Introduction
Six in ten adults in the United States have at least one chronic disease and four in ten adults have two or more [1]. These diseases are a major cause of morbidity and mortality and they put a significant burden on the healthcare system as well as the society at large [2]. Aging itself has been identified as a common driver of chronic diseases and an important target for extending human healthspan [3]. It has also been estimated that if we improve our collective healthspan by just one year the calculated savings are worth $38 trillion dollars, and if by 10 years those savings jump to $367 trillion dollars [4]. Biological age clocks, based on DNA methylation marks, have become important surrogate markers to assess the effectiveness of interventions at reducing biological age, with the expectation that biological age reductions will compress morbidity and extend mortality [5, 6].
Modifiable lifestyle factors, including concentrated exposure to dietary “epinutrients”, have been suggested to be able to favorably influence DNA methylation-based clocks and therefore have the potential to compress morbidity and extend mortality [7]. Epinutrients may be defined as dietary nutrients that provide either substrates or cofactors for DNA methylation activity or influence the expression or rate of activity of DNA methylation-related enzymes. Folate and betaine, for example, are cofactors in methylation biosynthetic pathways, alpha ketoglutarate, vitamin C, and vitamin A are ten-eleven translocation (TET) demethylase cofactors and modulators, and curcumin, epigallocatechin gallate (EGCG), rosmarinic acid, quercetin, and luteolin are known polyphenolic modulators of DNA methyl transferase (DMNT) enzymes [8, 9].
The modifiable lifestyle intervention used by participants in this case series was first investigated in a pilot clinical trial in which participants (all men between the ages of 50-72 years) reduced their biological age by an average of 3.23 years as compared to controls [7]. The case series reported on herein was conducted to further the investigation of a modifiable lifestyle intervention that was largely the same in other populations; importantly in women.
Results
Participants’ biological age change
Five of the six participants that followed the program saw a reduction in biological age, while one participant saw no change (Table 1) [7]. Of those six, the maximum reduction in biological age was 11.01 years, while the minimum reduction in biological age was 1.22 years, with a mean biological age reduction of 4.6 years.
Table 1. Participants’ biological age at baseline and endpoint.
| Participant | Chronological age | BioAgebaseline | BioAgeendpoint | Change |
| 1 | 62 | 57.33 | 46.32 | -11.01 |
| 2 | 56 | 53.85 | 49.78 | -4.07 |
| 3 | 56 | 52.79 | 45.39 | -7.40 |
| 4 | 65 | 61.63 | 60.41 | -1.22 |
| 5 | 62 | 60.30 | 60.30 | 0 |
| 6 | 46 | 49.09 | 45.15 | -3.94 |
A paired sample t-test was conducted to compare the biological age of the participants at baseline (BioAgebaseline) and the biological age of the participants at the end of week 8 (BioAgeendpoint). The results show a significant difference between mean baseline biological age (mbaseline = 51.23) and mean endpoint biological age (mendpoint = 55.83) (t-statistic = 2.783, p-value = 0.039) (Figure 1). Given the small sample size, we conducted a Shapiro-Wilk test of normality and found that participants’ biological age at baseline (Shapiro-Wilk Statistic = .959, p = .814) is normally distributed. However, biological age at endpoint is not (Shapiro-Wilk Statistic = .775, p = .035). Therefore, we performed a non-parametric Wilcoxon signed-rank test to compare the median biological age at baseline to the media biological age at endpoint. The Wilcoxon signed-rank test showed that the diet and lifestyle intervention elicited a significant change in the biological age of participants (Z = -2.023; p = .043) with the median biological age at baseline = 55.49 being significantly higher than the median biological age at the end of the 8-week intervention = 48.1 (Figure 1).
