Introduction
The progressive degeneration and functional decline of cells, tissues, and organs that accompany aging are unavoidable. To date, the most rigorous studies on aging have been conducted on the T-lymphoid immune organs, primarily comprising the reservoir for hematopoietic stem cells (HSCs) – the bone marrow (BM), and the immature T-cell cradle and educators – the thymus gland. These organs are all comprised of two main cellular components of different origins: lympho-hematopoietic — immature and mature T cells and non-lympho-hematopoietic — stromal or niche cells. These two cellular components interact with each other (lympho-stromal interaction) via soluble factors and cell-to-cell contacts that initiate gene expression to promote development of T-cell immune system and control the process of T-cell immune system aging. Although both lympho-hematopoietic and non-lymphohematopoietic components have been found not to be truly immortal, they are unlikely to develop age-related intrinsic defects at the same rate or show simultaneous mortality [1]. It is debatable if aging-induced failure to generate T cells, characterized by age-related thymic involution that reduces the naïve T-cell pool, is due to intrinsic defects in the lympho-hematopoietic progenitor/stem cells (LPCs or HSCs) themselves, or is adversely regulated by intrinsic defects of their niche cells, including mesenchymal stem cell-derived osteoblasts in the BM [2-4] and thymic epithelial cells (TECs) in the thymus [1, 5, 6]. It is important to clarify this clinically relevant issue so that selection of the right target(s) and development of proper strategies to rejuvenate the aged immune system can be implemented. Such an approach will hopefully delay and/or treat the onset of age-related immune deficient diseases.
It has been well documented that aging-induced decline of lymphocytes is not due to reduction of quantities in BM progenitors, because in mice, quantities of HSCs within BM are generally increased, instead of decreased (Six reports summarized in a recent review [7] and our own observation [1]), while in human, aged BM progenitor numbers are either decreased [8] or increased [9]. At least, in mice, quality, rather than quantity, of aged BM progenitors, is considered to contribute to decreased lymphopoiesis with aging. One of main changes of aged BM progenitor characteristics is a myeloid-skew (alteration in HSC differentiation), i.e. aged BM progenitors produce greater numbers of myeloid than lymphoid lineage cells compared to young BM progenitors [3, 10-12]. This seems that aging of the T-lymphoid system occurs due to primary and intrinsic changes in LPCs/HSCs. However, recently a few but intriguing studies have reported that the primary and dominant cause of lymphoid system aging may be attributed to their endogenous microenvironment, constituted by stem-cell niche cells (i.e. stromal cells), in the BM [2, 3] and thymus [1, 6]. Deterioration of these microenvironments induces LPC/HSC defects, and ultimately leads to a failure of the entire T-cell generation system.
The notion of aging-induced defects in the stem-cell niche rather than in the stem cells themselves, leading to system wide failure has been recently recognized in many systems, such as aging in oocytes/ovary [13-15], sperm/testis [16, 17], and muscles [18, 19]. However, this scenario has not been widely accepted in T-lymphoid system aging. This is in part due to the inadequate understanding of the divergent down-regulated lymphoid and up-regulated myeloid gene expression profile in aged BM progenitors [20], and the myeloid-biased cellular differentiation of aged BM progenitors [10, 12], despite the argument that these may arise from the loss or expansion potential from any particular niche [21, 22]. Therefore, it is of utmost importance to determine whether old niches in the BM and/or thymus can directly affect competence of young LPCs to generate T cells. Furthermore, it is important to delineate the interactions between LPCs and their niche via integrative analysis of both LPCs and their niche cells in the same developmental microenvironments.
A syngeneic bone marrow transplantation (BMT), where old BM progenitors are introduced into a young mouse environment and vice versa, is a common in vivo developmental setting to address this issue [23]. In this respect, BMT into irradiated host mice [10] was found to produce inaccurate results due to radiation-induced damage to stromal cells [12]. Likewise the use of non-irradiated wild-type (WT) host mice presented a different set of problems as host niches are predominantly occupied leaving only 0.1-1.0% of the HSC niches available for engraftment [24]. However, hosts with lympho-hematopoietic genetic mutation but possessing normal genetic (WT-equivalent) niche cells have been demonstrated to have an improved durability in stem cell engraftment for studying stem cell function in non-irradiation hosts [24, 25]. Although non-hematopoietc stromal cells in these lympho-hematopoietic deficient hosts are at pre-developmental state, due to lack of lympho-stromal crosstalk, these pre-developmental stromal cells will be soon re-programmed to provide normal environment for supporting hematopoietic cell development, as soon as normal HSCs are introduced in the system and participate in the crosstalk. The IL7R gene knockout mouse is one of these kind hosts. Therefore, conducting a syngeneic BMT in IL7R−/− host mice [26] instead of WT host mice, circumvents the problem of predominantly occupied host niches [27].
Purified subpopulations of T-cell precursors used in in vitro culture settings, such as fetal thymic organ culture (FTOC) [28] and OP9-DL1 monolayer culture [12, 29] is also a developmental setting to address the effects of aging on lympho-stromal interactions. However, in vitro manipulation of cells, such as flow cytometric sorting, may cause a defect in old LPCs, such as transplantability [30] and/or decreased adhesion to stroma [31]. In addition, since the potential thymus-seeding T-lymphocyte progenitors are complex and undefined [32], any single subpopulation isolated by cell sorting may not reflect the full differentiation potential of natural thymus-seeding LPCs. Therefore, recruitment of natural thymus-seeding LPCs in vivo using kidney capsule transplantation (KCT) of fetal thymic lobes was designed to overcome the above pitfalls [1].
In this study, we have circumvented the issues described above by developing several novel and comprehensive in vivo and in vitro models, such as using IL7R−/− [26] host mice for BMT, designing a cross-KCT (cKCT) model to provide the same microenvironment for competition, and using KCT-recruited young and old LPCs for a competitive co-culture on an OP9-DL1 stromal monolayer [29], to delineate the interactions between hematopoietic stem and their niche cells.
