Introduction
Sarcopenia is a common geriatric syndrome, defined as generalized, progressive loss of muscle mass and strength, occurring during normal aging with significant consequences on the quality of life for the elderly [1–4]. Age-related degradation of muscle leads to a slow but continuous loss in lean mass, starting after the age of 40 and thereafter is accelerated by age 70 [5, 6]. The prevalence of sarcopenia between age 60 to 70 is reported to be 5–13% but increases to 11–50% in people older than 80 [7, 8]. Sarcopenic patients face decreased strength and mobility, therefore are at higher risk for falls, bone fractures and increased mortality. Since the number and proportion of the global aging population is rapidly growing, preventing or delaying sarcopenia should have a great impact on the quality of life for elderly, as well as its socio-economic burden on individuals and society. Current strategies are mainly focused on exercise and nutrition, and despite some molecular pathways involved in aging have been suggested with potential for drug development, no specific medications have been developed with therapeutic impact in sarcopenia.
Underlying pathophysiologic mechanisms contributing to sarcopenia are highly complex and multifactorial. In addition to behavioral or “extrinsic” factors, such as a sedentary lifestyle, the remaining contributing/causal factors appear directly related to the aging process of muscle to include mitochondrial dysfunction with oxidative damage, lysosomal dysfunction, decreased protein synthesis, decreased anabolic hormones, inflammaging/immune-senescence and satellite cells dysfunction (decreased number and regenerative capacity). Mitochondria, as the “power-house” of cells, is considered as a central player in overall aging process, and certainly in muscle going through sarcopenic change. In a recent study using neurotrophin 3 (NT-3) gene therapy, we demonstrated that NT-3 increases muscle fiber diameter through direct activation of mTORC1 pathway [9]. In addition, NT-3 was shown to improve hypomyelination state, neuromuscular junction (NMJ) connectivity, have anti-inflammatory, antioxidant, antiapoptotic properties, and enhances mitochondria biogenesis [10–14]. This suggests a potential application of NT-3 gene therapy for muscle wasting conditions including age-related sarcopenia. In this study, we used a triple muscle-specific creatine kinase (tMCK) promoter to restrict NT-3 expression to the skeletal muscle and self-complimentary adeno-associated virus serotype 1 (scAAV1) as vector to assess the therapeutic efficacy of AAV1.NT-3 in wild type-aged C57BL/6J mice, a model for natural aging and sarcopenia. Quantitative histopathologic parameters served to address age-related changes in muscle, peripheral nerve and NMJ. These changes include muscle fiber size and fiber type switch, myelin thickness and the innervated status of the NMJ. Functional studies and in vivo muscle physiology were used to assess the motor strength of the mice. The results show that AAV1.NT-3 gene therapy in the wild type C57BL/6J mice at 2 years of age, unequivocally improved the function of sarcopenic muscle, increased muscle fiber size, myelin thickness and NMJ connectivity. In addition, we found attenuation of age-related kyphosis and coat changes as well.
Results
rAAV.NT-3 vector production and potency
scAAV1.tMCK.NT-3 design (Supplementary Figure 1A), and production followed previously described methods at Nationwide Children`s Hospital, Columbus [10]. scAAV1.tMCK.NT-3, at 1 × 1011 vg dose, was delivered to the gastrocnemius muscle of 18 months old C57BL/6 mice. Blood samples from terminally anesthetized treated and untreated mice were collected by cardiac puncture at six months post gene injection, and serum was assayed for NT-3 levels using a capture ELISA, as previously reported [10]. No detectable NT-3 levels were found in the untreated C57BL/6 mice at 2 years of age, in contrast to the treated cohort (Supplementary Figure 1B), showing significant improvements in functional and histologic outcome measures as described below. Similar to our previous observations in younger age groups, we found no effect of sex on serum NT-3 levels in the aged C57BL/6 mice.
Discussion
In this study, we provide strong evidence for the efficacy of AAV1.NT-3 gene therapy in sarcopenia and age related hypomyelination of peripheral nerves, as well as the maintenance of NMJ connectivity in 2-year-old C57BL/6 mouse, a model for natural aging. At 6 months post gene delivery, the NT-3 effects on the neuromuscular system manifested as significant histopathological improvements, reflecting upon functional performance and in vivo muscle physiology. Attenuation of other age related musculoskeletal and skin changes with treatment including kyphosis, dermatitis, and alopecia was also striking.
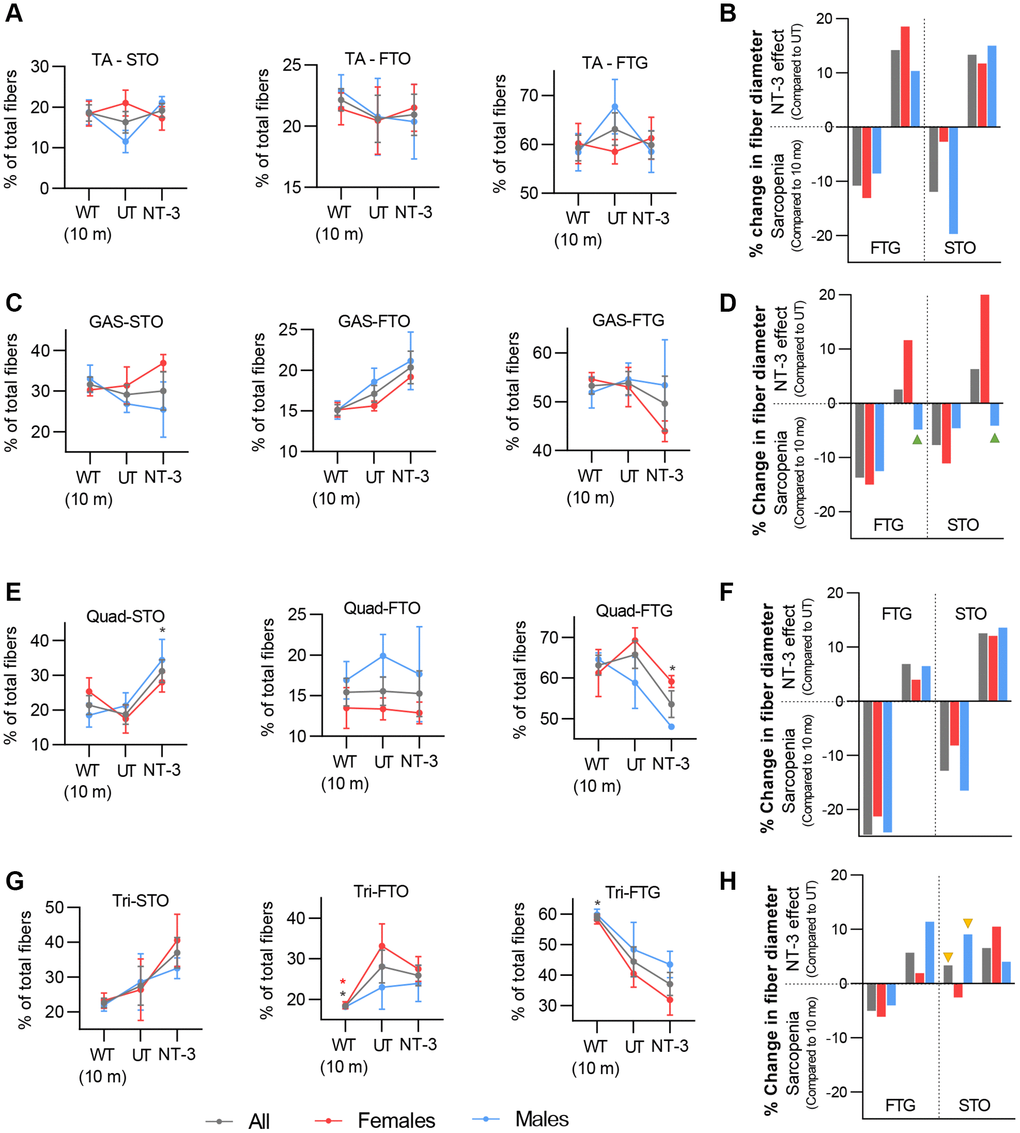
In the untreated C57BL/6 mice, detailed morphometric data from all four muscles, although confirmed the previous observations of type 2 fiber atrophy in general [24–26], revealed some differences between muscles in response to aging process. The most severe glycolytic fiber atrophy occurred in both male and female quadriceps, and the most severe oxidative fiber atrophy was present in male tibialis anterior muscles; triceps were the least affected muscle by sarcopenia. At 6 months post NT-3 gene therapy, quantitative data from all four muscles revealed an overall normalization of fiber size towards values observed in 10 months of age. In the quadriceps and triceps (proximal-extensor muscles) of the treated aged cohort, there was a switch to fatigue-resistant oxidative fibers occurred in both sexes, when compared to untreated. However, in distal hindlimb muscles (anterior compartment/flexor muscle vs. posterior compartment/extensor muscle), we found what appears to be a sex-dependent muscle remodeling. The switch from oxidative to glycolytic fiber type observed in the tibialis anterior muscle in females was associated with prominent FTG size increase. In the same muscle from males, oxidative fiber size increase was greater than glycolytic, along with an increase in oxidative fiber type. Contrary to the tibialis anterior, in the female gastrocnemius muscle, there was a switch from glycolytic to oxidative fiber type, in addition to a prominent STO size increase. We were intrigued with these observations that the most prominent muscle fiber size increase occurred in the direction of fiber type switch; to oxidative vs. to glycolytic. Interestingly however, male gastrocnemius muscle failed to show any size increase in STO or FTG, while a small FTO percentage gain was detected along in addition to a meager size increase in this fiber subtype. Overall, these observations show the presence of a muscle- and sex-dependent remodeling with aging, as well as response to NT-3 treatment.