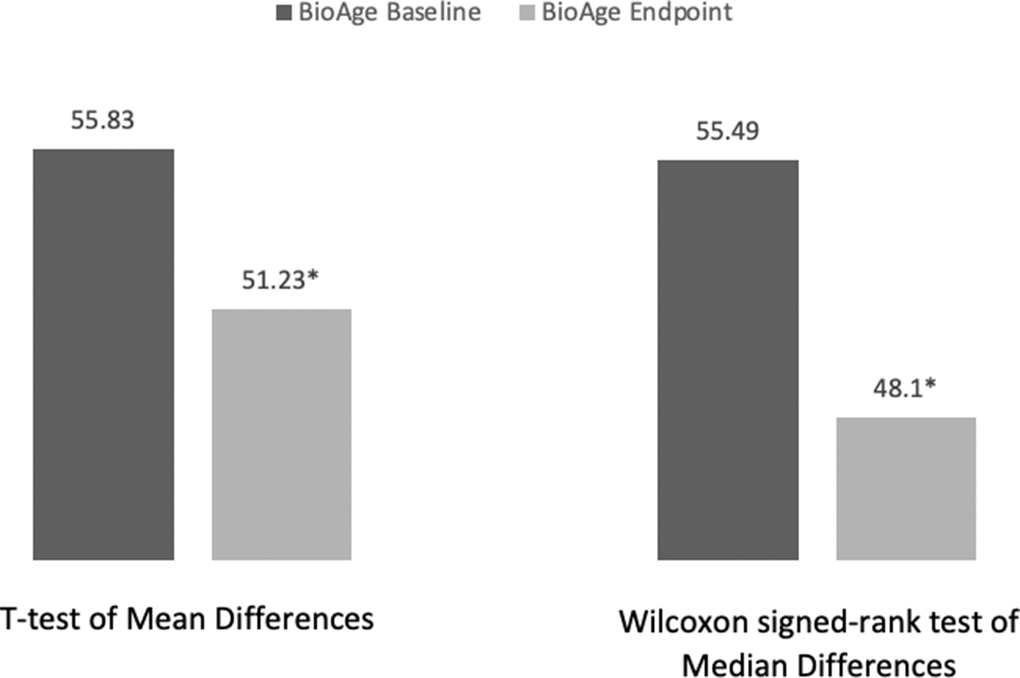
Figure 1. Participants’ average biological age change analysis. *Significant difference with p-value < .05.
Participant adherence to the program
For the diet and lifestyle goals listed in Table 2, participants checked a box to “Mark as done” if they met the listed goal. Over the 8 weeks of the study, data reporting was relatively high with an average of 6.44 days reported per week across all six participants. Adherence for each goal was calculated based on the percentage of days reported that a participant checked a goal. Average individual adherence across all goals varied between 71.2% and 97.2%, with an average of 81.7%. Average adherence according to week had less variance, with the lowest adherence average across participants observed for week 8 (76.0%) and highest such adherence observed at week 6 (86.7%). For the six participants, average individual adherence to diet and supplements goals (84.4%) seems to have been easier than adherence to lifestyle goals (77.0%), with the breathing exercise being most challenging (average adherence-breathing exercise = 57.5%) and intermittent fasting being least challenging (average adherence-intermittent fasting = 93.7%).
Table 2. Summary of dietary and lifestyle interventions delivered through the app.
| “Today’s foods” | “Today’s supplements” |
|
|
| “Healthy habits daily recap” | “Water goal” |
| 8 cups per day |
Discussion
Significance of biological age change
The findings of this case series of 6 women following the methylation-supportive diet and lifestyle program include changes to DNA methylation patterns leading to an average reversal of epigenetic biological age of 4.60 years with a statistically significant difference between baseline and endpoint biological age (p=.039). The original pilot study of a dietary and lifestyle program that was largely the same as this also resulted in an average reversal of biological age of 3.23 years as compared to controls (p=.018), accounting for any normal variability in epigenetic methylation [7]. Both studies used the same Horvath DNAmAge clock (2013) to measure biological age at baseline and the end of the intervention period. Participants in this case series were all women, between the ages of 45 and 65, unlike the original pilot cohort which were men between the ages of 50-72. Taken together, these data suggest that a methylation-supportive diet and lifestyle intervention may favorably influence biological age in both sexes during middle age and older.
Participant adherence to the intervention
As suggested by analysis of adherence data in this case series, 100% adherence to the program is not required to achieve positive outcomes, since the improvements in biological age were observed with goals-based adherence of between 71.2% and 97.2% (average 81.7%). We attribute the relatively high participant adherence in this case series in part to the nutrition coaching support provided. The level of adherence is notable given participants engaged in the program during the months of November through January, a time when many Americans celebrate food-centric holidays, and adherence to dietary programs can be more challenging.