Results
Aged LPCs can compete with their young counterparts in a cKCT setting, although they cannot do so in a conventional BMT setting
It is well known that co-transplantation of young and old BM progenitor cells into a young WT host in conventional BMT, old BM progenitor cells could not compete with their young counterparts in generating T-lineage cells, no matter whether the hosts are irradiated [10] or non-irradiated [12]. The conventional BMT has an in vitro manipulation process of BM progenitors. Evidence showed that when aged BM progenitors stay in vitro, they display decreased adhesion to stroma [31]. We asked whether aged BM progenitors recruited in vivo by a grafted fetal thymus are able to compete with their young counterparts, because when we examined in vivo repopulation in the fetal thymus with aged LPCs over 1 to 4 weeks in a KCT model and in a time-course manner [1, 34], we found that LPCs from old BM were able to generate the same number of thymocytes (Fig. 2A) with similar differentiation profiles as young BM progenitor did (Fig. 2B). The longer the progenitors remained in the fetal microenvironment, the less distinction was observed between the young and old groups as defined by CD4 vs. CD8 differentiation profiles (Fig. 2B). These results were independent of whether cells matured in dGUO-treated or intact fetal thymus at any of the sampled time points. This encourages us to develop a novel in vivo competitive method based on KCT.
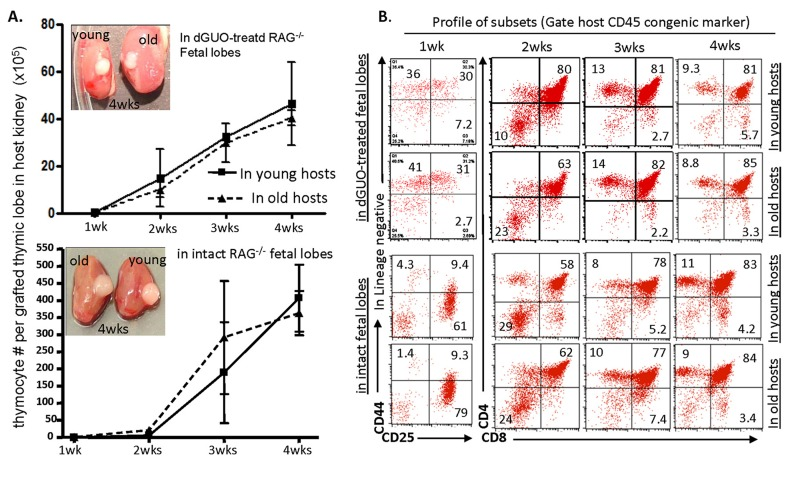
Figure 2. Competence of thymic T-lymphopoiesis from aged- and young-LPCs in repopulation of grafted fetal thymic lobes in vivo in a time course manner (A) Thymocyte number in dGUO-treated (top panel) or intact (bottom panel) grafted fetal thymic lobes, at various weeks after transplanted under the kidney capsules of young (~2 months) and old (20 - 22 months) WT mice. The images shown are representative results of grafted thymic lobe size in the hosts? kidney capsules. Each data point (triangle or square) represents 2-3 host mice. (B) A representative result shows differentiation profiles (CD44 vs. CD25 at one-week time point, and CD4 vs. CD8 at all other longer time points) of thymocytes from grafted fetal thymic lobes under the young and old kidney capsules.
We, therefore, developed a cross-KCT (cKCT) model (Supplemental Fig. S2) by transplanting dGUO-treated CD45.1+CD45.2+ RAG−/− fetal thymic lobes, which are a good in vivo model to accumulate WT LPCs [1, 34], into young CD45.1+ or old CD45.2+ mice (first hosts). Five days after the first host KCT, cKCT was performed by transplanting the fetal thymic lobes carrying seeded LPCs from the first hosts into age-reversed and CD45-subtype-reverseed second hosts to recruit LPCs from the second hosts for additional 5 days. The lobes containing “young and old” or “old and young” LPCs were then subjected to FTOC for one week. The later seeded LPCs from the second hosts of either young or old mice became a dominant population in the lobes after FTOC (Fig. 3A, open triangles and open circles). The KCT young LPCs were then compared with KCT old LPCs (both from the first hosts), while the cKCT young LPCs were compared with the cKCT old LPCs (both from the second hosts). In this in vivo competitive experiment we found no significant difference in T-lineage cell numbers generated (Fig. 3A) and in the differentiation of CD4+ and CD8+ single positive T cells (Fig. 3B) derived from young and old LPCs in the fetal thymic lobes. Taken together, our results indicate that the old LPCs did not lose their competence for T-cell generation and were equal in this respect with young LPCs when allowed to naturally seed the thymus but were not subjected to in vitro manipulation.
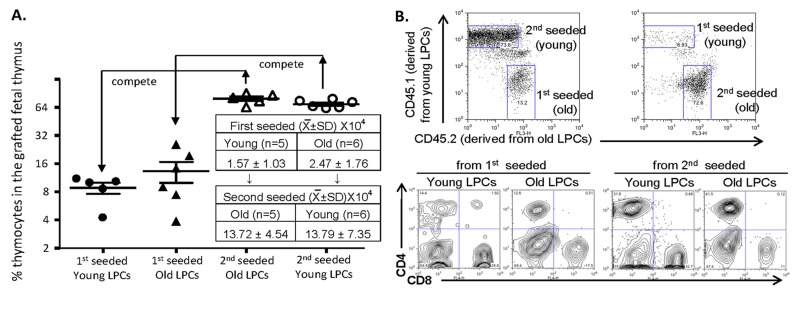
Figure 3. Comparison of competence for thymic T-lymphopoiesis from aged- and young-LPCs’ in competitive repopulation of grafted fetal thymus (A) Results of competitive repopulation of grafted fetal thymus by “young and old” or “old and young” natural thymus-seeding cells from the first and second hosts. Left panel shows % T-lineage thymocytes (gated on DP, CD4+ and CD8+ SPs) in the grafted thymus naturally seeded by young (~2 month old, circles) and old (22 months old, triangles) BM progenitors in the different seeding orders (initial seeding: filled circles or triangles; subsequent/second seeding: open circles or triangles). Each triangle or circle represents one animal. An unpaired Student's t-test shows p > 0.05 (no significant). The table in the A panel shows absolute cell numbers per grafted lobe (each host mouse was grafted with 2-3 fetal thymic lobes, n = animal number). (B) A representative result of differentiated CD4+ and CD8+ T cells from the grafted fetal thymic lobes (bottom panels). The thymocytes are derived from first and second seeded young- and old-BM progenitors, identified by CD45.1 and CD45.2 congenic markers (top panels).