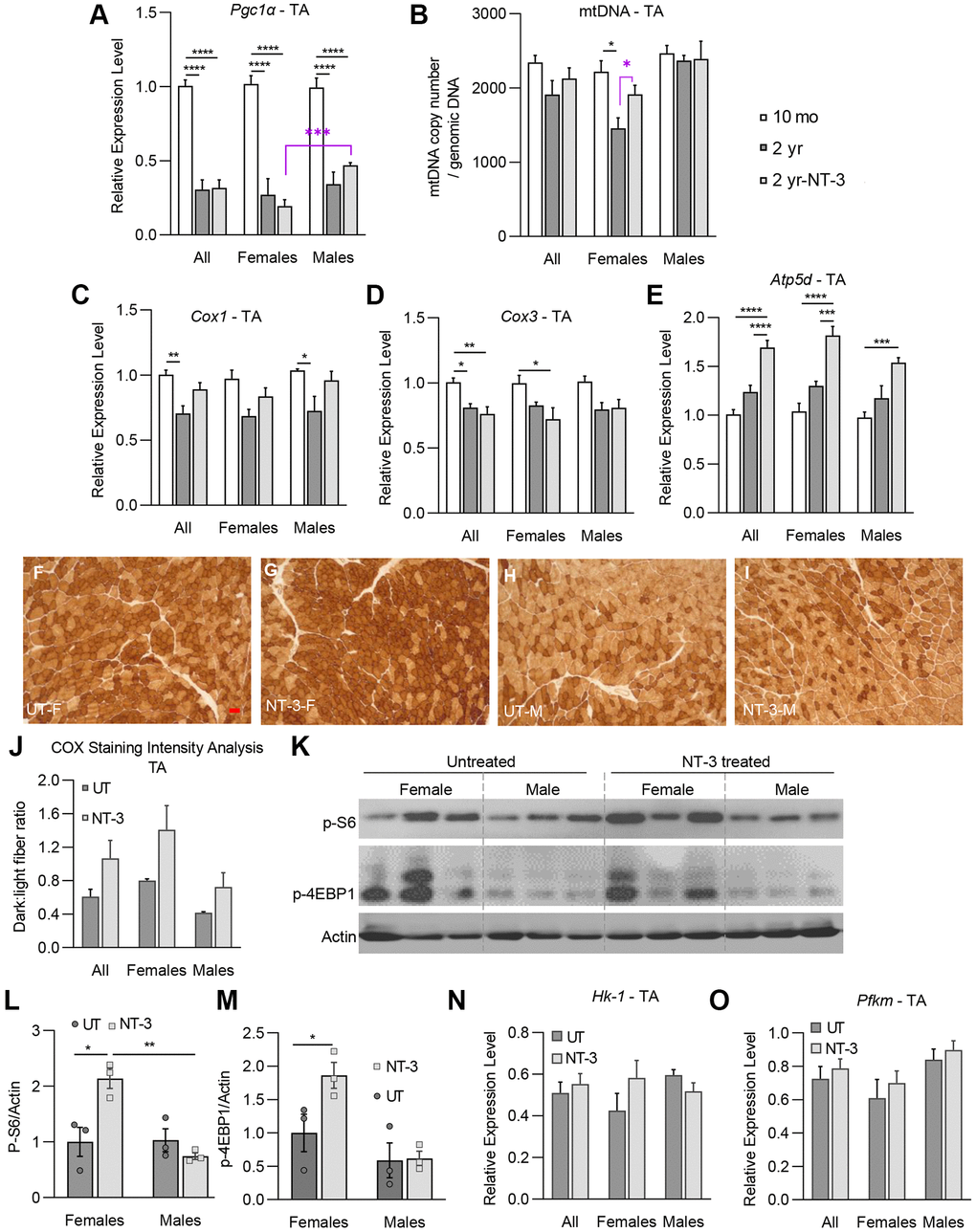
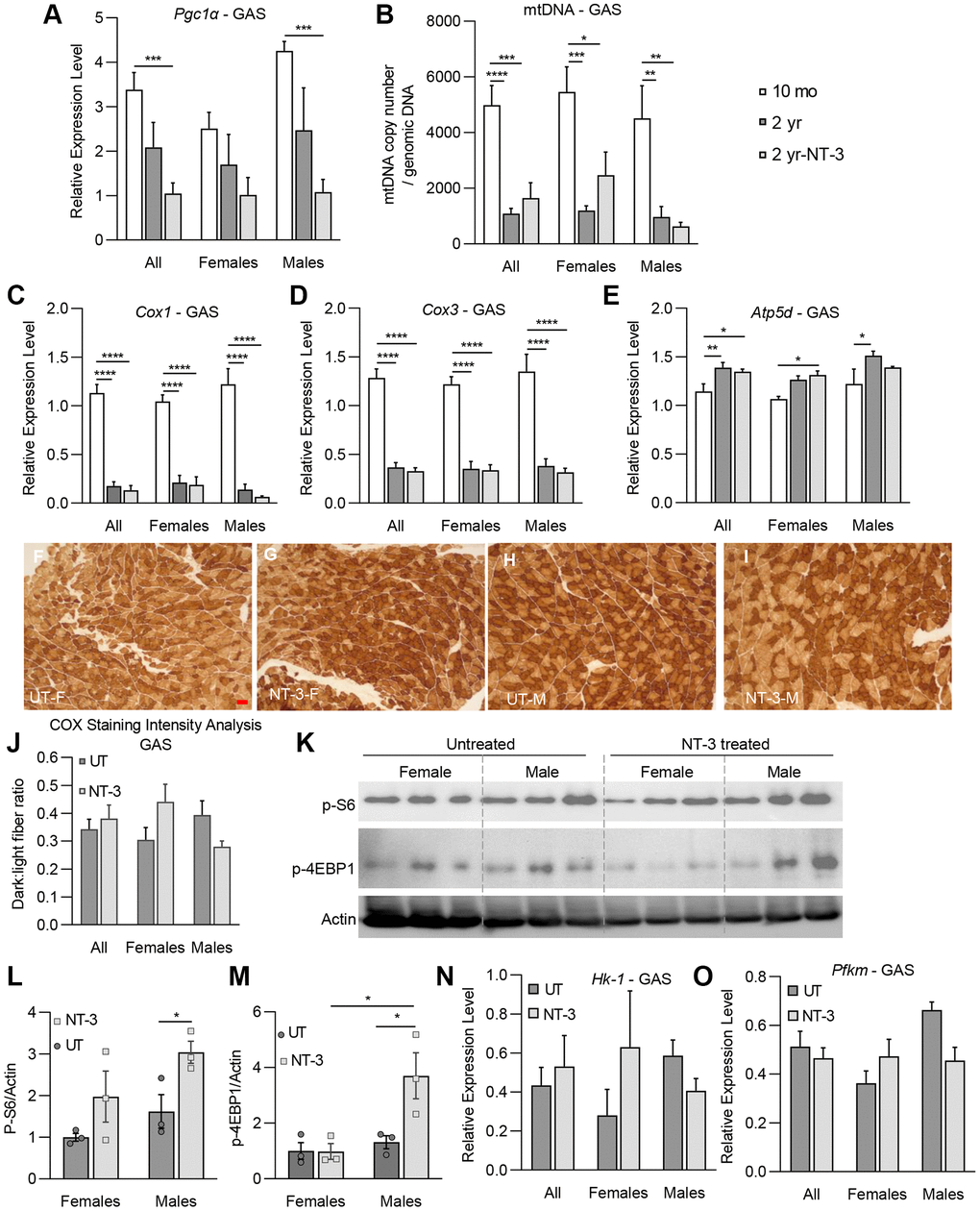
The results from the molecular studies assessing the NT-3 effect on the oxidative state of distal hind limb muscles, accompanied by western blot analyses for mTORC1 activation are in accordance with the histologic findings. We like to emphasize here that NT-3 effect is not directed to well-differentiated or normal functioning cells, but rather is operative upon remodeled-cell metabolism that may result from a pathological process [9, 27]. In this context, it is not surprising to see NT-3, through mTORC1 activation, exerts an anabolic effect in the female tibialis anterior by increasing FTG fiber size and the percentage of glycolytic fibers, and by doing so, overcoming the age-related remodeling by normalizing the muscle towards to a younger age. Even though Pgc1α transcripts at this time point was found not elevated, we showed evidence that NT-3 prevented the age-related mitochondria loss by increasing mitochondria copy number towards 10-month levels. This was associated with significantly increased Atp5d transcripts and COX stain intensity, suggesting increased mitochondrial function. Decreased Pgc1α levels can be interpreted as correlative of decreased STO percentage, which was also associated with an increase in FTG percentage along with a significant size increase of FTG fibers. We believe that the total mtDNA increase in tissue might be linked to these changes in FTG subtype. In contrast, NT-3 in male tibialis anterior showed no mTORC1 activation, but preserved mitochondria content and function. This was supported by an increase in Pgc1α and Atp5d transcripts as well as COX stain intensity, which was reflected in muscle histology as fiber type switch from glycolytic, back to the oxidative levels, as in the 10-month-old samples. The findings in gastrocnemius, when contrasted with tibialis anterior muscle, appear unique, as males were less responsive to NT-3. Even though with treatment, mTORC1 was activated, the male gastrocnemius muscle failed to respond with size increase in any of the fiber types. NT-3 also did not alter mitochondria copy number, or markers of oxidative phosphorylation expression significantly. In female gastrocnemius, although did not reach to significance level (likely due to small n number), NT-3 lead to some increases in the phosphorylated S6 protein, as well as in mitochondria content, which were associated with STO greater than FTG fiber size increase, along with increased STO percentage.
Studies have shown that mtDNA and mRNA abundance and mitochondrial ATP production, all decline with advancing age [21]. PGC1α is an important transcriptional coactivator of mitochondrial biogenesis and respiration [28]. Previous studies have shown that PGC1α drives the formation of fatigue resistant STO muscle fibers [29]. Decreased Pgc1α expression in sarcopenic muscle from old rodents has been reported [30], as well as with aging in sedentary persons compared with physically active individuals [31, 32]. Recent transcriptomic data from skeletal muscle found reduced expression of genes related to all electron transport complexes and pyruvate dehydrogenase complexes with aging in humans [33]. Our molecular studies from untreated aged C57BL/6 mice agree with these previous reports by showing decreased Pgc1α expression and mtDNA content as well as reduced Cox1 and Cox3 transcripts compared to 10-month-old muscle. Lack of Pgc1α increase at 6 months post gene injection, although seemingly not fitting with the finding of increased mitochondria content in female tibialis, might be explained with the possibility of Pgc1α activation in an earlier time point followed by an equilibrium state. In a previous study of NT-3 gene therapy in young male trembler J (TrJ) mice, at 4 months post AAV.NT-3 injection we found increased Pgc1α expression, accompanied by increased phosphorylation levels of 4E-BP1 and S6 proteins as evidence of mTORC1 activation in the gastrocnemius muscle [9]. AAV.NT-3 treatment was capable of reversing the defective expression levels of Pgc1α seen in the TrJ neurogenic muscle along with