Introduction
Incorrect segregation of sister chromatids during mitosis may lead to aneuploidy which is found in many types of tumors [1, 2]. The Budding Uninhibited By Benzimidazoles (BUB1, BUB1B, and BUB3) gene family plays a central role in spindle checkpoint during mitosis [3–6].
BUB1 is required for chromosome congression, kinetochore localization, and the establishment and/or maintenance of efficient bipolar attachment to spindle microtubules [7, 8]. As a paralogous gene of BUB1, BUB1B associates with unattached/incorrectly attached kinetochores, stabilizes kinetochore-microtubule attachment, and helps ensure proper chromosome alignment [9, 10]. BUB3 works when binding with BUB1. When the spindle assembly checkpoint is activated, the BUB1/BUB3 complex is formed. It inhibits the ubiquitin ligase of APC/C (anaphase promoting complex C) by phosphorylation of its activator - CDC20 [11, 12].
In the process of mitosis, human cells show a protein phosphorylation peak that changes the behavior of a large group of proteins. In the meantime, process of mitosis also promotes the dramatic transformation of cell morphology, intracellular structure, and biochemistry [6]. Accurate chromosomal segregation is essential to mitosis. It ensures that chromosomes are properly aligned and attached to microtubules with their kinetochores at metaphase. Errors in this process may result in chromosomal mis-segregation and subsequent mitotic catastrophes, leading to chromosomal instability and the formation of tetraploid or aneuploid cells [13]. As a key mediator of the spindle checkpoint, deregulation of BUBs has been shown to be highly associated with various malignancies such as leukemia, gastric cancer, breast cancer, and liver cancer [6, 14–19].
Sarcoma is a malignant cancer consisting of a heterogeneous group of mesenchymal neoplasms [20]. The degradation of the mitotic checkpoint kinase BUB1 has been shown in Kaposi’s sarcoma [21]. However, bioinformatic analysis has not been applied to explore the role of the BUB family in other types of sarcomas. A comprehensive analysis of BUBs in sarcomas may provide a novel tool for the prognosis of the disease and may potentially be valuable therapeutic targets.
Through analysis of gene expression data from publicly-available datasets, we explored the expression of different BUB factors in patients with sarcoma to determine their expression patterns, distinct prognostic values, and potential functions in sarcoma.
Results
Transcriptional levels of BUBs in patients with sarcoma
Three BUB family members have been identified in patients with different types of sarcoma: BUB1, BUB1B and BUB3. They are overexpressed in all samples of sarcoma compared to normal tissue. The transcriptional levels of BUBs were compared between sarcoma and normal samples using Oncomine databases (Figure 1). The mRNA levels of these BUBs were significantly upregulated in patients with sarcoma in two datasets.
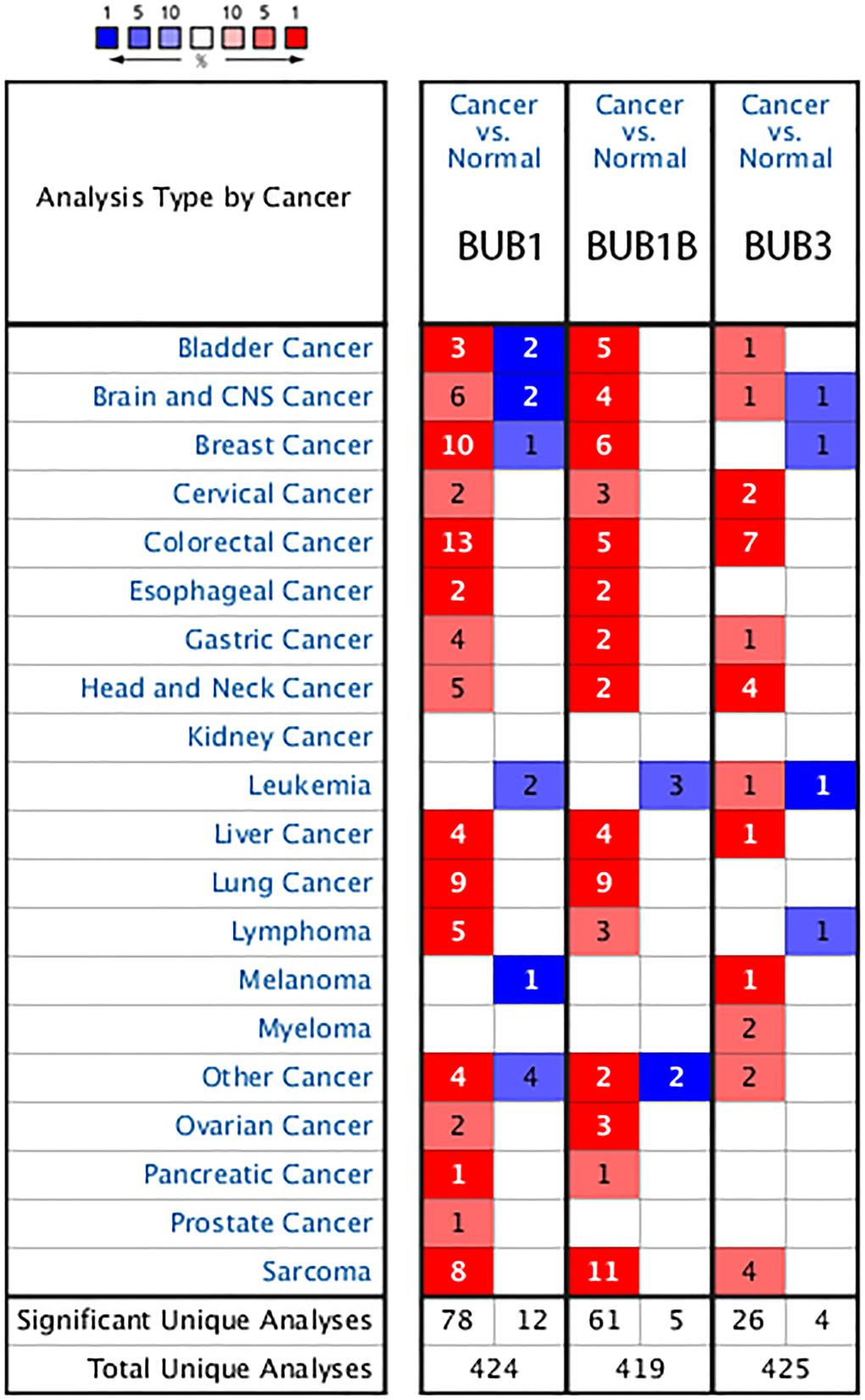
Figure 1. The transcription levels of BUB factors in different types of cancers (Oncomine). The BUB family of genes are overexpressed in many types of cancers including Sarcoma. The red cells represent evidence of gene overexpression. The blue cells represent evidence of decreased gene expression. The number in each cell represents the amount of evidence. The deeper the color, the higher the significance.