Aged BM progenitors, recruited by grafted fetal thymic lobes in vivo, compete with young BM progenitors even subjected subsequently to in vitro culture
We were curious if natural thymus-seeding aged BM progenitors, accumulated in vivo during fetal thymus KCT, can compete in T-cell generation with their young counterparts in an in vitro culture model. To test this, we designed and performed competitive cultures on an OP9-DL1 stromal cell monolayer (workflow in Fig. 4A). After young CD45.1+ and old CD45.2+ natural thymus-seeding cells were recruited in vivo from dGUO-treated CD45.1+CD45.2+ RAG−/− fetal thymic lobes in KCT of young CD45.1+ and old CD45.2+ host mice for 7 days, we selected CD4−8− DN population from the lobes, and loaded the DN cells at equal numbers on the same monolyayer co-culture for 10 days. The results showed that LPCs from young- and old-sources had the capacity to generate equal numbers and proportions of T-lineage cells (Figs. 4B and 4C) and an equal % of CD4+CD8+ double positive (DP) thymocytes (Figs. 4D and 4E). It suggests two possible scenarios: 1) Aged BM contains mixed population of defective and functional progenitors, and only the functional progenitors can naturally seed the fetal thymus. Therefore they can compete with young LPCs; 2) The competence of T-cell generation from the natural thymus-seeding old BM progenitors can be recovered to the young progenitor levels after the exposure to the fetal thymic microenvironment for a certain period of time.
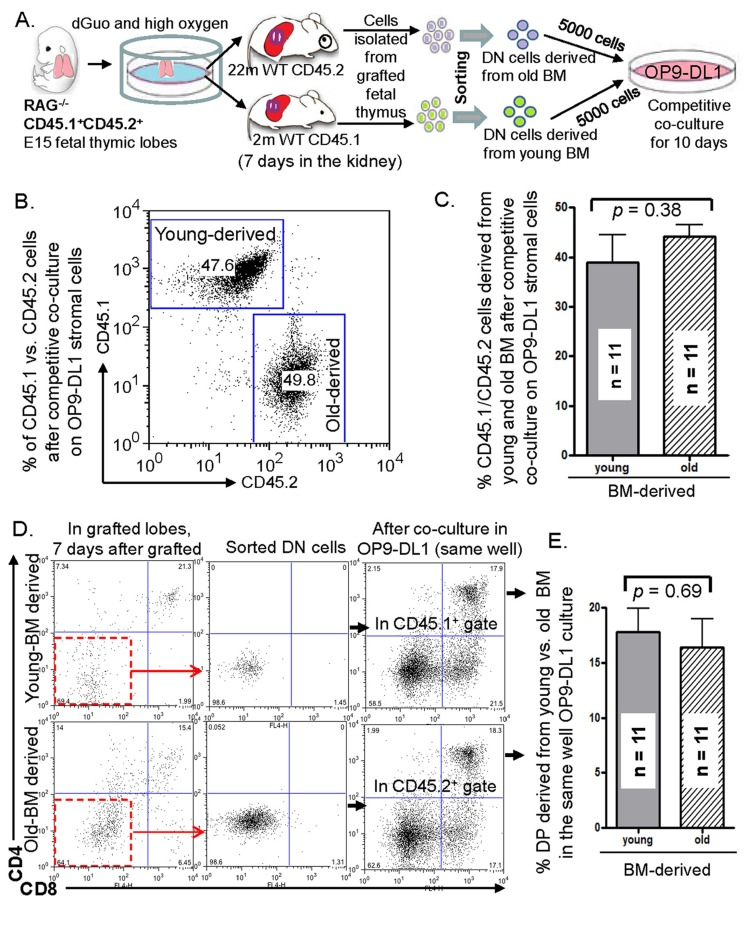
Figure 4. In vitro competition between aged and young LPCs accumulated by grafted fetal thymic lobes in vivo under old or young mouse kidney capsules (A) Schematic workflow of the comprehensive competitive culture assay showing the recruitment of old and young natural thymus-seeding cells in vivo to the in vitro competitive co-culture on OP9-DL1 monolayer stromal cells. (B) A representative result of % T-lineage cells derived from old (CD45.2+) and young (CD45.1+) thymus-seeding LPCs after competitive co-culture on OP9-DL1 stromal cell monolayer. (C) A summary of % T-lineage cells derived from old (CD45.2+) and young (CD45.1+) thymus-seeding LPCs after competitive co-culture on OP9-DL1 stromal cells. (D) A representative flow cytometry dot-plot shows CD4 vs. CD8 profile of T-lineage cells from the grafted fetal thymic lobes 7 days after KCT (left panels); purification of DN cells after negative-selection with beads (middle panels); and CD4 vs. CD8 profile of T-lineage cells after competitive co-culture on OP9-DL1 stromal cells (right panels). (E) A summary of % CD4+CD8+ (DP) cells derived from old (CD45.2+) and young (CD45.1+) thymus-seeding LPCs after competitive co-culture on OP9-DL1 stromal cells. Data in C and E panels show mean ± SEM, n = competitive co-culture wells; total host animal number is 5 young and 5 old WT mice. The experiment was conducted 5 times (i.e. 5 FTOC, 5 sorts, and 5 cultures).
