Introduction
With the social trend to delay childbearing because of careers, the treatment of older infertile female has become a major challenge for today’s fertility specialists. Female fertility decreases with age. A decline in oocyte quality plays a major role in reproductive problems in older women. Whether advanced age is related to the decline of endometrial receptivity (ER) remains controversial. One study reported that in women aged >40 years, both embryo viability and, to a lesser extent, ER were decreased [1].
Oocyte donation (OD) affords a scientific model to study the impact of advanced age on ER. Animal experiments have shown age-related changes in the uterus, accompanied by a decrease in implantation and pregnancy rates. Despite transferring embryos from younger animals, older animals were ultimately unable to conceive [2]. When a similar experiment was carried out in humans by OD from young healthy women to older infertile recipients, conflicting results were obtained. Some investigators have concluded that a satisfactory embryo implantation rate (IR) in women with advanced maternal age (AMA) implies that the uterine factors are not involved [3–9], while others have found decreased pregnancy rate, implantation rate, and increased miscarriage rate in recipients with advanced age, suggesting that fertility does not depend merely on oocyte age and quality, but also on uterine age [10–14].
Many clinical trials have studied the impact of AMA on the ER and results after OD. There have been no systematic reviews and meta-analyses on this issue.
Currently, there is a pressing need for a systematic review and meta-analysis to evaluate the clinical question of the relationship between AMA and the decline of ER with the OD model.
Results
Studies selection and characteristics
In total, 352 articles were obtained using this research strategy. Of these, 317 articles were excluded because they were found irrelevant after reviewing their titles and abstracts. Of the remaining 35 studies, 17 were excluded for different reasons: 15 were reviews and 2 had incomplete data. Finally, 18 studies were included in the present study (Figure 1).
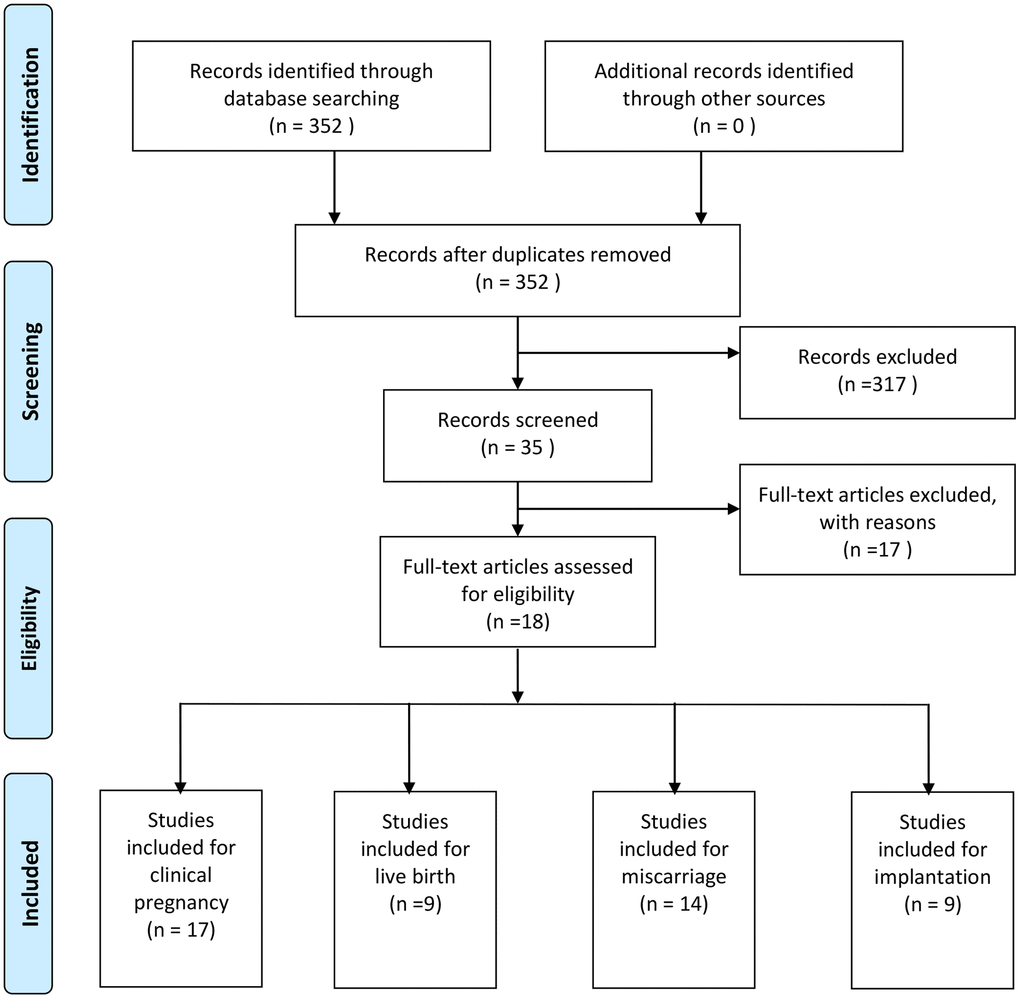
Figure 1. Flow chart showing study selection process.