In the Detwiller Sarcoma dataset, BUB1 was upregulated in malignant fibrous histiocytoma, fibrosarcoma, leiomyosarcoma, round cell liposarcoma, and pleomorphic liposarcoma with 12.013 (P = 1.59E-09), 14.408 (P = 2.70E-08), 11.066 (P = 3.42E-07), 9.248 (P = 2.85E-06), and 10.159 (P = 6.81E-05) fold changes, respectively. BUB1B was upregulated in fibrosarcoma, pleomorphic liposarcoma, malignant fibrous histiocytoma, round cell liposarcoma, synovial sarcoma, and leiomyosarcoma. The fold changes for upregulation of each sarcoma are as follows: 8.908 (P = 2.52E-07), 4.687 (P = 6.25E-06), 5.849 (P = 3.01E-07), 4.734 (P = 1.99E-06), 3.933 (P = 1.13E-05), and 5.471 (P = 2.31E-05), respectively. BUB3 was upregulated in synovial sarcoma, fibrosarcoma, and malignant fibrous histiocytoma. The fold changes are 2.999 (P = 1.14E-05), 2.164 (P = 2.64E-05), and 2.350 (P = 7.05E-05), respectively. In the Barretina Sarcoma dataset, BUB1 was upregulated in myxofibrosarcoma, pleomorphic liposarcoma, and leiomyosarcoma with 3.085 (P = 6.53E-14), 2.926 (P = 1.22E-09), and 2.878 (P = 5.95E-09) fold changes, respectively. BUB1B was upregulated in dedifferentiated liposarcoma, myxofibrosarcoma, pleomorphic liposarcoma, leiomyosarcoma, and myxoid/round cell liposarcoma with fold changes of 3.628 (P = 2.20E-17), 7.544 (P = 1.11E-17), 5.121 (P = 8.66E-13), 4.838 (P = 1.46E-10), and 2.842 (P = 1.88E-10), respectively. BUB3 was upregulated in myxofibrosarcoma with a 2.514 (P = 1.57E-12) fold change (Table 1).
Table 1. The significant changes of BUBs expression in transcription level between different types of sarcomas (ONCOMINE database).
| Gene ID | Types of Sarcoma versus Normal | Fold Change | p-Value | t-Test | References |
| BUB1 | Malignant Fibrous Histiocytoma vs. Normal | 12.013 | 1.59E–09 | 9.949 | Detwiller Sarcoma |
| Fibrosarcoma vs. Normal | 14.408 | 2.70E–08 | 10.002 | Detwiller Sarcoma | |
| Leiomyosarcoma vs. Normal | 11.066 | 3.42E–07 | 9.000 | Detwiller Sarcoma | |
| Round Cell Liposarcoma vs. Normal | 9.248 | 2.85E–06 | 8.909 | Detwiller Sarcoma | |
| Pleomorphic Liposarcoma vs. Normal | 10.159 | 6.81E–05 | 8.788 | Detwiller Sarcoma | |
| Myxofibrosarcoma vs. Normal | 3.085 | 6.53E–14 | 11.255 | Barretina Sarcoma | |
| Pleomorphic Liposarcoma vs. Normal | 2.926 | 1.22E–09 | 8.750 | Barretina Sarcoma | |
| Leiomyosarcoma vs. Normal | 2.878 | 5.95E–09 | 7.802 | Barretina Sarcoma | |
| BUB1B | Dedifferentiated Liposarcoma vs. Normal | 3.628 | 2.20E–17 | 12.579 | Barretina Sarcoma |
| Myxofibrosarcoma vs. Normal | 7.544 | 1.11E–17 | 16.533 | Barretina Sarcoma | |
| Pleomorphic Liposarcoma vs. Normal | 5.121 | 8.66E–13 | 13.250 | Barretina Sarcoma | |
| Leiomyosarcoma vs. Normal | 4.838 | 1.46E–10 | 9.806 | Barretina Sarcoma | |
| Myxoid/Round Cell Liposarcoma vs. Normal | 2.842 | 1.88E–10 | 10.778 | Barretina Sarcoma | |
| Fibrosarcoma vs. Normal | 8.908 | 2.52E–07 | 7.540 | Detwiller Sarcoma | |
| Pleomorphic Liposarcoma vs. Normal | 4.687 | 6.25E–06 | 6.348 | Detwiller Sarcoma | |
| Malignant Fibrous Histiocytoma vs. Normal | 5.849 | 3.01E–07 | 7.079 | Detwiller Sarcoma | |
| Round Cell Liposarcoma vs. Normal | 4.734 | 1.99E–06 | 6.976 | Detwiller Sarcoma | |
| Synovial Sarcoma vs. Normal | 3.933 | 1.13E–05 | 5.772 | Detwiller Sarcoma | |
| Leiomyosarcoma vs. Normal | 5.471 | 2.31E–05 | 5.623 | Detwiller Sarcoma | |
| BUB3 | Myxofibrosarcoma vs. Normal | 2.514 | 1.57E–12 | 10.808 | Barretina Sarcoma |
| Synovial Sarcoma vs. Normal | 2.999 | 1.14E–05 | 5.854 | Detwiller Sarcoma | |
| Fibrosarcoma vs. Normal | 2.164 | 2.64E–05 | 5.391 | Detwiller Sarcoma | |
| Malignant Fibrous Histiocytoma vs. Normal | 2.350 | 7.05E–05 | 4.687 | Detwiller Sarcoma |
The expression patterns of BUBs in sarcoma samples were further compared to normal tissue using the GEPIA database. Notably, the expressions of BUB1 and BUB1B are significantly higher in sarcomas compared to normal tissues (Figure 2A–2E). The overexpression of BUB3 does not reach significance, possibly due to the low number of patients in the control group.
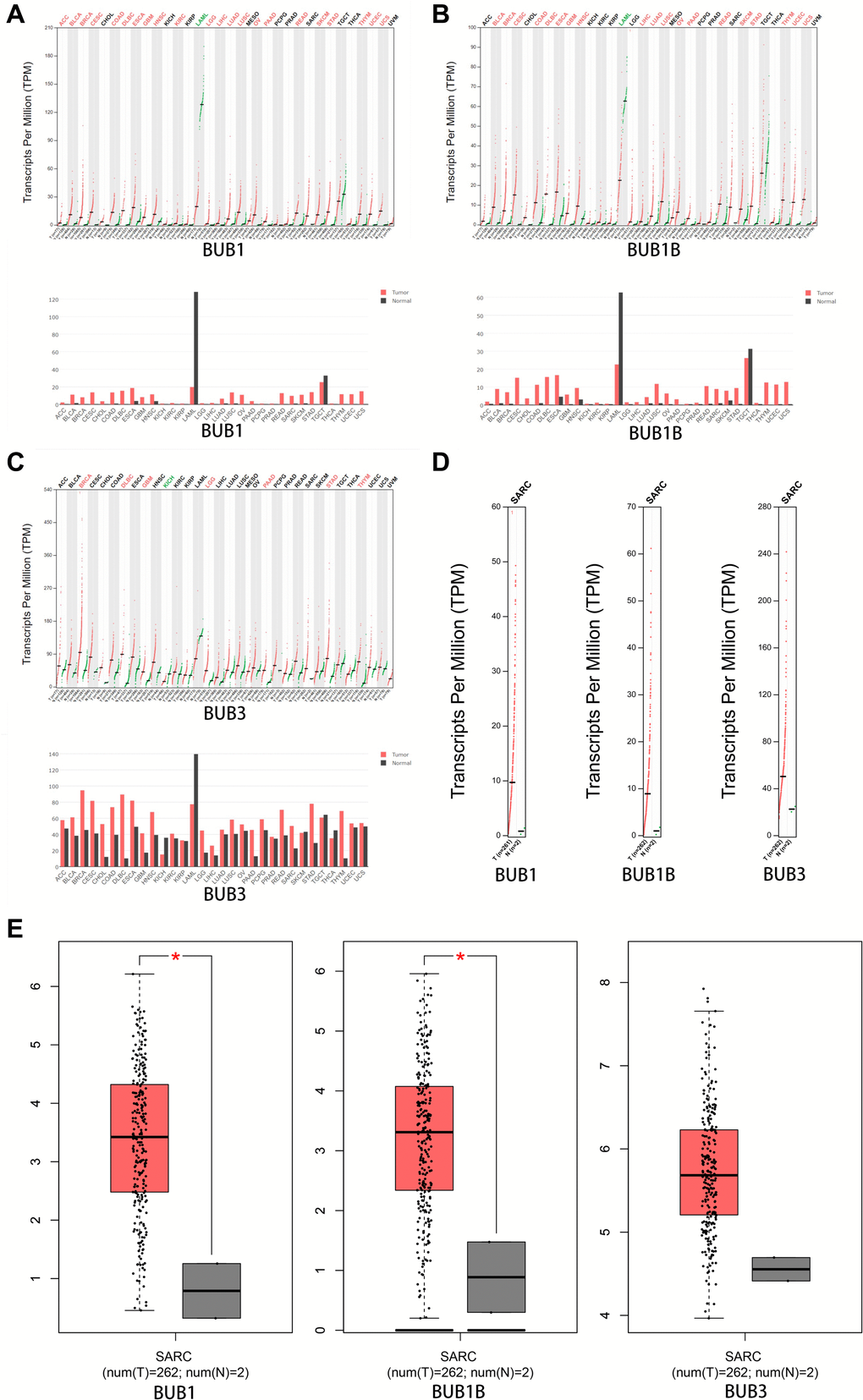
Figure 2. The expression of BUBs in different types of cancers (GEPIA). The expression of (A) BUB1, (B) BUB1B, and (C) BUB3 in different types of cancers. (D, E) The expression of BUBs in sarcomas. Each dot represents a sample. Cancer names were abbreviated. Details were listed in Supplementary Materials. *P < 0.05.
