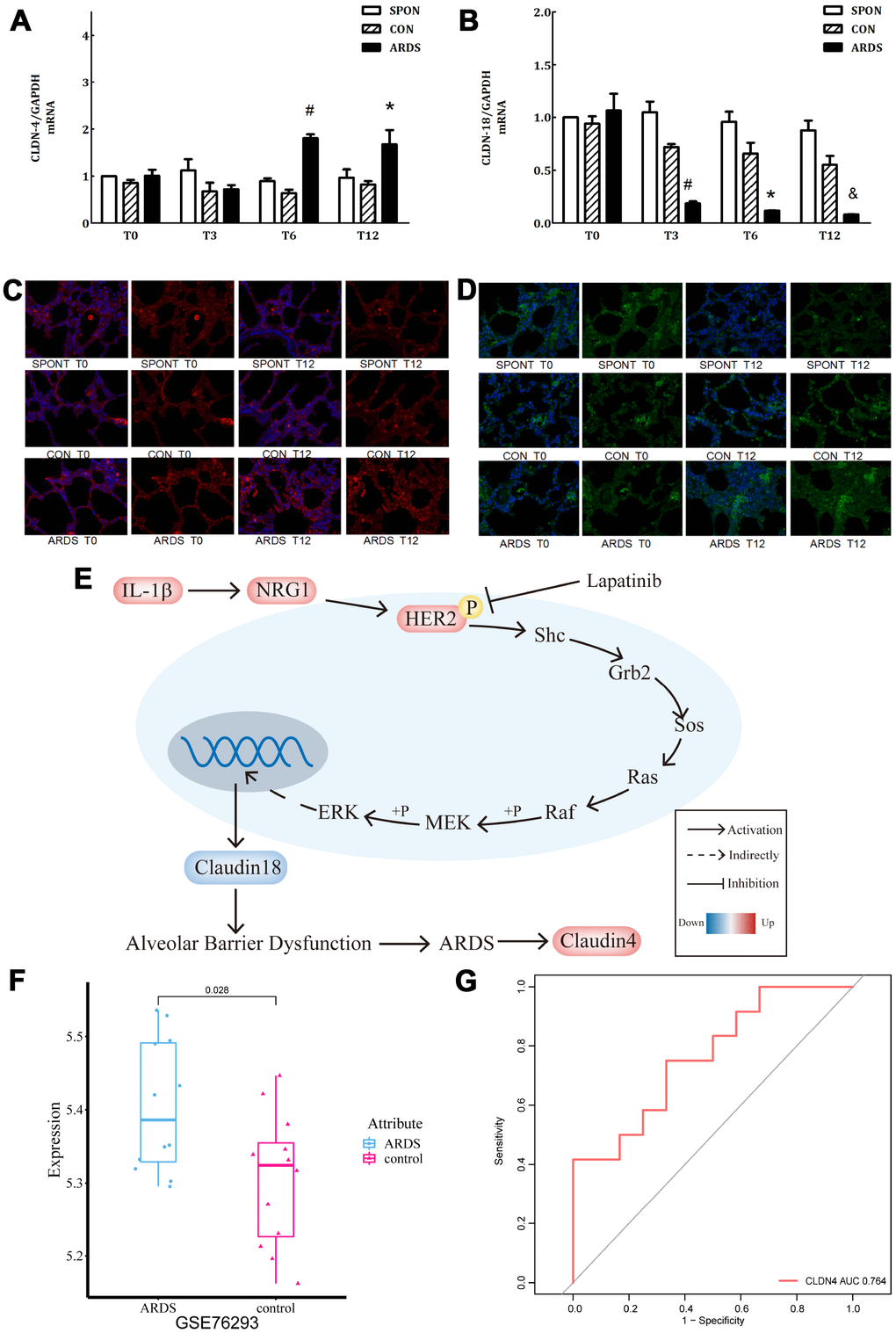
Figure 5.Further verification of claudin4/18 expression and localization in the ARDS model. (A) Comparison of expression levels of claudin4, claudin4 expression significantly increased at 8 hours and 12 hours in ARDS. (B) Claudin18 expression in lung tissue decreased significantly at 3 hours, 6 hours, and 12 hours of ARDS. (C). Three groups of 0, 3, 6 and 12 hours of lung tissue. Claudin4 immunofluorescence, purple fluorescence is the nucleus, red fluorescence is claudin4 protein, and the fluorescence and fluorescence intensity around the cells are compared. The fluorescence around the cells in the ARDS group was significantly stronger than that in the other groups at 12 hours. The expression of claudin4 protein was increased during the induction of lung tissue in the ARDS model. (D) The lung tissues of three groups at 0, 3, 6 and 12 hours showed immunofluorescence. The nucleus was stained with blue fluorescence and the protein was stained with red fluorescence. The 12-hour peripheral fluorescence of ARDS group was significantly weaker than that of other groups. During the induction of ARDS model, the expression of lung tissue decreased and the location of protein on cell membrane decreased; (E) IL-1β activated HER2 signaling pathway by up-regulating NRG1, which regulates the expression of claudin18 protein, promoting the development of ARDS. Furthermore, in turn, this would initiate a compensatory protective mechanism, promoting the expression of claudin4, strengthening the repair of lung barrier function. The series of reactions would be reversed by the addition of lapatinib, a HER2 blocker. (F) Claudin4 was up-regulated in ARDS compared to control in GSE76293. (G) ROC curve results indicated that claudin4 can distinguish ARDS and control in GSE76293.