Eighteen eligible studies were published from 1991 to 2005, including 11 retrospective studies, 5 prospective studies, and 2 studies without a study design. The sample size ranged from 22 to 3089. Of the 18 studies, 17, 8, 14, and 9th studies reported the impact of AMA on clinical pregnancy rate (CPR), IR, miscarriage rate (MR), and live birth rate (LBR) (Table 1).
Table 1. Characteristics of included studies.
| Study | Country | Design | Model | Protocol for COS | Protocol for ET | Sample size(cycles) | Group | Age for AMA | Outcomes |
| Soares 2005 | Spain | Retro | OD | A long protocol | HRT with or without GnRH-a | 3089 | <40y; 40-44y; 45-49y; >49y | 45 | CPR, IR, MR, obstetric outcomes |
| Moomjy 1999 | USA | Retro | OD | GnRH-a long protocol | HRT with or without GnRH-a | 370 | ≤34y; 35-42y;≥ 43y | 42 | CPR, MR |
| Yaron 1998 | Israel | Pros | OD | HMG alone or with GnRH-a | HRT | 1001 | <30y; 31-40y; >40y | 40 | CPR, MR, LBR |
| Abdalla 1997 | UK | Retro | OD, 2 recipients from different age group shared oocytes from a donor | Intranasal Buserelin + HMG | HRT with or without GnRH-a | 104 | ≤39y; ≥40y | 40 | CPR, IR, LBR |
| Borini 1996 | Italy | Retro | OD, Recipients of different ages shared oocytes from single donor | Buserelin/LA+FSH/HMG | HRT | 114 | ≤39y; 40-49y | 40 | CPR, IR, MR |
| Cano 1995 | Spain | Pros | OD, Recipients of different ages shared oocytes from same cohort of follicles | Long protocol with LA+HMG/FSH | HRT | 90 | <40y; ≥40y | 40 | CPR, IR, MR |
| Legro 1995 | USA | Retro | OD | Long protocol with LA + HMG | HRT | 307 | ≤42y; >42y | 42 | Ongoing-PR, MR |
| Rosewaks 1995 | USA | / | Younger IVF-ET donor to older recipients | / | HRT | 48 | / | / | Ongoing-PR, IR |
| Balmaceda1994 | USA | Retro | Donor to recipients with POF or poor responder | Long protocol with LA + HMG | HRT | 258<189 fresh and 69 frozen) | ≤30y;31-35y;36-40y;41-45y; 46-53y | 40y | CPR, IR, MR |
| Check 1994 | New Jersey | Retro | Infertile women undergoing IVF shared 50% oocytes to recipients | Long protocol with LA +HMG | HRT | 121 | <40y; ≥40y | 40y | CPR, LBR |