Transcriptional level of BUBs in sarcoma cell lines
To further explore the expression level of BUBs in sarcoma cell lines, we queried the CCLE database. The expressions of BUB1, BUB1B and BUB3 were highly upregulated (Figure 3) compared to normal control cell lines.
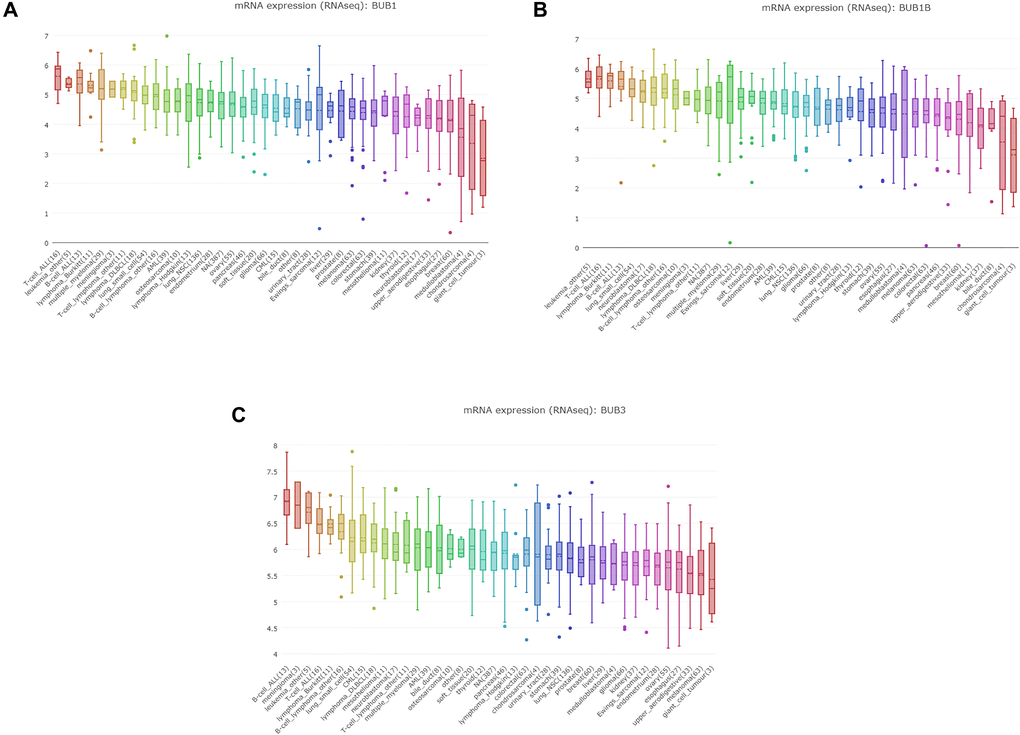
Figure 3. The expression of BUBs in cancer cell lines (CCLE). The mRNA expression of (A) BUB1, (B) BUB1B, and (C) BUB3 in sarcoma cell lines, analyzed by CCLE. Lineages are composed of a number of cell lines from the same area or system of the body. The number next to the lineage name indicates how many cell lines are in the lineage. The highest average distribution is on the left and is colored red. The dashed line within a box is the mean.
The prognostic values of BUBs in sarcoma
To investigate the clinical prognostic value of BUBs in sarcoma, we analyzed survival data of sarcoma patients using Kaplan-Meier Plotter and the GEPIA database (Figure 4). Both overall survival and disease-free survival of sarcoma patients are negatively associated with BUBs in the datasets. In the GEPIA dataset, the hazard ratios of BUB1 are 1.4 (P = 0.096) for overall survival and 1.6 (P = 0.015) for disease-free survival. As for BUB1B, they are 1.8 (P = 0.0038) and 1.6 (P = 0.0064) for overall and disease-free survivals, respectively. For BUB3, they are 1.5 (P = 0.046) and 1.6 (P = 0.015) for overall and disease-free survivals, respectively. In the Kaplan-Meier Plotter dataset, the hazard ratios of BUB1 are 2.18 (P = 0.0023) for overall survival and 2.26 (P = 0.0012) for disease-free survival. As for BUB1B, they are 2 (P = 7e-04) and 2.54 (P = 0.00083) for overall and disease-free survivals, respectively. For BUB3, they are 2.16 (P = 0.0048) and 1.96 (P = 0.027) for overall and disease-free survivals, respectively.
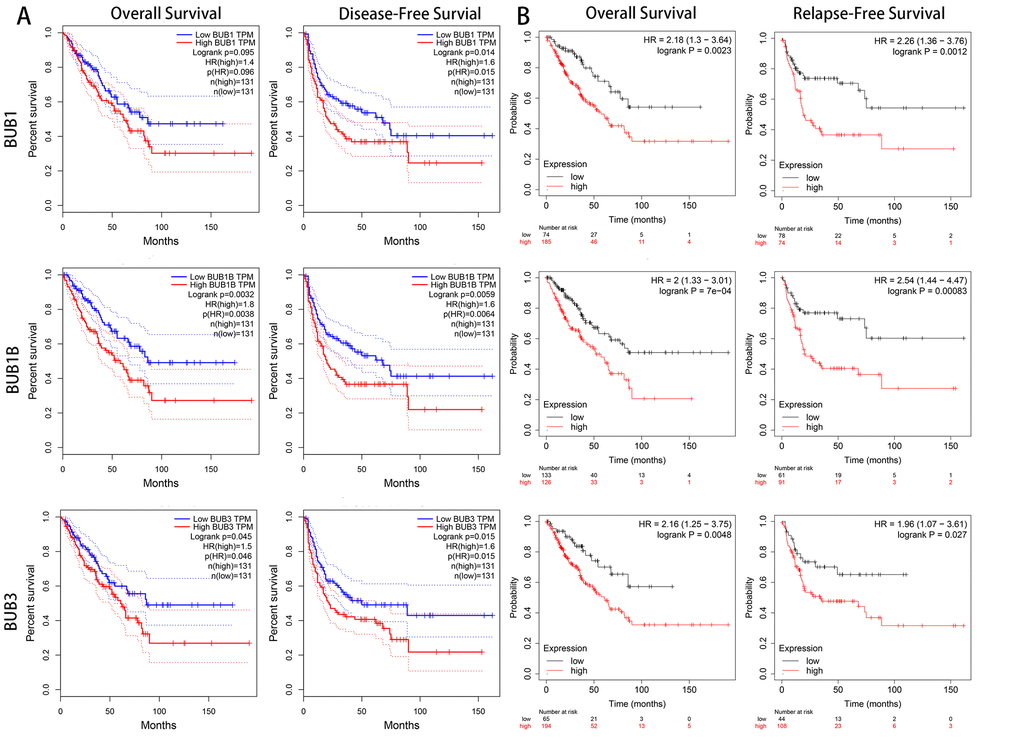
Figure 4. The prognostic value of mRNA level of BUB factors in sarcoma patients (GEPIA and kaplan meier plotter). The prognostic value of mRNA level of BUB factors in sarcoma patients was analyzed by (A) GEPIA and (B) Kaplan Meier Plotter. Higher expression of BUBs is associated with worse survival. HR, hazard ratio; TPM, Transaction per million.
