Introduction
Mitochondrial open reading frame of the 12S ribosomal RNA type-c (MOTS-c) is a mitochondrially-encoded 16-amino-acid biopeptide that functions as an exercise-induced regulator of metabolic homeostasis [1, 2] and a modulator of obesity-, diet-, and aging-dependent metabolic function by acting as a systemic, endocrine-acting mitokine [3, 4]. MOTS-c dynamically translocates to the nucleus in response to metabolic stress to directly regulate the expression of a broad spectrum of genes, including antioxidant response element-containing target genes [5]. By targeting folate-dependent de novo purine biosynthesis, MOTS-c boosts the levels of the endogenous AMP analog 5-aminomidazole-4-carboxamide ribonucleotide (AICAR), which in turn activates the master energy sensor AMP-activated protein kinase (AMPK). Skeletal muscle cell-targeted involvement of the folate-AICAR-AMPK pathway constitutes the mechanistic basis of MOTS-c as an endogenous “exercise mimetic”, which can stimulate glucose utilization and fat oxidation and suppress inflammation [4].
Exercise interventions show promise as effective adjunct strategies to prevent and/or attenuate chemotherapy-associated toxicity (e.g., cardiotoxicity and cardiopulmonary dysfunction) in patients with early-stage breast cancer (BC) [6, 7]. Exploratory studies have demonstrated that exercise interventions might also modulate host- and tumor-related pathways in patients on standard chemotherapy [8]. Indeed, several ongoing and planned interventional studies (e.g., Neo-ACT NCT05184582, Neo-Train NCT04623554) have been designed to examine whether physical exercise interventions during the neoadjuvant chemotherapy period can bolster treatment efficacy [9, 10]. Pharmacological therapeutics that partially mimic the systemic impact of exercise have also been proposed for those cancer patients for whom exercise training may not be an option [11–13]. One putative exercise mimetic, metformin, shares many mechanistic features with MOTS-c, including: (a) insulin sensitization, (b) enhancing glucose utilization, (c) suppressing mitochondrial respiration, and (d) targeting the folate-AICAR-AMPK pathway [4, 14–16]. To date, however, no study has examined whether metformin can influence the expression of MOTS-c in cancer patients. Here, we explored the impact of metformin on circulating MOTS-c levels in the METTEN study (EudraCT number 2011-000490-30), a phase 2 clinical trial of women with HER2-positive BC randomized to receive either metformin (850 mg twice daily) for 24 weeks concurrently with 12 cycles of weekly paclitaxel plus trastuzumab, followed by four cycles of 3-weekly FE75C plus trastuzumab (arm A), or an equivalent regimen without metformin (arm B), before surgery [17].
The present study was conducted with paired baseline and post-treatment serum samples collected from 38 patients (n=19 in each arm) belonging to the intention-to-treat population of the METTEN trial (Figure 1A, left), which included randomly assigned patients receiving at least one dose of study medication. All samples were evaluated in parallel for circulating MOTS-c using a commercially available competitive ELISA (CEX132Hu; CloudClone Corp., Wuhan, China) [18, 19]. Within- and between-group data were assessed by paired t-test and post hoc Tukey multiple comparison tests on repeated measures ANOVA. No statistically-significant differences were found between the pre- and post-levels of circulating MOTS-c irrespective of the treatment arm (Figure 1A, right). Similarly, changes in circulating MOTS-c levels failed to reach statistical significance when comparing patients achieving pathological complete response (pCR), defined as the absence of invasive tumor cells in post-neoadjuvant therapy surgical histopathology of the complete resected breast specimen, including sample regional lymph nodes [17], and patients with non-pCR, irrespective of the treatment arm (Figure 1B).