| Navot 1994 | USA | Pros | Younger donor to older recipients | HMG or concomitant menotropins and a GnRH-a | HRT | 89 | 42.7±0.3 vs. 33.4±0.7 | / | CPR, IR, MR, LBR |
| Sauer 1994 | USA | Retro | Fertile young women donate Oocyte to women with different age | COH with LA and HMG | HRT | 300 | <30y;30-39y;40-49y.50-59y | 40 | IR, LBR |
| Abdalla 1993 | UK | Retro | Oocyte donation | Intra-nasal buserelin and HMG | HRT with or without GnRH-a | 371 | 25-29y;30-34y;35-39y;40-44y;45-49y | 40 | CPR, MR |
| Flamigni 1993 | Italy | Retro | Oocyte donation | COH with buserelin/LA+FSH/HMG | HRT | 141 | 21-35y; 36-40y; 41-49y; 50-61y | 40y | CPR, IR, MR |
| Meldrum 1993 | USA | / | Oocyte donation | COH with LA+HMG | HRT with or without LA | 52 | <40y; ≥40y | 40y | CPR |
| Yaron 1993 | Israel | Retro | Oocyte donation | HMG | HRT | 458 | <40y; ≥40y | 40y | CPR, MR |
| Navot 1991 | USA | Pros | Oocyte donation | HMG or concomitant menotropins and GnRH-a | HRT | 89 | <35y; >40y | 40y | CPR, LBR |
| Sauer 1991 | USA | Pros | Oocyte donation | COH with leuprolide and HMG | HRT | 22 | <40y;40-44y | 40y | CPR, IR, LBR |
Meta-analysis
First, we evaluated the impact of AMA on CPR in infertile women undergoing assisted reproductive technology treatment using the OD model. Seventeen studies were included in this meta-analysis. The results showed a slightly lower CPR in women with AMA than in younger women (RR, 0.92; 95% CI, 0.82, 1.03; P=0.16). I2, which was used to describe the heterogeneity of the included studies, was 52%, indicating statistical heterogeneity in the results (P=0.007). Therefore, the random-effects model was used (Figure 2).
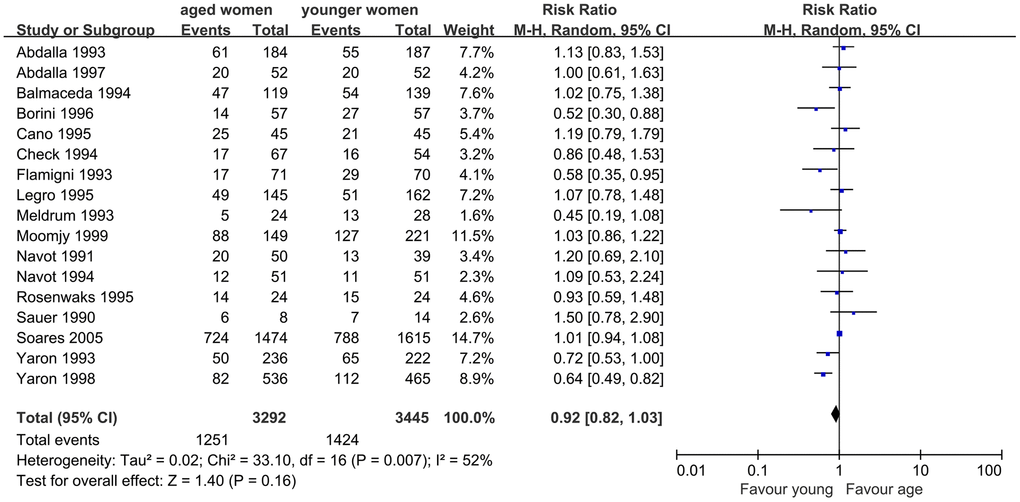
Figure 2. Forest plot showing the results of meta-analysis of studies comparing the effect of AMA on clinical pregnancy rate after OD treatment.
Similarly, nine studies were included to assess the impact of AMA on embryo implantation. The results of the meta-analysis showed similar IR in women with AMA and younger women (RR, 0.85; 95% CI, 0.69, 1.05; P=0.14). I2 was 59%, indicating moderate heterogeneity (P=0.01), and a random-effects model was used (Figure 3).
