Introduction
Aging is generally considered the result of time-dependent deterioration due to stochastic, accumulative ‘wear and tear’ causing gradual degeneration. Therefore, time is the prevailing determinant in age-related processes [1, 2]. However, although aging is highly correlated with time, additional factors significantly influence the rate of aging and as a consequence, individual aging differs greatly [3-6]. Moreover, the rate of age-related deterioration and functional decline varies within every individual in a tissue-specific manner [7-12]. In humans, lifespan ranges from less than 10 years for the severe progeria patients [13, 14] to over 100 years for centenarians. Many pro-aging factors are likely controlled to some extent by genetic variation [2, 15]. However, even in genetically identical, inbred animals aging rate varies substantially among individuals [4, 16, 17]. This indicates that other factors besides time are of significance. Genomic instability due to accumulation of stochastic damage in DNA over time [16-28] causing cell death and cellular senescence is believed to be one of the drivers of aging [29-43]. It has proved difficult to mechanistically dissect processes involved in individual and tissue-specific aging.
Acknowledgments
The work presented here was in part financially supported by IOP Genomics IGE03009, NIH/NIA (3PO1 AG017242), STW Grant STW-LGC.6935 and Netherlands Bioinformatics Center (NBIC) BioRange II – BR4.1. Support was also obtained from Markage (FP7-Health-2008-200880), LifeSpan (LSHG-CT-2007-036894), European Research Council (ERC advanced scientist grant JHJH).
Conflicts of Interest
The authors of this manuscript declare no conflicts of interest.
References
- 1. Ressler S, Bartkova J, Niederegger H, Bartek J, Scharffetter-Kochanek K, Jansen-Durr P, Wlaschek M. p16INK4A is a robust in vivo biomarker of cellular aging in human skin. Aging cell. 2006; 5: 379 -389. [PubMed] .
- 2. Vijg J. 2007; Aging of the Genome, The dual role of DNA in life and death Oxford University Press .
- 3. Cherkas LF, Aviv A, Valdes AM, Hunkin JL, Gardner JP, Surdulescu GL, Kimura M, Spector TD. The effects of social status on biological aging as measured by white-blood-cell telomere length. Aging cell. 2006; 5: 361 -365. [PubMed] .
- 4. Jonker MJ, Melis JP, Kuiper RV, van der Hoeven TV, Wackers PF, Robinson J, van der Horst GT, Dolle ME, Vijg J, Breit TM, Hoeijmakers JH, van Steeg H. Life spanning murine gene expression profiles in relation to chronological and pathological aging in multiple organs. Aging cell. 2013; 12: 901 -909. [PubMed] .
- 5. Tomas-Loba A, Bernardes de Jesus B, Mato JM, Blasco MA. A metabolic signature predicts biological age in mice. Aging cell. 2013; 12: 93 -101. [PubMed] .
- 6. Holly AC, Melzer D, Pilling LC, Henley W, Hernandez DG, Singleton AB, Bandinelli S, Guralnik JM, Ferrucci L, Harries LW. Towards a gene expression biomarker set for human biological age. Aging cell. 2013; 12: 324 -326. [PubMed] .
- 7. Busuttil R, Bahar R, Vijg J. Genome dynamics and transcriptional deregulation in aging. Neuroscience. 2007; 145: 1341 -1347. [PubMed] .
- 8. Fu C, Hickey M, Morrison M, McCarter R, Han ES. Tissue specific and non-specific changes in gene expression by aging and by early stage CR. Mechanisms of ageing and development. 2006; 127: 905 -916. [PubMed] .
- 9. Glass D, Vinuela A, Davies MN, Ramasamy A, Parts L, Knowles D, Brown AA, Hedman AK, Small KS, Buil A, Grundberg E, Nica AC, Di Meglio P, et al. Gene expression changes with age in skin, adipose tissue, blood and brain. Genome biology. 2013; 14: R75 [PubMed] .
- 10. Park SK, Kim K, Page GP, Allison DB, Weindruch R, Prolla TA. Gene expression profiling of aging in multiple mouse strains: identification of aging biomarkers and impact of dietary antioxidants. Aging cell. 2009; 8: 484 -495. [PubMed] .