enhanced levels of activated 4E-BP1. In the skeletal muscle, mTORC1 can regulate mitochondrial biogenesis and metabolism through 4E-BP1/PGC1α [34]. Therefore, it is conceivable that NT-3 might also promote oxidative phosphorylation through activation of 4E-BP1 and PGC1α in the muscle. PGC1α is known to co-regulate several genes, and its expression alone is considered sufficient to increase mitochondrial mass [35]; however, it was also reported that PGC1α is dispensable for exercise induced mitochondrial biogenesis in skeletal muscle [36]. Therefore, further studies are needed to assess the link between NT-3 and Pgc1α expression in the sarcopenic muscle, including earlier time point experiments in this model.
Physical exercise, aerobic or anaerobic, has been well accepted to be a countermeasure for sarcopenia, but also recognized as an important practice in the prevention and treatment of not only chronic and degenerative diseases, but also age-associated multisystem diseases, including cardiovascular, neurodegenerative, chronic pulmonary diseases, cancer, diabetes, and morbid obesity. Aerobic/endurance exercise helps to maintain and improve cardiovascular fitness, respiratory function and muscle oxidative capacity, whereas strength/resistance-exercise programs primarily increase type 2 muscle fiber size, muscle strength, and function. Although the underlying sequential events that lead to these muscle adaptations are poorly understood, it has been emphasized that the mechanisms that regulate these processes involve the “quality” of skeletal muscle mitochondria [37]. Cellular antioxidant capacity and oxidative stress are postulated to be critical factors in the aging process. In one study, whole body resistance exercise training in aged men and women was shown to induce significantly higher complex IV activity and decreased oxidative stress markers, compared to before training [38]. Notably, there were no apparent changes in normal mtDNA content or mtDNA deletion products, suggesting that regular resistance exercise decreases oxidative stress, but does not affect mtDNA. It was also postulated that increases in complex IV of the electron transport chain may have an indirect antioxidant effect in older adults [39]. We think these observations are relevant to this current study regarding the similarities between exercise and NT-3-induced changes in sarcopenic muscle. COX stain intensity quantification obtained from tibialis and gastrocnemius muscles in our study appears to be a sensitive indicator of functioning mitochondria, as in the case of resistance exercise, correlating well with treadmill functional tests being more prominent in females. Activation of the mTORC1 pathway leading to radial growth in glycolytic fiber type, that we see in female tibialis muscle, is also a supportive finding. Instead, an overall increase of oxidative fibers in quadriceps and triceps of both sexes, and in female gastrocnemius muscle with NT-3, is reminiscent to the effects of aerobic/endurance exercise. Moreover, exercise induced mechanical loading has been shown to induce production of IGF-1, VEGF, and hepatocyte growth factor in osteocytes cell lines, which may play roles in regulating muscle growth [40]. In addition, muscle NT-3 levels increased by exercise training have been shown to contribute to improvement in various conditions [40–44]. Moreover, although it has not been studied in the context of sarcopenia, we predict that NT-3, via its known anti-inflammatory and immunomodulatory properties, [14, 45–47] may also have an attenuating effect on age-related inflammation; presumably a contributing factor to sarcopenia. Exercise intervention, despite its effectiveness as therapeutic regime for sarcopenia, is only available to patients or elderly who are reasonably mobile and can participate in such intervention without safety concerns. When considering the burden of sarcopenia on the lifestyle of elderly, and on the healthcare system, we believe this preclinical study is providing strong support for AAV.NT-3 gene therapy in the successful management of sarcopenia, as a serious and plausible option in the future.