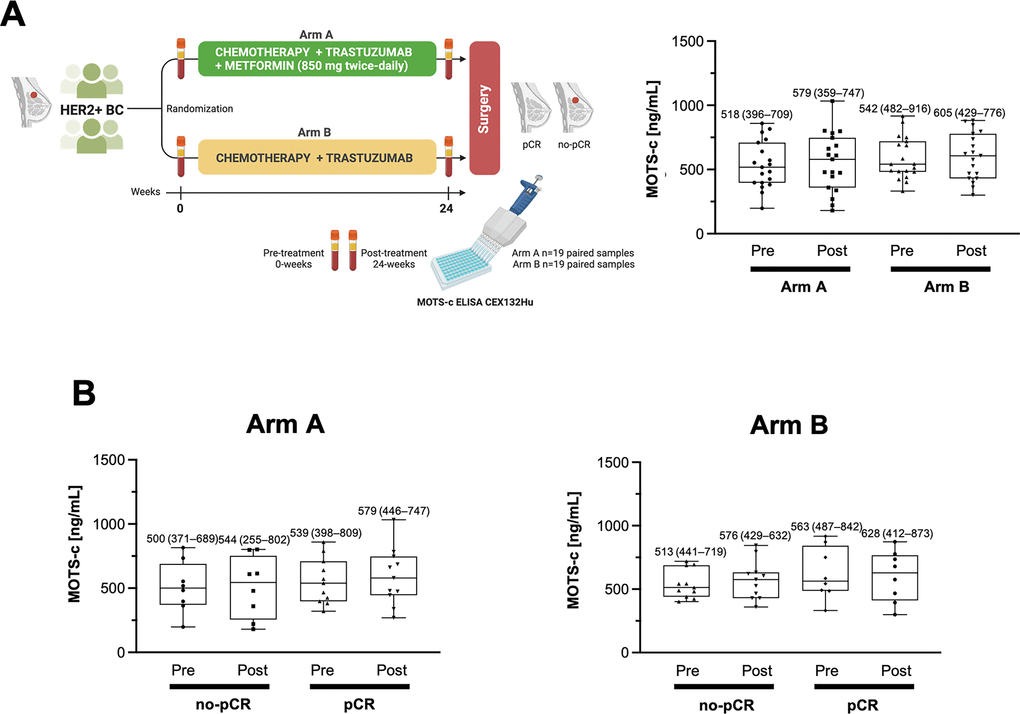
Figure 1. Circulating levels of MOTS-c in patients with HER2+ breast cancer treated with neoadjuvant metformin. (A) Left. METTEN study design. Circulating MOTS-c levels were determined through blood draws obtained at pre- (0 weeks) and post- (24 weeks) treatment using a commercial ELISA kit (CloudClone Corp., Wuhan, China; Catalog No. CEX132Hu). Right. Box plot (median, 25%–75% quartiles and minimal and maximal values) of the pre- and post-treatment distribution of circulating MOTS-c in women randomized to arms A (metformin-containing) and B (without metformin). (B) Box plot (median, 25%–75% quartiles and minimal and maximal values) of the pre- and post-treatment distribution of circulating MOTS-c in non-pCR and pCR groups. No between-group comparisons reached statistical significance in A and B.
Our findings indicate that metformin does not operate as an exercise mimetic to augment the circulating levels of MOTS-c in patients with BC treated with neoadjuvant therapy. It is possible that the lack of effect relates to different target tissues of metformin and MOTS-c. We have recently learned that an expansion of the skeletal muscle-derived MOTS-c protein pool occurs concomitantly with increases in mitochondrial DNA [20], which might suggest that circulating MOTS-c could operate as a surrogate marker of phenotypic and functional shifts of mitochondrial networks [21]. Metformin is known to activate AMPK when targeting the liver, kidney, and intestine but not skeletal muscle [22] –the major tissue for MOTS-c production and secretion–, which might prevent any regulatory effect on circulating MOTS-c. Moreover, we are accumulating evidence that metformin does not enhance (and instead dampens) the beneficial strength gains and muscle activation in response to exercise training in healthy elderly people [23–26]. The mechanisms of MOTS-c production, secretion, distribution, and metabolism in the human body remain to be fully elucidated. Likewise, the extent of involvement of various tissue targets and/or the effects of metformin on skeletal muscle metabolism and how they determine the pharmacodynamics and endogenous serum levels of MOTS-c in patients with BC await evaluation in future studies. One should acknowledge that plasma and muscle MOTS-c show opposing responses to aging in older men, thereby suggesting that the primary source of circulating MOTS-c is not skeletal muscle or the pharmacokinetics of MOTS-c changes with age [27]. Similarly, the ability of muscle cells to release MOTS-c can be impaired due to changes in the export process and/or to the exhausted capacity of muscle (or hepatic) cells to tolerate or adapt to systemic metabolic stress occurring in cancer patients. Therefore, we cannot exclude the possibility that serum circulating MOTS-c and muscle MOTS-c can be differentially regulated by metformin, as aging does [27].