- 11. Swindell WR. Comparative analysis of microarray data identifies common responses to caloric restriction among mouse tissues. Mechanisms of ageing and development. 2008; 129: 138 -153. [PubMed] .
- 12. Zahn JM, Poosala S, Owen AB, Ingram DK, Lustig A, Carter A, Weeraratna AT, Taub DD, Gorospe M, Mazan-Mamczarz K, Lakatta EG, Boheler KR, Xu X, et al. AGEMAP: a gene expression database for aging in mice. PLoS genetics. 2007; 3: e201 [PubMed] .
- 13. Cleaver JE, Lam ET, Revet I. Disorders of nucleotide excision repair: the genetic and molecular basis of heterogeneity. Nature reviews Genetics. 2009; 10: 756 -768. .
- 14. Kraemer KH, Patronas NJ, Schiffmann R, Brooks BP, Tamura D, DiGiovanna JJ. Xeroderma pigmentosum, trichothiodystrophy and Cockayne syndrome: a complex genotype-phenotype relationship. Neuroscience. 2007; 145: 1388 -1396. [PubMed] .
- 15. Hoeijmakers JH. DNA damage, aging, and cancer. The New England journal of medicine. 2009; 361: 1475 -1485. [PubMed] .
- 16. Hollander CF, Solleveld HA, Zurcher C, Nooteboom AL, Van Zwieten MJ. Biological and clinical consequences of longitudinal studies in rodents: their possibilities and limitations. An overview. Mechanisms of ageing and development. 1984; 28: 249 -260. [PubMed] .
- 17. Melis JP, Wijnhoven SW, Beems RB, Roodbergen M, van den Berg J, Moon H, Friedberg E, van der Horst GT, Hoeijmakers JH, Vijg J, van Steeg H. Mouse models for xeroderma pigmentosum group A and group C show divergent cancer phenotypes. Cancer research. 2008; 68: 1347 -1353. [PubMed] .
- 18. Barzilai A, Rotman G, Shiloh Y. ATM deficiency and oxidative stress: a new dimension of defective response to DNA damage. DNA repair. 2002; 1: 3 -25. [PubMed] .
- 19. Bucholtz N and Demuth I. DNA-repair in mild cognitive impairment and Alzheimer's disease. DNA repair. 2013; .
- 20. Canugovi C, Misiak M, Ferrarelli LK, Croteau DL, Bohr VA. The role of DNA repair in brain related disease pathology. DNA repair. 2013; 12: 578 -587. [PubMed] .
- 21. Diderich K, Alanazi M, Hoeijmakers JH. Premature aging and cancer in nucleotide excision repair-disorders. DNA repair. 2011; 10: 772 -780. [PubMed] .
- 22. Dolle ME, Snyder WK, Gossen JA, Lohman PH, Vijg J. Distinct spectra of somatic mutations accumulated with age in mouse heart and small intestine. Proceedings of the National Academy of Sciences of the United States of America. 2000; 97: 8403 -8408. [PubMed] .
- 23. Giese H, Dolle ME, Hezel A, van Steeg H, Vijg J. Accelerated accumulation of somatic mutations in mice deficient in the nucleotide excision repair gene XPA. Oncogene. 1999; 18: 1257 -1260. [PubMed] .
- 24. Melis JP, Kuiper RV, Zwart E, Robinson J, Pennings JL, van Oostrom CT, Luijten M, van Steeg H. Slow accumulation of mutations in Xpc mice upon induction of oxidative stress. DNA repair. 2013; in press .
- 25. Melis JP, Luijten M, Mullenders LH, van Steeg H. The role of XPC: implications in cancer and oxidative DNA damage. Mutation research. 2011; 728: 107 -117. [PubMed] .
- 26. Melis JP, van Steeg H, Luijten M. Oxidative DNA damage and nucleotide excision repair. Antioxidants & redox signaling. 2013; 18: 2409 -2419. [PubMed] .
