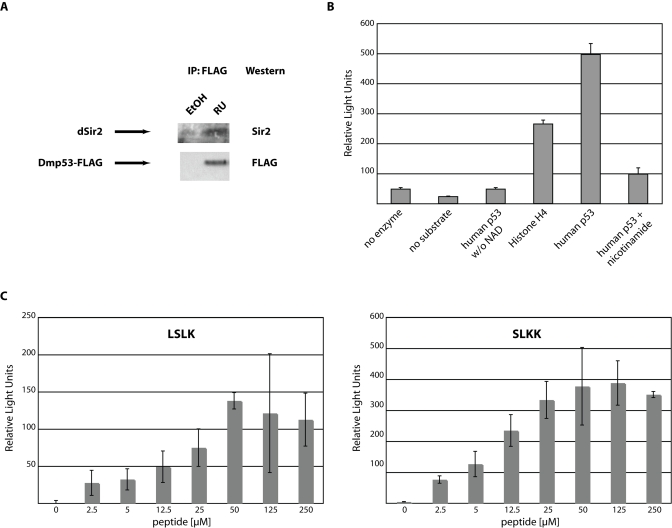
Figure 3.Functional interaction between dSir2 and Dmp53.(A)
Endogenous dSir2 physically interacts with Dmp53. A FLAG-tagged version of wild
type Dmp53 was expressed in females during the first 10 days of adult life using
the ELAV-Switch driver. Head extracts were then immunoprecipitated with
anti-FLAG antibody. Western blot analysis with an antibody against dSir2 shows
efficient co-immunoprecipitation of endogenous dSir2 with the over expressed
wild type Dmp53-FLAG construct. (B) Recombinant dSir2 deacetylates human
substrates. Recombinant purified dSir2 was incubated with the indicated
substrates (5μM) in triplicate and released fluorescence was measured as
Relative Light Units. No deacetylation activity was observed when no NAD was
added or the Sir2 inhibitor nicotinamide was added. Shown is a representative of
at least three independent experiments. (C) Recombinant dSir2 deacetylates
Dmp53-derived peptides. Recombinant purified dSir2 was incubated in triplicate
with the indicated Dmp53-derived peptides. Deacetylation activity is
dose-dependent and reaches saturation at higher substrate concentrations. The
SLKK substrate gets deacetylated with similar efficiency as the human p53
peptide and about twice as efficiently as the LSLK peptide, suggesting substrate
specificity of dSir2. The experiments shown are background corrected for non-NAD
containing reactions. Shown are representatives of at least three independent
experiments.