Co-expression profile of BUBs in sarcoma
Genes co-expressed with BUBs in sarcoma were analyzed using the Oncomine database. In the Chibon Sarcoma datasets [22], we found BUB1 expression level was positively correlated with levels of CCNB1, AURKA, UBE2C, DLGAP5, NCAPG, CENPA, BIRC5, KIF4A, CCNB2, TPX2, KIF18B, KIF2C, CDC20, CDCA5, CENPE, CKS2, and MELK. For BUB1B, its expression level was positively correlated with levels of CACS5, NUSAP1, KIF20A, TOP2A, PRC1, KIF14, PTTG1, RACGAP1, CKS2, MELK, CENPE, CDCA5, BUB1, CCNB1, and AURKA. In the Schaefer Sarcoma dataset, we found that BUB3 expression level was positively correlated with levels of DLAT, PRPS1, MRPS7, TIMM23, PPIF, MAPK1, TFDP1, NUP98, ATP6V1A, MED6, CAPRIN1, CYP51P2, TMX1, EXOC5, MAT2A, and ACLY (Figure 5A). The association between BUB1, BUB1B, and BUB3 was analyzed using the GEPIA (Figure 5B) and Linked Omics databases (Figure 5C). The correlation coefficients of BUB1 and BUB1B were 0.8 (GEPIA) and 0.8988 (Linked Omics). For BUB1 and BUB3, they were 0.62 (GEPIA) and 0.5179 (Linked Omics). For BUB1B and BUB3, they were 0.6 (GEPIA) and 0.5293 (Linked Omics). All correlations were found to be statistically significant.
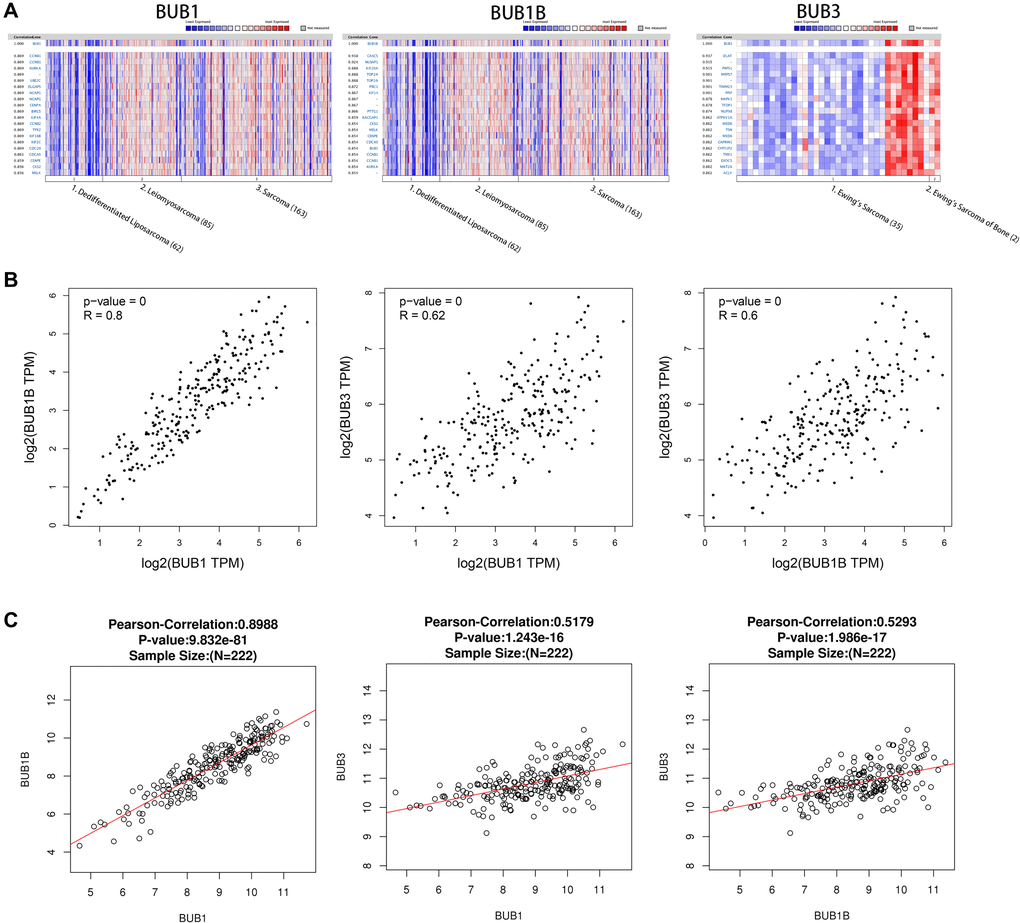
Figure 5. Co-expressed genes of BUBs, correlations between BUBs (ONCOMINE, GEPIA, and LinkedOmics). Co-expressed genes of BUBs in sarcomas, analyzed by (A) ONCOMINE. For each BUB factor, the top twenty co-expressed genes were displayed. The correlations between BUBs in sarcomas were analyzed by (B) GEPIA and (C) LinkedOmics. BUB1, BUB1B and BUB3 are positively correlated with each other. TPM, Transaction per million.
Predicted functions and pathways of BUBs in sarcomas
The functional roles of BUBs were evaluated from three perspectives: biological processes, molecular functions, and cellular components. We found that BUBs are involved in the biological processes of meiotic sister chromatid cohesion, meiotic cell cycle, cell division, cellular protein metabolic process, anaphase-promoting complex-dependent catabolic process, cell population proliferation, mitotic spindle assembly checkpoint, apoptotic process, protein phosphorylation, sister chromatid segregation, and nuclear division. Regarding molecular function, protein serine/threonine kinase activity was found to be significantly controlled by BUBs. Cellular components including protein-containing complex, nuclear lumen, cytosol, condensed, chromosome outer kinetochore, condensed nuclear chromosome kinetochore were all found to be associated with the function of BUBs. KEGG pathways analysis showed that BUBs are involved in Human T-lymphotropic virus 1 infection and cell cycle pathways (Table 2 and Figure 6).