- 27. Ono T, Miyamura Y, Ikehata H, Yamanaka H, Kurishita A, Yamamoto K, Suzuki T, Nohmi T, Hayashi M, Sofuni T. Spontaneous mutant frequency of lacZ gene in spleen of transgenic mouse increases with age. Mutation research. 1995; 338: 183 -188. [PubMed] .
- 28. Stevnsner T, Nyaga S, de Souza-Pinto NC, van der Horst GT, Gorgels TG, Hogue BA, Thorslund T, Bohr VA. Mitochondrial repair of 8-oxoguanine is deficient in Cockayne syndrome group B. Oncogene. 2002; 21: 8675 -8682. [PubMed] .
- 29. Beltrami E, Ruggiero A, Busuttil R, Migliaccio E, Pelicci PG, Vijg J, Giorgio M. Deletion of p66Shc in mice increases the frequency of size-change mutations in the lacZ transgene. Aging cell. 2013; 12: 177 -183. [PubMed] .
- 30. Garinis GA, Uittenboogaard LM, Stachelscheid H, Fousteri M, van Ijcken W, Breit TM, van Steeg H, Mullenders LH, van der Horst GT, Bruning JC, Niessen CM, Hoeijmakers JH, Schumacher B. Persistent transcription-blocking DNA lesions trigger somatic growth attenuation associated with longevity. Nature cell biology. 2009; 11: 604 -615. .
- 31. Hasty P and Vijg J. Accelerating aging by mouse reverse genetics: a rational approach to understanding longevity. Aging cell. 2004; 3: 55 -65. [PubMed] .
- 32. Kyng KJ, May A, Stevnsner T, Becker KG, Kolvra S, Bohr VA. Gene expression responses to DNA damage are altered in human aging and in Werner Syndrome. Oncogene. 2005; 24: 5026 -5042. [PubMed] .
- 33. Liu B, Wang Z, Ghosh S, Zhou Z. Defective ATM-Kap-1-mediated chromatin remodeling impairs DNA repair and accelerates senescence in progeria mouse model. Aging cell. 2013; 12: 316 -318. [PubMed] .
- 34. Maslov AY, Ganapathi S, Westerhof M, Quispe-Tintaya W, White RR, Van Houten B, Reiling E, Dolle ME, van Steeg H, Hasty P, Hoeijmakers JH, Vijg J. DNA damage in normally and prematurely aged mice. Aging cell. 2013; 12: 467 -477. [PubMed] .
- 35. Matheu A, Maraver A, Klatt P, Flores I, Garcia-Cao I, Borras C, Flores JM, Vina J, Blasco MA, Serrano M. Delayed ageing through damage protection by the Arf/p53 pathway. Nature. 2007; 448: 375 -379. [PubMed] .
- 36. Rufini A, Tucci P, Celardo I, Melino G. Senescence and aging: the critical roles of p53. Oncogene. 2013; in press .
- 37. Sepe S, Payan-Gomez C, Milanese C, Hoeijmakers JH, Mastroberardino PG. Nucleotide excision repair in chronic neurodegenerative diseases. DNA repair. 2013; 12: 568 -577. [PubMed] .
- 38. Seviour EG and Lin SY. The DNA damage response: Balancing the scale between cancer and ageing. Aging. 2010; 2: 900 -907. [PubMed] .
- 39. Suh Y and Vijg J. Maintaining genetic integrity in aging: a zero sum game. Antioxidants & redox signaling. 2006; 8: 559 -571. [PubMed] .
- 40. Wang C, Jurk D, Maddick M, Nelson G, Martin-Ruiz C, von Zglinicki T. DNA damage response and cellular senescence in tissues of aging mice. Aging cell. 2009; 8: 311 -323. [PubMed] .
- 41. Campisi J. Senescent cells, tumor suppression, and organismal aging: good citizens, bad neighbors. Cell. 2005; 120: 513 -522. [PubMed] .
- 42. Campisi J and Yaswen P. Aging and cancer cell biology, 2009. Aging cell. 2009; 8: 221 -225. [PubMed] .
- 43. Vijg J and Campisi J. Puzzles, promises and a cure for ageing. Nature. 2008; 454: 1065 -1071. [PubMed] .