Materials and Methods
Animals and treatment groups
Naturally aged C57BL/6 mice were included in the study (JAX stock #000664) and all animal experiments were performed according to the guidelines approved by The Research Institute at Nationwide Children’s Hospital Animal Care and Use Committee that operates in full accordance with the Animal Welfare Act and the Health Research Extension Act (IACUC approval number = AR18-00076). 18 months old C57BL/6 (6 males and 6 females, n = 12) mice received 1 × 1011 vg dose of AAV1.tMCK.NT-3, via intramuscular (IM) injection into the gastrocnemius muscle. Age- and sex-matched C57BL/6 (6 males and 6 females, n = 12) mice were injected with Ringer’s lactate as controls. Mice were tested functionally with treadmill, rotarod, and in vivo muscle contractility assay and they were sacrificed six months post-injection by an over-dosage of xylazine/ketamine anesthesia for harvesting blood, sciatic nerves, as well as upper and lower limb muscles including lumbricals, at six months post gene injection.
AAV1.tMCK.NT-3 vector production and potency
Construct of self-complimentary (sc) AAV serotype 1 vector with muscle specific tMCK promoter was described previously [10]. The vector was produced in our Viral Vector Core at Nationwide Children’s Hospital, Columbus (Andelyn Biosciences). Aliquots of virus were stored at −80°C until used. Serum was separated from blood samples that were collected by cardiac puncture at six months post gene injection and serum NT-3 levels were detected by ELISA as previously reported (NT-3, n = 9; UT, n = 8) [10].
Run to exhaustion test
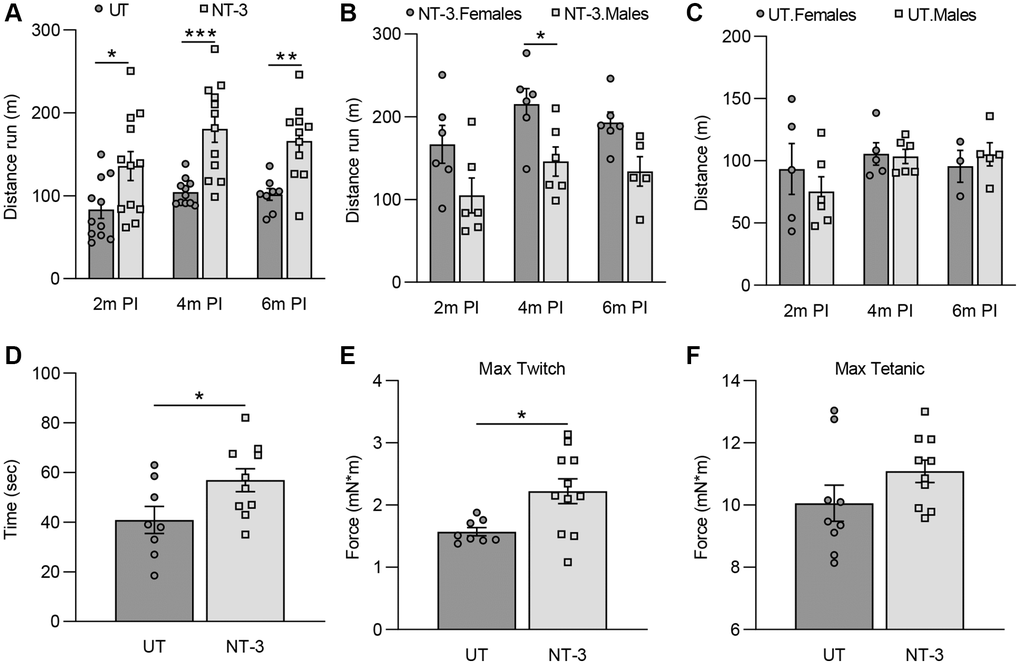
Run to exhaustion treadmill performance test was performed at two-, four- and six-months post injection. Mice were exercised to exhaustion via treadmill (Columbus Instruments, Exer-6M Treadmill), as described previously [15]. Mice were acclimated to the treadmill prior to data collection. Mice were run to exhaustion with increasing treadmill speed by 1 meter/min each minute, starting at an initial 7 meter per minute velocity. Lanes have a shock plate that pulses at a frequency of ~3 Hz. Mice were considered at “exhaustion” level when they were unable to re-engage the treadmill for 3 seconds after resting on the shock-plate. Run duration was recorded and used to calculate the distance ran (2 months post-injection: NT-3, n = 12; UT, n = 11; 4 months post-injection, NT-3, n = 12; UT, n = 11; 6 months post-injection: NT-3, n = 11; UT, n = 8).
In vivo muscle contractility assay
In vivo muscle contractility assay was performed at the endpoint, as described previously [15]. Hind paw of the anesthetized mouse was placed on footplate, which was attached to a dual-mode lever, and the tibia was aligned perpendicular to the lever. Subcutaneous EMG electrodes were used to stimulate the tibial nerve. Gastrocnemius muscle torque around the ankle joint was measured by muscle physiology apparatus (Aurora Scientific, ON, Canada) using isometric contraction (maximum twitch response) and fatigue (maximum tetanic response) protocols (Max twitch: NT-3, n = 11; UT, n = 8; Max tetanic: NT-3, n = 10; UT, n = 9).
Rotarod
Rotarod test was performed at six months post-injection to assess motor function and balance of the mice. Mice were acclimated to rotarod apparatus (Columbus Instruments, Ohio, USA) prior to data collection. Rotarod protocol included a 5-rpm run with a constant acceleration of 0.2 rpm/s. The averages of the best two out of three runs were calculated (NT-3, n = 10; UT, n = 8).
Musculoskeletal and skin changes
Age-related musculoskeletal and skin changes including kyphosis, dermatitis, and alopecia were documented semi-quantitatively. Six mice, from both treated and untreated cohorts, were photographed at the end point and the severity of kyphosis, dermatitis, and alopecia were scored. Severe changes were scored as 1, mild-moderate changes as 0.5, and no changes as 0. For kyphosis, arbitrary lines were drawn tangential to the scoliosis curvature to delineate the scoliosis angle (Supplementary Figure 3A). Angle range 90°–110° is considered as severe, 11°–150° as mild-moderate and >150° as none.