There are several limitations to this study. Endogenous levels of circulating MOTS-c have been shown to vary significantly (from 154 pg/mL to 584 ng/mL) depending on the assay method used [5, 18, 19, 28–30]. In our series of patients, the endogenous serum levels of MOTS-c ranged from 181 ng/mL to 1033 ng/mL. Overall, these findings would strongly suggest that the immunoreactive species of circulating MOTS-c detected using different kits are not identical. Our previous analysis confirmed that treatment of non-diabetic patients with HER2+ BC with oral metformin (850 mg twice daily) for 24 weeks produced blood levels of circulating metformin of ~7 μmol/L, equivalent to those generally achieved in diabetic patients with the usual clinical doses and schedule [17]. The exercise-induced augmentation of circulating MOTS-c in young subjects was found to return to baseline after only 4 hours of resting [4]. As we measured circulating MOTS-c in blood that was not strictly timed in relation to the last preceding oral dose of metformin [17], our data need to be viewed cautiously in terms of association between metformin treatment, achieved serum concentration of MOTS-c, and probability of pCR in BC patients. Moreover, the METTEN trial was conducted in patients with the HER2+ subtype of breast cancer, which leaves open the question of whether the circulating levels of MOTS-c and/or the regulatory activity of metformin on MOTS-c might be different in patients with other BC subtypes, such as luminal A, HER2-negative luminal B or triple negative [31]. Nonetheless, this is a retrospective study in a small sample size for which the evaluation of MOTS-c was not part of the original study design. Care should therefore be taken in interpreting and generalizing these findings. With an ever-growing recognition of the role of MOTS-c on age-related diseases including diabetes, obesity, osteoporosis, cardiovascular, and neurodegenerative diseases [32, 33], further studies are needed to elucidate the nature of the interaction between metformin and MOTS-c in cancer and non-cancer patients.
Author Contributions
Conceptualization, E.C. and J.A.M.; methodology, E.C. and J.A.M.; formal analysis, E.C., S.V., and J.A.M.; investigation, E.C., S.V., B. M-C., and J.A.M.; validation, E.C., S.V., and J.A.M.; data curation, E.C., S.V., and J.A.M.; resources, E.C., B. M-C., and J.A.M.; visualization, E.C., S.V., and J.A.M.; writing—original draft preparation, E.C., B. M-C., and J.A.M.; writing—review and editing, J.A.M.; supervision, E.C. and J.A.M.; project administration, E.C.; funding acquisition, E.C., B. M-C., and J.A.M. All authors have read and agreed to the published version of the manuscript.
Conflicts of Interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
Work in the Menendez laboratory is supported by the Spanish Ministry of Science and Innovation (Grant PID2019-10455GB-I00, Plan Nacional de l+D+I, founded by the European Regional Development Fund, Spain). Elisabet Cuyàs holds a “Miguel Servet” research contract (CP20/00003) from the Instituto de Salud Carlos III (Spain) and is supported by the Spanish Ministry of Science and Innovation (Grant PI22/00297, Proyectos de I+D+I en Salud, Acción Estratégica en Salud 2021-2023, founded by the European Regional Development Fund, Spain).