- 44. Kim J and Eberwine J. RNA: state memory and mediator of cellular phenotype. Trends in cell biology. 2010; 20: 311 -318. [PubMed] .
- 45. de Magalhaes JP, Curado J, Church GM. Meta-analysis of age-related gene expression profiles identifies common signatures of aging. Bioinformatics. 2009; 25: 875 -881. [PubMed] .
- 46. Swindell WR. Gene expression profiling of long-lived dwarf mice: longevity-associated genes and relationships with diet, gender and aging. BMC genomics. 2007; 8: 353 [PubMed] .
- 47. Swindell WR. Genes and gene expression modules associated with caloric restriction and aging in the laboratory mouse. BMC genomics. 2009; 10: 585 [PubMed] .
- 48. Swindell WR. Dietary restriction in rats and mice: a meta-analysis and review of the evidence for genotype-dependent effects on lifespan. Ageing research reviews. 2012; 11: 254 -270. [PubMed] .
- 49. Swindell WR, Johnston A, Sun L, Xing X, Fisher GJ, Bulyk ML, Elder JT, Gudjonsson JE. Meta-profiles of gene expression during aging: limited similarities between mouse and human and an unexpectedly decreased inflammatory signature. PloS one. 2012; 7: e33204 [PubMed] .
- 50. Southworth LK, Owen AB, Kim SK. Aging mice show a decreasing correlation of gene expression within genetic modules. PLoS genetics. 2009; 5: e1000776 [PubMed] .
- 51. Melis JPM, et al. 2011; Nucleotide Excision Repair and Cancer Vengrova DS. DNA Repair and Human Health. Rijeka, Croatia InTech 121 -144. .
- 52. Melis JPM. 2012; Nucleotide excision repair in aging & cancer. Department of Toxicogenetics. Leiden Leiden University Medical Center 158 .
- 53. Katic M, Kennedy AR, Leykin I, Norris A, McGettrick A, Gesta S, Russell SJ, Bluher M, Maratos-Flier E, Kahn CR. Mitochondrial gene expression and increased oxidative metabolism: role in increased lifespan of fat-specific insulin receptor knock-out mice. Aging cell. 2007; 6: 827 -839. [PubMed] .
- 54. Komarova EA, Antoch MP, Novototskaya LR, Chernova OB, Paszkiewicz G, Leontieva OV, Blagosklonny MV, Gudkov AV. Rapamycin extends lifespan and delays tumorigenesis in heterozygous p53+/− mice. Aging. 2012; 4: 709 -714. [PubMed] .
- 55. Linford NJ, Beyer RP, Gollahon K, Krajcik RA, Malloy VL, Demas V, Burmer GC, Rabinovitch PS. Transcriptional response to aging and caloric restriction in heart and adipose tissue. Aging cell. 2007; 6: 673 -688. [PubMed] .
- 56. Steelman LS, Chappell WH, Abrams SL, Kempf RC, Long J, Laidler P, Mijatovic S, Maksimovic-Ivanic D, Stivala F, Mazzarino MC, Donia M, Fagone P, Malaponte G, et al. Roles of the Raf/MEK/ERK and PI3K/PTEN/Akt/mTOR pathways in controlling growth and sensitivity to therapy-implications for cancer and aging. Aging. 2011; 3: 192 -222. [PubMed] .
- 57. Bluher M, Kahn BB, Kahn CR. Extended longevity in mice lacking the insulin receptor in adipose tissue. Science. 2003; 299: 572 -574. [PubMed] .
- 58. Edgar D and Trifunovic A. The mtDNA mutator mouse: Dissecting mitochondrial involvement in aging. Aging. 2009; 1: 1028 -1032. [PubMed] .
- 59. Holzenberger M, Dupont J, Ducos B, Leneuve P, Geloen A, Even PC, Cervera P, Le Bouc Y. IGF-1 receptor regulates lifespan and resistance to oxidative stress in mice. Nature. 2003; 421: 182 -187. [PubMed] .
- 60. Keniry M and Parsons R. The role of PTEN signaling perturbations in cancer and in targeted therapy. Oncogene. 2008; 27: 5477 -5485. [PubMed] .