Table 2. The functions of BUBs and genes significantly associated with BUBs alterations.
| Category | Term | Name | Count | FDR | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BP | GO:0051754 | meiotic sister chromatid cohesion, centromeric | 2 | 1.42e–06 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BP | GO:0051321 | meiotic cell cycle | 3 | 4.97e–06 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BP | GO:0051301 | cell division | 3 | 4.22e–05 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BP | GO:0044267 | cellular protein metabolic process | 3 | 0.0107 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BP | GO:0031145 | anaphase-promoting complex-dependent catabolic process | 2 | 3.01e–05 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BP | GO:0008283 | cell population proliferation | 2 | 0.0062 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BP | GO:0007094 | mitotic spindle assembly checkpoint | 3 | 2.16e–07 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BP | GO:0006915 | apoptotic process | 2 | 0.0107 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BP | GO:0006468 | protein phosphorylation | 2 | 0.0108 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BP | GO:0000819 | sister chromatid segregation | 3 | 1.51e–06 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BP | GO:0000280 | nuclear division | 3 | 8.96e–06 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MF | GO:0004674 | protein serine/threonine kinase activity | 2 | 0.0429 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CC | GO:0032991 | protein-containing complex | 3 | 0.0386 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CC | GO:0031981 | nuclear lumen | 3 | 0.0277 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CC | GO:0005829 | cytosol | 3 | 0.0402 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CC | GO:0000940 | condensed chromosome outer kinetochore | 2 | 1.03e–05 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CC | GO:0000778 | condensed nuclear chromosome kinetochore | 2 | 1.03e–05 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CC | GO:0000777 | condensed chromosome kinetochore | 3 | 6.68e–06 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| KEGG Pathways | hsa05166 | HTLV-I infection | 2 | 0.00098 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| KEGG Pathways | hsa04110 | Cell cycle | 3 | 1.04e–06 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| The functions of BUBs and the genes significantly associated with BUBs alterations were predicted by analyzing Gene Ontology (GO) and the Kyoto Encyclopedia of Genes and Genomes (KEGG) using Database for Annotation, Visualization and Integrated Discovery (DAVID). Biological Processes (BP), Molecular Functions (MF), Cellular Components (CC). | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
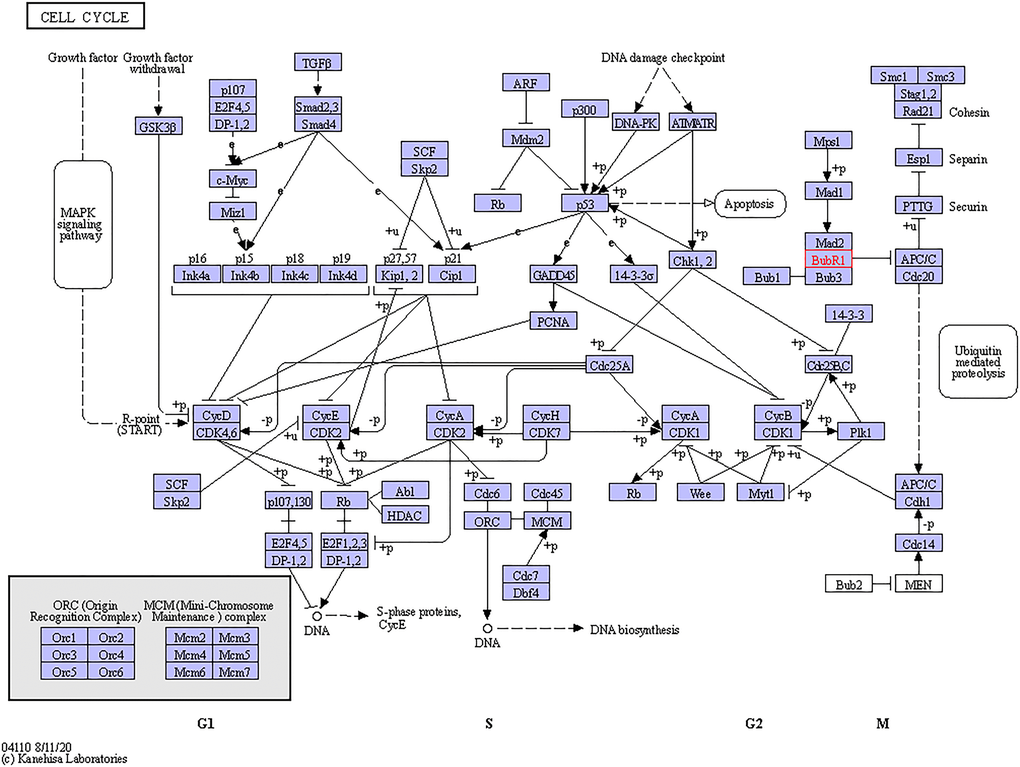
Figure 6. Cell cycle regulated by the BUBs alteration in sarcomas. The cell cycle regulated by the BUBs alteration in sarcomas are shown. Figure was downloaded from the KEGG database.
Discussion
Dysregulation in BUB genes have been reported in many cancers [6, 14–19, 21, 23, 24]. However, the role that BUB factors play in sarcoma remains poorly understood. The present study was the first to explore the expression patterns of BUBs in sarcoma, while investigating its prognostic value and clinical relevance.
BUB1 and BUB1B are two paralogous genes that play important roles in mitosis. The catalytic function of BUB1 is required for chromosome arm resolution and positioning of the chromosomal passenger complex. BUB1B plays a role in the inhibition of the APC/C, delaying the onset of anaphase and ensuring proper chromosome segregation. Deviations from their normal expression level may cause chromosome mis-segregation, aneuploidy, and cancer predisposition. Mouse embryonic fibroblasts lacking BUB1 catalytic activity showed reduced phosphorylation of centromeric substrates of AURKB, which participate in the regulation of alignment and segregation of chromosomes during mitosis and meiosis through association with microtubules [25]. Delocalization of AURKB strongly compromises the cells' ability to efficiently correct spindle attachment errors which results in an increased rate of chromosome alignment defects [26].
The roles of BUB1/BUB1B in cancer cells are still controversial with contradicting results from studies [8]. Low expression of these genes has been shown to be associated with poor survival and cancer metastasis [27, 28], while other studies have demonstrated correlation to delayed tumor growth with prolonged survival [29, 30]. On the other hand, studies have shown that overexpression of BUB1/BUB1B is related to progression and recurrence of various tumors including glioblastoma [29], pancreatic ductal adenocarcinoma [31–33], prostate cancer [34], gastric adenocarcinoma [35], and hepatocellular carcinoma [36]. In the present study, analysis using the Oncomine and GEPIA datasets revealed that the expression of BUB1 and BUB1B were higher in human sarcomas than in normal tissues. CCLE databases also showed that BUBs were highly expressed in human sarcoma cell lines. In addition, using the Kaplan-Meier Plotter and GEPIA databases, we determined that the overall and disease-free survival of sarcoma patients were significantly lower in patients with overexpressed BUB1 and BUB1B.
BUB3 is another member of the BUB family which works by binding to BUB1. Positive correlations were found among BUB1, BUB1B, and BUB3, which is in accordance with the fact that BUBs work as a complex during mitosis. The BUB1/BUB3 complex inhibits the action of the APC, preventing early entry of anaphase and exit of mitosis, which is crucial for the fidelity of chromosomal segregation (Figure 6). This effect was produced by phosphorylation of its activator CDC20 [37]. Overexpression of BUB3 was detected in oral cancer [38], gastric cancer [14], prostate cancer [39], and lung cancer [40]. Inhibition of BUB3 could enhance tumor chemosensitivity [38]. However, in a study by Elin Ersvær et al, it was demonstrated that decreased levels of nuclear BUB3 increased the risk of tumor recurrence [39], and da Silva et al. proposed that BUB3 could exert the role of a tumor suppressor through a kinetochore-independent mechanism [41].
