences can easily be realized in vivo in the lower dermis. Later, the same group has shown that UVA in similar doses also modulates the “stemness” of ADSCs [42].
These results demonstrate that adipocytes can react even to low doses of UVR with a suppression of PPARγ expression and adipogenic differentiation as well as with a reduced accumulation of triglycerides in mature adipocytes. This additionally supports the idea that not only systemic pathways, but also direct local responses in dWAT, can be involved in the reaction of sWAT to UVR in vivo, as observed in [37].
UVR can cause a reduction of dWAT and trigger cutaneous fibrosis
Chronic (five times weekly, 20 weeks, 16.3 J/cm2 per session) UVA irradiation of hairless mice produced some thickening of the epidermis but no effects in either the upper or lower dermis [43]. However, this irradiation caused the disappearance of dermal adipocytes, the production of fibrosis and a significant increase of hyaluronan content in the lower dermis compared to control animals. Since it was possible to prevent the appearance of cutaneous fibrosis by hydrocortisone, it was concluded that such modifications of the skin structure were caused by an inflammatory reaction induced by UVR.
Similar results were obtained in [44], where histological changes in the skin were investigated in the 11- and 16-week-old female C57BL/6J mice which were chronically (three times weekly, 2-8 weeks, 24 J/cm2) exposed to UVA radiation. Whereas no significant anatomical changes were observed after 2-4 weeks of irradiation, a substantial reduction of dWAT and increased accumulation of collagen fibers were observed after 8 weeks of UVR. The amount of insoluble collagen in the dermis of these mice was found to be approximately 37% higher than in control mice. At the same time, the dermal thickness in 8 week-old UVA-irradiated mice was not statistically different from corresponding age controls which points at the replacement of dWAT layer with cutaneous fibrosis.
These earlier results must be re-analyzed, taking into account the recently discovered property of dermal adipocytes to undergo transition into the mesenchymal cells (AMT) [25]. This transformation was recognized to be an important pathophysiological step in cutaneous fibrosis. Later it was reported that resistin-like molecule α (RELMα/FIZZ1) can suppress adipocyte-specific genes, triggering a de-differentiation of these cells; at the same time, RELMα/FIZZ1 induced α-smooth muscle actin and type I collagen expression which points to phenotypic transformation of adipocytes into the myofibroblasts [45]. AMT in [25] was induced by subcutaneous injections of bleomycin. AMT has universal features seen under many circumstances, and can be induced by different physical and chemical factors, among them also UVR. From this point of view, the results obtained in [43,44] can be interpreted as AMT caused by UV exposure (Fig. 2). This effect can be assumed not to be as pronounced as after bleomycin injection. Furthermore, the dWAT cellularity at the time of UVR application should be sufficiently high. This correlates with observations that the replacement of dWAT with cutaneous fibrosis after UVR was observed after sufficiently long UVR exposures as the mice reached the stage of adulthood, which associated with an expanded dWAT depot [31].
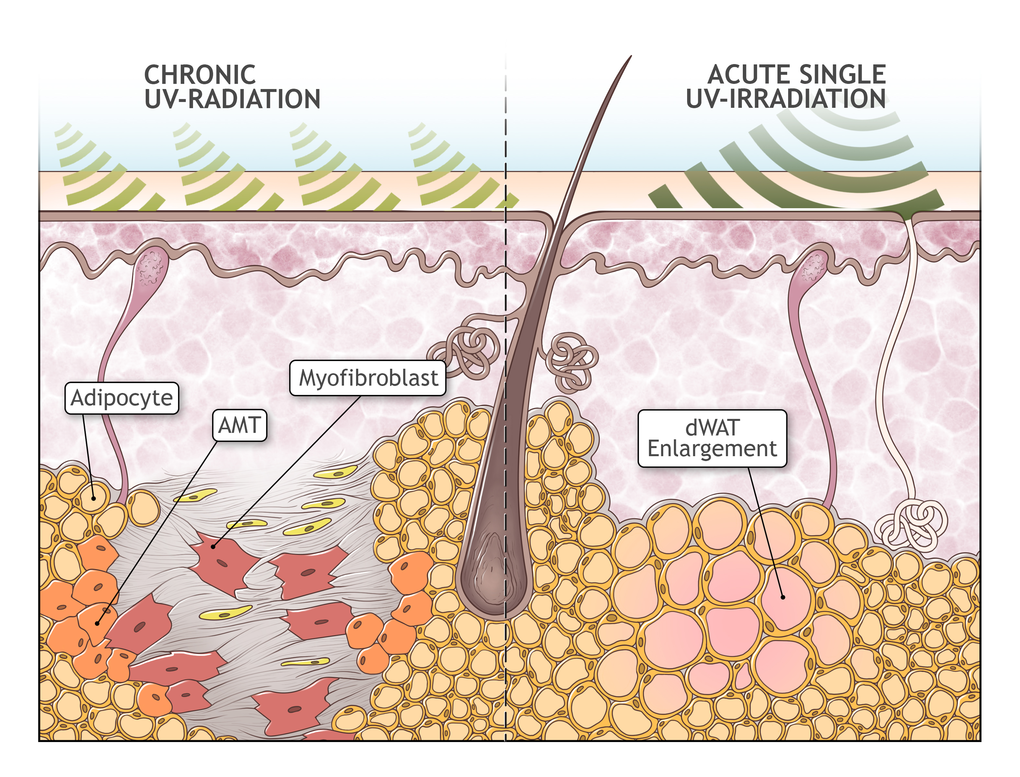
Figure 2. Possible role of adipocyte-myofibroblast transition in extrinsic aging. Absorption of UV radiation in the skin causes acute enlargement of the dWAT layer. However, upon chronic overexposure to UV radiation, it causes the depletion of dWAT and a concurrent development of cutaneous fibrosis, presumably through adipocyte-myofibroblast transition (AMT). Replacement of dWAT volume with fibrosis leads to production of mechanically heterogeneous skin structures and to the loss of the effective skin volume.
Replacement of dWAT with fibrosis through AMT should lead to the production of spatially heterogeneous skin structures. Additionally, this process should prompt the loss of effective skin volume, taking into account the total volume of dermis and dWAT, corresponding to the skin modifications observed in extrinsic aging.
This important suggestion can be directly proven with the help of the recently developed “adipochaser” mouse model [46,47], which allows to trace the fate of adipocytes in the tissue. If correct, this effect can, at least in extrinsic aging, shift the point of interest from connective tissue to dWAT. We cannot fully exclude that the AMT mechanism is also somehow involved in the intrinsic aging.
Skin surface lipids and their role in skin reactions to UVR
Skin reactions to UVR are not only dependent on the type and the dose of irradiation, but also on the content of the skin surface lipids (among them prominently α-tocopherol). These lipids are commonly referred to as photo-protectants [48]. The content of these lipids in the skin is significantly reduced after a single UVR application. This can lead to a stronger UV absorption in the skin during a next round of UVR and thus accelerate skin aging. These lipids are believed to be mainly produced and secreted by sebaceous glands which were recognized as the major physiological source for their delivery to the skin surface [49]. At the same time, it was estimated that approximately 90% of the total amount of vitamin E is stored in the lipid droplets of adipocytes and about two-thirds of this content exists in the form of α-tocopherol [50]. Moreover, it is known that α-tocopherol can stimulate the expression of PPARγ and along with that lipid accumulation during adipogenic differentiation [51]. This PPARγ stimulation seems to be realized indirectly through inhibition of its antagonists [52]. Moreover, α-tocopherol can induce the expression of adiponectin, which is in line with its adipogenic effects [53]. These results provide an additional link to dWAT involvement in the skin reaction to UVR.