Significance of studying healthy individuals
Studying individuals who are considered generally healthy at baseline, as is the case in this case series as well as the original pilot study, may have removed potential confounding from disease-accelerated biological aging and instead limited the investigation to the influence of the intervention on aging itself. It has been documented that those with chronic disease, such as type 2 diabetes, have epigenetic methylation patterns that are consistent with being biologically older than their disease-free counterparts, and that those with a higher biological age who are presently disease-free are at greater risk of developing a chronic disease [10, 11]. In addition, it has been observed in large data sets that positive lifestyle factors as well as effective medical treatment correlate with a reduced biological age in those with type 2 diabetes (Preprint) [11]. The participants in the original pilot study of this intervention were considered to be healthy as defined by an exclusion of diagnosed illness [7]. In this case series, five (out of six) participants may be considered healthy as is suggested by their baseline biological age being less than their chronological age [12].
One participant who did not complete the program
There was a seventh, male participant in this case series who started the intervention, but who dropped out before completing the program due to a reported family emergency. As such he was not eligible to be included in the final analysis. His baseline DNAmAge was 57.59 and chronological age was 71. Even though he did not complete the program, he did obtain his DNAmAge at the eight-week time point, where it had increased to 61.58. Sudden acute acceleration in biological age due to diverse stressful events (which is then reversed following recovery from the event) has been documented (Preprint) [13].
Conclusions
The findings of this case series add to the existing evidence suggesting that widely-accessible, cost-effective dietary and lifestyle interventions, that are designed to support DNA methylation and are widely considered to be safe, may be able to reduce measures of biological aging and have the potential to impact healthspan, lifespan, and the economic burden of aging. This case series of women participants extends the previous pilot study of this intervention in men, indicating that favorable biological age changes may be achievable in both sexes. In addition, the investigation of otherwise-healthy individuals, rather than those with diagnosed disease, suggests an influence directly on underlying mechanisms of aging instead of disease-driven aging.
A significant limitation of this case series is its small cohort size. Other limitations include its use of a biological age assessment based on the first-generation Horvath multi-tissue clock and the lack of a control group that allows for ruling out changes in biological age due to factors outside the intervention. Future research will need to include larger, more diverse cohorts, the use of more advanced clocks and a control group. Such future research should also examine the relationship between diet and lifestyle program adherence and biological age change.
Materials and Methods
Study participants
The six participants included in the final analysis of this study, all female between the ages of 46 and 65, were the first and only individuals to complete the 8-week diet and lifestyle program and obtain comparable baseline and endpoint biological age testing as measured by the Horvath DNAmAge clock. Recruitment of study participants was conducted via advertisement on various websites and social media platforms maintained by Dr. Kara Fitzgerald and participants completed the program between November 2021 and January 2022.
After the 7th case (the individual referenced above who stopped following the program before completion), due to change in laboratory methodology, we were unable to obtain further comparable Horvath DNAmAge data from additional participants. Therefore, within the constraints of comparable data on the Horvath DNAmAge clock at baseline and endpoint, no cases were omitted.
Study design
Participants followed an intervention that included a specific set of dietary recommendations high in known epinutrients. Simple carbohydrates were restricted, and the diet was largely plant centered but included key nutrient-dense animal protein from 5-10 eggs per week, 6 oz of animal protein daily, and three 3-ounce servings of liver per week (or an encapsulated liver supplement). Participants were also asked to eat all food within a 12-hour window each day to incorporate a basic level of intermittent fasting. Different from the original study, participants were encouraged to track their water consumption aiming for 8-cups of water per day.
Dietary supplements consisted of a probiotic containing 40 million CFU of Lactobacillus plantarum 299v. L. plantarum (UltraFlora® Intensive Care, Metagenics Inc. Aliso Viejo, CA, USA) and a fruit and vegetable powder, rich in additional polyphenolic compounds, twice a day (PhytoGanix®, Metagenics Inc. Aliso Viejo, CA, USA).
Lifestyle modifications that participants were asked to incorporate included a minimum of 30 minutes of physical activity at least 5 days a week, at an intensity of 60-80 percent of maximum perceived exertion. All participants were encouraged to get a minimum of seven hours of sleep per night, participate in two, 10-minute breathing sessions per day designed to elicit the relaxation response (a meditation video was provided).