- 61. Lenaz G, Baracca A, Fato R, Genova ML, Solaini G. New insights into structure and function of mitochondria and their role in aging and disease. Antioxidants & redox signaling. 2006; 8: 417 -437. [PubMed] .
- 62. Merry BJ. Oxidative stress and mitochondrial function with aging--the effects of calorie restriction. Aging cell. 2004; 3: 7 -12. [PubMed] .
- 63. Pulliam DA, Bhattacharya A, Van Remmen H. Mitochondrial Dysfunction in Aging and Longevity: A Causal or Protective Role? Antioxidants & redox signaling. 2012; .
- 64. Scialo F, Mallikarjun V, Stefanatos R, Sanz A. Regulation of Lifespan by the Mitochondrial Electron Transport Chain: Reactive Oxygen Species-Dependent and Reactive Oxygen Species-Independent Mechanisms. Antioxidants & redox signaling. 2012; .
- 65. Seo K, Choi E, Lee D, Jeong DE, Jang SK, Lee SJ. Heat shock factor 1 mediates the longevity conferred by inhibition of TOR and insulin/IGF-1 signaling pathways in C. elegans. Aging cell. 2013; in press .
- 66. Solari F, Bourbon-Piffaut A, Masse I, Payrastre B, Chan AM, Billaud M. The human tumour suppressor PTEN regulates longevity and dauer formation in Caenorhabditis elegans. Oncogene. 2005; 24: 20 -27. [PubMed] .
- 67. Zeng N, Yang KT, Bayan JA, He L, Aggarwal R, Stiles JW, Hou X, Medina V, Abad D, Palian BM, Al-Abdullah I, Kandeel F, Johnson DL, et al. PTEN controls beta-cell regeneration in aged mice by regulating cell cycle inhibitor p16. Aging cell. 2013; in press .
- 68. Busuttil RA, Garcia AM, Reddick RL, Dolle ME, Calder RB, Nelson JF, Vijg J. Intra-organ variation in age-related mutation accumulation in the mouse. PloS one. 2007; 2: 9 e876 [PubMed] .
- 69. Wang J, Clauson CL, Robbins PD, Niedernhofer LJ, Wang Y. The oxidative DNA lesions 8,5'-cyclopurines accumulate with aging in a tissue-specific manner. Aging cell. 2012; 11: 714 -716. [PubMed] .
- 70. Zhan M, Yamaza H, Sun Y, Sinclair J, Li H, Zou S. Temporal and spatial transcriptional profiles of aging in Drosophila melanogaster. Genome research. 2007; 17: 1236 -1243. [PubMed] .
- 71. Huang T, Zhang J, Xie L, Dong X, Zhang L, Cai YD, Li YX. Crosstissue coexpression network of aging. Omics : a journal of integrative biology. 2011; 15: 665 -671. [PubMed] .
- 72. Ludlow AT, Witkowski S, Marshall MR, Wang J, Lima LC, Guth LM, Spangenburg EE, Roth SM. Chronic exercise modifies age-related telomere dynamics in a tissue-specific fashion. The journals of gerontology Series A, Biological sciences and medical sciences. 2012; 67: 911 -926. .
- 73. Vijg J, Busuttil RA, Bahar R, Dolle ME. Aging and genome maintenance. Annals of the New York Academy of Sciences. 2005; 1055: 35 -47. [PubMed] .