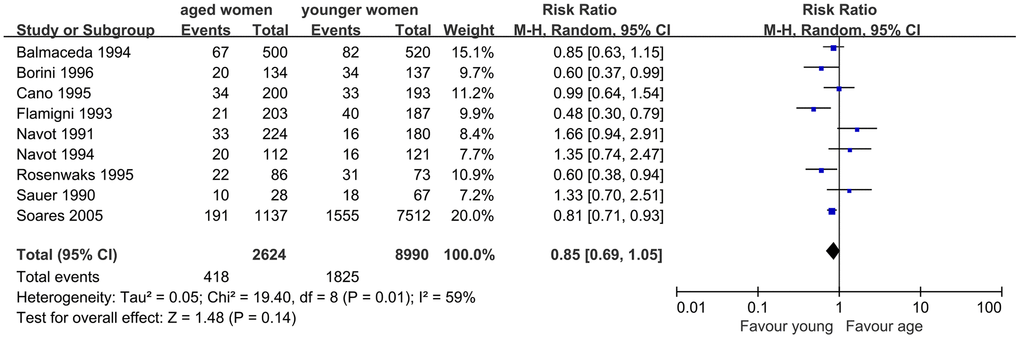
Figure 3. Forest plot showing the results of meta-analysis of studies comparing the effect of AMA on embryo implantation rate after OD treatment.
When we evaluated the impact of AMA on MR, 14 studies were included. The results indicated a significantly higher MR in infertile women with AMA than in younger women. The Q statistic P > 0.1, indicated the homogeneity of the studies (I2=0%, P=0.56). The fixed effects model was used and the pooled RR was 1.37 (95% CI, 1.13, 1.67; P=0.002) (Figure 4).
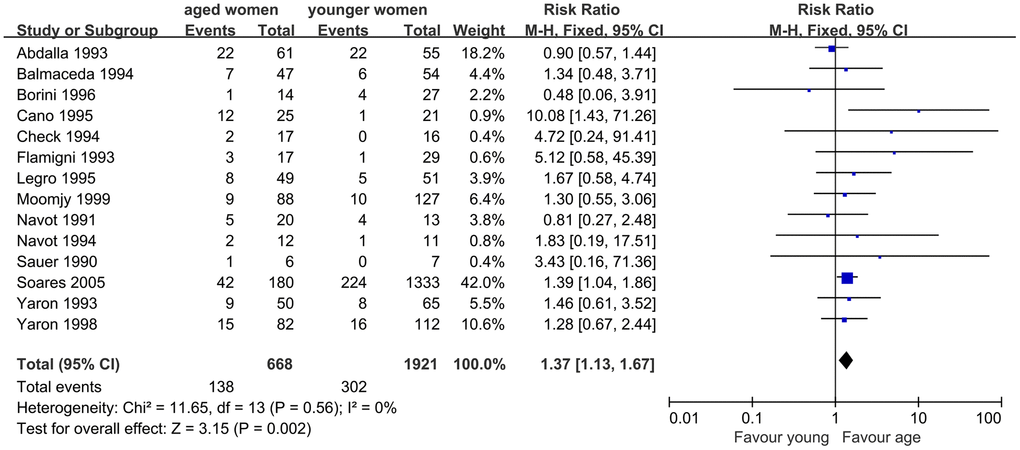
Figure 4. Forest plot showing the results of meta-analysis of studies comparing the effect of AMA on miscarriage rate after OD treatment.
Additionally, LBR was evaluated, and nine studies were included. The results of meta-analysis showed no significant difference in LBR between women with AMA and younger women. Good homogeneity was observed in the results (I2=22%, P=0.25). The fixed effect model combined RR was 0.77 (95% CI, 0.65, 0.91, P=0.002) (Figure 5).
