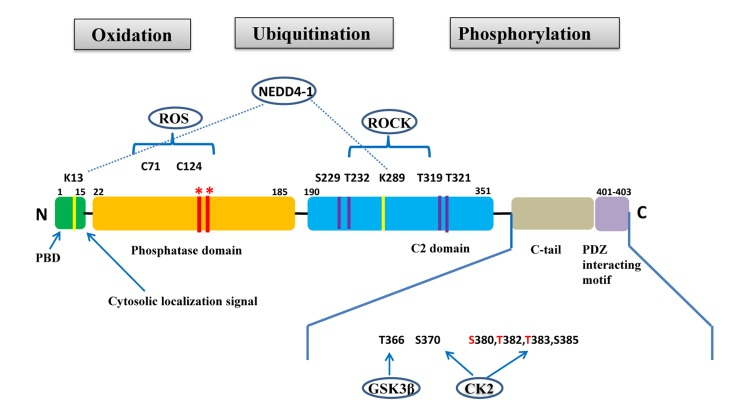
Figure 1.PTEN gene and protein structuresThe PTEN protein is composed of 403 amino acids and contains an N-terminal PIP2-binding domain (PBD), a phosphatase domain, a C2 domain, a C-terminal tail containing proline–glutamic acid–serine–threonine sequences, and a PDZ interacting motif at the end. *Mutations on the phosphatase domain that disrupt PTEN's phosphatase activity include the C124S mutation, which abrogates both the lipid and protein phosphatase activity of PTEN, and the G129E mutation, which abrogates only the lipid phosphatase activity of PTEN. The C-terminal tail residues phosphorylated by glycogen synthase kinase 3β (GSK3β) and casein kinase 2 (CK2) are shown. Mutations of S380, T382, and T383 (referred to as the STT) can destabilize PTEN and increase its phosphatase activity. The PIP2-binding site and adjacent cytoplasmic localization signal are located at the N-terminal. The N-terminal poly-basic region appears to selectively interact with PIP2 and contribute to the nuclear accumulation of PTEN. Ubiquitination of PTEN has also been found on K13 and K289.