References
- 1. Lee C, Kim KH, Cohen P. MOTS-c: A novel mitochondrial-derived peptide regulating muscle and fat metabolism. Free Radic Biol Med. 2016; 100:182–7. https://doi.org/10.1016/j.freeradbiomed.2016.05.015 [PubMed]
- 2. Benayoun BA, Lee C. MOTS-c: A Mitochondrial-Encoded Regulator of the Nucleus. Bioessays. 2019; 41:e1900046. https://doi.org/10.1002/bies.201900046 [PubMed]
- 3. Lee C, Zeng J, Drew BG, Sallam T, Martin-Montalvo A, Wan J, Kim SJ, Mehta H, Hevener AL, de Cabo R, Cohen P. The mitochondrial-derived peptide MOTS-c promotes metabolic homeostasis and reduces obesity and insulin resistance. Cell Metab. 2015; 21:443–54. https://doi.org/10.1016/j.cmet.2015.02.009 [PubMed]
- 4. Reynolds JC, Lai RW, Woodhead JST, Joly JH, Mitchell CJ, Cameron-Smith D, Lu R, Cohen P, Graham NA, Benayoun BA, Merry TL, Lee C. MOTS-c is an exercise-induced mitochondrial-encoded regulator of age-dependent physical decline and muscle homeostasis. Nat Commun. 2021; 12:470. https://doi.org/10.1038/s41467-020-20790-0 [PubMed]
- 5. Kim KH, Son JM, Benayoun BA, Lee C. The Mitochondrial-Encoded Peptide MOTS-c Translocates to the Nucleus to Regulate Nuclear Gene Expression in Response to Metabolic Stress. Cell Metab. 2018; 28:516–24.e7. https://doi.org/10.1016/j.cmet.2018.06.008 [PubMed]
- 6. Hornsby WE, Douglas PS, West MJ, Kenjale AA, Lane AR, Schwitzer ER, Ray KA, Herndon JE 2nd, Coan A, Gutierrez A, Hornsby KP, Hamilton E, Wilke LG, et al. Safety and efficacy of aerobic training in operable breast cancer patients receiving neoadjuvant chemotherapy: a phase II randomized trial. Acta Oncol. 2014; 53:65–74. https://doi.org/10.3109/0284186X.2013.781673 [PubMed]
- 7. Jones LW, Fels DR, West M, Allen JD, Broadwater G, Barry WT, Wilke LG, Masko E, Douglas PS, Dash RC, Povsic TJ, Peppercorn J, Marcom PK, et al. Modulation of circulating angiogenic factors and tumor biology by aerobic training in breast cancer patients receiving neoadjuvant chemotherapy. Cancer Prev Res (Phila). 2013; 6:925–37. https://doi.org/10.1158/1940-6207.CAPR-12-0416 [PubMed]
- 8. Loughney L, West MA, Kemp GJ, Grocott MP, Jack S. Exercise intervention in people with cancer undergoing neoadjuvant cancer treatment and surgery: A systematic review. Eur J Surg Oncol. 2016; 42:28–38. https://doi.org/10.1016/j.ejso.2015.09.027 [PubMed]
- 9. Ginzac A, Bernadach M, Molnar I, Duclos M, Thivat E, Durando X. Adapted Physical Activity for Breast Cancer Patients Treated with Neoadjuvant Chemotherapy and Trastuzumab Against HER2 (APACAN2): A Protocol for a Feasibility Study. Front Oncol. 2021; 11:744609. https://doi.org/10.3389/fonc.2021.744609 [PubMed]
- 10. Guerrieri D, Moon HY, van Praag H. Exercise in a Pill: The Latest on Exercise-Mimetics. Brain Plast. 2017; 2:153–69. https://doi.org/10.3233/BPL-160043 [PubMed]
- 11. Fan W, Evans RM. Exercise Mimetics: Impact on Health and Performance. Cell Metab. 2017; 25:242–7. https://doi.org/10.1016/j.cmet.2016.10.022 [PubMed]
- 12. Hawley JA, Joyner MJ, Green DJ. Mimicking exercise: what matters most and where to next? J Physiol. 2021; 599:791–802. https://doi.org/10.1113/JP278761 [PubMed]