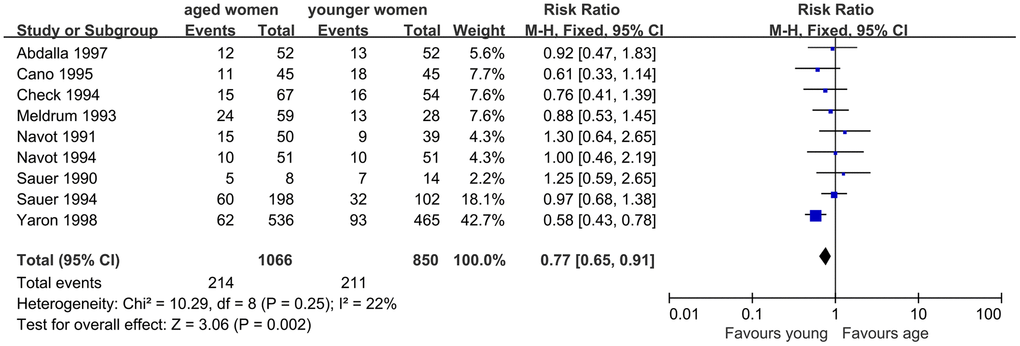
Figure 5. Forest plot showing the results of meta-analysis of studies comparing the effect of AMA on live birth rate after OD treatment.
The results included in this meta-analysis scored medium to high on the Newcastle-Ottawa Scale (not shown). The funnel plots evaluating the impact of AMA on CPR, IR, MR, and LBR suggest a lack of publication bias due to their symmetrical shape (Supplementary Figures 1–4).
To the best of our knowledge, this study is the first systematic review and meta-analysis to assess the impact of AMA on ER using an OD model. The results showed a trend toward a lower IR and CPR without significance; however, significantly increased MR and decreased LBR were observed in women with AMA, indicating that ER was negatively affected by advanced maternal age.
It is well known that fecundity declines in women with AMA are associated with decreased oocyte quality. However, there is no consistent conclusion regarding whether ER is also impaired in women with AMA. OD is considered a perfect model for ascertaining the extent of this relationship. Some studies have evaluated the impact of AMA on endometrial tissues using an OD model.
Early in 1990, one study explored whether ER decreased in older women. In this study, women aged 40-44 years with ovarian failure were enrolled and underwent embryo transfer with OD. These results suggest that the endometrium retains the ability to respond to gonadal steroids and receptivity for embryo implantation, even in older women [6]. Other similar studies also suggested that the age-related decline in female fertility has been attributed to oocyte quality and can be corrected by OD [4], showed similar PR, cumulative PR, and LBR in different age groups, and failed to detect any impact of age on pregnancy outcome in the OD model [15–18].
Other studies have compared pregnancy and implantation rates in oocyte recipients of different ages and showed significant differences in pregnancy and implantation rates according to the age of recipients, suggesting that the ER decreased with age [9, 10, 13]. Some studies reported significantly decreased PR [19] and IR [20, 21], significantly increased MR [11], and worse obstetric outcome [8] in women of advanced age. These discrepancies may be attributed to differences in patients’ age, body mass index (BMI), country, indication of OD, analysis method, and study design.
Although there were discrepancies among the studies, the pooled results suggest that AMA may have a negative effect on the ER. As donated oocytes are obtained from young women, the age-related decline in LBR and increased MR with OD cannot be attributed to oocyte quality. Possible explanations for our findings are as follows.
First, there is an age-related decline in ER. An in vitro experiment found that the expression of HOXA10, a marker of ER, was inversely correlated with uterine age [22]. An animal study compared mRNA levels of endometrial cells in vitro obtained from young and aged cows using next generation sequencing (NGS) and polymerase chain reaction (PCR), and found that endometrial cells of aged cows have higher levels of inflammatory, IFN-signaling, and cell division dysfunction than those of young cows [23]. In human, it has also been reported that placenta in women with AMA is associated with premature senescence during placentation due to SIRT1 deficiency, which promotes epithelial-mesenchymal transition of trophoblast cells and enhances the invasion of trophoblast cells by regulating vimentin acetylation [24]. Older women with decreased serum anti-mullerian hormone (AMH) and antral follicle count (AFC) levels showed significantly lower endometrial vascularization index, flow index, and vascularization flow index, and lower CPR and ongoing PR, indicating impaired ER [25].