The intervention program was delivered through the beta testing of a HIPAA-compliant digital application [herein the app]. The app provided video and written instruction, daily tracking tools, optional recipes, a shopping list, and reminders for participants to complete. A summary of dietary and lifestyle interventions delivered through the app, displayed as “Today’s Foods”, “Today’s Supplements”, “Water Goal”, and “Healthy Habits Daily Recap” are shown in Table 2. An overview of the intervention is shown in Table 3.
Table 3. Overview of the dietary and lifestyle intervention*.
| Intervention category | Details | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dietary Prescription | Guidance per week: | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
3 servings of liver
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
5-10 eggs
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Guidance per day: | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
2 cups of dark leafy greens
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
2 cups cruciferous vegetables
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 3 additional cups colorful vegetables of your choosing (excluding white potatoes, sweetcorn) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 1-2 medium beet | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 4 tbsp (1/4 cup) pumpkin seeds (or pumpkin seed butter) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 4 tbsp (1/4 cup) sunflower seeds (or sunflower seed butter) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
1+ serving methylation adaptogens, choose from:
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
6 oz animal protein
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 2 servings of low glycemic fruit | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| General guidance: | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Organic preferred over conventional | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stay hydrated | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Don’t eat between 7pm and 7am | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Include “healthy” oils
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Avoid added sugar/candy, dairy, grains, legumes/beans | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Minimize plastic food containers | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Supplement Prescription | PhytoGanix®, a combination of organic vegetables, fruits, seeds, herbs, plant enzymes, prebiotics and probiotics at a dose of 2 servings daily, divided UltraFlora® Intensive Care, containing Lactobacillus plantarum 299v at a dose of 2 capsules daily, divided | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Exercise Prescription | Minimum of 30 minutes of exercise per day for at least 5 days per week, at an intensity of 60-80% of maximum perceived exertion | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sleep Prescription | Average a minimum of 7 hours of sleep per night | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Stress Management Prescription A | Breathing exercise Steps to Elicit the Relaxation Response developed by Herbert Benson MD, twice daily | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AStress Management Recommendations were updated from the original Study Protocol as listed on ClinicalTrials.gov. All updates were IRB approved. *Patent pending. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
As in the original pilot study, adherence to the program was supported by regular coaching sessions conducted by trained nutritionists, delivered weekly during the first four weeks, and then at least every other week thereafter. Nutrition coaches had a predefined list of questions that covered adherence to intervention guidelines and queried any changes to medications. Participants had access to nutrition coaches between sessions via texting within the app, with an expected response time of 24-48 hours.
Laboratory testing and DNAmAge analysis
Biological age testing was conducted at baseline and at week 8 for all six participants by TruDiagnostc Laboratory. To calculate the Horvath DNAmAge clock, the agep() function in the wateRmelon R package was used. Beta values were normalized using the SeSAMe pipeline [a].
For consistency with the first pilot study of this diet and lifestyle intervention, this case series also used Horvath’s original DNAmAge Clock (2013), also known as the Pan-Tissue clock, which continues to be widely accepted, even as second- and third- generation clocks have since been developed. While it was trained against chronological age, multiple studies have shown acceleration of this clock to be associated with age-related disease risk and all-cause mortality [8].
Author Contributions
KF and TC developed the manuscript. SM contributed to the data analysis and results reporting. RH assisted with reviewing and editing the manuscript. All authors approved the manuscript.
Acknowledgments
We would like to acknowledge and thank the following for their assistance: Nutritionist coaches: Karen Herb MS CNS LDN, Amanda Turton-Huff, MS, Melissa Parker MS CNS CDN, Miranda Kusi MS CNS CDN, Olivia Moeller MS CNS, Despina Giannopoulou MS IFMCP, Sally Logan MS CNS CKNS, Melissa Twedt MS CNS LDN, Rachel Suprenant, ND IFMCP, Darisa Espinal ND, MS and Josette Herdell MS CNS LDN. Suggestic staff: Victor Chapela, Shai Rozen, Erica Sanders and Ives Laurent. TruDiagnostic staff: Ryan Smith and Varun Dwaraka PHD.
Conflicts of Interest
The authors declare that they have no conflicts of interest. Kara Fitzgerald is the owner of a patent application for the intervention used in this study.