- 13. Corominas-Faja B, Quirantes-Piné R, Oliveras-Ferraros C, Vazquez-Martin A, Cufí S, Martin-Castillo B, Micol V, Joven J, Segura-Carretero A, Menendez JA. Metabolomic fingerprint reveals that metformin impairs one-carbon metabolism in a manner similar to the antifolate class of chemotherapy drugs. Aging (Albany NY). 2012; 4:480–98. https://doi.org/10.18632/aging.100472 [PubMed]
- 14. Cuyàs E, Fernández-Arroyo S, Buxó M, Pernas S, Dorca J, Álvarez I, Martínez S, Pérez-Garcia JM, Batista-López N, Rodríguez-Sánchez CA, Amillano K, Domínguez S, Luque M, et al. Metformin induces a fasting- and antifolate-mimicking modification of systemic host metabolism in breast cancer patients. Aging (Albany NY). 2019; 11:2874–88. https://doi.org/10.18632/aging.101960 [PubMed]
- 15. Cuyàs E, Verdura S, Martin-Castillo B, Menendez JA. Metformin: Targeting the Metabolo-Epigenetic Link in Cancer Biology. Front Oncol. 2021; 10:620641. https://doi.org/10.3389/fonc.2020.620641 [PubMed]
- 16. Tramonti A, Cuyàs E, Encinar JA, Pietzke M, Paone A, Verdura S, Arbusà A, Martin-Castillo B, Giardina G, Joven J, Vazquez A, Contestabile R, Cutruzzolà F, Menendez JA. Metformin Is a Pyridoxal-5'-phosphate (PLP)-Competitive Inhibitor of SHMT2. Cancers (Basel). 2021; 13:4009. https://doi.org/10.3390/cancers13164009 [PubMed]
- 17. Martin-Castillo B, Pernas S, Dorca J, Álvarez I, Martínez S, Pérez-Garcia JM, Batista-López N, Rodríguez-Sánchez CA, Amillano K, Domínguez S, Luque M, Stradella A, Morilla I, et al. A phase 2 trial of neoadjuvant metformin in combination with trastuzumab and chemotherapy in women with early HER2-positive breast cancer: the METTEN study. Oncotarget. 2018; 9:35687–704. https://doi.org/10.18632/oncotarget.26286 [PubMed]
- 18. Yang B, Yu Q, Chang B, Guo Q, Xu S, Yi X, Cao S. MOTS-c interacts synergistically with exercise intervention to regulate PGC-1α expression, attenuate insulin resistance and enhance glucose metabolism in mice via AMPK signaling pathway. Biochim Biophys Acta Mol Basis Dis. 2021; 1867:166126. https://doi.org/10.1016/j.bbadis.2021.166126 [PubMed]
- 19. Du C, Zhang C, Wu W, Liang Y, Wang A, Wu S, Zhao Y, Hou L, Ning Q, Luo X. Circulating MOTS-c levels are decreased in obese male children and adolescents and associated with insulin resistance. Pediatr Diabetes. 2018. [Epub ahead of print]. https://doi.org/10.1111/pedi.12685 [PubMed]
- 20. Malik AN, Czajka A. Is mitochondrial DNA content a potential biomarker of mitochondrial dysfunction? Mitochondrion. 2013; 13:481–92. https://doi.org/10.1016/j.mito.2012.10.011 [PubMed]
- 21. Hyatt JK. MOTS-c increases in skeletal muscle following long-term physical activity and improves acute exercise performance after a single dose. Physiol Rep. 2022; 10:e15377. https://doi.org/10.14814/phy2.15377 [PubMed]
- 22. Zhang CS, Li M, Wang Y, Li X, Zong Y, Long S, Zhang M, Feng JW, Wei X, Liu YH, Zhang B, Wu J, Zhang C, et al. The aldolase inhibitor aldometanib mimics glucose starvation to activate lysosomal AMPK. Nat Metab. 2022; 4:1369–401. https://doi.org/10.1038/s42255-022-00640-7 [PubMed]