Second, another possible explanation for our results may be embryo quality. The oocytes were donated by younger women, which did not contribute to poor embryo quality. However, advanced paternal age may be a reason for poor embryo quality. New dominant mutations, which may be embryologically fatal, are now known to be common in men of advanced age. Thus, it is reasonable to assume that the male partners of older recipients are more likely to be older, and such new dominant mutation may lead to decreased embryo quality [19].
Third, the risk of adverse pregnancy outcomes, such as gestational diabetes, preeclampsia, stillbirth, intrauterine growth restriction, and placenta previa, markedly increased among women with AMA. The increased complications may be related to impaired placentation function and progressive uterine vascular endothelial damage with aging [26–28].
The strength of this study is that it is the first systematic review and meta-analysis to describe the relationship between AMA and ER decline. The sample size was very large (7037 women), which provided an excellent precision for estimates with pooled RRs.
This study had several limitations. First, there was significant between-study heterogeneity, such as different study designs (prospective or retrospective studies), varied definition for AMA (40 years, 42 years, or 45 years), and different endometrium preparation protocol. In addition, most of the studies were retrospective in design, and there were residual confounding factors. Finally, some of the included studies had a small sample size. Despite these drawbacks, this systematic review and meta-analysis provide a valuable analysis and summary of the relevant literatures.
In conclusion, this study found that AMA is related to a decline in ER. Because of the small sample size and the possibility of aneuploidy embryos, further prospective cohort studies using the preimplanation genetic testing-Aneuploid (PGT-A) model are needed to observe the impact of AMA and analyze the possible causes.
Materials and Methods
This study was conducted in accordance with the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. As the data were extracted from previously published studies, and our paper did not include animal and human studies, institutional review board approval was an exemption.
Search strategy
A comprehensive search of PubMed, EMBASE, and Google Scholar was conducted from their inception dates until May 2022. The keywords used for the search were as follows: a term including advanced age (advanced maternal age, older woman, aging woman) and a term that included the endometrial receptivity (uterine receptivity). These subsets were summarized with “AND” to obtain the most complete literatures related to our study object. Cohort, retrospective, and prospective studies published in English were included. Full-text review and data extraction were completed by two separate reviewers, and any disagreements were resolved by consensus or by a third reviewer.
Study selection and data extraction
After reviewing the retrieved titles and abstracts, irrelevant studies were assessed using the Newcastle-Ottawa Quality Assessment Scale. Data were extracted by two authors independently using pre-defined criteria. Data extraction included the research features and results.
Statistical analysis
Review Manager Version 5.3 was used for meta-analysis. Categorical variables were calculated using the Mantel-Haenszel statistical method and expressed as risk ratio (RR); Forest plots were used to assess the heterogeneity of the included studies, and I2 was used to quantify the heterogeneity between studies. A fixed or random-effect model was used to calculate RR and its 95% confidence interval (CI). Because of the low power of the χ2 test for heterogeneity with a small sample size, a P value of 0.10 rather than 0.05 was considered to be statistically significant.
Supplementary Materials
Author Contributions
Jing Zhao participated in the design of the study and the acquisition of data, performed the statistical analysis, drafted the article and revised it critically. Yanping Li contributed to conception and design. Bixia Huang and Ning Li conducted acquisition of data and analysis and interpretation of data. Xiaofei Wang and Bin Xu participated in the interpretation of the data and the revision of the article. All authors read and approved the final manuscript.
Acknowledgments
We would like to thank Editage (https://www.editage.com) for English language editing.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
This project was supported by Natural Science Foundation of Hunan Province of China (No. 2021JJ31128).