Histological analysis
Muscle histology
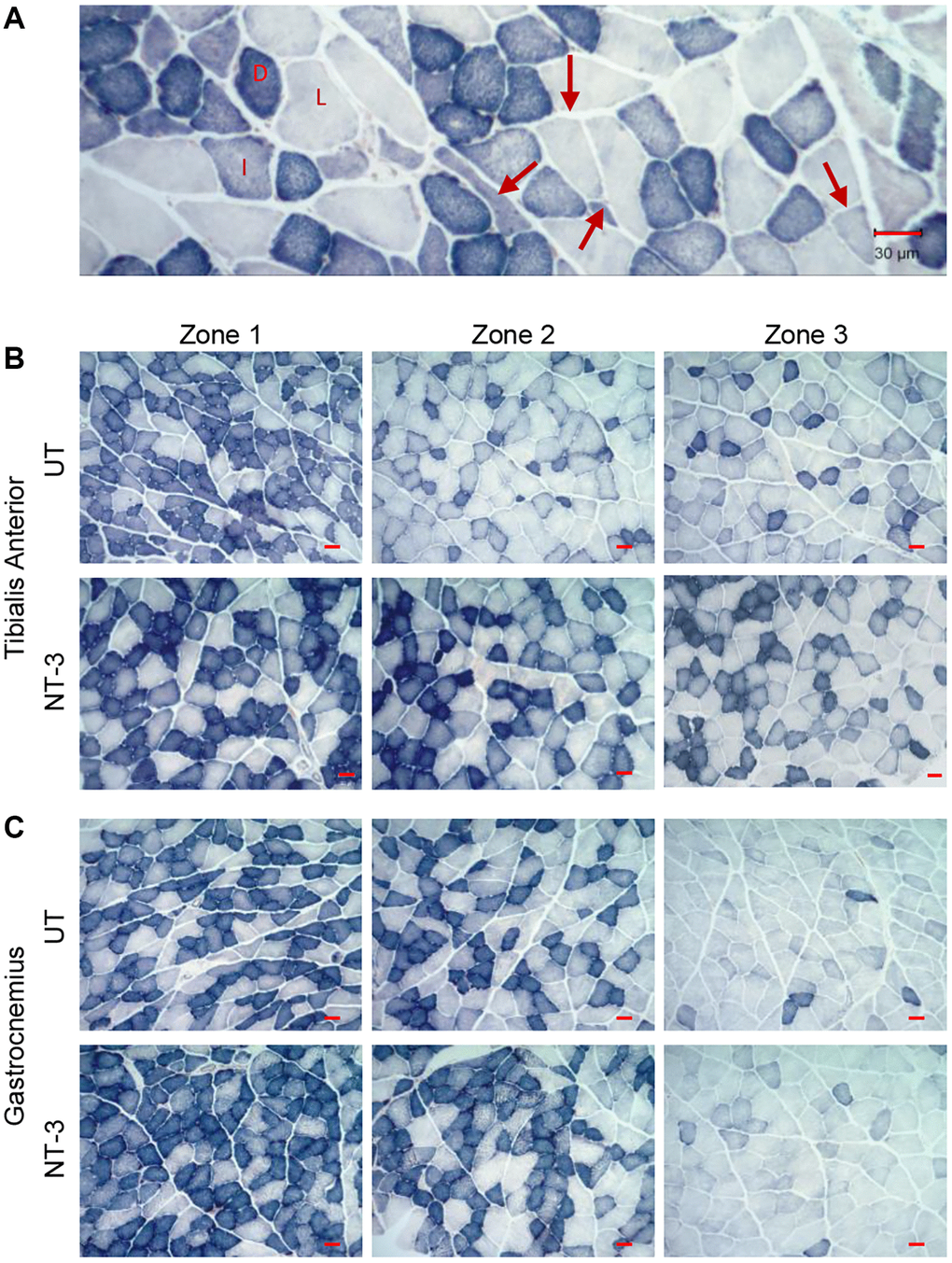
Tibialis anterior, gastrocnemius, quadriceps, and triceps muscles from treated and untreated mice were collected and 12 μm thick cross cryostat-sections were cut. Succinic dehydrogenase (SDH) enzyme histochemistry was performed as previously described to assess metabolic fiber type distribution and myofiber size changes in the aging muscle [15]. Deep, intermediate, and superficial zones of the muscles were analyzed to represent fibers at various oxidative states equally. One representative area from each zone were photographed at 20× magnification using an Olympus BX41 microscope and SPOT Insight 12 Mp sCMOS camera. Sample selection was based on the suitability of the tissue sections, including staining quality, contrast, and lack of artifacts and not based on outcomes of behavioral or physiological analyses. Shortest distance across the muscle fiber was measured as fiber diameter (Zeiss Axiovision LE4 software V4.9.1.0) and mean fiber diameter (mean ± SEM) was calculated for each fiber type (STO, FTO, FTG) as well as for combination of all fiber types. Fiber type percent distribution of total fibers was determined for each mouse from each treatment group. Data were obtained from a total of 2747 (n = 9), 2959 (n = 9), 1722 (n = 5), and 1357 (n = 4) fibers of the treated cohort, and 2815 (n = 8), 2557 (n = 8), 2040 (n = 6), and 2164 (n = 6) fibers of the untreated cohort for tibialis anterior, triceps, gastrocnemius and quadriceps muscles respectively.
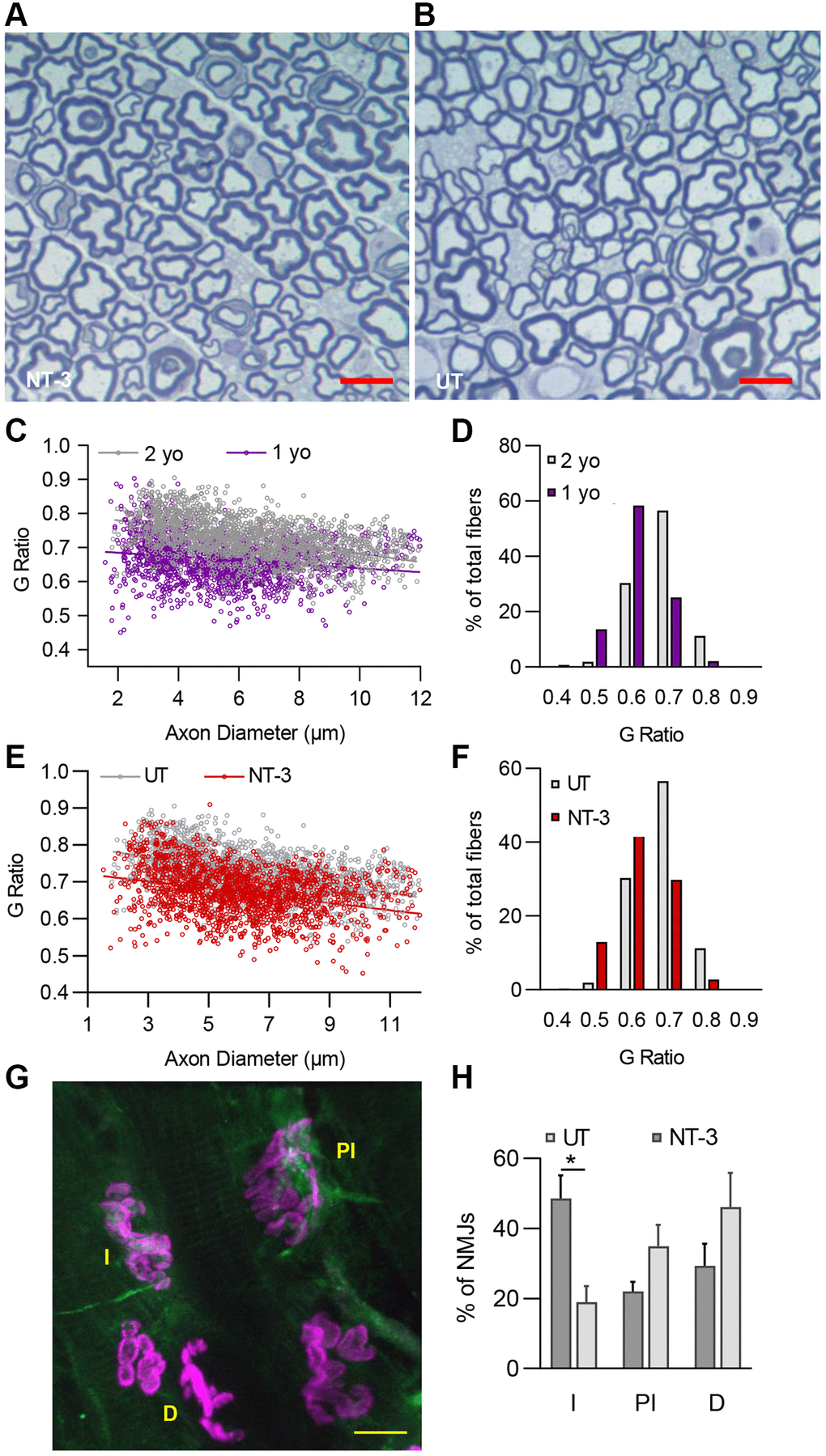
G ratio of the myelinated fibers
G ratio was calculated to assess the myelin thickness of fibers in tibialis nerve, as described previously [48]. Semithin, toluidine blue-stained cross sections of tibial nerve were prepared for each mouse (n = 6 for both treated and untreated 2-year-old mice, n = 4 for 1-year-old control mice, with even sex distribution) and three randomly selected nonoverlapping areas were photographed randomly at 100× magnification using an Olympus BX41 microscope and SPOT Insight 12Mp sCMOS camera. Myelin interior and exteriors were outlined in Axiovision (AxioVs40 × 64 V 4.9.1.0) to determine the area, which was used to calculate diameters to determine g ratio. A total of 1833 fibers for treated, 1928 for untreated and 1658 for 1-year-old control mice were measured to generate scattergrams and the percent g ratio distribution histograms. Slopes of treated vs. untreated and 1-year-old vs. 2-year-old mice were compared using GraphPad Prism (9.0.0).