Adipose tissue displays the slowest turnover for its stored α-tocopherol with an average half-life of 184 days [54]. It is not known whether UVR can speed up the release of α-tocopherol from adipocytes located in dWAT. We would however predict that such an enhanced turnover rate should indeed occur, especially in the case of repeated UVRs that continuously deplete the α-tocopherol pool in the skin and thus force an additional outflow of this lipid from the adipocytes located near the skin surface.
Hence, chronic UVR should deplete the pool of α-tocopherol in dermal adipocytes and thus also suppress the adipogenic differentiation potential and triglyceride accumulation in these cells. This would provide an explanation for the observed continuous reduction of dWAT in the skin chronically irradiated with UV. In contrast, a single UVR may induce a compensatory expansion of dWAT to provide a short-term pool of skin lipids. The reaction of dWAT to UVR should therefore be strongly dependent on the irradiation schedule.
Immuno-modulators influence adipogenesis and accelerate skin aging
One of the most intriguing phenomena in skin aging is the acceleration of this process through application of different drugs. This effect is known, for example, for immuno-modulators such as cyclosporine A (CsA). Whereas chronic exposure of the Skh-1 hairless mice to non-erythemal doses of UVB induced skin wrinkles after 6-7 weeks of irradiation, concomitant systemic application of CsA reduced the onset time of wrinkling down to 4 weeks [55]. In contrast, HRS/J hairless mice demonstrate no wrinkles after 10 weeks of the same UVB irradiation, and display no wrinkles after 7 weeks of combined application of UVB with CsA.
These results should be re-analyzed taking into account that the dWAT structure and activity in murine skin is dependent on the genetic strain and on the stage of the HF cycle [19]. The main genetic mice strains used in skin aging experiments are female C57BL/6J mice as well as female Skh-1, HRS/J and albino hairless mice. DWAT in C57BL/6J mice demonstrates a cyclic evolution, which was described in detail in [19]. A mutation of hr gene leading to skin baldness causes a disruption of the integrity of HFs as well as the production of utriculi (open comedones) and cysts in the lower dermis and subcutis [56,57]. Whereas both the Skh-1 [58] and the HRS/J [59] hairless mice have pronounced dWAT layers, HRS/J mice also demonstrate accelerated thymus atrophy [60].
CsA belongs to the group of immuno-suppressors which alter the production of cytokines and influence adipogenesis. This drug can specifically inhibit calcineurin, which is upstream of the nuclear factor of activated T cells (NFAT) transcription factor [61–63]. If NFAT takes part in premature skin aging as observed in [55], dWAT can also be affected, since this pathway is involved in the differentiation of preadipocytes as well [64,65]. Indeed, chronic application of some immuno-suppressors can reduce both adipocyte size and number [66] and it was proposed that CsA inhibits adipogenic differentiation through prevention of the nuclear localization of NFAT [64]. Hairless mice with thymus atrophy have a marked deficiency in functional T cells, including iNKTs which represent the resident population in adipose tissue [67]. Whereas it is actually not known which level of NFAT expression is typical for dermal adipocytes in athymic nude mice, we assume its expression is altered compared to wild-type animals. This can lead to impaired adipogenic differentiation in the dermis of these animals. This may be one of the reasons for the qualitative differences in effects of CsA on skin aging in different genetic strains observed in [55].
Sexual dimorphism in dWAT and skin aging
Murine dWAT demonstrates pronounced sexual dimorphism: females have dWAT layers which can be an order of magnitude thicker than in males, whereas the total skin thickness is higher in intact males [30]. After gonadectomy, the dWAT thickness significantly increases both in males and females, whereas treatment of these animals with dihydrotestosterone, 17β-estradiol or dehydroepiandrosterone markedly depletes this depot. Such effects were connected with ability of androgens to inhibit the adipogenic differentiation of stem cells and preadipocytes [68,69].
A sexually dimorphic response is also known for the response of mice to UVR: males demonstrate a reduced responsiveness to UVA radiation compared to females [70]. Such a response correlates with the lower thickness of dWAT in non-irradiated males and with the ability of UVA to reach the panniculus carnosus to affect dermal adipocytes. This again points to the involvement of dermal adipocytes minimally in the context of extrinsic skin aging. Whereas the sexual dimorphism of dWAT in humans was not clearly demonstrated, its gender difference is likely to be present in humans as well. This effect can at least partly explain the difference in the skin aging processes in male and female subjects [71] and will need further intensive investigation.
Histological evidence for the reaction of dWAT to light irradiation in murine skin
Production of reactive oxygen species (ROS) is one of the primary responses in the skin reaction to UVR [72,73]. Despite the fact that ROS are often considered to be generally harmful, they in fact able both to stimulate and to suppress the cellular processes, such as the proliferation and differentiation of adipose tissue-derived stem cells (ADSCs) [74]. For example, whereas a high-dose UVB is able to suppress the proliferation of ADSCs, low-dose UVB can increase survival of these cells and up-regulate the expression of different growth factors [75]. Consequently, dWAT and sWAT should react to UVR in a dose-dependent and bi-phasic manner.
The dWAT depot in rodents can demonstrate a quick and significant modulation of its thickness in response to application of different physical factors [27]. In [44], dWAT in C57BL/6J mice was shown to be significantly reduced after UVA irradiation (three times weekly, 8 weeks, 24 J/cm2 per session). Similar results were obtained in [76] where the 6-week-old C57BL/6J female mice were exposed to a low-dose UVB radiation (four times weekly, 30 weeks, gradually increasing doses). Histological pictures of the skin in these mice demonstrated a significant reduction of dWAT thickness with a corresponding thickening of dWAT-free dermis layer. Similarly, in [77], dWAT in Skh-1 hairless mice was reduced after UVB irradiation (10 weeks, 3 times weekly, gradually increasing doses from 20 mJ/cm2 up to 180 mJ/cm2).
At the same time, in [78], the 6-week-old albino hairless mice received a single low-dose UVR (275-380 nm, 200 mJ/cm2), which was equal to the minimal erythemal dose for these animals. 72 h after irradiation, skin biopsies clearly demonstrated thickening of the dermis, which was mainly connected with expansion of the dWAT layer. Similarly, UVR (6 weeks, 3 times weekly, minimal erythemal dose) of the 8-week-old HR-1 hairless mice with the wavelengths of 274-380 nm provided significant increase in the thickness and cellularity of dWAT [79]. These results demonstrate a qualitative difference in modification of dWAT structure in C57BL/6J and different models of hairless mice, but they will need to be re-approved taking into account the spatial heterogeneity of dWAT described in [32].
Infrared (IR) radiation with wavelengths up to 1 mm is also able to induce extrinsic skin aging [80,81]. These light waves have much higher penetration depths than UVR and can thus reach the superficial area of sWAT. From this point of view, it would be interesting to compare the modification of dWAT in IR- and UV-irradiated murine skin. In [82], male Wistar rats were irradiated with IR (1.100-1.800 nm) of 40 J/cm2. Skin histology demonstrated abrupt appearance of dermal adipocytes on day 7 after irradiation, with subsequent gradual decrease of their number up to day 180. During the whole observation period, the number of dermal adipocytes was significantly higher in irradiated than in controls. Whereas the action mechanisms of IR and UVR on the skin should to be very different, both types of light seem to have the ability to modify dWAT.