Discussion
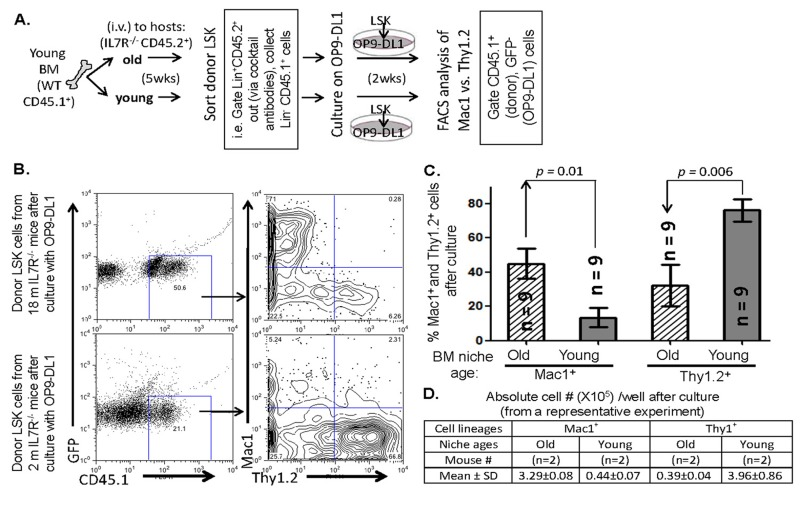
In the BM and thymus, lympho-hematopoietic and non-hematopoietic (stem-cell niche or stromal) cellular compartments tightly regulate competence for T-cell generation, while aging clearly induces defects in this competence [36]. It is still controversial whether the defect originates in the LPC/HSC itself or in the supportive microenvironment. To understand the cellular mechanism of aging in the T-cell generation it is necessary to determine which cellular compartment develops the primary/intrinsic defect. We demonstrate here that this defect originates primarily from the endogenous microenvironment, as evidenced by the fact that the aged BM niches could induce the same defects in BM progenitors from young mice (Fig. 1). Our findings also show that in mice, LPCs seem to lack an intrinsic defect prior to 20-22 months of age, (comparable to ~80-year-old humans), since they were able to repopulate the dGUO-treated and intact fetal thymic lobes in vivo over either short (1 week) or long (4 weeks) time period, comparable to LPCs from young mice (Fig. 2). Natural thymus-seeding LPCs from aged and young mouse BM, acquired in vivo by a grafted fetal thymic lobe, compete well each other to generate T-lineage cells in a common young microenvironment (Fig. 3). These natural thymus-seeding LPCs from aged BM were also able to compete with corresponding young LPCs even in subsequent in vitro culture (Fig. 4). These findings do not imply that LPCs do not age. In fact LPCs progressively lost their capacity to generate T-lineage cells with increasing age as demonstrated in a BMT setting, but they out-competed the ability of their niche cells to generate T-lineage with the passage of time (Fig. 5).
The mechanism by which young BM progenitor cells in the old BM-niches differentiate along the same pathway as old progenitor cells to make a myeloid skew (Fig. 1) is not clear. It is possible related to levels of reactive oxygen species (ROS) produced by BM niches since HSCs from a high oxygen milieu showed myeloid-skewed differentiation [37]. However, the central role of osteoblast-rich niches in this differentiation has been defined. This similar observation was reported [38], in which loss of osteoblasts in a gene knockout mouse model increased myelopoiesis. Another possible mechanism is hyperactivation of the mammalian target of rapamycin (mTOR) during aging [39, 40]. Although over-activation of mTOR in aging was found in stem cells [40], it is possible due to defects in stem cell regulation by the niche. A recently publication demonstrated the mechanism of calorie restriction (CR) on mammalian intestinal stem cells (ISC) during aging, in which CR, similar to Rapamycin administration, inhibits activity of mechanistic target of rapamycin complex 1 (mTORC1) in Paneth cells, a key constitute of the ISC niche. It results in promoting a more favorable stem-cell microenvironment, and then the changed microenvironment normalizes the stem cells [41].
Interestingly, when old BM progenitor cells were introduced into young mice, the young BM-niche did not rejuvenate differentiation profile of the old progenitor cells along the same pathway as young LPCs [23]. However, when old LPCs were recruited into young thymic niches in vivo (within KCT environment), the old LPCs were then directed to differentiate along the same differentiation pathway as young LPCs (Fig. 2). We believe this may be accounted for in two ways. 1) The BM of aged mice may contain both defective as well as functional hematopoietic progenitors, but only the functional progenitors are able to colonize the thymus. Thus, although aged BM has a reduced functional stem cell pool, there is still sufficient population of individual normal functional LPCs on a per cell basis [21] in aged mice. Because the thymic niche is gated [42] and number of the LPC recruitment is limited [an estimated ~10-100 new progenitor cells enter the young adult mouse thymus per day [43, 44]], the normal functional LPCs from aged BM seeding fetal thymus in vivo should be sufficient to provide the daily thymus recruitment requirement. Although the young functional BM stem cell pool is larger than the aged one, the total number of thymus-seeding LPCs from both pools should be the same at any given time. Therefore, grafted fetal thymic lobe-recruited LPCs from both aged and young mice are similar and produce the same number of T-lineage cells.
Nevertheless, when in vitro isolated aged LPCs were introduced into the young BM microenvironment via conventional BMT, and allowed to co-exist with young LPCs [10, 12, 45, 46], the aged LPCs are unable to compete, while in a cKCT microenvironment they are able to compete with young LPCs. This finding may be accounted for by 2) the reduced transplant efficacy of aged BM progenitors. For example, when removing aged BM progenitors from osteoblastic niches, they showed decreased adhesion to stroma [31]. In vitro manipulation of HSCs has been suggested to undergo replicative stress [47] or reduction of transplant efficiency [30]. Therefore, the defect in LPCs is only encountered with transplanted aged BM cells in conventional BMT [12], while this is not observed in physiological thymus-seeding aged BM cells (Figs. 2, 3, and 4) [1, 34]. Thus aged BM progenitors are not likely to have an intrinsic defect, but possess the stress from aged microenvironment, since it can be avoided by the in vivo collection (Figs. 2, 3, and 4).