Immunohistochemical analysis of neuromuscular junctions (NMJ)
Lumbrical muscles collected from treated and untreated mice (n = 4 for both cohorts with equal sex distribution) were processed as described previously [11]. Muscles were fixed and stained with primary antibodies (Acetylcholine receptor (AChR) antibody, α Bungarotoxin, T1175, 1:500; Anti Neurofilament 200 antibody, N4142, 1:500; SV2 antibody, AB_2315387, 1:50) [49, 50], followed by incubation with secondary antibodies (Alexa Fluor 488 conjugated anti-rabbit and anti-mouse IgG, 1:500). Samples were imaged at 60× magnification using a Zeiss LSM 800 confocal microscope. NMJs were considered to be innervated when nerve completely overlapped the AChRs, as partially innervated when some parts of the AChRs were not overlapping with nerve, and as denervated when there was not any nerve co localizing with AChRs [11, 51]. An average of 41.3 NMJs per mouse were evaluated from NT-3-treated and UT-mice (n = 4 mice per group with equal sex distribution).
Expression analysis
Protein extraction and western blot analysis
Twenty micrometer thick sections from frozen TA and GAS muscle blocks (20 section per block, n = 3 per group) were put into 2 ml centrifuge tubes and homogenized in homogenization buffer [125 mM Tris-HCL pH6.8, 4% SDS, 4 M Urea solution with 1X Halt protease inhibitor (ThermoFisher) and 1× phosphatase inhibitor (Sigma)] using a disposable pestle. The lysate was then incubated on a rotary spin cycle at 4°C for 2 hours, followed by centrifugation at 10,000 g for 10 min at 4°C. The supernatant was then transferred to a new tube.
Protein samples were run in Novex 10–20% Tricine mini protein gel (ThermoFisher) and transferred to PDVF membranes (GE Healthcare). Membranes were blocked for 1 h at room temperature with 5% milk in TBS-T (TBS buffer with 0.05% Tween-20). Membranes were then incubated with primary antibodies in TBS-T buffer overnight at 4°C. After 5 min of three times wash with TBS-T, membranes were incubated with secondary antibodies in 5% milk in TBS-T for 1 h. Membranes were washed again with TBS-T for 3 times and TBS for 2 times with 5 min each wash. ECL WesternSure premium chemiluminescent substrate (LI-COR) was used for band detection followed by exposure using Chemidoc Imaging system (Bio-Rad) and band intensities were quantified using ImageJ (NIH). Primary antibodies: anti-phospho S6 protein Ser235/236 (#4858), and anti-phospho 4EBP1 thr37/46 (#2855) were from Cell Signaling Technology and anti-actin antibody (sc-47778) was from Santa Cruz; secondary antibody: HRP-linked anti-rabbit/mouse IgG (#7074/7076) was purchased from Cell Signaling Technology.
RNA isolation and mRNA expression
Total RNA was extracted from frozen muscle blocks (20 micrometer thick sections, 20 section per block; NT-3, n = 9; UT, n = 8; WT, n = 8) using Mini RNeasy Plus Universal Kit (Qiagen). cDNAs were synthesized using ProtoScript II First Strand cDNA Synthesis Kit (BioLabs). Primer sets (synthesized by IDT) for Pgc-1α, Cox1, Cox3 and Atp5d were obtained from previous publications [52–54] and new primers were designed for Pfkm (F-GAAGATACCAACTCGGACCAC, R-ATGACCCATGAAGAGCATCA) and Hk-1 genes (F-CGGAATGGGGAGCCTTTGG, R-GCCTTCCTTATCCGTTTCAATGG). All qPCR were performed using PowerUp SYBR Green Master Mix (ThermoFisher) according to the manufacturer’s instructions. qPCR assays were performed using QuantStudio 6 Flex (Applied Biosystem). Expression data were normalized to mouse Gapdh mRNA level and data were analyzed by ΔΔCt method.
COX staining density analysis
Histochemical enzyme activity of cytochrome c oxidase (COX) was assessed by quantifying the COX stain intensity in tibialis anterior and gastrocnemius muscles from treated and untreated mice. 12 μm thick-fresh frozen sections were stained using COX enzyme histochemistry protocol established in our clinical neuromuscular pathology laboratory. Muscle sections representing deep, intermediate, and superficial zones from gastrocnemius (n = 8 treated, n = 7 untreated) and tibialis anterior (n = 6 treated, n = 6 untreated) muscles were photographed at 10× magnification, using an Olympus BX41 microscope and SPOT camera, with even distribution of sex. Image data were collated and processed utilizing the Python programming language. [55] Images were calibrated using representative selections from each image to establish ranges of intensity for each fiber-type; dark (oxidative/higher mitochondria content/type 1) and light (glycolytic/lower mitochondria content/type 2) fibers, including COX deficient pale fibers. Ratio of dark to light fibers was calculated and presented as mean ± SEM.
Statistics
Adequate sample size was determined according to our previous studies that performed analogous experiments [11, 15]. All statistical analyses were performed in GraphPad Prism 9.0 software. Two tail Student t-test, one-way ANOVA with Tukey’s multiple comparison test, two-way ANOVA with Sidak’s multiple comparison test or linear regression analysis were performed when applicable, and significance level was set at P ≤ 0.05. The tests that meet the best assumptions of the data were chosen. Results were given as mean ± SEM in all experiments and the number of animals was mentioned in figure legends along with the name of the statistical analysis performed. Other than the functional tests and quantitative muscle analysis, no blinding was used.
Author Contributions
B.O.: formal analysis, data curation, visualization, original draft preparation, review and editing; L.T.: formal analysis, data curation, original draft preparation; M.M.: data curation; K.M.: data curation; A.R.: data curation; Z.S.: conceptualization, methodology, visualization, original draft preparation, review and editing, supervision, funding acquisition.
Acknowledgments
AAV vector used in this study was produced in Andelyn Biosciences.
Conflicts of Interest
The authors declare no conflicts of interest related to this study.
Ethical Statement
Naturally aged C57BL/6 mice were included in the study (JAX stock #000664) and all animal experiments were performed according to the guidelines approved by The Research Institute at Nationwide Children’s Hospital Animal Care and Use Committee that operates full accordance with the Animal Welfare Act and the Health Research Extension Act (IACUC approval number = AR18-00076).
Funding
This study was funded by Sarepta Therapeutics, Inc.