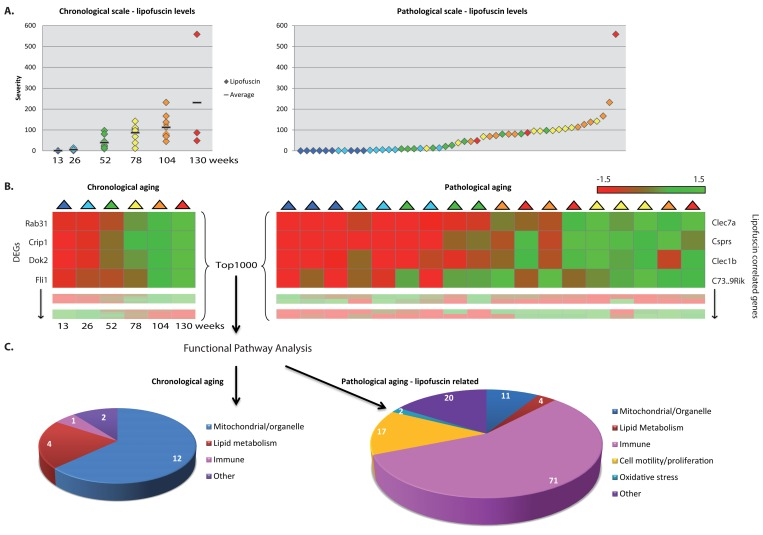
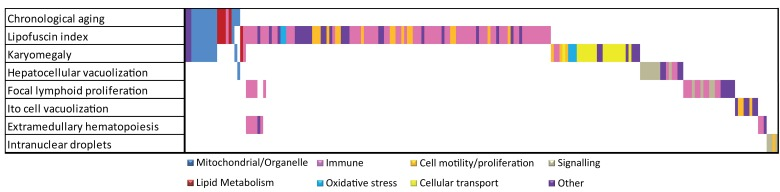
- 74. Brunk UT, Jones CB, Sohal RS. A novel hypothesis of lipofuscinogenesis and cellular aging based on interactions between oxidative stress and autophagocytosis. Mutation research. 1992; 275: 395 -403. [PubMed] .
- 75. Georgakopoulou EA, Tsimaratou K, Evangelou K, Fernandez Marcos PJ, Zoumpourlis V, Trougakos IP, Kletsas D, Bartek J, Serrano M, Gorgoulis VG. Specific lipofuscin staining as a novel biomarker to detect replicative and stress-induced senescence. A method applicable in cryo-preserved and archival tissues. Aging. 2013; 5: 37 -50. [PubMed] .
- 76. Goel SK, Lalwani ND, Reddy JK. Peroxisome proliferation and lipid peroxidation in rat liver. Cancer research. 1986; 46: 1324 -1330. [PubMed] .
- 77. Hohn A and Grune T. Lipofuscin: formation, effects and role of macroautophagy. Redox biology. 2013; 1: 140 -144. [PubMed] .
- 78. Jung T, Bader N, Grune T. Lipofuscin: formation, distribution, and metabolic consequences. Annals of the New York Academy of Sciences. 2007; 1119: 97 -111. [PubMed] .
- 79. Kurz T, Eaton JW, Brunk UT. Redox activity within the lysosomal compartment: implications for aging and apoptosis. Antioxidants & redox signaling. 2010; 13: 511 -523. [PubMed] .
- 80. Melis JP, Speksnijder EN, Kuiper RV, Salvatori DC, Schaap MM, Maas S, Robinson J, Verhoef A, van Benthem J, Luijten M, van Steeg H. Detection of genotoxic and non-genotoxic carcinogens in Xpc(−/−)p53(+/−) mice. Toxicology and applied pharmacology. 2013; 266: 289 -297. [PubMed] .
- 81. Moreira PI, Santos MS, Oliveira CR. Alzheimer's disease: a lesson from mitochondrial dysfunction. Antioxidants & redox signaling. 2007; 9: 1621 -1630. [PubMed] .
- 82. Moriya K, Nakagawa K, Santa T, Shintani Y, Fujie H, Miyoshi H, Tsutsumi T, Miyazawa T, Ishibashi K, Horie T, Imai K, Todoroki T, Kimura S, et al. Oxidative stress in the absence of inflammation in a mouse model for hepatitis C virus-associated hepatocarcinogenesis. Cancer research. 2001; 61: 4365 -4370. [PubMed] .
- 83. Perse M, Injac R, Erman A. Oxidative status and lipofuscin accumulation in urothelial cells of bladder in aging mice. PloS one. 2013; 8: e59638 [PubMed] .
- 84. Terman A and Brunk UT. Oxidative stress, accumulation of biological ‘garbage’, and aging. Antioxidants & redox signaling. 2006; 8: 197 -204. [PubMed] .