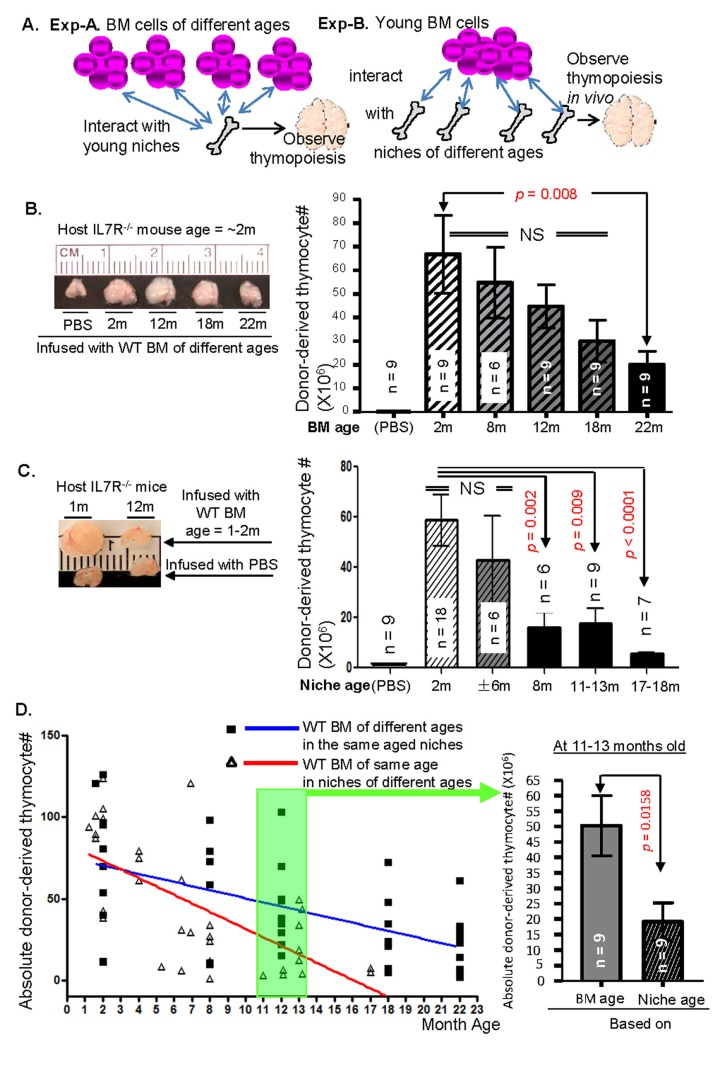
Although LPCs/HSCs are not exempt from aging [48, 49], their aging occurs at a much slower rate than the aging of their niche cells, as measured by comparing the competence of thymic T-lymphopoiesis in a time-course manner based on BM progenitor ages or niche ages in a IL7R−/− host BMT microenvironment (Fig. 5). Cells of both hematopoietic and non-hematopoietic origin undergo age-related deterioration from replicative stress and epigenetic changes [50] that influence DNA integrity [48], or exhaustion of their stem cell pools. However, during the process of natural aging, LPCs/HSCs outlive their niche cells, as evidenced by the onset in reduction in thymic T-lymphopoiesis of aged BM progenitors when repopulating the non-irradiated young IL7R−/− thymus (at the age of 22 months). Concurrently, the niches lose their ability to support the production of functional T cells from the repopulating young BM progenitors in the same setting by “middle age” (~12 months of age in the mice).
In conclusion, the experiments in this study employed comprehensive in vivo and in vitro models to answer several pertinent questions. Which cellular component, HSC itself or HSC niche cell, determines HSCs to take age-related myeloid-skew developmental profile? Can the aged BM-niche influence competence of young BM progenitors to generate T-lineage cells by providing signals that are different from those provided by young niche cells? Why aged LPCs, derived from isolated BM progenitors, cannot compete with their young counterparts in a conventional BMT environment, while they can do so using in vivo collected natural thymus-seeding LPCs? Which cell type (LPCs or niche cells) primarily develops age-related inability in T-cell development in a time-course manner? We have answered these questions by showing clear evidence that the dominant and primary defect arising from aging of T-lymphopoiesis lies in a dysfunction of the niche cells [51] rather than in the T-cell progenitor pools. Further exploration of these issues will provide the foundation for gene-, pharmaceutical-, and stem-cell-based therapies by focusing on the right cellular targets and optimizing the timing for rejuvenation of reduced T-lymphopoiesis in order to treat aging-related onset of T-lymphocyte deficiency.
Methods
Murine models and animal care
Mice from the C57BL/6 genetic background that expressed CD45.2+ or CD45.1+ congenic markers on the hematopoietic cell surface were used. Young (±2-month), early middle-aged (6~9-month), middle-aged (±12-month) and aged (over 18-month) (ages indicated in each figure) wild-type (WT), IL7R−/− mice [26], and RAG−/− fetal mice were originally purchased from the National Cancer Institute, National Institute for Aging /National Institutes of Health (NIH) (Bethesda, MD), and Jackson Laboratory (Bar Harbor, ME), respectively, and bred in our SPF animal facility. E15 indicates fetal mouse at embryonic day-15. All animal experiments were in compliance with the protocols approved by the Institutional Animal Care and Use Committee of the University of Texas Health Science Center at Tyler and the University of North Texas Health Science Center at Fort Worth, in accordance with guidelines of the NIH, USA.