References
- 1. Wiedmer P, Jung T, Castro JP, Pomatto LCD, Sun PY, Davies KJA, Grune T. Sarcopenia - Molecular mechanisms and open questions. Ageing Res Rev. 2021; 65:101200. https://doi.org/10.1016/j.arr.2020.101200 [PubMed]
- 2. Cruz-Jentoft AJ, Landi F, Schneider SM, Zúñiga C, Arai H, Boirie Y, Chen LK, Fielding RA, Martin FC, Michel JP, Sieber C, Stout JR, Studenski SA, et al. Prevalence of and interventions for sarcopenia in ageing adults: a systematic review. Report of the International Sarcopenia Initiative (EWGSOP and IWGS). Age Ageing. 2014; 43:748–59. https://doi.org/10.1093/ageing/afu115 [PubMed]
- 3. Janssen I, Shepard DS, Katzmarzyk PT, Roubenoff R. The healthcare costs of sarcopenia in the United States. J Am Geriatr Soc. 2004; 52:80–5. https://doi.org/10.1111/j.1532-5415.2004.52014.x [PubMed]
- 4. Clark BC, Manini TM. Functional consequences of sarcopenia and dynapenia in the elderly. Curr Opin Clin Nutr Metab Care. 2010; 13:271–6. https://doi.org/10.1097/MCO.0b013e328337819e [PubMed]
- 5. Mitchell WK, Williams J, Atherton P, Larvin M, Lund J, Narici M. Sarcopenia, dynapenia, and the impact of advancing age on human skeletal muscle size and strength; a quantitative review. Front Physiol. 2012; 3:260. https://doi.org/10.3389/fphys.2012.00260 [PubMed]
- 6. Murton AJ. Muscle protein turnover in the elderly and its potential contribution to the development of sarcopenia. Proc Nutr Soc. 2015; 74:387–96. https://doi.org/10.1017/S0029665115000130 [PubMed]
- 7. Porter MM. The effects of strength training on sarcopenia. Can J Appl Physiol. 2001; 26:123–41. https://doi.org/10.1139/h01-009 [PubMed]
- 8. Vandervoot AA, Symons TB. Functional and metabolic consequences of sarcopenia. Can J Appl Physiol. 2001; 26:90–101. https://doi.org/10.1139/h01-007 [PubMed]
- 9. Yalvac ME, Amornvit J, Chen L, Shontz KM, Lewis S, Sahenk Z. AAV1.NT-3 gene therapy increases muscle fiber diameter through activation of mTOR pathway and metabolic remodeling in a CMT mouse model. Gene Ther. 2018; 25:129–38. https://doi.org/10.1038/s41434-018-0009-8 [PubMed]
- 10. Sahenk Z, Galloway G, Clark KR, Malik V, Rodino-Klapac LR, Kaspar BK, Chen L, Braganza C, Montgomery C, Mendell JR. AAV1.NT-3 gene therapy for charcot-marie-tooth neuropathy. Mol Ther. 2014; 22:511–21. https://doi.org/10.1038/mt.2013.250 [PubMed]
- 11. Ozes B, Moss K, Myers M, Ridgley A, Chen L, Murrey D, Sahenk Z. AAV1.NT-3 gene therapy in a CMT2D model: phenotypic improvements in GarsP278KY/+ mice. Brain Commun. 2021; 3:fcab252. https://doi.org/10.1093/braincomms/fcab252 [PubMed]
- 12. Yalvac ME, Arnold WD, Hussain SA, Braganza C, Shontz KM, Clark KR, Walker CM, Ubogu EE, Mendell JR, Sahenk Z. VIP-expressing dendritic cells protect against spontaneous autoimmune peripheral polyneuropathy. Mol Ther. 2014; 22:1353–63. https://doi.org/10.1038/mt.2014.77 [PubMed]
- 13. Sahenk Z, Yalvac ME, Amornvit J, Arnold WD, Chen L, Shontz KM, Lewis S. Efficacy of exogenous pyruvate in TremblerJ mouse model of Charcot-Marie-Tooth neuropathy. Brain Behav. 2018; 8:e01118. https://doi.org/10.1002/brb3.1118 [PubMed]
- 14. Yalvac ME, Arnold WD, Braganza C, Chen L, Mendell JR, Sahenk Z. AAV1.NT-3 gene therapy attenuates spontaneous autoimmune peripheral polyneuropathy. Gene Ther. 2016; 23:95–102. https://doi.org/10.1038/gt.2015.67 [PubMed]
- 15. Sahenk Z, Ozes B, Murrey D, Myers M, Moss K, Yalvac ME, Ridgley A, Chen L, Mendell JR. Systemic delivery of AAVrh74.tMCK.hCAPN3 rescues the phenotype in a mouse model for LGMD2A/R1. Mol Ther Methods Clin Dev. 2021; 22:401–14. https://doi.org/10.1016/j.omtm.2021.06.010 [PubMed]
- 16. Gheller BJ, Riddle ES, Lem MR, Thalacker-Mercer AE. Understanding Age-Related Changes in Skeletal Muscle Metabolism: Differences Between Females and Males. Annu Rev Nutr. 2016; 36:129–56. https://doi.org/10.1146/annurev-nutr-071715-050901 [PubMed]
- 17. Ceballos D, Cuadras J, Verdú E, Navarro X. Morphometric and ultrastructural changes with ageing in mouse peripheral nerve. J Anat. 1999; 195:563–76. https://doi.org/10.1046/j.1469-7580.1999.19540563.x [PubMed]
- 18. Verdú E, Ceballos D, Vilches JJ, Navarro X. Influence of aging on peripheral nerve function and regeneration. J Peripher Nerv Syst. 2000; 5:191–208. https://doi.org/10.1046/j.1529-8027.2000.00026.x [PubMed]
- 19. Chung T, Park JS, Kim S, Montes N, Walston J, Höke A. Evidence for dying-back axonal degeneration in age-associated skeletal muscle decline. Muscle Nerve. 2017; 55:894–901. https://doi.org/10.1002/mus.25267 [PubMed]
- 20. Gonzalez-Freire M, de Cabo R, Studenski SA, Ferrucci L. The Neuromuscular Junction: Aging at the Crossroad between Nerves and Muscle. Front Aging Neurosci. 2014; 6:208. https://doi.org/10.3389/fnagi.2014.00208 [PubMed]
- 21. Short KR, Bigelow ML, Kahl J, Singh R, Coenen-Schimke J, Raghavakaimal S, Nair KS. Decline in skeletal muscle mitochondrial function with aging in humans. Proc Natl Acad Sci U S A. 2005; 102:5618–23. https://doi.org/10.1073/pnas.0501559102 [PubMed]
- 22. Barchiesi A, Vascotto C. Transcription, Processing, and Decay of Mitochondrial RNA in Health and Disease. Int J Mol Sci. 2019; 20:2221. https://doi.org/10.3390/ijms20092221 [PubMed]
- 23. Hebert SL, Lanza IR, Nair KS. Mitochondrial DNA alterations and reduced mitochondrial function in aging. Mech Ageing Dev. 2010; 131:451–62. https://doi.org/10.1016/j.mad.2010.03.007 [PubMed]
- 24. Lexell J, Taylor CC, Sjöström M. What is the cause of the ageing atrophy? Total number, size and proportion of different fiber types studied in whole vastus lateralis muscle from 15- to 83-year-old men. J Neurol Sci. 1988; 84:275–94. https://doi.org/10.1016/0022-510x(88)90132-3 [PubMed]
- 25. Joseph AM, Adhihetty PJ, Buford TW, Wohlgemuth SE, Lees HA, Nguyen LM, Aranda JM, Sandesara BD, Pahor M, Manini TM, Marzetti E, Leeuwenburgh C. The impact of aging on mitochondrial function and biogenesis pathways in skeletal muscle of sedentary high- and low-functioning elderly individuals. Aging Cell. 2012; 11:801–9. https://doi.org/10.1111/j.1474-9726.2012.00844.x [PubMed]
- 26. Lexell J, Taylor CC. Variability in muscle fibre areas in whole human quadriceps muscle: effects of increasing age. J Anat. 1991; 174:239–49. [PubMed]
- 27. Young C, Miller E, Nicklous DM, Hoffman JR. Nerve growth factor and neurotrophin-3 affect functional recovery following peripheral nerve injury differently. Restor Neurol Neurosci. 2001; 18:167–75. [PubMed]
- 28. Handschin C, Spiegelman BM. Peroxisome proliferator-activated receptor gamma coactivator 1 coactivators, energy homeostasis, and metabolism. Endocr Rev. 2006; 27:728–35. https://doi.org/10.1210/er.2006-0037 [PubMed]
- 29. Lin J, Wu H, Tarr PT, Zhang CY, Wu Z, Boss O, Michael LF, Puigserver P, Isotani E, Olson EN, Lowell BB, Bassel-Duby R, Spiegelman BM. Transcriptional co-activator PGC-1 alpha drives the formation of slow-twitch muscle fibres. Nature. 2002; 418:797–801. https://doi.org/10.1038/nature00904 [PubMed]