Additional correlations between UVR and dWAT
Possible role of vitamin D modulation in dWAT caused by UVR
Vitamin D can be strongly induced in the skin by UVB radiation [83]. On the other hand, vitamin D is involved in skin aging, since skin aging demonstrates a U-shaped dependence on vitamin D content [84]. It was also shown that vitamin D receptor knockout mice (VDR-/-) show a number of signs of premature aging, among them wrinkling skin and a significantly thinner dWAT layer relative to wild type mice [85]. Interestingly, VDR-/- mice demonstrate highly increased expression of uncoupling protein in sWAT, leading to “beige” fat [86,87], which correlates with a local loss of sWAT volume. Even more intriguingly, it was shown that these adipocytes can express the vitamin D receptor and autonomously express 1,25-dihydroxyvitamin D3 [88], suggesting that adipocytes participate in vitamin D production after UVR.
The dermal adipose layer in murine skin can be depleted by application of 1,25-dihydroxyvitamin D3 in high doses; at the same time, this fat depot significantly expands in the absence of vitamin D [89]. This effect can be connected with the ability of 1,25-dihydroxyvitamin D3 to inhibit the differentiation of murine preadipocytes through suppression of PPARγ [90]. This reflects the well-known fact that VDR and PPAR signaling pathways are interconnected [91,92]. Contrarily, both 25-hydroxyvitamin D3 and 1,25-dihydroxyvitamin D3 can promote differentiation of human preadipocytes [93] and mesenchymal cells [94]. This apparently contradictive influence of vitamin D on the differentiation of murine and human adipocytes still needs to be explained.
Whereas it is actually not known whether dermal adipocytes can produce vitamin D, especially after UVR, it is likely that these cells can do so. In this case, it can be supposed that UVB can modulate the dWAT structure also indirectly through induction of vitamin D production in the skin.
Possible role of hyaluronan in dWAT modulation
DWAT can be also modulated indirectly through modification of hyaluronan (HA) content in the skin. HA is expressed during adipocyte differentiation; the depletion of HA content was shown to reduce the adipogenic differentiation of preadipocytes in vitro as well as the abdominal fat accumulation in C57BL/6J mice [95]. Multiple applications of a hyaluronidase result in significant (up to 35%) reduction of fat mass in the same mouse strain with a simultaneous reduction of the adipocyte size [96]. Not only the high-molecular hyaluronan, but also its enzymatic fragments inhibit adipocyte differentiation [97]. Many of these finding have been summarized in [98].
UVR can significantly modulate the HA content in different compartments of the skin [99]. It was shown that low dose UVB can increase the epidermal synthesis of HA [100], whereas chronic UVB exposure causes loss of HA from the dermis, which is primarily connected with a down-regulation of hyaluronan synthase [101]. Such behavior of HA in the skin correlates with observed expansion of dWAT after low doses of single UVR exposure and depletion of this depot after chronic exposure of the skin to UVR described above.
Ethnic differences in skin aging – is dWAT involved?
Whereas the intrinsic aging is believed to occur similarly in different ethnic groups [2], there are well-known ethnic differences in extrinsic skin aging [102–104]. They show higher rates of these processes in Caucasians than in Asians [103,105], which was generally connected with different melanin content and composition in these ethnic groups [2]. For example, the middle-aged Caucasian women demonstrate a much higher appearance of wrinkles as the Asian women from the same age group [103]. Severe wrinkles on the upper lip were found in 38% and 10% of French and Japanese women aged between 50 and 64 years, respectively. Some types of wrinkles were shown to appear approximately 15 years earlier in French than in Japanese women [103]. Similar differences were observed between German and Japanese women [106]. If dermal adipocytes are involved in the skin aging processes, these differences should be at least partly connected with ethnic variations in dWAT content.
Whereas differences in the skin structure between ethnic groups are well-known [107], direct information about the content of dermal adipocytes in these groups is currently absent. Some further analysis can be done according to the ethnic prevalence of hypertrophic scarring, which correlates with amount of “dermal cone” structures in the skin [16]. Recently, it was shown that the appearance of severe hypertrophic scars indeed significantly varies with race [108], demonstrating 45% higher prevalence among Asians and 78% higher prevalence among African Americans compared with Caucasians. On the other hand, the African Americans have bigger sebaceous glands and produce much more lipids than Asians, followed by Caucasians [107]. This correlates with the appearance of hypertrophic scars in these ethnic groups and can at least partly be connected with their dWAT structure.
All this can be an indication for increased content of dWAT in the form of “dermal cones” in the skin of Asians comparing to the same age group of Caucasians, which can, on the other side, be reflected in slower processes of skin aging in the Asian skin.
Local interaction between fibroblasts and adipocytes
Modification of dWAT caused by extrinsic or intrinsic aging can influence the function of fibroblasts and thus induce the change of the dermis structure. Enlarged adipocytes significantly suppress the synthetic activity of fibroblasts, whereas small adipocytes did not demonstrate such effect [109]. Very recently, it was also shown that the expansion of sWAT correlates with a decrease of elastic fiber content in the dermis [110]. Consequently, not only chronological or photo-induced aging, but also the local expansion of the adjacent adipose tissue can induce a relatively quick degradation of the elastic fibers in the dermis, which under normal conditions can persist there for many decades [111–113].
Discussion
During the last few years, it was convincingly demonstrated that dWAT is involved in different physiological and pathological processes in the skin. Remarkably, all observed reactions were connected with significant modulation of the dWAT structure and function. Whereas physiological skin reactions (e.g., immune reaction to pathogens, thermoregulation, etc.) normally cause an expansion of this special adipose layer realized both through hypertrophy and hyperplasia of dermal adipocytes, pathological processes (such as cutaneous fibrosis) were found to correlate with significant dWAT involution.
A number of groups have demonstrated that adipocytes from various superficial and deep fat depots can react to UVR. This interaction can be of direct or indirect nature (e.g., through activation of paracrine signaling or the VDR pathway), depending on the depth of the location of the adipocytes in relation to the penetration depth of UVR. Reduction of triglycerides in sWAT of sun exposed skin and a decrease of adiponectin levels in remote adipose tissue support the indirect mechanism, whereas pronounced histological changes in dWAT after UVR encourage a direct effect of UVR on the adipocytes.
The sexual dimorphism of dWAT and ethnic differences in skin aging provide additional arguments for the involvement of dWAT in the skin aging processes. Female mice have a thicker dWAT layer and demonstrate much higher responsiveness to UVR than males from the same genetic background. On the other hand, ethnic differences in skin aging correlate with the appearance of hypertrophic scars. Taking into account the known spatial correlation between hypertrophic scars and local content of dermal adipocytes, we assume that dWAT should vary in different ethnic groups. This variation may be one of the reasons for the different rates of skin aging in these groups. Verification of this relationship will require a comparison of “dermal cone” structures in different ethnic groups.
Since UVR was shown to cause depletion of dWAT and the concurrent appearance of dermal fibrosis, one can assume that recently discovered mechanism of adipocyte-to-myofibroblast transition should be involved in the skin reaction to UVR. Whereas this transition in experimental models was mainly induced by subcutaneous bleomycin injections, such effects may also be possible as a result of UVR. In this case, dermal adipocytes contribute at least to the extrinsic skin aging processes. This important assumption will need further experimental verification.