Comparison of myeloid vs. lymphoid fates in age-associated BM microenvironments
Erythrocyte-depleted BM cells (RBC lysing buffer from Sigma), from a pool of young CD45.1+ WT donor mice, were retro-orbitally transplanted (BMT) into 2- and 18-month-old CD45.2+ IL7R−/− host mice (2 × 106 cells/mouse). The donor LPCs [donor CD45.1+, lineage-negative, host CD45.2-negative, ckit+, andSca1+ BM population, i.e. LSK cells [52]] were sorted (FACS-Aria, Supplemental Fig. S1) from the young or old CD45.2+ IL7R−/− hosts, 5 weeks after the BMT. These cells were cultured on an 80% confluent OP9-DL1 (kindly provided by Dr. J.C. Zuniga-Pflucker)[33] stromal cell monolayer (~2000 sorted donor LSK cells per culture well), in presence of Flt3 ligand (5 ng/ml) and IL-7 (5 ng/ml) (R&D). On the culture day 7, non-adherent cells were transferred to a fresh OP9-DL1 monolayer, and after 14 days in culture the cells were harvested and analyzed via flow cytometry for the differentiation of Mac1+ and Thy1.2+ cells derived from these sorted LSK cells (workflow in Fig. 1A).
Kidney capsule transplantation (KCT)
Single-host KCT was performed as previously described [1] with the following modifications: (i) In group one the grafted fetal thymic lobes prior to KCT were treated with dGUO (2’-Deoxyguanosine, Sigma) for 5 days in fetal thymic organ culture (FTOC) for depleting hematopoietic cells in the lobes, while in group two the grafted fetal thymic lobes were intact prior to KCT; (ii) Thymocyte numbers and thymocyte differentiation profiles in the grafted fetal thymic lobes were monitored over time (1, 2, 3, and 4 weeks after the KCT). For cross KCT (cKCT), RAG−/− mouse (CD45.1+CD45.2+ double congenic marker positive) E15 fetal thymic lobes from same littermate were subjected to KCT into first hosts of young (~2-month) CD45.1+ or aged (~22-month) CD45.2+ WT mice for 5 days. And then, cKCT was performed by transplanting the fetal thymic lobes carrying seeded LPCs from the first hosts into age-reversed and CD45-subtype-reversed second hosts to recruit natural thymus-seeding LPCs from the second hosts for additional 5 days. The thymic lobes contain young and aged (group-1) or aged and young (group-2) LPCs were harvested and cultured in vitro in 95% oxygen FTOC system [53] for ~7 days to expand the recruited LPCs before flow cytometric analysis (detailed workflow is shown in supplemental Fig. S2).
Competitive reconstitution of natural thymus-seeding young and aged LPCs through an in vivo recruitment
The dGUO-treated fetal thymic lobes (E15 CD45.1+CD45.2+RAG−/−) were transplanted under the kidney capsules of young (~2-month) WT (CD45.1+) and aged (22-month) WT (CD45.2+) host mice, respectively, to recruit natural thymus-seeding LPCs over a 7-day period. The CD4 and CD8 double negative (DN, with CD3−) thymocytes from the grafted thymic lobe were then sorted by negative selection using magnetic beads (IMagTM streptavidin particles, BD bioscience). The sorted cells (1:1 ratio of young and aged sources ~5000 cells each) were loaded on 80% confluent monolayers of OP9-DL1 stromal cells [33]in 12-well plates and culture in the presence of exogenously supplied IL-7 (5ng/ml) and Flt3 ligand (5ng/ml) (R&D). On the 5th day of the culture non-adherent cells were transferred to a fresh OP9-DL1 monolayers, and ten days after the co-culture, the cells were isolated and analyzed by flow cytometry for CD45.1 vs. CD45.2, and CD4 vs. CD8 cell populations (workflow in Fig. 4A).
Competence of BM progenitors or their niche cells of different ages to generate T cells
Experiment-A: to test the competence for thymic T-lymphopoiesis by BM progenitor cells from various ages, BM cells were obtained from CD45.2+ WT mice of ~2-, 8-, 12-, 18-, and 22-months of ages. About 2 × 106 erythrocyte-depleted BM cells were retro-orbitally injected into non-irradiated CD45.1+ young IL7R−/− host mice to reconstitute the donor T-cell pool in the host thymus. Experiment-B: to test competence for thymic T-lymphopoiesis in LPC niches (both BM and thymic niches) of different ages, ~2 × 106 erythrocyte-depleted BM cells from a pool of young CD45.1+ WT mice were retro-orbitally injected into ~2-, 6-, 8, 12~13-, and 18-month-old non-irradiated CD45.2+ IL7R−/− host mice. Five weeks after the injection, the donor-derived thymic T-lymphopoiesis was analyzed by flow cytometry (workflow in Fig. 5A).
Flow cytometric assay
Cell counting was carried out using the BioRad TC10TM automated cell counter or a hemacytometer. For flow cytometric analysis, thymocytes isolated with a cell-strainer were blocked with anti-FcR (clone 2.4G2) to diminish nonspecific binding, and then stained with combinations of fluorochrome-conjugated antibodies (as indicated in each figure). Data were acquired by a dual-laserFACS-Calibur system and analyzed with CellQuest and FlowJo software.
Statistics
Statistical significance was analyzed by unpaired Student's t-test with Welch's correction (if variances shown unequal) or Mann-Whitney test (if data shown non-parameter). Linear regression is used to fit straight lines for the two groups. By using a dummy variable to fit two regression lines in a single equation, the equality of the two slopes is tested based on a t-test. Differences were considered statistically significant at values of p < 0.05.
Supplementary Materials
Acknowledgments
This work was supported by NIA/NIH (R21AG031880) and NIAID/NIH (R01AI081995) grants to D-M.S. The authors have no competing financial interests.
We greatly thank Dr. J.C. Zuniga-Pflucker (University of Toronto) for kindly providing the OP9-DL1 stromal cell line; Dr. H.T. Petrie (Scripps Florida) for critical suggestions on the initial experimental design; and Drs. Dan Dimitrijevich (UNT-HSC at Ft.W.) for carefully reading the manuscript, as well as Drs. Jian Fu and Zhuang Wu (UTHSCT) for assisting in our transition.
Conflicts of Interest
The authors of this manuscript have no conflict of interests to declare.
References
- 1. Zhu X, Gui J, Dohkan J, Cheng L, Barnes PF, Su DM. Lymphohematopoietic progenitors do not have a synchronized defect with age-related thymic involution. Aging Cell. 2007; 6: 663 -672. [PubMed] .