- 30. Kang C, Chung E, Diffee G, Ji LL. Exercise training attenuates aging-associated mitochondrial dysfunction in rat skeletal muscle: role of PGC-1α. Exp Gerontol. 2013; 48:1343–50. https://doi.org/10.1016/j.exger.2013.08.004 [PubMed]
- 31. Krämer DK, Ahlsén M, Norrbom J, Jansson E, Hjeltnes N, Gustafsson T, Krook A. Human skeletal muscle fibre type variations correlate with PPAR alpha, PPAR delta and PGC-1 alpha mRNA. Acta Physiol (Oxf). 2006; 188:207–16. https://doi.org/10.1111/j.1748-1716.2006.01620.x [PubMed]
- 32. Lanza IR, Short DK, Short KR, Raghavakaimal S, Basu R, Joyner MJ, McConnell JP, Nair KS. Endurance exercise as a countermeasure for aging. Diabetes. 2008; 57:2933–42. https://doi.org/10.2337/db08-0349 [PubMed]
- 33. Su J, Ekman C, Oskolkov N, Lahti L, Ström K, Brazma A, Groop L, Rung J, Hansson O. A novel atlas of gene expression in human skeletal muscle reveals molecular changes associated with aging. Skelet Muscle. 2015; 5:35. https://doi.org/10.1186/s13395-015-0059-1 [PubMed]
- 34. Tsai S, Sitzmann JM, Dastidar SG, Rodriguez AA, Vu SL, McDonald CE, Academia EC, O'Leary MN, Ashe TD, La Spada AR, Kennedy BK. Muscle-specific 4E-BP1 signaling activation improves metabolic parameters during aging and obesity. J Clin Invest. 2015; 125:2952–64. https://doi.org/10.1172/JCI77361 [PubMed]
- 35. Wu Z, Puigserver P, Andersson U, Zhang C, Adelmant G, Mootha V, Troy A, Cinti S, Lowell B, Scarpulla RC, Spiegelman BM. Mechanisms controlling mitochondrial biogenesis and respiration through the thermogenic coactivator PGC-1. Cell. 1999; 98:115–24. https://doi.org/10.1016/S0092-8674(00)80611-X [PubMed]
- 36. Rowe GC, El-Khoury R, Patten IS, Rustin P, Arany Z. PGC-1α is dispensable for exercise-induced mitochondrial biogenesis in skeletal muscle. PLoS One. 2012; 7:e41817. https://doi.org/10.1371/journal.pone.0041817 [PubMed]
- 37. Barbieri E, Agostini D, Polidori E, Potenza L, Guescini M, Lucertini F, Annibalini G, Stocchi L, De Santi M, Stocchi V. The pleiotropic effect of physical exercise on mitochondrial dynamics in aging skeletal muscle. Oxid Med Cell Longev. 2015; 2015:917085. https://doi.org/10.1155/2015/917085 [PubMed]
- 38. Parise G, Brose AN, Tarnopolsky MA. Resistance exercise training decreases oxidative damage to DNA and increases cytochrome oxidase activity in older adults. Exp Gerontol. 2005; 40:173–80. https://doi.org/10.1016/j.exger.2004.09.002 [PubMed]
- 39. Melov S, Tarnopolsky MA, Beckman K, Felkey K, Hubbard A. Resistance exercise reverses aging in human skeletal muscle. PLoS One. 2007; 2:e465. https://doi.org/10.1371/journal.pone.0000465 [PubMed]
- 40. Juffer P, Jaspers RT, Lips P, Bakker AD, Klein-Nulend J. Expression of muscle anabolic and metabolic factors in mechanically loaded MLO-Y4 osteocytes. Am J Physiol Endocrinol Metab. 2012; 302:E389–95. https://doi.org/10.1152/ajpendo.00320.2011 [PubMed]
- 41. Ying Z, Roy RR, Edgerton VR, Gómez-Pinilla F. Exercise restores levels of neurotrophins and synaptic plasticity following spinal cord injury. Exp Neurol. 2005; 193:411–9. https://doi.org/10.1016/j.expneurol.2005.01.015 [PubMed]
- 42. Wang BL, Jin H, Han XQ, Xia Y, Liu NF. Involvement of brain-derived neurotrophic factor in exercise-induced cardioprotection of post-myocardial infarction rats. Int J Mol Med. 2018; 42:2867–80. https://doi.org/10.3892/ijmm.2018.3841 [PubMed]
- 43. Domínguez-Sanchéz MA, Bustos-Cruz RH, Velasco-Orjuela GP, Quintero AP, Tordecilla-Sanders A, Correa-Bautista JE, Triana-Reina HR, García-Hermoso A, González-Ruíz K, Peña-Guzmán CA, Hernández E, Peña-Ibagon JC, Téllez-T LA, et al. Acute Effects of High Intensity, Resistance, or Combined Protocol on the Increase of Level of Neurotrophic Factors in Physically Inactive Overweight Adults: The BrainFit Study. Front Physiol. 2018; 9:741. https://doi.org/10.3389/fphys.2018.00741 [PubMed]
- 44. Sharma NK, Ryals JM, Gajewski BJ, Wright DE. Aerobic exercise alters analgesia and neurotrophin-3 synthesis in an animal model of chronic widespread pain. Phys Ther. 2010; 90:714–25. https://doi.org/10.2522/ptj.20090168 [PubMed]
- 45. Sekimoto M, Tsuji T, Matsuzaki J, Chamoto K, Koda T, Nemoto K, Degawa M, Nishimura S, Nishimura T. Functional expression of the TrkC gene, encoding a high affinity receptor for NT-3, in antigen-specific T helper type 2 (Th2) cells. Immunol Lett. 2003; 88:221–6. https://doi.org/10.1016/s0165-2478(03)00080-4 [PubMed]
- 46. Beutner C, Lepperhof V, Dann A, Linnartz-Gerlach B, Litwak S, Napoli I, Prinz M, Neumann H. Engineered stem cell-derived microglia as therapeutic vehicle for experimental autoimmune encephalomyelitis. Gene Ther. 2013; 20:797–806. https://doi.org/10.1038/gt.2012.100 [PubMed]
- 47. Yang J, Yan Y, Xia Y, Kang T, Li X, Ciric B, Xu H, Rostami A, Zhang GX. Neurotrophin 3 transduction augments remyelinating and immunomodulatory capacity of neural stem cells. Mol Ther. 2014; 22:440–50. https://doi.org/10.1038/mt.2013.241 [PubMed]
- 48. Ozes B, Myers M, Moss K, Mckinney J, Ridgley A, Chen L, Bai S, Abrams CK, Freidin MM, Mendell JR, Sahenk Z. AAV1.NT-3 gene therapy for X-linked Charcot-Marie-Tooth neuropathy type 1. Gene Ther. 2022; 29:127–37. https://doi.org/10.1038/s41434-021-00231-3 [PubMed]
- 49. Lin W, Burgess RW, Dominguez B, Pfaff SL, Sanes JR, Lee KF. Distinct roles of nerve and muscle in postsynaptic differentiation of the neuromuscular synapse. Nature. 2001; 410:1057–64. https://doi.org/10.1038/35074025 [PubMed]
- 50. Valdez G, Heyer MP, Feng G, Sanes JR. The role of muscle microRNAs in repairing the neuromuscular junction. PLoS One. 2014; 9:e93140. https://doi.org/10.1371/journal.pone.0093140 [PubMed]
- 51. Seburn KL, Nangle LA, Cox GA, Schimmel P, Burgess RW. An active dominant mutation of glycyl-tRNA synthetase causes neuropathy in a Charcot-Marie-Tooth 2D mouse model. Neuron. 2006; 51:715–26. https://doi.org/10.1016/j.neuron.2006.08.027 [PubMed]
- 52. Wang L, Xu X, Jiang C, Ma G, Huang Y, Zhang H, Lai Y, Wang M, Ahmed T, Lin R, Guo W, Luo Z, Li W, et al. mTORC1-PGC1 axis regulates mitochondrial remodeling during reprogramming. FEBS J. 2020; 287:108–21. https://doi.org/10.1111/febs.15024 [PubMed]
- 53. Chaignat E, Yahya-Graison EA, Henrichsen CN, Chrast J, Schütz F, Pradervand S, Reymond A. Copy number variation modifies expression time courses. Genome Res. 2011; 21:106–13. https://doi.org/10.1101/gr.112748.110 [PubMed]
- 54. Jousse C, Muranishi Y, Parry L, Montaurier C, Even P, Launay JM, Carraro V, Maurin AC, Averous J, Chaveroux C, Bruhat A, Mallet J, Morio B, Fafournoux P. Perinatal protein malnutrition affects mitochondrial function in adult and results in a resistance to high fat diet-induced obesity. PLoS One. 2014; 9:e104896. https://doi.org/10.1371/journal.pone.0104896 [PubMed]
- 55. Python Software Foundation. 2022. https://www.python.org/.