Despite the uncertain roles of BUB3 in cancers seen in previous studies, our study shows a positive correlation between BUB3 and human sarcomas, as well as sarcoma cell lines, especially in osteosarcoma, chondrosarcoma and Ewing sarcoma. Furthermore, higher expression of BUB3 is associated with decreased disease-free survival and overall survival.
In the present study, we systematically analyzed the expression and prognostic value of BUBs in sarcoma. Our results indicated that BUB1, BUB1B and BUB3 may serve as oncogenes in sarcomas and have significant prognostic values. Specifically, the investigation of these genes could help ensure the diagnosis of sarcomas and predict the chance of survival in patients. Highly expressed BUBs may indicate higher malignancy in sarcomas. For these patients, more radical treatment is recommended. A disadvantage of the study is that analyses were solely based on sarcoma tissue samples but not blood samples from patients. It is unknown whether BUBs are overexpressed and can be detected in the blood of sarcoma patients or not. Further exploration could be focused on the expression level of BUBs in blood samples, which may become a more convenient screening method for the diagnosis of sarcoma. In the meantime, gene therapy targeting the BUB family can be a promising approach in the treatment of sarcomas.
Materials and Methods
Ethics statement
The current study was approved by the Academic Committee of Central South University and was conducted in accordance with the principles of the Declaration of Helsinki. All data sets are retrieved from published literature, all written informed consents have been obtained.
Data collection and processing
We first researched the expression level of the BUB gene family in tumors, especially sarcoma and normal tissues using the Oncomine database (https://www.oncomine.org/), Gene Expression Profiling Interactive Analysis (GEPIA, http://gepia.cancer-pku.cn/) and Cancer Cell Line Encyclopedia (CCLE, https://www.broadinstitute.org/ccle). Then, the prognostic value of BUBs was analyzed using GEPIA and Kaplan-Meier Plotter (https://kmplot.com/analysis/). Next, we analyzed the co-expression of BUBs in Oncomine and the correlations among BUBs in GEPIA and LinkedOmics (http://www.linkedomics.org/login.php). Lastly, the predicted functions and pathways of BUBs were analyzed in the Kyoto Encyclopedia of Genes and Genomes (KEGG, https://www.genome.jp/kegg/), Gene Ontology (GO, http://geneontology.org/), and Database for Annotation, Visualization and Integrated Discovery (DAVID, http://david.ncifcrf.gov) (Figure 7).
Figure 7. The flowchart of this study.
ONCOMINE analysis
The transcription levels of the BUB family in different sarcomas were analyzed using Oncomine gene expression array datasets (https://www.oncomine.org/), an online cancer microarray database. Student’s t test was conducted to compare the mRNA expressions of the BUB family in clinical cancer specimens and normal controls [42]. The cutoff of fold change was defined as > 2. Statistical significance was set at P value < 0.01. Co-expressed genes of BUBs in sarcoma were analyzed using ONCOMINE.
GEPIA dataset
Gene Expression Profiling Interactive Analysis (GEPIA, http://gepia.cancer-pku.cn/) is an interactive web platform that uses standard processing procedures to analyze the RNA sequencing expression data of 9736 tumors and 8587 normal samples from the Cancer Genome Atlas and Genotype Tissue Expression Project [43]. The expression levels of BUB family members were compared between tumor tissues and normal tissues. P < 0.05 was considered statistically significant.
CCLE dataset
The Cancer Cell Line Encyclopedia (CCLE, https://www.broadinstitute.org/ccle) project is a collaboration between the Broad Institute and the Novartis Institute of Biomedicine and the Institute of Genomics of the Novartis Research Foundation to carry out detailed genetics and pharmacologic characterization on a large group of human cancer models, to develop integrated computational analysis linking different pharmacological weaknesses with genomic patterns, and to transform cell line genomics into cancer patient stratification. CCLE provides public access to genomic data, analysis and visualization for approximately 1,000 cell lines [44]. The BUB family’s expressions in cancer cell lines were confirmed by the CCLE database.
Kaplan-Meier plotter
Kaplan Meier Plotter (https://kmplot.com/analysis/) contained the expression data of 54000 genes and the survival information of 21 cancer types including sarcoma. In order to analyze the overall survival and relapse-free survival of patients with sarcoma, the samples were divided into two groups according to median expression (BUBs high and low expression) and assessed by the Kaplan-Meier survival chart, based on hazard ratios (HR) and log-rank P-value [45].
KEGG and GO enrichment analyses of DEGs
The Database for Annotation, Visualization and Integrated Discovery (DAVID, http://david.ncifcrf.gov) (version 6.7) is an online biological information database that provides a comprehensive set of functional annotation information of genes and proteins [46]. The Kyoto Encyclopedia of Genes and Genomes (KEGG, https://www.genome.jp/kegg/) is a database resource which contains large-scale molecular datasets of high-level functions and biological systems generated by high-throughput experimental technologies [47]. Gene ontology (GO) (http://geneontology.org/) is a bioinformatics tool which can annotate genes and analyze their biological processes [48]. To further analyze the function of BUBs, pathway analyses were also performed using KEGG online database.
Supplementary Materials
Author Contributions
Conceptualization, W.G., L.-Z.L., T.-Q.Y.; Investigation, W.G., W. T., T.-Q.Y.; Writing, L.-Z.L., C.-L.A., T.-Q.Y., W.-W.C.; Review & Editing, L.-Z.L., W.G., C.-L.A., W. T., W.-W.C.; Visualization, L.-Z.L., W.-W.C., W. T.; Supervision, W.-W.C.; Funding Acquisition, W.-W.C.
Acknowledgments
I would like to express my gratitude to all the authors that contributed to the present manuscript. This study would not be finished without the efforts of all my co-authors.
Conflicts of Interest
The authors declare no competing interests.