- 2. Lo Celso C and Scadden DT. The haematopoietic stem cell niche at a glance. J Cell Sci. 2011; 124: 3529 -3535. [PubMed] .
- 3. Morrison SJ, Wandycz AM, Akashi K, Globerson A, Weissman IL. The aging of hematopoietic stem cells. Nat Med. 1996; 2: 1011 -1016. [PubMed] .
- 4. Zhang J and Li L. Stem cell niche: microenvironment and beyond. J Biol Chem. 2008; 283: 9499 -9503. [PubMed] .
- 5. Dorshkind K and Swain S. Age-associated declines in immune system development and function: causes, consequences, and reversal. Curr Opin Immunol. 2009; 21: 404 -407. [PubMed] .
- 6. Sun L, Guo J, Brown R, Amagai T, Zhao Y, Su DM. Declining expression of a single epithelial cell-autonomous gene accelerates age-related thymic involution. Aging Cell. 2010; 9: 347 -357. [PubMed] .
- 7. Chinn IK, Blackburn CC, Manley NR, Sempowski GD. Changes in primary lymphoid organs with aging. Semin Immunol. 2012; .
- 8. Tuljapurkar SR, McGuire TR, Brusnahan SK, Jackson JD, Garvin KL, Kessinger MA, Lane JT, BJ OK, Sharp JG. Changes in human bone marrow fat content associated with changes in hematopoietic stem cell numbers and cytokine levels with aging. J Anat. 2011; 219: 574 -581. [PubMed] .
- 9. Kuranda K, Vargaftig J, de la Rochere P, Dosquet C, Charron D, Bardin F, Tonnelle C, Bonnet D, Goodhardt M. Age-related changes in human hematopoietic stem/progenitor cells. Aging Cell. 2011; 10: 542 -546. [PubMed] .
- 10. Sudo K, Ema H, Morita Y, Nakauchi H. Age-associated characteristics of murine hematopoietic stem cells. J Exp Med. 2000; 192: 1273 -1280. [PubMed] .
- 11. Waterstrat A and Van Zant G. Effects of aging on hematopoietic stem and progenitor cells. Curr Opin Immunol. 2009; 21: 408 -413. [PubMed] .
- 12. Zediak VP, Maillard I, Bhandoola A. Multiple prethymic defects underlie age-related loss of T progenitor competence. Blood. 2007; 110: 1161 -1167. [PubMed] .
- 13. Niikura Y, Niikura T, Tilly JL. Aged mouse ovaries possess rare premeiotic germ cells that can generate oocytes following transplantation into a young host environment. Aging (Albany NY). 2009; 1: 971 -978. [PubMed] .
- 14. Pan L, Chen S, Weng C, Call G, Zhu D, Tang H, Zhang N, Xie T. Stem cell aging is controlled both intrinsically and extrinsically in the Drosophila ovary. Cell Stem Cell. 2007; 1: 458 -469. [PubMed] .
- 15. Massasa E, Costa XS, Taylor HS. Failure of the stem cell niche rather than loss of oocyte stem cells in the aging ovary. Aging (Albany NY). 2010; 2: 1 -2. [PubMed] .
- 16. Boyle M, Wong C, Rocha M, Jones DL. Decline in self-renewal factors contributes to aging of the stem cell niche in the Drosophila testis. Cell Stem Cell. 2007; 1: 470 -478. [PubMed] .
- 17. Ryu BY, Orwig KE, Oatley JM, Avarbock MR, Brinster RL. Effects of aging and niche microenvironment on spermatogonial stem cell self-renewal. Stem Cells. 2006; 24: 1505 -1511. [PubMed] .
- 18. Gopinath SD and Rando TA. Stem cell review series: aging of the skeletal muscle stem cell niche. Aging Cell. 2008; 7: 590 -598. [PubMed] .
- 19. Conboy IM, Conboy MJ, Wagers AJ, Girma ER, Weissman IL, Rando TA. Rejuvenation of aged progenitor cells by exposure to a young systemic environment. Nature. 2005; 433: 760 -764. [PubMed] .
- 20. Rossi DJ, Bryder D, Zahn JM, Ahlenius H, Sonu R, Wagers AJ, Weissman IL. Cell intrinsic alterations underlie hematopoietic stem cell aging. Proc Natl Acad Sci U S A. 2005; 102: 9194 -9199. [PubMed] .
- 21. Wagner W, Horn P, Bork S, Ho AD. Aging of hematopoietic stem cells is regulated by the stem cell niche. Exp Gerontol. 2008; 43: 974 -980. [PubMed] .
- 22. Ergen AV and Goodell MA. Mechanisms of hematopoietic stem cell aging. Exp Gerontol. 2009; 45: 286 -290. [PubMed] .
- 23. Mackall CL, Punt JA, Morgan P, Farr AG, Gress RE. Thymic function in young/old chimeras: substantial thymic T cell regenerative capacity despite irreversible age-associated thymic involution. Eur J Immunol. 1998; 28: 1886 -1893. [PubMed] .
- 24. Bhattacharya D, Rossi DJ, Bryder D, Weissman IL. Purified hematopoietic stem cell engraftment of rare niches corrects severe lymphoid deficiencies without host conditioning. J Exp Med. 2006; 203: 73 -85. [PubMed] .
- 25. Waskow C, Madan V, Bartels S, Costa C, Blasig R, Rodewald HR. Hematopoietic stem cell transplantation without irradiation. Nat Methods. 2009; 6: 267 -269. [PubMed] .
- 26. Peschon JJ, Morrissey PJ, Grabstein KH, Ramsdell FJ, Maraskovsky E, Gliniak BC, Park LS, Ziegler SF, Williams DE, Ware CB, Meyer JD, Davison BL. Early lymphocyte expansion is severely impaired in interleukin 7 receptor-deficient mice. J Exp Med. 1994; 180: 1955 -1960. [PubMed] .
- 27. Prockop SE and Petrie HT. Regulation of thymus size by competition for stromal niches among early T cell progenitors. J Immunol. 2004; 173: 1604 -1611. [PubMed] .