References
- 1. Chetkowski RJ, Rode RA, Burruel V, Nass TE. The effect of pituitary suppression and the women’s age on embryo viability and uterine receptivity. Fertil Steril. 1991; 56:1095–103. [PubMed]
- 2. Sauer MV. The impact of age on reproductive potential: lessons learned from oocyte donation. Maturitas. 1998; 30:221–5. https://doi.org/10.1016/s0378-5122(98)00077-2 [PubMed]
- 3. Balmaceda JP, Bernardini L, Ciuffardi I, Felix C, Ord T, Sueldo CE, Asch RH. Oocyte donation in humans: a model to study the effect of age on embryo implantation rate. Hum Reprod. 1994; 9:2160–3. https://doi.org/10.1093/oxfordjournals.humrep.a138410 [PubMed]
- 4. Abdalla HI, Burton G, Kirkland A, Johnson MR, Leonard T, Brooks AA, Studd JW. Age, pregnancy and miscarriage: uterine versus ovarian factors. Hum Reprod. 1993; 8:1512–7. https://doi.org/10.1093/oxfordjournals.humrep.a138289 [PubMed]
- 5. Navot D, Bergh PA, Williams MA, Garrisi GJ, Guzman I, Sandler B, Grunfeld L. Poor oocyte quality rather than implantation failure as a cause of age-related decline in female fertility. Lancet. 1991; 337:1375–7. https://doi.org/10.1016/0140-6736(91)93060-m [PubMed]
- 6. Sauer MV, Paulson RJ, Lobo RA. A preliminary report on oocyte donation extending reproductive potential to women over 40. N Engl J Med. 1990; 323:1157–60. https://doi.org/10.1056/NEJM199010253231702 [PubMed]
- 7. Navot D, Drews MR, Bergh PA, Guzman I, Karstaedt A, Scott RT
Jr , Garrisi GJ, Hofmann GE. Age-related decline in female fertility is not due to diminished capacity of the uterus to sustain embryo implantation. Fertil Steril. 1994; 61:97–101. https://doi.org/10.1016/s0015-0282(16)56459-0 [PubMed] - 8. Soares SR, Troncoso C, Bosch E, Serra V, Simón C, Remohí J, Pellicer A. Age and uterine receptiveness: predicting the outcome of oocyte donation cycles. J Clin Endocrinol Metab. 2005; 90:4399–404. https://doi.org/10.1210/jc.2004-2252 [PubMed]
- 9. Abdalla HI, Wren ME, Thomas A, Korea L. Age of the uterus does not affect pregnancy or implantation rates; a study of egg donation in women of different ages sharing oocytes from the same donor. Hum Reprod. 1997; 12:827–9. https://doi.org/10.1093/humrep/12.4.827 [PubMed]
- 10. Check JH, Askari HA, Fisher C, Vanaman L. The use of a shared donor oocyte program to evaluate the effect of uterine senescence. Fertil Steril. 1994; 61:252–6. https://doi.org/10.1016/s0015-0282(16)56512-1 [PubMed]
- 11. Cano F, Simón C, Remohí J, Pellicer A. Effect of aging on the female reproductive system: evidence for a role of uterine senescence in the decline in female fecundity. Fertil Steril. 1995; 64:584–9. https://doi.org/10.1016/s0015-0282(16)57797-8 [PubMed]
- 12. Flamigni C, Borini A, Violini F, Bianchi L, Serrao L. Oocyte donation: comparison between recipients from different age groups. Hum Reprod. 1993; 8:2088–92. https://doi.org/10.1093/oxfordjournals.humrep.a137986 [PubMed]
- 13. Yaron Y, Botchan A, Amit A, Kogosowski A, Yovel I, Lessing JB. Endometrial receptivity: the age-related decline in pregnancy rates and the effect of ovarian function. Fertil Steril. 1993; 60:314–8. https://doi.org/10.1016/s0015-0282(16)56104-4 [PubMed]
- 14. Borini A, Bianchi L, Violini F, Maccolini A, Cattoli M, Flamigni C. Oocyte donation program: pregnancy and implantation rates in women of different ages sharing oocytes from single donor. Fertil Steril. 1996; 65:94–7. https://doi.org/10.1016/s0015-0282(16)58033-9 [PubMed]