- 23. Sharoff CG, Hagobian TA, Malin SK, Chipkin SR, Yu H, Hirshman MF, Goodyear LJ, Braun B. Combining short-term metformin treatment and one bout of exercise does not increase insulin action in insulin-resistant individuals. Am J Physiol Endocrinol Metab. 2010; 298:E815–23. https://doi.org/10.1152/ajpendo.00517.2009 [PubMed]
- 24. Malin SK, Gerber R, Chipkin SR, Braun B. Independent and combined effects of exercise training and metformin on insulin sensitivity in individuals with prediabetes. Diabetes Care. 2012; 35:131–6. https://doi.org/10.2337/dc11-0925 [PubMed]
- 25. Walton RG, Dungan CM, Long DE, Tuggle SC, Kosmac K, Peck BD, Bush HM, Villasante Tezanos AG, McGwin G, Windham ST, Ovalle F, Bamman MM, Kern PA, Peterson CA. Metformin blunts muscle hypertrophy in response to progressive resistance exercise training in older adults: A randomized, double-blind, placebo-controlled, multicenter trial: The MASTERS trial. Aging Cell. 2019; 18:e13039. https://doi.org/10.1111/acel.13039 [PubMed]
- 26. Kulkarni AS, Peck BD, Walton RG, Kern PA, Mar JC, Windham ST, Bamman MM, Barzilai N, Peterson CA. Metformin alters skeletal muscle transcriptome adaptations to resistance training in older adults. Aging (Albany NY). 2020; 12:19852–66. https://doi.org/10.18632/aging.104096 [PubMed]
- 27. D’Souza RF, Woodhead JST, Hedges CP, Zeng N, Wan J, Kumagai H, Lee C, Cohen P, Cameron-Smith D, Mitchell CJ, Merry TL. Increased expression of the mitochondrial derived peptide, MOTS-c, in skeletal muscle of healthy aging men is associated with myofiber composition. Aging (Albany NY). 2020; 12:5244–58. https://doi.org/10.18632/aging.102944 [PubMed]
- 28. Ramanjaneya M, Bettahi I, Jerobin J, Chandra P, Abi Khalil C, Skarulis M, Atkin SL, Abou-Samra AB. Mitochondrial-Derived Peptides Are Down Regulated in Diabetes Subjects. Front Endocrinol (Lausanne). 2019; 10:331. https://doi.org/10.3389/fendo.2019.00331 [PubMed]
- 29. Dieli-Conwright CM, Sami N, Norris MK, Wan J, Kumagai H, Kim SJ, Cohen P. Effect of aerobic and resistance exercise on the mitochondrial peptide MOTS-c in Hispanic and Non-Hispanic White breast cancer survivors. Sci Rep. 2021; 11:16916. https://doi.org/10.1038/s41598-021-96419-z [PubMed]
- 30. Knoop A, Thomas A, Thevis M. Development of a mass spectrometry based detection method for the mitochondrion-derived peptide MOTS-c in plasma samples for doping control purposes. Rapid Commun Mass Spectrom. 2019; 33:371–80. https://doi.org/10.1002/rcm.8337 [PubMed]
- 31. Martin-Castillo B, Lopez-Bonet E, Cuyàs E, Viñas G, Pernas S, Dorca J, Menendez JA. Cancer stem cell-driven efficacy of trastuzumab (Herceptin): towards a reclassification of clinically HER2-positive breast carcinomas. Oncotarget. 2015; 6:32317–38. https://doi.org/10.18632/oncotarget.6094 [PubMed]
- 32. Mohtashami Z, Singh MK, Salimiaghdam N, Ozgul M, Kenney MC. MOTS-c, the Most Recent Mitochondrial Derived Peptide in Human Aging and Age-Related Diseases. Int J Mol Sci. 2022; 23:11991. https://doi.org/10.3390/ijms231911991 [PubMed]
- 33. Miller B, Kim SJ, Kumagai H, Yen K, Cohen P. Mitochondria-derived peptides in aging and healthspan. J Clin Invest. 2022; 132:e158449. https://doi.org/10.1172/JCI158449 [PubMed]