- 28. Min H, Montecino-Rodriguez E, Dorshkind K. Reduction in the developmental potential of intrathymic T cell progenitors with age. J Immunol. 2004; 173: 245 -250. [PubMed] .
- 29. Zuniga-Pflucker JC. T-cell development made simple. Nat Rev Immunol. 2004; 4: 67 -72. [PubMed] .
- 30. Min D, Panoskaltsis-Mortari A, Kuro OM, Hollander GA, Blazar BR, Weinberg KI. Sustained thymopoiesis and improvement in functional immunity induced by exogenous KGF administration in murine models of aging. Blood. 2007; 109: 2529 -2537. [PubMed] .
- 31. Kohler A, Schmithorst V, Filippi MD, Ryan MA, Daria D, Gunzer M, Geiger H. Altered cellular dynamics and endosteal location of aged early hematopoietic progenitor cells revealed by time-lapse intravital imaging in long bones. Blood. 2009; 114: 290 -298. [PubMed] .
- 32. Bhandoola A, von Boehmer H, Petrie HT, Zuniga-Pflucker JC. Commitment and developmental potential of extrathymic and intrathymic T cell precursors: plenty to choose from. Immunity. 2007; 26: 678 -689. [PubMed] .
- 33. Schmitt TM and Zuniga-Pflucker JC. Induction of T cell development from hematopoietic progenitor cells by delta-like-1 in vitro. Immunity. 2002; 17: 749 -756. [PubMed] .
- 34. Gui J, Zhu X, Dohkan J, Cheng L, Barnes PF, Su DM. The aged thymus shows normal recruitment of lymphohematopoietic progenitors but has defects in thymic epithelial cells. Int Immunol. 2007; 19: 1201 -1211. [PubMed] .
- 35. Swain S, Clise-Dwyer K, Haynes L. Homeostasis and the age-associated defect of CD4 T cells. Semin Immunol. 2005; 17: 370 -377. [PubMed] .
- 36. Geiger H and Van Zant G. The aging of lympho-hematopoietic stem cells. Nat Immunol. 2002; 3: 329 -333. [PubMed] .
- 37. Jang YY and Sharkis SJ. A low level of reactive oxygen species selects for primitive hematopoietic stem cells that may reside in the low-oxygenic niche. Blood. 2007; 110: 3056 -3063. [PubMed] .
- 38. Visnjic D, Kalajzic Z, Rowe DW, Katavic V, Lorenzo J, Aguila HL. Hematopoiesis is severely altered in mice with an induced osteoblast deficiency. Blood. 2004; 103: 3258 -3264. [PubMed] .
- 39. Blagosklonny MV. Aging, stem cells, and mammalian target of rapamycin: a prospect of pharmacologic rejuvenation of aging stem cells. Rejuvenation Res. 2008; 11: 801 -808. [PubMed] .
- 40. Chen C, Liu Y, Zheng P. mTOR regulation and therapeutic rejuvenation of aging hematopoietic stem cells. Sci Signal. 2009; 2: ra75 [PubMed] .
- 41. Yilmaz OH, Katajisto P, Lamming DW, Gultekin Y, Bauer-Rowe KE, Sengupta S, Birsoy K, Dursun A, Yilmaz VO, Selig M, Nielsen GP, Mino-Kenudson M, Zukerberg LR, et al. mTORC1 in the Paneth cell niche couples intestinal stem-cell function to calorie intake. Nature. 2012; 486: 490 -495. [PubMed] .
- 42. Foss DL, Donskoy E, Goldschneider I. The importation of hematogenous precursors by the thymus is a gated pheno-menon in normal adult mice. J Exp Med. 2001; 193: 365 -374. [PubMed] .
- 43. Petrie HT. Role of thymic organ structure and stromal composition in steady-state postnatal T-cell production. Immunol Rev. 2002; 189: 8 -19. [PubMed] .
- 44. Shortman K, Egerton M, Spangrude GJ, Scollay R. The generation and fate of thymocytes. Semin Immunol. 1990; 2: 3 -12. [PubMed] .
- 45. Geiger H and Rudolph KL. Aging in the lympho-hematopoietic stem cell compartment. Trends Immunol. 2009; 30: 360 -365. [PubMed] .
- 46. Chambers SM, Shaw CA, Gatza C, Fisk CJ, Donehower LA, Goodell MA. Aging hematopoietic stem cells decline in function and exhibit epigenetic dysregulation. PLoS Biol. 2007; 5: e201 [PubMed] .
- 47. Kamminga LM and de Haan G. Cellular memory and hematopoietic stem cell aging. Stem Cells. 2006; 24: 1143 -1149. [PubMed] .
- 48. Geiger H, Rennebeck G, Van Zant G. Regulation of hematopoietic stem cell aging in vivo by a distinct genetic element. Proc Natl Acad Sci U S A. 2005; 102: 5102 -5107. [PubMed] .
- 49. Marciniak-Czochra A, Stiehl T, Wagner W. Modeling of replicative senescence in hematopoietic development. Aging (Albany NY). 2009; 1: 723 -732. [PubMed] .
- 50. Issa JP. Age-related epigenetic changes and the immune system. Clin Immunol. 2003; 109: 103 -108. [PubMed] .
- 51. Wilson A and Trumpp A. Bone-marrow haematopoietic-stem-cell niches. Nat Rev Immunol. 2006; 6: 93 -106. [PubMed] .
- 52. Schwarz BA and Bhandoola A. Circulating hematopoietic progenitors with T lineage potential. Nat Immunol. 2004; 5: 953 -960. [PubMed] .
- 53. Su DM, Wang J, Lin Q, Cooper MD, Watanabe T. Interferons alpha/beta inhibit IL-7-induced proliferation of CD4- CD8- CD3- CD44+ CD25+ thymocytes, but do not inhibit that of CD4- CD8- CD3- CD44- CD25- thymocytes. Immunology. 1997; 90: 543 -549. [PubMed] .