The recently proposed involvement of adipogenesis in the long-term effects of the soft-tissue fillers [26] indirectly assumes that the pre-stimulation of such structures can improve the treatment results. In light of all these observations, dermal adipocytes may be an effective target in stand-alone and combinational skin anti-aging therapies.
Conclusion
There are strong indications that the transition of adipocytes to mesenchymal cells substantially contributes to the development of cutaneous fibrosis. It may be an important part of extrinsic skin aging, whereby both the reduction of dWAT and substitution of dWAT volume with fibrotic structures contribute. Since intrinsic (chronological) skin aging is also connected with a progressive reduction of the dWAT layer, it cannot be excluded that an adipocyte-myofibroblast transition is also involved in this type of skin aging. In the future, this may make dermal adipocytes new interesting targets in anti-aging strategies.
Acknowledgements
We thank Min Kim and Travis Vandergriff for their generous help with the histological samples. P.E.S. is supported by NIH grants R01-DK55758, P01-DK088761 and R01-DK099110, as well as Cancer Prevention and Research Institute of Texas (CPRIT) grant RP140412. Figure2 was illustrated by Richard Howdy, certified medical illustrator, at Visually Medical (VisuallyMedical.com).
Conflicts of Interest
ILK is the managing partner of Wellcomet GmbH. No methods or devices of Wellcomet GmbH were used in this publication. PES declares no conflict of interests.
References
- 1. Farage MA, Miller KW, Elsner P, Maibach HI. Intrinsic and extrinsic factors in skin ageing: a review. Int J Cosmet Sci. 2008; 30:87–95. https://doi.org/10.1111/j.1468-2494.2007.00415.x [PubMed]
- 2. Vierkötter A and Krutmann J. Environmental influences on skin aging and ethnic-specific manifestations. Dermatoendocrinol. 2012; 4:227–31. https://doi.org/10.4161/derm.19858 [PubMed]
- 3. Fisher GJ, Wang ZQ, Datta SC, Varani J, Kang S, Voorhees JJ. Pathophysiology of premature skin aging induced by ultraviolet light. N Engl J Med. 1997; 337:1419–28. https://doi.org/10.1056/NEJM199711133372003 [PubMed]
- 4. Flament F, Bazin R, Laquieze S, Rubert V, Simonpietri E, Piot B. Effect of the sun on visible clinical signs of aging in Caucasian skin. Clin Cosmet Investig Dermatol. 2013; 6:221–32. https://doi.org/10.2147/CCID.S44686 [PubMed]
- 5. Mays PK, Bishop JE, Laurent GJ. Age-related changes in the proportion of types I and III collagen. Mech Ageing Dev. 1988; 45:203–12. https://doi.org/10.1016/0047-6374(88)90002-4 [PubMed]
- 6. Massudi H, Grant R, Braidy N, Guest J, Farnsworth B, Guillemin GJ. Age-associated changes in oxidative stress and NAD+ metabolism in human tissue. PLoS One. 2012; 7:e42357. https://doi.org/10.1371/journal.pone.0042357 [PubMed]
- 7. Fisher GJ, Kang S, Varani J, Bata-Csorgo Z, Wan Y, Datta S, Voorhees JJ. Mechanisms of photoaging and chronological skin aging. Arch Dermatol. 2002; 138:1462–70. https://doi.org/10.1001/archderm.138.11.1462 [PubMed]
- 8. Brennan M, Bhatti H, Nerusu KC, Bhagavathula N, Kang S, Fisher GJ, Varani J, Voorhees JJ. Matrix metalloproteinase-1 is the major collagenolytic enzyme responsible for collagen damage in UV-irradiated human skin. Photochem Photobiol. 2003; 78:43–48. https://doi.org/10.1562/0031-8655(2003)078<0043:MMITMC>2.0.CO;2 [PubMed]
- 9. Calderwood SK, Murshid A, Prince T. The shock of aging: molecular chaperones and the heat shock response in longevity and aging--a mini-review. Gerontology. 2009; 55:550–58. https://doi.org/10.1159/000225957 [PubMed]
- 10. Tchkonia T, Morbeck DE, Von Zglinicki T, Van Deursen J, Lustgarten J, Scrable H, Khosla S, Jensen MD, Kirkland JL. Fat tissue, aging, and cellular senescence. Aging Cell. 2010; 9:667–84. https://doi.org/10.1111/j.1474-9726.2010.00608.x [PubMed]
- 11. Jacques SL. Optical properties of biological tissues: a review. Phys Med Biol. 2013; 58:R37–61. https://doi.org/10.1088/0031-9155/58/11/R37 [PubMed]
- 12. Bruls WA, Slaper H, van der Leun JC, Berrens L. Transmission of human epidermis and stratum corneum as a function of thickness in the ultraviolet and visible wavelengths. Photochem Photobiol. 1984; 40:485–94. https://doi.org/10.1111/j.1751-1097.1984.tb04622.x [PubMed]
- 13. Meinhardt M, Krebs R, Anders A, Heinrich U, Tronnier H. Absorption spectra of human skin in vivo in the ultraviolet wavelength range measured by optoacoustics. Photochem Photobiol. 2009; 85:70–77. https://doi.org/10.1111/j.1751-1097.2008.00398.x [PubMed]
- 14. Pellacani G and Seidenari S. Variations in facial skin thickness and echogenicity with site and age. Acta Derm Venereol. 1999; 79:366–69. https://doi.org/10.1080/000155599750010283 [PubMed]
- 15. Driskell RR, Jahoda CA, Chuong CM, Watt FM, Horsley V. Defining dermal adipose tissue. Exp Dermatol. 2014; 23:629–31. https://doi.org/10.1111/exd.12450 [PubMed]
- 16. Matsumura H, Engrav LH, Gibran NS, Yang TM, Grant JH, Yunusov MY, Fang P, Reichenbach DD, Heimbach DM, Isik FF. Cones of skin occur where hypertrophic scar occurs. Wound Repair Regen. 2001; 9:269–77. https://doi.org/10.1046/j.1524-475X.2001.00269.x [PubMed]
- 17. Engrav LH, Tuggle CK, Kerr KF, Zhu KQ, Numhom S, Couture OP, Beyer RP, Hocking AM, Carrougher GJ, Ramos ML, Klein MB, Gibran NS. Functional genomics unique to week 20 post wounding in the deep cone/fat dome of the Duroc/Yorkshire porcine model of fibroproliferative scarring. PLoS One. 2011; 6:e19024. https://doi.org/10.1371/journal.pone.0019024 [PubMed]
- 18. Wojciechowicz K, Gledhill K, Ambler CA, Manning CB, Jahoda CA. Development of the mouse dermal adipose layer occurs independently of subcutaneous adipose tissue and is marked by restricted early expression of FABP4. PLoS One. 2013; 8:e59811. https://doi.org/10.1371/journal.pone.0059811 [PubMed]