- 15. Legro RS, Wong IL, Paulson RJ, Lobo RA, Sauer MV. Recipient’s age does not adversely affect pregnancy outcome after oocyte donation. Am J Obstet Gynecol. 1995; 172:96–100. https://doi.org/10.1016/0002-9378(95)90091-8 [PubMed]
- 16. Noyes N, Hampton BS, Berkeley A, Licciardi F, Grifo J, Krey L. Factors useful in predicting the success of oocyte donation: a 3-year retrospective analysis. Fertil Steril. 2001; 76:92–7. https://doi.org/10.1016/s0015-0282(01)01823-4 [PubMed]
- 17. Paulson RJ, Hatch IE, Lobo RA, Sauer MV. Cumulative conception and live birth rates after oocyte donation: implications regarding endometrial receptivity. Hum Reprod. 1997; 12:835–9. https://doi.org/10.1093/humrep/12.4.835 [PubMed]
- 18. Sauer MV, Paulson RJ, Ary BA, Lobo RA. Three hundred cycles of oocyte donation at the University of Southern California: assessing the effect of age and infertility diagnosis on pregnancy and implantation rates. J Assist Reprod Genet. 1994; 11:92–6. https://doi.org/10.1007/BF02215994 [PubMed]
- 19. Yaron Y, Ochshorn Y, Amit A, Kogosowski A, Yovel I, Lessing JB. Oocyte donation in Israel: a study of 1001 initiated treatment cycles. Hum Reprod. 1998; 13:1819–24. https://doi.org/10.1093/humrep/13.7.1819 [PubMed]
- 20. Moomjy M, Cholst I, Mangieri R, Rosenwaks Z. Oocyte donation: insights into implantation. Fertil Steril. 1999; 71:15–21. https://doi.org/10.1016/s0015-0282(98)00420-8 [PubMed]
- 21. Meldrum DR. Female reproductive aging--ovarian and uterine factors. Fertil Steril. 1993; 59:1–5. https://doi.org/10.1016/s0015-0282(16)55608-8 [PubMed]
- 22. Fogle RH, Li A, Paulson RJ. Modulation of HOXA10 and other markers of endometrial receptivity by age and human chorionic gonadotropin in an endometrial explant model. Fertil Steril. 2010; 93:1255–9. https://doi.org/10.1016/j.fertnstert.2008.11.002 [PubMed]
- 23. Tanikawa N, Ohtsu A, Kawahara-Miki R, Kimura K, Matsuyama S, Iwata H, Kuwayama T, Shirasuna K. Age-associated mRNA expression changes in bovine endometrial cells in vitro. Reprod Biol Endocrinol. 2017; 15:63. https://doi.org/10.1186/s12958-017-0284-z [PubMed]
- 24. Xiong L, Ye X, Chen Z, Fu H, Li S, Xu P, Yu J, Wen L, Gao R, Fu Y, Qi H, Kilby MD, Saffery R, et al. Advanced Maternal Age-associated SIRT1 Deficiency Compromises Trophoblast Epithelial-Mesenchymal Transition through an Increase in Vimentin Acetylation. Aging Cell. 2021; 20:e13491. https://doi.org/10.1111/acel.13491 [PubMed]
- 25. Wang L, Lv S, Mao W, Bai E, Yang X. Fecundity disorders in older women: declines in follicular development and endometrial receptivity. BMC Womens Health. 2020; 20:115. https://doi.org/10.1186/s12905-020-00979-7 [PubMed]
- 26. Cleary-Goldman J, Malone FD, Vidaver J, Ball RH, Nyberg DA, Comstock CH, Saade GR, Eddleman KA, Klugman S, Dugoff L, Timor-Tritsch IE, Craigo SD, Carr SR, et al, and FASTER Consortium. Impact of maternal age on obstetric outcome. Obstet Gynecol. 2005; 105:983–90. https://doi.org/10.1097/01.AOG.0000158118.75532.51 [PubMed]
- 27. Crawford NM, Steiner AZ. Age-related infertility. Obstet Gynecol Clin North Am. 2015; 42:15–25. https://doi.org/10.1016/j.ogc.2014.09.005 [PubMed]
- 28. Ethics Committee of the American Society for Reproductive Medicine. Electronic address: [email protected], and Ethics Committee of the American Society for Reproductive Medicine. Oocyte or embryo donation to women of advanced reproductive age: an Ethics Committee opinion. Fertil Steril. 2016; 106:e3–e7. https://doi.org/10.1016/j.fertnstert.2016.07.002 [PubMed]