- 85. Terman A, Kurz T, Navratil M, Arriaga EA, Brunk UT. Mitochondrial turnover and aging of long-lived postmitotic cells: the mitochondrial-lysosomal axis theory of aging. Antioxidants & redox signaling. 2010; 12: 503 -535. [PubMed] .
- 86. Xu H, Chen M, Manivannan A, Lois N, Forrester JV. Age-dependent accumulation of lipofuscin in perivascular and subretinal microglia in experimental mice. Aging cell. 2008; 7: 58 -68. [PubMed] .
- 87. Brunk UT and Terman A. Lipofuscin: mechanisms of age-related accumulation and influence on cell function. Free radical biology & medicine. 2002; 33: 611 -619. [PubMed] .
- 88. Droge W and Schipper HM. Oxidative stress and aberrant signaling in aging and cognitive decline. Aging cell. 2007; 6: 361 -370. [PubMed] .
- 89. Go YM and Jones DP. Redox control systems in the nucleus: mechanisms and functions. Antioxidants & redox signaling. 2010; 13: 489 -509. [PubMed] .
- 90. Hearps AC, Martin GE, Angelovich TA, Cheng WJ, Maisa A, Landay AL, Jaworowski A, Crowe SM. Aging is associated with chronic innate immune activation and dysregulation of monocyte phenotype and function. Aging cell. 2012; 11: 867 -875. [PubMed] .
- 91. Jackaman C, Radley-Crabb HG, Soffe Z, Shavlakadze T, Grounds MD, Nelson DJ. Targeting macrophages rescues age-related immune deficiencies in C57BL/6J geriatric mice. Aging cell. 2013; 12: 345 -357. [PubMed] .
- 92. Jun JI and Lau LF. Cellular senescence controls fibrosis in wound healing. Aging. 2010; 2: 627 -631. [PubMed] .
- 93. Labunskyy VM and Gladyshev VN. Role of Reactive Oxygen Species-Mediated Signaling in Aging. Antioxidants & redox signaling. 2013; 19: 1362 -1372. [PubMed] .
- 94. Liu D and Xu Y. p53, oxidative stress, and aging. Antioxidants & redox signaling. 2011; 15: 1669 -1678. [PubMed] .
- 95. Ponnappan S and Ponnappan U. Aging and immune function: molecular mechanisms to interventions. Antioxidants & redox signaling. 2011; 14: 1551 -1585. [PubMed] .
- 96. Qian F, Wang X, Zhang L, Chen S, Piecychna M, Allore H, Bockenstedt L, Malawista S, Bucala R, Shaw AC, Fikrig E, Montgomery RR. Age-associated elevation in TLR5 leads to increased inflammatory responses in the elderly. Aging cell. 2012; 11: 104 -110. [PubMed] .
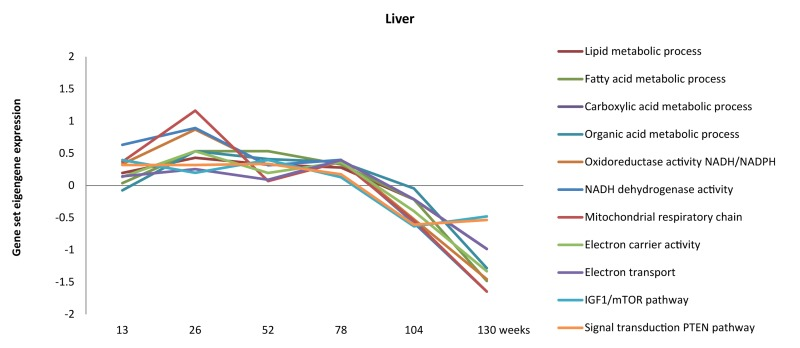
- 97. Sheedfar F, Biase SD, Koonen D, Vinciguerra M. Liver diseases and aging: friends or foes? Aging cell. 2013; in press .
- 98. Swindell WR, Johnston A, Xing X, Little A, Robichaud P, Voorhees JJ, Fisher G, Gudjonsson JE. Robust shifts in S100a9 expression with aging: a novel mechanism for chronic inflammation. Scientific reports. 2013; 3: 1215 [PubMed] .