- 19. Festa E, Fretz J, Berry R, Schmidt B, Rodeheffer M, Horowitz M, Horsley V. Adipocyte lineage cells contribute to the skin stem cell niche to drive hair cycling. Cell. 2011; 146:761–71. https://doi.org/10.1016/j.cell.2011.07.019 [PubMed]
- 20. Chen CC, Plikus MV, Tang PC, Widelitz RB, Chuong CM. The modulatable stem cell niche: tissue interactions during hair and feather follicle regeneration. J Mol Biol. 2016; 428:1423–40. https://doi.org/10.1016/j.jmb.2015.07.009 [PubMed]
- 21. Schmidt BA and Horsley V. Intradermal adipocytes mediate fibroblast recruitment during skin wound healing. Development. 2013; 140:1517–27. https://doi.org/10.1242/dev.087593 [PubMed]
- 22. Kasza I, Suh Y, Wollny D, Clark RJ, Roopra A, Colman RJ, MacDougald OA, Shedd TA, Nelson DW, Yen MI, Yen CL, Alexander CM. Syndecan-1 is required to maintain intradermal fat and prevent cold stress. PLoS Genet. 2014; 10:e1004514. https://doi.org/10.1371/journal.pgen.1004514 [PubMed]
- 23. Alexander CM, Kasza I, Yen CL, Reeder SB, Hernando D, Gallo RL, Jahoda CA, Horsley V, MacDougald OA. Dermal white adipose tissue: a new component of the thermogenic response. J Lipid Res. 2015; 56:2061–69. https://doi.org/10.1194/jlr.R062893 [PubMed]
- 24. Zhang LJ, Guerrero-Juarez CF, Hata T, Bapat SP, Ramos R, Plikus MV, Gallo RL. Innate immunity. Dermal adipocytes protect against invasive Staphylococcus aureus skin infection. Science. 2015; 347:67–71. https://doi.org/10.1126/science.1260972 [PubMed]
- 25. Marangoni RG, Korman BD, Wei J, Wood TA, Graham LV, Whitfield ML, Scherer PE, Tourtellotte WG, Varga J. Myofibroblasts in murine cutaneous fibrosis originate from adiponectin-positive intradermal progenitors. Arthritis Rheumatol. 2015; 67:1062–73. https://doi.org/10.1002/art.38990 [PubMed]
- 26. Kruglikov IL and Wollina U. Soft tissue fillers as non-specific modulators of adipogenesis: change of the paradigm? Exp Dermatol. 2015; 24:912–15. https://doi.org/10.1111/exd.12852 [PubMed]
- 27. Kruglikov IL and Scherer PE. Dermal adipocytes: from irrelevance to metabolic targets? Trends Endocrinol Metab. 2016; 27:1–10. https://doi.org/10.1016/j.tem.2015.11.002 [PubMed]
- 28. Kruglikov IL, Scherer PE, Wollina U. Are dermal adipocytes involved in psoriasis? Exp Dermatol. 2016; •••: Epub ahead of print. https://doi.org/10.1111/exd.12996 [PubMed]
- 29. Longo C, Casari A, Beretti F, Cesinaro AM, Pellacani G. Skin aging: in vivo microscopic assessment of epidermal and dermal changes by means of confocal microscopy. J Am Acad Dermatol. 2013; 68:e73–82. https://doi.org/10.1016/j.jaad.2011.08.021 [PubMed]
- 30. Azzi L, El-Alfy M, Martel C, Labrie F. Gender differences in mouse skin morphology and specific effects of sex steroids and dehydroepiandrosterone. J Invest Dermatol. 2005; 124:22–27. [PubMed]
- 31. Wang Y, Marshall KL, Baba Y, Gerling GJ, Lumpkin EA. Hyperelastic material properties of mouse skin under compression. PLoS One. 2013; 8:e67439. https://doi.org/10.1371/journal.pone.0067439 [PubMed]
- 32. Kruglikov IL and Scherer PE. Dermal adipocytes and hair cycling: is spatial heterogeneity a characteristic feature of the dermal adipose tissue depot? Exp Dermatol. 2016; 25:258–62. https://doi.org/10.1111/exd.12941 [PubMed]
- 33. Bilkei-Gorzo A, Drews E, Albayram Ö, Piyanova A, Gaffal E, Tueting T, Michel K, Mauer D, Maier W, Zimmer A. Early onset of aging-like changes is restricted to cognitive abilities and skin structure in Cnr1⁻/⁻ mice. Neurobiol Aging. 2012; 33:200.e11–22. https://doi.org/10.1016/j.neurobiolaging.2010.07.009 [PubMed]
- 34. Kunstyr I and Leuenberger HG. Gerontological data of C57BL/6J mice. I. Sex differences in survival curves. J Gerontol. 1975; 30:157–62. https://doi.org/10.1093/geronj/30.2.157 [PubMed]
- 35. Sun LQ, Lee DW, Zhang Q, Xiao W, Raabe EH, Meeker A, Miao D, Huso DL, Arceci RJ. Growth retardation and premature aging phenotypes in mice with disruption of the SNF2-like gene, PASG. Genes Dev. 2004; 18:1035–46. https://doi.org/10.1101/gad.1176104 [PubMed]
- 36. Tyner SD, Venkatachalam S, Choi J, Jones S, Ghebranious N, Igelmann H, Lu X, Soron G, Cooper B, Brayton C, Park SH, Thompson T, Karsenty G, et al. p53 mutant mice that display early ageing-associated phenotypes. Nature. 2002; 415:45–53. https://doi.org/10.1038/415045a [PubMed]
- 37. Kim EJ, Kim YK, Kim JE, Kim S, Kim MK, Park CH, Chung JH. UV modulation of subcutaneous fat metabolism. J Invest Dermatol. 2011; 131:1720–26. https://doi.org/10.1038/jid.2011.106 [PubMed]
- 38. Li WH, Pappas A, Zhang L, Ruvolo E, Cavender D. IL-11, IL-1α, IL-6, and TNF-α are induced by solar radiation in vitro and may be involved in facial subcutaneous fat loss in vivo. J Dermatol Sci. 2013; 71:58–66. https://doi.org/10.1016/j.jdermsci.2013.03.009 [PubMed]
- 39. Matsui S, Yamane T, Kobayashi-Hattori K, Oishi Y. Ultraviolet B irradiation reduces the expression of adiponectin in ovarial adipose tissues through endocrine actions of calcitonin gene-related peptide-induced serum amyloid A. PLoS One. 2014; 9:e98040. https://doi.org/10.1371/journal.pone.0098040 [PubMed]
- 40. Geldenhuys S, Hart PH, Endersby R, Jacoby P, Feelisch M, Weller RB, Matthews V, Gorman S. Ultraviolet radiation suppresses obesity and symptoms of metabolic syndrome independently of vitamin D in mice fed a high-fat diet. Diabetes. 2014; 63:3759–69. https://doi.org/10.2337/db13-1675 [PubMed]
- 41. Lee J, Lee J, Jung E, Kim YS, Roh K, Jung KH, Park D. Ultraviolet A regulates adipogenic differentiation of human adipose tissue-derived mesenchymal stem cells via up-regulation of Kruppel-like factor 2. J Biol Chem. 2010; 285:32647–56. https://doi.org/10.1074/jbc.M110.135830 [PubMed]
- 42. Lee J, Jung E, Hyun JW, Park D. Ultraviolet A regulates the stemness of human adipose tissue-derived mesenchymal stem cells through downregulation of the HIF-1α via activation of PGE(2)-cAMP signaling. J Cell Biochem. 2012; 113:3681–91. https://doi.org/10.1002/jcb.24241 [PubMed]