Ethical Statement
This retrospective case series was completed with National University of Natural Medicine as the IRB of record, but this particular submission is exempted from requiring approval, as it falls under “small case series automatic exemption”.
Funding
No funding was received for this work.
References
- 1. About chronic disease. Center for Disease Control. 2022. https://www.cdc.gov/chronicdisease/about/index.htm.
- 2. Woolf SH, Schoomaker H. Life Expectancy and Mortality Rates in the United States, 1959-2017. JAMA. 2019; 322:1996–2016. https://doi.org/10.1001/jama.2019.16932 [PubMed]
- 3. Kennedy BK, Berger SL, Brunet A, Campisi J, Cuervo AM, Epel ES, Franceschi C, Lithgow GJ, Morimoto RI, Pessin JE, Rando TA, Richardson A, Schadt EE, et al. Geroscience: linking aging to chronic disease. Cell. 2014; 159:709–13. https://doi.org/10.1016/j.cell.2014.10.039 [PubMed]
- 4. Scott AJ, Ellison M, Sinclair DA. The economic value of targeting aging. Nat Aging. 2021; 1:616–23. https://doi.org/10.1038/s43587-021-00080-0
- 5. Bergsma T, Rogaeva E. DNA Methylation Clocks and Their Predictive Capacity for Aging Phenotypes and Healthspan. Neurosci Insights. 2020; 15:2633105520942221. https://doi.org/10.1177/2633105520942221 [PubMed]
- 6. Belsky DW, Caspi A, Corcoran DL, Sugden K, Poulton R, Arseneault L, Baccarelli A, Chamarti K, Gao X, Hannon E, Harrington HL, Houts R, Kothari M, et al. DunedinPACE, a DNA methylation biomarker of the pace of aging. Elife. 2022; 11:e73420. https://doi.org/10.7554/eLife.73420 [PubMed]
- 7. Fitzgerald KN, Hodges R, Hanes D, Stack E, Cheishvili D, Szyf M, Henkel J, Twedt MW, Giannopoulou D, Herdell J, Logan S, Bradley R. Potential reversal of epigenetic age using a diet and lifestyle intervention: a pilot randomized clinical trial. Aging (Albany NY). 2021; 13:9419–32. https://doi.org/10.18632/aging.202913 [PubMed]
- 8. Hore TA. Modulating epigenetic memory through vitamins and TET: implications for regenerative medicine and cancer treatment. Epigenomics. 2017; 9:863–71. https://doi.org/10.2217/epi-2017-0021 [PubMed]
- 9. Arora I, Sharma M, Tollefsbol TO. Combinatorial Epigenetics Impact of Polyphenols and Phytochemicals in Cancer Prevention and Therapy. Int J Mol Sci. 2019; 20:4567. https://doi.org/10.3390/ijms20184567 [PubMed]
- 10. An S, Ahn C, Moon S, Sim EJ, Park SK. Individualized Biological Age as a Predictor of Disease: Korean Genome and Epidemiology Study (KoGES) Cohort. J Pers Med. 2022; 12:505. https://doi.org/10.3390/jpm12030505 [PubMed]
- 11. Cortez BN, Pan H, Aguayo-Mazzucato C. DNA methylation supports accelerated biological age in Type 2 Diabetes which can be reversed with pharmacological treatments: Retrospective Cohort Study. bioRxiv. 2022.09.14.507908. [Epub ahead of print]. https://doi.org/10.1101/2022.09.14.507908
- 12. Chen BH, Marioni RE, Colicino E, Peters MJ, Ward-Caviness CK, Tsai PC, Roetker NS, Just AC, Demerath EW, Guan W, Bressler J, Fornage M, Studenski S, et al. DNA methylation-based measures of biological age: meta-analysis predicting time to death. Aging (Albany NY). 2016; 8:1844–65. https://doi.org/10.18632/aging.101020 [PubMed]
- 13. Poganik JR, Zhang B, Baht GP, Kerepesi C, Yim SH, Lu AT, Haghani A, Gong T, Hedman AM, Andolf E, Pershagen G, Almqvist C, White JP, et al. Biological age is increased by stress and restored upon recovery. bioRxiv. 2022.05.04.490686. [Epub ahead of print]. https://doi.org/10.1101/2022.05.04.490686