References
- 1. Mitelman F. Catalogue of chromosome aberrations in cancer. Cytogenet Cell Genet. 1983; 36:1–515. https://doi.org/10.1159/000131930 [PubMed]
- 2. Santaguida S, Amon A. Short- and long-term effects of chromosome mis-segregation and aneuploidy. Nat Rev Mol Cell Biol. 2015; 16:473–85. https://doi.org/10.1038/nrm4025 [PubMed]
- 3. Klebig C, Korinth D, Meraldi P. Bub1 regulates chromosome segregation in a kinetochore-independent manner. J Cell Biol. 2009; 185:841–58. https://doi.org/10.1083/jcb.200902128 [PubMed]
- 4. Kiyomitsu T, Obuse C, Yanagida M. Human Blinkin/AF15q14 is required for chromosome alignment and the mitotic checkpoint through direct interaction with Bub1 and BubR1. Dev Cell. 2007; 13:663–76. https://doi.org/10.1016/j.devcel.2007.09.005 [PubMed]
- 5. Qi W, Yu H. KEN-box-dependent degradation of the Bub1 spindle checkpoint kinase by the anaphase-promoting complex/cyclosome. J Biol Chem. 2007; 282:3672–79. https://doi.org/10.1074/jbc.M609376200 [PubMed]
- 6. Zhu LJ, Pan Y, Chen XY, Hou PF. BUB1 promotes proliferation of liver cancer cells by activating SMAD2 phosphorylation. Oncol Lett. 2020; 19:3506–12. https://doi.org/10.3892/ol.2020.11445 [PubMed]
- 7. Johnson VL, Scott MI, Holt SV, Hussein D, Taylor SS. Bub1 is required for kinetochore localization of BubR1, Cenp-E, Cenp-F and Mad2, and chromosome congression. J Cell Sci. 2004; 117:1577–89. https://doi.org/10.1242/jcs.01006 [PubMed]
- 8. Meraldi P, Sorger PK. A dual role for Bub1 in the spindle checkpoint and chromosome congression. EMBO J. 2005; 24:1621–33. https://doi.org/10.1038/sj.emboj.7600641 [PubMed]
- 9. Warren CD, Brady DM, Johnston RC, Hanna JS, Hardwick KG, Spencer FA. Distinct chromosome segregation roles for spindle checkpoint proteins. Mol Biol Cell. 2002; 13:3029–41. https://doi.org/10.1091/mbc.e02-04-0203 [PubMed]
- 10. Bernard P, Hardwick K, Javerzat JP. Fission yeast bub1 is a mitotic centromere protein essential for the spindle checkpoint and the preservation of correct ploidy through mitosis. J Cell Biol. 1998; 143:1775–87. https://doi.org/10.1083/jcb.143.7.1775 [PubMed]
- 11. Jiang H, He X, Wang S, Jia J, Wan Y, Wang Y, Zeng R, Yates J
3rd , Zhu X, Zheng Y. A microtubule-associated zinc finger protein, BuGZ, regulates mitotic chromosome alignment by ensuring Bub3 stability and kinetochore targeting. Dev Cell. 2014; 28:268–81. https://doi.org/10.1016/j.devcel.2013.12.013 [PubMed] - 12. Toledo CM, Herman JA, Olsen JB, Ding Y, Corrin P, Girard EJ, Olson JM, Emili A, DeLuca JG, Paddison PJ. BuGZ is required for Bub3 stability, Bub1 kinetochore function, and chromosome alignment. Dev Cell. 2014; 28:282–94. https://doi.org/10.1016/j.devcel.2013.12.014 [PubMed]
- 13. Wang X, Cheung HW, Chun AC, Jin DY, Wong YC. Mitotic checkpoint defects in human cancers and their implications to chemotherapy. Front Biosci. 2008; 13:2103–14. [PubMed]
- 14. Grabsch H, Takeno S, Parsons WJ, Pomjanski N, Boecking A, Gabbert HE, Mueller W. Overexpression of the mitotic checkpoint genes BUB1, BUBR1, and BUB3 in gastric cancer--association with tumour cell proliferation. J Pathol. 2003; 200:16–22. https://doi.org/10.1002/path.1324 [PubMed]
- 15. Hernando E, Orlow I, Liberal V, Nohales G, Benezra R, Cordon-Cardo C. Molecular analyses of the mitotic checkpoint components hsMAD2, hBUB1 and hBUB3 in human cancer. Int J Cancer. 2001; 95:223–27. https://doi.org/10.1002/1097-0215(20010720)95:4<223::aid-ijc1038>3.0.co;2-l [PubMed]
- 16. Grabsch HI, Askham JM, Morrison EE, Pomjanski N, Lickvers K, Parsons WJ, Boecking A, Gabbert HE, Mueller W. Expression of BUB1 protein in gastric cancer correlates with the histological subtype, but not with DNA ploidy or microsatellite instability. J Pathol. 2004; 202:208–14. https://doi.org/10.1002/path.1499 [PubMed]
- 17. Mukherjee A, Joseph C, Craze M, Chrysanthou E, Ellis IO. The role of BUB and CDC proteins in low-grade breast cancers. Lancet. 2015 (Suppl 1); 385:S72. https://doi.org/10.1016/S0140-6736(15)60387-7 [PubMed]
- 18. Ru HY, Chen RL, Lu WC, Chen JH. hBUB1 defects in leukemia and lymphoma cells. Oncogene. 2002; 21:4673–79. https://doi.org/10.1038/sj.onc.1205585 [PubMed]
- 19. Ohshima K, Haraoka S, Yoshioka S, Hamasaki M, Fujiki T, Suzumiya J, Kawasaki C, Kanda M, Kikuchi M. Mutation analysis of mitotic checkpoint genes (hBUB1 and hBUBR1) and microsatellite instability in adult T-cell leukemia/lymphoma. Cancer Lett. 2000; 158:141–50. https://doi.org/10.1016/s0304-3835(00)00512-7 [PubMed]
- 20. Skubitz KM, D'Adamo DR. Sarcoma. Mayo Clin Proc. 2007; 82:1409–32. https://doi.org/10.4065/82.11.1409 [PubMed]
- 21. Sun Z, Xiao B, Jha HC, Lu J, Banerjee S, Robertson ES. Kaposi's sarcoma-associated herpesvirus-encoded LANA can induce chromosomal instability through targeted degradation of the mitotic checkpoint kinase Bub1. J Virol. 2014; 88:7367–78. https://doi.org/10.1128/JVI.00554-14 [PubMed]
- 22. Chibon F, Lagarde P, Salas S, Perot G, Brouste V, Tirode F, Lucchesi C, de Reynies A, Kauffmann A, Bui B, Terrier P, Bonvalot S, Le Cesne A, et al. Validated prediction of clinical outcome in sarcomas and multiple types of cancer on the basis of a gene expression signature related to genome complexity. Nat Med. 2010; 16:781–87. https://doi.org/10.1038/nm.2174 [PubMed]
- 23. Qiu J, Zhang S, Wang P, Wang H, Sha B, Peng H, Ju Z, Rao J, Lu L. BUB1B promotes hepatocellular carcinoma progression via activation of the mTORC1 signaling pathway. Cancer Med. 2020; 9:8159–72. https://doi.org/10.1002/cam4.3411 [PubMed]
- 24. Morales AG, Pezuk JA, Brassesco MS, de Oliveira JC, de Paula Queiroz RG, Machado HR, Carlotti CG
Jr , Neder L, de Oliveira HF, Scrideli CA, Tone LG. BUB1 and BUBR1 inhibition decreases proliferation and colony formation, and enhances radiation sensitivity in pediatric glioblastoma cells. Childs Nerv Syst. 2013; 29:2241–48. https://doi.org/10.1007/s00381-013-2175-8 [PubMed] - 25. Ricke RM, Jeganathan KB, Malureanu L, Harrison AM, van Deursen JM. Bub1 kinase activity drives error correction and mitotic checkpoint control but not tumor suppression. J Cell Biol. 2012; 199:931–49. https://doi.org/10.1083/jcb.201205115 [PubMed]