- 43. Mitani H, Koshiishi I, Toyoda H, Toida T, Imanari T. Alterations of hairless mouse skin exposed to chronic UV irradiation and its prevention by hydrocortisone. Photochem Photobiol. 1999; 69:41–46. https://doi.org/10.1111/j.1751-1097.1999.tb05304.x [PubMed]
- 44. Sayama A, Soushin T, Okada T, Doi K, Nakayama H. Morphological and biochemical changes during aging and photoaging of the skin of C57BL/6J mice. J Toxicol Pathol. 2010; 23:133–39. https://doi.org/10.1293/tox.23.133 [PubMed]
- 45. Martins V, Gonzalez De Los Santos F, Wu Z, Capelozzi V, Phan SH, Liu T. FIZZ1-induced myofibroblast transdifferentiation from adipocytes and its potential role in dermal fibrosis and lipoatrophy. Am J Pathol. 2015; 185:2768–76. https://doi.org/10.1016/j.ajpath.2015.06.005 [PubMed]
- 46. Wang QA, Tao C, Gupta RK, Scherer PE. Tracking adipogenesis during white adipose tissue development, expansion and regeneration. Nat Med. 2013; 19:1338–44. https://doi.org/10.1038/nm.3324 [PubMed]
- 47. Wang QA and Scherer PE. The AdipoChaser mouse: A model tracking adipogenesis in vivo. Adipocyte. 2014; 3:146–50. https://doi.org/10.4161/adip.27656 [PubMed]
- 48. K Gratz I and Kofler B. UV irradiation-induced inflammation, what is the trigger? Exp Dermatol. 2015; 24:916–17. https://doi.org/10.1111/exd.12849 [PubMed]
- 49. Thiele JJ, Weber SU, Packer L. Sebaceous gland secretion is a major physiologic route of vitamin E delivery to skin. J Invest Dermatol. 1999; 113:1006–10. https://doi.org/10.1046/j.1523-1747.1999.00794.x [PubMed]
- 50. Landrier JF, Marcotorchino J, Tourniaire F. Lipophilic micronutrients and adipose tissue biology. Nutrients. 2012; 4:1622–49. https://doi.org/10.3390/nu4111622 [PubMed]
- 51. Uto-Kondo H, Ohmori R, Kiyose C, Kishimoto Y, Saito H, Igarashi O, Kondo K. Tocotrienol suppresses adipocyte differentiation and Akt phosphorylation in 3T3-L1 preadipocytes. J Nutr. 2009; 139:51–57. https://doi.org/10.3945/jn.108.096131 [PubMed]
- 52. Kawahara A, Haraguchi N, Tsuchiya H, Ikeda Y, Hama S, Kogure K. Peroxisome proliferator-activated receptor γ (PPARγ)-independent specific cytotoxicity against immature adipocytes induced by PPARγ antagonist T0070907. Biol Pharm Bull. 2013; 36:1428–34. https://doi.org/10.1248/bpb.b13-00024 [PubMed]
- 53. Landrier JF, Gouranton E, El Yazidi C, Malezet C, Balaguer P, Borel P, Amiot MJ. Adiponectin expression is induced by vitamin E via a peroxisome proliferator-activated receptor γ-dependent mechanism. Endocrinology. 2009; 150:5318–25. https://doi.org/10.1210/en.2009-0506 [PubMed]
- 54. Novotny JA, Fadel JG, Holstege DM, Furr HC, Clifford AJ. This kinetic, bioavailability, and metabolism study of RRR-α-tocopherol in healthy adults suggests lower intake requirements than previous estimates. J Nutr. 2012; 142:2105–11. https://doi.org/10.3945/jn.112.166462 [PubMed]
- 55. Moloney SJ and Learn DB. The effect of systemic cyclosporin A on a hairless mouse model of photoaging. Photochem Photobiol. 1992; 56:495–504. https://doi.org/10.1111/j.1751-1097.1992.tb02193.x [PubMed]
- 56. Panteleyev AA, van der Veen C, Rosenbach T, Müller-Röver S, Sokolov VE, Paus R. Towards defining the pathogenesis of the hairless phenotype. J Invest Dermatol. 1998; 110:902–07. https://doi.org/10.1046/j.1523-1747.1998.00219.x [PubMed]
- 57. Golovatenko-Abramov PK, Zhirkova EA, Kolokol’chikova EG, Bocharova VS, Platonov ES. [Dermal cysts participate in reparative regeneration of epidermis in Hr(hr)/Hr(hr) mice]. Ontogenez. 2010; 41:285–91. [PubMed]
- 58. Benavides F, Oberyszyn TM, VanBuskirk AM, Reeve VE, Kusewitt DF. The hairless mouse in skin research. J Dermatol Sci. 2009; 53:10–18. https://doi.org/10.1016/j.jdermsci.2008.08.012 [PubMed]
- 59. Tadini KA and Campos PM. In vivo skin effects of a dimethylaminoethanol (DMAE) based formulation. Pharmazie. 2009; 64:818–22. [PubMed]
- 60. San José I, García-Suárez O, Hannestad J, Cabo R, Gauna L, Represa J, Vega JA. The thymus of the hairless rhino-j (hr/rh-j) mice. J Anat. 2001; 198:399–406. https://doi.org/10.1046/j.1469-7580.2001.19840399.x [PubMed]
- 61. Clipstone NA and Crabtree GR. Identification of calcineurin as a key signalling enzyme in T-lymphocyte activation. Nature. 1992; 357:695–97. https://doi.org/10.1038/357695a0 [PubMed]
- 62. Horsley V, Aliprantis AO, Polak L, Glimcher LH, Fuchs E. NFATc1 balances quiescence and proliferation of skin stem cells. Cell. 2008; 132:299–310. https://doi.org/10.1016/j.cell.2007.11.047 [PubMed]
- 63. Pan MG, Xiong Y, Chen F. NFAT gene family in inflammation and cancer. Curr Mol Med. 2013; 13:543–54. https://doi.org/10.2174/1566524011313040007 [PubMed]
- 64. Ho IC, Kim JH, Rooney JW, Spiegelman BM, Glimcher LH. A potential role for the nuclear factor of activated T cells family of transcriptional regulatory proteins in adipogenesis. Proc Natl Acad Sci USA. 1998; 95:15537–41. https://doi.org/10.1073/pnas.95.26.15537 [PubMed]
- 65. Neal JW and Clipstone NA. Calcineurin mediates the calcium-dependent inhibition of adipocyte differentiation in 3T3-L1 cells. J Biol Chem. 2002; 277:49776–81. https://doi.org/10.1074/jbc.M207913200 [PubMed]
- 66. Rovira J, Marcelo Arellano E, Burke JT, Brault Y, Moya-Rull D, Bañón-Maneus E, Ramírez-Bajo MJ, Gutiérrez-Dalmau A, Revuelta I, Quintana LF, Campistol JM, Diekmann F. Effect of mTOR inhibitor on body weight: from an experimental rat model to human transplant patients. Transpl Int. 2008; 21:992–98. https://doi.org/10.1111/j.1432-2277.2008.00710.x [PubMed]
- 67. Lynch L, Michelet X, Zhang S, Brennan PJ, Moseman A, Lester C, Besra G, Vomhof-Dekrey EE, Tighe M, Koay HF, Godfrey DI, Leadbetter EA, Sant’Angelo DB, et al. Regulatory iNKT cells lack expression of the transcription factor PLZF and control the homeostasis of T(reg) cells and macrophages in adipose tissue. Nat Immunol. 2015; 16:85–95. https://doi.org/10.1038/ni.3047 [PubMed]
- 68. Gupta V, Bhasin S, Guo W, Singh R, Miki R, Chauhan P, Choong K, Tchkonia T, Lebrasseur NK, Flanagan JN, Hamilton JA, Viereck JC, Narula NS, et al. Effects of dihydrotestosterone on differentiation and proliferation of human mesenchymal stem cells and preadipocytes. Mol Cell Endocrinol. 2008; 296:32–40. https://doi.org/10.1016/j.mce.2008.08.019 [PubMed]
- 69. Blouin K, Nadeau M, Perreault M, Veilleux A, Drolet R, Marceau P, Mailloux J, Luu-The V, Tchernof A. Effects of androgens on adipocyte differentiation and adipose tissue explant metabolism in men and women. Clin Endocrinol (Oxf). 2010; 72:176–88. https://doi.org/10.1111/j.1365-2265.2009.03645.x [PubMed]
- 70. Reeve VE, Allanson M, Domanski D, Painter N. Gender differences in UV-induced inflammation and immunosuppression in mice reveal male unresponsiveness to UVA radiation. Photochem Photobiol Sci. 2012; 11:173–79. https://doi.org/10.1039/C1PP05224A [PubMed]
- 71. Farage MA, Miller KW, Zouboulis CC, Piérard GE, Maibach HI. Gender differences in skin Aging and the changing profile of the sex hormones with age. J Steroids Horm Sci. 2012; 3:109. https://doi.org/10.4172/2157-7536.1000109
- 72. Viña J, Borras C, Abdelaziz KM, Garcia-Valles R, Gomez-Cabrera MC. The free radical theory of aging revisited: the cell signaling disruption theory of aging. Antioxid Redox Signal. 2013; 19:779–87. https://doi.org/10.1089/ars.2012.5111 [PubMed]
- 73. Gragnani A, Mac Cornick S, Chominski V, de Noronha SM, de Noronha SA, Ferreira LM. Review of major theories of skin aging. Adv Aging Res. 2014; 3:265–84. https://doi.org/10.4236/aar.2014.34036
- 74. Park SG, Kim JH, Xia Y, Sung JH. Generation of reactive oxygen species in adipose-derived stem cells: friend or foe? Expert Opin Ther Targets. 2011; 15:1297–306. https://doi.org/10.1517/14728222.2011.628315 [PubMed]
- 75. Jeong YM, Sung YK, Kim WK, Kim JH, Kwack MH, Yoon I, Kim DD, Sung JH. Ultraviolet B preconditioning enhances the hair growth-promoting effects of adipose-derived stem cells via generation of reactive oxygen species. Stem Cells Dev. 2013; 22:158–68. https://doi.org/10.1089/scd.2012.0167 [PubMed]
- 76. Sarchio SN, Scolyer RA, Beaugie C, McDonald D, Marsh-Wakefield F, Halliday GM, Byrne SN. Pharmacologically antagonizing the CXCR4-CXCL12 chemokine pathway with AMD3100 inhibits sunlight-induced skin cancer. J Invest Dermatol. 2014; 134:1091–100. https://doi.org/10.1038/jid.2013.424 [PubMed]
- 77. Yano K, Oura H, Detmar M. Targeted overexpression of the angiogenesis inhibitor thrombospondin-1 in the epidermis of transgenic mice prevents ultraviolet-B-induced angiogenesis and cutaneous photo-damage. J Invest Dermatol. 2002; 118:800–05. https://doi.org/10.1046/j.1523-1747.2002.01752.x [PubMed]
- 78. Jin XJ, Kim EJ, Oh IK, Kim YK, Park CH, Chung JH. Prevention of UV-induced skin damages by 11,14,17-eicosatrienoic acid in hairless mice in vivo. J Korean Med Sci. 2010; 25:930–37. https://doi.org/10.3346/jkms.2010.25.6.930 [PubMed]
- 79. Hwang IS, Kim JE, Choi SI, Lee HR, Lee YJ, Jang MJ, Son HJ, Lee HS, Oh CH, Kim BH, Lee SH, Hwang DY. UV radiation-induced skin aging in hairless mice is effectively prevented by oral intake of sea buckthorn (Hippophae rhamnoides L.) fruit blend for 6 weeks through MMP suppression and increase of SOD activity. Int J Mol Med. 2012; 30:392–400. [PubMed]
- 80. Kim HH, Lee MJ, Lee SR, Kim KH, Cho KH, Eun HC, Chung JH. Augmentation of UV-induced skin wrinkling by infrared irradiation in hairless mice. Mech Ageing Dev. 2005; 126:1170–77. https://doi.org/10.1016/j.mad.2005.06.003 [PubMed]
- 81. Cho S, Shin MH, Kim YK, Seo JE, Lee YM, Park CH, Chung JH. Effects of infrared radiation and heat on human skin aging in vivo. J Investig Dermatol Symp Proc. 2009; 14:15–19. https://doi.org/10.1038/jidsymp.2009.7 [PubMed]
- 82. Tanaka Y, Matsuo K, Yuzuriha S. Near-infrared irradiation non-thermally affects subcutaneous adipocytes and bones. Eplasty. 2011; 11:e12. [PubMed]
- 83. Engelsen O. The relationship between ultraviolet radiation exposure and vitamin D status. Nutrients. 2010; 2:482–95. https://doi.org/10.3390/nu2050482 [PubMed]
- 84. Reichrath J. Unravelling of hidden secrets: the role of vitamin D in skin aging. Dermatoendocrinol. 2012; 4:241–44. https://doi.org/10.4161/derm.21312 [PubMed]
- 85. Keisala T, Minasyan A, Lou YR, Zou J, Kalueff AV, Pyykkö I, Tuohimaa P. Premature aging in vitamin D receptor mutant mice. J Steroid Biochem Mol Biol. 2009; 115:91–97. https://doi.org/10.1016/j.jsbmb.2009.03.007 [PubMed]
- 86. Wong KE, Szeto FL, Zhang W, Ye H, Kong J, Zhang Z, Sun XJ, Li YC. Involvement of the vitamin D receptor in energy metabolism: regulation of uncoupling proteins. Am J Physiol Endocrinol Metab. 2009; 296:E820–28. https://doi.org/10.1152/ajpendo.90763.2008 [PubMed]
- 87. Malloy PJ and Feldman BJ. Cell-autonomous regulation of brown fat identity gene UCP1 by unliganded vitamin D receptor. Mol Endocrinol. 2013; 27:1632–42. https://doi.org/10.1210/me.2013-1037 [PubMed]
- 88. Mutt SJ, Hyppönen E, Saarnio J, Järvelin MR, Herzig KH. Vitamin D and adipose tissue-more than storage. Front Physiol. 2014; 5:228. https://doi.org/10.3389/fphys.2014.00228 [PubMed]
- 89. Sakai Y, Kishimoto J, Demay MB. Metabolic and cellular analysis of alopecia in vitamin D receptor knockout mice. J Clin Invest. 2001; 107:961–66. https://doi.org/10.1172/JCI11676 [PubMed]
- 90. Lee H, Bae S, Yoon Y. Anti-adipogenic effects of 1,25-dihydroxyvitamin D3 are mediated by the maintenance of the wingless-type MMTV integration site/β-catenin pathway. Int J Mol Med. 2012; 30:1219–24. [PubMed]
- 91. Pérez E, Bourguet W, Gronemeyer H and de Lera AR. Modulation of RXR function through ligand design. Biochim Biophys Acta. 2012;1821:57–69.