- 26. Baron AP, von Schubert C, Cubizolles F, Siemeister G, Hitchcock M, Mengel A, Schröder J, Fernández-Montalván A, von Nussbaum F, Mumberg D, Nigg EA. Probing the catalytic functions of Bub1 kinase using the small molecule inhibitors BAY-320 and BAY-524. Elife. 2016; 5:e12187. https://doi.org/10.7554/eLife.12187 [PubMed]
- 27. Shichiri M, Yoshinaga K, Hisatomi H, Sugihara K, Hirata Y. Genetic and epigenetic inactivation of mitotic checkpoint genes hBUB1 and hBUBR1 and their relationship to survival. Cancer Res. 2002; 62:13–17. [PubMed]
- 28. Park HY, Jeon YK, Shin HJ, Kim IJ, Kang HC, Jeong SJ, Chung DH, Lee CW. Differential promoter methylation may be a key molecular mechanism in regulating BubR1 expression in cancer cells. Exp Mol Med. 2007; 39:195–204. https://doi.org/10.1038/emm.2007.22 [PubMed]
- 29. Yu H, Zhang S, Ibrahim AN, Deng Z, Wang M. Serine/threonine kinase BUB1 promotes proliferation and radio-resistance in glioblastoma. Pathol Res Pract. 2019; 215:152508. https://doi.org/10.1016/j.prp.2019.152508 [PubMed]
- 30. Siemeister G, Mengel A, Fernández-Montalván AE, Bone W, Schröder J, Zitzmann-Kolbe S, Briem H, Prechtl S, Holton SJ, Mönning U, von Ahsen O, Johanssen S, Cleve A, et al. Inhibition of BUB1 Kinase by BAY 1816032 Sensitizes Tumor Cells toward Taxanes, ATR, and PARP Inhibitors In Vitro and In Vivo. Clin Cancer Res. 2019; 25:1404–14. https://doi.org/10.1158/1078-0432.CCR-18-0628 [PubMed]
- 31. Piao J, Zhu L, Sun J, Li N, Dong B, Yang Y, Chen L. High expression of CDK1 and BUB1 predicts poor prognosis of pancreatic ductal adenocarcinoma. Gene. 2019; 701:15–22. https://doi.org/10.1016/j.gene.2019.02.081 [PubMed]
- 32. Dong S, Huang F, Zhang H, Chen Q. Overexpression of BUB1B, CCNA2, CDC20, and CDK1 in tumor tissues predicts poor survival in pancreatic ductal adenocarcinoma. Biosci Rep. 2019; 39:BSR20182306. https://doi.org/10.1042/BSR20182306 [PubMed]
- 33. Tian X, Wang N. Upregulation of ASPM, BUB1B and SPDL1 in tumor tissues predicts poor survival in patients with pancreatic ductal adenocarcinoma. Oncol Lett. 2020; 19:3307–15. https://doi.org/10.3892/ol.2020.11414 [PubMed]
- 34. Fu X, Chen G, Cai ZD, Wang C, Liu ZZ, Lin ZY, Wu YD, Liang YX, Han ZD, Liu JC, Zhong WD. Overexpression of BUB1B contributes to progression of prostate cancer and predicts poor outcome in patients with prostate cancer. Onco Targets Ther. 2016; 9:2211–20. https://doi.org/10.2147/OTT.S101994 [PubMed]
- 35. Stahl D, Braun M, Gentles AJ, Lingohr P, Walter A, Kristiansen G, Gütgemann I. Low BUB1 expression is an adverse prognostic marker in gastric adenocarcinoma. Oncotarget. 2017; 8:76329–39. https://doi.org/10.18632/oncotarget.19357 [PubMed]
- 36. Zhuang L, Yang Z, Meng Z. Upregulation of BUB1B, CCNB1, CDC7, CDC20, and MCM3 in Tumor Tissues Predicted Worse Overall Survival and Disease-Free Survival in Hepatocellular Carcinoma Patients. Biomed Res Int. 2018; 2018:7897346. https://doi.org/10.1155/2018/7897346 [PubMed]
- 37. Gayyed MF, El-Maqsoud NM, Tawfiek ER, El Gelany SA, Rahman MF. A comprehensive analysis of CDC20 overexpression in common malignant tumors from multiple organs: its correlation with tumor grade and stage. Tumour Biol. 2016; 37:749–62. https://doi.org/10.1007/s13277-015-3808-1 [PubMed]
- 38. Silva PMA, Delgado ML, Ribeiro N, Florindo C, Tavares ÁA, Ribeiro D, Lopes C, do Amaral B, Bousbaa H, Monteiro LS. Spindly and Bub3 expression in oral cancer: Prognostic and therapeutic implications. Oral Dis. 2019; 25:1291–301. https://doi.org/10.1111/odi.13089 [PubMed]
- 39. Ersvær E, Kildal W, Vlatkovic L, Cyll K, Pradhan M, Kleppe A, Hveem TS, Askautrud HA, Novelli M, Wæhre H, Liestøl K, Danielsen HE. Prognostic value of mitotic checkpoint protein BUB3, cyclin B1, and pituitary tumor-transforming 1 expression in prostate cancer. Mod Pathol. 2020; 33:905–15. https://doi.org/10.1038/s41379-019-0418-2 [PubMed]
- 40. Haruki N, Saito H, Harano T, Nomoto S, Takahashi T, Osada H, Fujii Y, Takahashi T. Molecular analysis of the mitotic checkpoint genes BUB1, BUBR1 and BUB3 in human lung cancers. Cancer Lett. 2001; 162:201–05. https://doi.org/10.1016/s0304-3835(00)00675-3 [PubMed]
- 41. Morais da Silva S, Moutinho-Santos T, Sunkel CE. A tumor suppressor role of the Bub3 spindle checkpoint protein after apoptosis inhibition. J Cell Biol. 2013; 201:385–93. https://doi.org/10.1083/jcb.201210018 [PubMed]
- 42. Rhodes DR, Yu J, Shanker K, Deshpande N, Varambally R, Ghosh D, Barrette T, Pandey A, Chinnaiyan AM. ONCOMINE: a cancer microarray database and integrated data-mining platform. Neoplasia. 2004; 6:1–6. https://doi.org/10.1016/s1476-5586(04)80047-2 [PubMed]
- 43. Tang Z, Li C, Kang B, Gao G, Li C, Zhang Z. GEPIA: a web server for cancer and normal gene expression profiling and interactive analyses. Nucleic Acids Res. 2017; 45:W98–102. https://doi.org/10.1093/nar/gkx247 [PubMed]
- 44. Ghandi M, Huang FW, Jané-Valbuena J, Kryukov GV, Lo CC, McDonald ER
3rd , Barretina J, Gelfand ET, Bielski CM, Li H, Hu K, Andreev-Drakhlin AY, Kim J, et al. Next-generation characterization of the Cancer Cell Line Encyclopedia. Nature. 2019; 569:503–08. https://doi.org/10.1038/s41586-019-1186-3 [PubMed] - 45. Sun CC, Li SJ, Hu W, Zhang J, Zhou Q, Liu C, Li LL, Songyang YY, Zhang F, Chen ZL, Li G, Bi ZY, Bi YY, et al. Comprehensive Analysis of the Expression and Prognosis for E2Fs in Human Breast Cancer. Mol Ther. 2019; 27:1153–65. https://doi.org/10.1016/j.ymthe.2019.03.019 [PubMed]
- 46. Huang DW, Sherman BT, Tan Q, Collins JR, Alvord WG, Roayaei J, Stephens R, Baseler MW, Lane HC, Lempicki RA. The DAVID Gene Functional Classification Tool: a novel biological module-centric algorithm to functionally analyze large gene lists. Genome Biol. 2007; 8:R183. https://doi.org/10.1186/gb-2007-8-9-r183 [PubMed]
- 47. Kanehisa M. The KEGG database. Novartis Found Symp. 2002; 247:91–101. https://doi.org/10.1002/0470857897.ch8 [PubMed]
- 48. Ashburner M, Ball CA, Blake JA, Botstein D, Butler H, Cherry JM, Davis AP, Dolinski K, Dwight SS, Eppig JT, Harris MA, Hill DP, Issel-Tarver L, et al. Gene ontology: tool for the unification of biology. The Gene Ontology Consortium. Nat Genet. 2000; 25:25–29. https://doi.org/10.1038/75556 [PubMed]