- 92. Matsuda S and Kitagishi Y. Peroxisome proliferator-activated receptor and vitamin d receptor signaling pathways in cancer cells. Cancers (Basel). 2013; 5:1261–70. https://doi.org/10.3390/cancers5041261 [PubMed]
- 93. Nimitphong H, Holick MF, Fried SK, Lee MJ. 25-hydroxyvitamin D₃ and 1,25-dihydroxyvitamin D₃ promote the differentiation of human subcutaneous preadipocytes. PLoS One. 2012; 7:e52171. https://doi.org/10.1371/journal.pone.0052171 [PubMed]
- 94. Narvaez CJ, Simmons KM, Brunton J, Salinero A, Chittur SV, Welsh JE. Induction of STEAP4 correlates with 1,25-dihydroxyvitamin D3 stimulation of adipogenesis in mesenchymal progenitor cells derived from human adipose tissue. J Cell Physiol. 2013; 228:2024–36. https://doi.org/10.1002/jcp.24371 [PubMed]
- 95. Ji E, Jung MY, Park JH, Kim S, Seo CR, Park KW, Lee EK, Yeom CH, Lee S. Inhibition of adipogenesis in 3T3-L1 cells and suppression of abdominal fat accumulation in high-fat diet-feeding C57BL/6J mice after downregulation of hyaluronic acid. Int J Obes. 2014; 38:1035–43. https://doi.org/10.1038/ijo.2013.202 [PubMed]
- 96. Kang L, Lantier L, Kennedy A, Bonner JS, Mayes WH, Bracy DP, Bookbinder LH, Hasty AH, Thompson CB, Wasserman DH. Hyaluronan accumulates with high-fat feeding and contributes to insulin resistance. Diabetes. 2013; 62:1888–96. https://doi.org/10.2337/db12-1502 [PubMed]
- 97. Park BG, Lee CW, Park JW, Cui Y, Park YS, Shin WS. Enzymatic fragments of hyaluronan inhibit adipocyte differentiation in 3T3-L1 pre-adipocytes. Biochem Biophys Res Commun. 2015; 467:623–28. https://doi.org/10.1016/j.bbrc.2015.10.104 [PubMed]
- 98. Zhu Y, Crewe C, Scherer PE. Hyaluronan in adipose tissue: beyond dermal filler and therapeutic carrier. Sci Transl Med. 2016; 8:323ps4. https://doi.org/10.1126/scitranslmed.aad6793 [PubMed]
- 99. Averbeck M, Gebhardt CA, Voigt S, Beilharz S, Anderegg U, Termeer CC, Sleeman JP, Simon JC. Differential regulation of hyaluronan metabolism in the epidermal and dermal compartments of human skin by UVB irradiation. J Invest Dermatol. 2007; 127:687–97. https://doi.org/10.1038/sj.jid.5700614 [PubMed]
- 100. Rauhala L, Hämäläinen L, Salonen P, Bart G, Tammi M, Pasonen-Seppänen S, Tammi R. Low dose ultraviolet B irradiation increases hyaluronan synthesis in epidermal keratinocytes via sequential induction of hyaluronan synthases Has1-3 mediated by p38 and Ca2+/calmodulin-dependent protein kinase II (CaMKII) signaling. J Biol Chem. 2013; 288:17999–8012. https://doi.org/10.1074/jbc.M113.472530 [PubMed]
- 101. Dai G, Freudenberger T, Zipper P, Melchior A, Grether-Beck S, Rabausch B, de Groot J, Twarock S, Hanenberg H, Homey B, Krutmann J, Reifenberger J, Fischer JW. Chronic ultraviolet B irradiation causes loss of hyaluronic acid from mouse dermis because of down-regulation of hyaluronic acid synthases. Am J Pathol. 2007; 171:1451–61. https://doi.org/10.2353/ajpath.2007.070136 [PubMed]
- 102. Halder RM and Ara CJ. Skin cancer and photoaging in ethnic skin. Dermatol Clin. 2003; 21:725–32, x. https://doi.org/10.1016/S0733-8635(03)00085-8 [PubMed]
- 103. Tschachler E, Morizot F. Ethnic differences in skin aging. In: Gilchrest BA, Krutmann J (eds). Skin Aging (pp. 23-31). Springer, Heidelberg-Berlin, 2006.
- 104. Alexis AF and Alam M. Racial and ethnic differences in skin aging: implications for treatment with soft tissue fillers. J Drugs Dermatol. 2012; 11:s30–32. [PubMed]
- 105. Nouveau-Richard S, Yang Z, Mac-Mary S, Li L, Bastien P, Tardy I, Bouillon C, Humbert P, de Lacharrière O. Skin ageing: a comparison between Chinese and European populations. A pilot study. J Dermatol Sci. 2005; 40:187–93. https://doi.org/10.1016/j.jdermsci.2005.06.006 [PubMed]
- 106. Perner D, Vierkötter A, Sugiri D, Matsui M, Ranft U, Esser C, Ethève S, Goralczyk R, Kaneko N, Yamamoto A, Morita A, Krutmann J, Krämer U. Association between sun-exposure, smoking behaviour and plasma antioxidant levels with the different manifestation of skin ageing signs between Japanese and German women--a pilot study. J Dermatol Sci. 2011; 62:138–40. https://doi.org/10.1016/j.jdermsci.2011.02.010 [PubMed]
- 107. Rawlings AV. Ethnic skin types: are there differences in skin structure and function? Int J Cosmet Sci. 2006; 28:79–93. https://doi.org/10.1111/j.1467-2494.2006.00302.x [PubMed]
- 108. Sood RF, Hocking AM, Muffley LA, Ga M, Honari S, Reiner AP, Rowhani-Rahbar A, Gibran NS. Race and melanocortin 1 receptor polymorphism R163Q are associated with post-burn hypertrophic scarring: a prospective cohort study. J Invest Dermatol. 2015; 135:2394–401. https://doi.org/10.1038/jid.2015.197 [PubMed]
- 109. Ezure T and Amano S. Negative regulation of dermal fibroblasts by enlarged adipocytes through release of free fatty acids. J Invest Dermatol. 2011; 131:2004–09. https://doi.org/10.1038/jid.2011.145 [PubMed]
- 110. Ezure T and Amano S. Increment of subcutaneous adipose tissue is associated with decrease of elastic fibres in the dermal layer. Exp Dermatol. 2015; 24:924–29. https://doi.org/10.1111/exd.12816 [PubMed]
- 111. Sherratt MJ. Tissue elasticity and the ageing elastic fibre. Age (Dordr). 2009; 31:305–25. https://doi.org/10.1007/s11357-009-9103-6 [PubMed]
- 112. Kruglikov IL. Neocollagenesis in non-invasive aesthetic treatments. Journal of Cosmetics Dermatological Sciences and Applications. 2013; 3:1–5. https://doi.org/10.4236/jcdsa.2013.31A001
- 113. Sherratt MJ. Body mass index and dermal remodelling. Exp Dermatol. 2015; 24:922–23. https://doi.org/10.1111/exd.12851 [PubMed]



