Introduction
Low back pain (LBP) is a leading cause of disability which affects more than 70% of the worldwide population [1]. Analysis of records from 1997 to 2017 shows LBP has been a common symptom that presents as a debilitating health problem that carries very high socioeconomic burden in all countries [2, 3]. Although many potential factors have been found to be related to LBP, intervertebral disc degeneration (IVDD) is one of the most important [4]. The intervertebral disc (IVD) is the key component of human spine which allows movement and links the superior and inferior vertebral bodies via the cartilaginous endplates (CEP) and is composed of three distinct components: the central highly hydrated gelatinous nucleus pulposus (NP), highly organized peripheral annulus fibrous (AF) and the CEP.
IVDD is known to involve both age-related changes and tissue damage caused by diverse stresses [5], and is characterized by chronic decreases in the number and function of NP cells along with associated loss of extracellular matrix (ECM). Understanding of these fundamental changes has focused attention towards biological treatments that facilitate the recovery of ECM function including gene therapy and other means to enable disc regeneration such as stem cell therapies [6]. Indeed, prior research has identified that nucleus pulposus mesenchymal stem cells (NP-MSCs) exist within the IVD [7] and that these cells participate in the regeneration and homeostasis of the IVD [8]. These endogenic stem cells self-renew and differentiate into NP cells that are responsible for self-regeneration and when activated can promote regeneration of the ECM [9].
The IVD represents the largest non-vascular tissue in the body [10]. Its main energy supply comes from the capillaries of the vertebral bodies, which nourish NP cells by diffusion through the cartilage endplate [5] but this process is inhibited when the disk and cartilage endplates undergo degeneration [11]. This leads to the changes in the IVD microenvironment through the accumulation of metabolites leading to hypoxia and acidosis (low pH) [12]. Regarding the latter, the pH of healthy discs is 7.1 [13] but it drops to 6.5 or even lower in severely degenerated discs [14, 15]. Importantly, an acidic microenvironment plays a key role in IVD degeneration since it causes loss of chondrocytes and consequent reductions in ECM [16, 17].
Acid-sensing ion channels (ASICs), a membrane H+-gated subgroup of the degenerin/epithelial Na+ channel (DEG/ENaC) family, have emerged as key receptors for extracellular protons in central and peripheral neurons. Four genes (Accn1, Accn2, Accn3 and Accn4) encode at least seven ASIC isoforms including ASIC1a, ASIC1b, ASIC1b2, ASIC2a, ASIC2b, ASIC3, ASIC4 [18–20], and each ASIC is comprised of three subunits which can form functional homo- or hetero- trimeric channels [21]. ASICs are widely distributed in various tissues of mammals [22–24] and exert diverse roles in acid sensation, taste, learning and memory, proprioception, Na/H2O transport and mechanosensation in many tissues. ASICs are expressed in nervous and joint tissues and have been implicated in neurodegenerative diseases including spinal cord injury, multiple sclerosis, Parkinson’s disease and Huntington’s disease [25–30]. Notably, ASICs are activated by acidosis, lactate and arachidonic acid and highly acidic conditions increase ASIC expression to promote survival of NP cells [31]. Instructively, blockade of ASICs by Amiloride, Psalmotoxin 1 (PcTx1), APETx2 or specific small interfering RNA (siRNA) can restrain acid-induced apoptosis in endplate chondrocytes of IVDs [32–35]. These results serve to illustrate the connection between acidic pH and ASIC function during IVD degeneration. However, the functional status of ASICs in NP-MSCs, a critical cell population required for IVD maintenance and regeneration, remains unclear.
In this report we studied the expression of ASICs in the IVD with different degrees of degeneration together with the interrelationship between ASIC function and acidosis. We found ASIC1 and ASIC3 were selectively increased in freshly isolated IVD tissues according to their degree of degeneration and that the physiological upregulation of ASIC1/ASIC3 could be mimicked in vitro by subjecting NP-MSCs to acidic culture conditions to simulate acidosis. Moreover, such treatment led to cell cycle and entry into cellular senescence [36–39] and we identified a number of potential senescence programming pathways involved. Consistently, we demonstrate that ASIC inhibitors prevent senescence induction in NP-MSCs, a finding which supports their potential as therapeutics for IVDD.
Results
Characterization of nucleus pulposus stem cells
Primary NP-MSCs isolated from human degenerated IVDs were cultured with low-glucose DMEM medium and displayed the morphological appearance of long-spindle shape cells that were strongly adherent to the plastic substrate (Figure 1A). The cells reached ~80% confluence approximately three weeks later and maintained a consistent appearance (Figure 1B). The identity of the cultured cells from IVDDs was confirmed by flow cytometric analyses demonstrating high expression of CD105, CD90 and CD73, with very low or negative expression of CD34, CD45 and HLA-DR (Figure 1C), consistent with the known phenotypes of NP-MSCs [40].
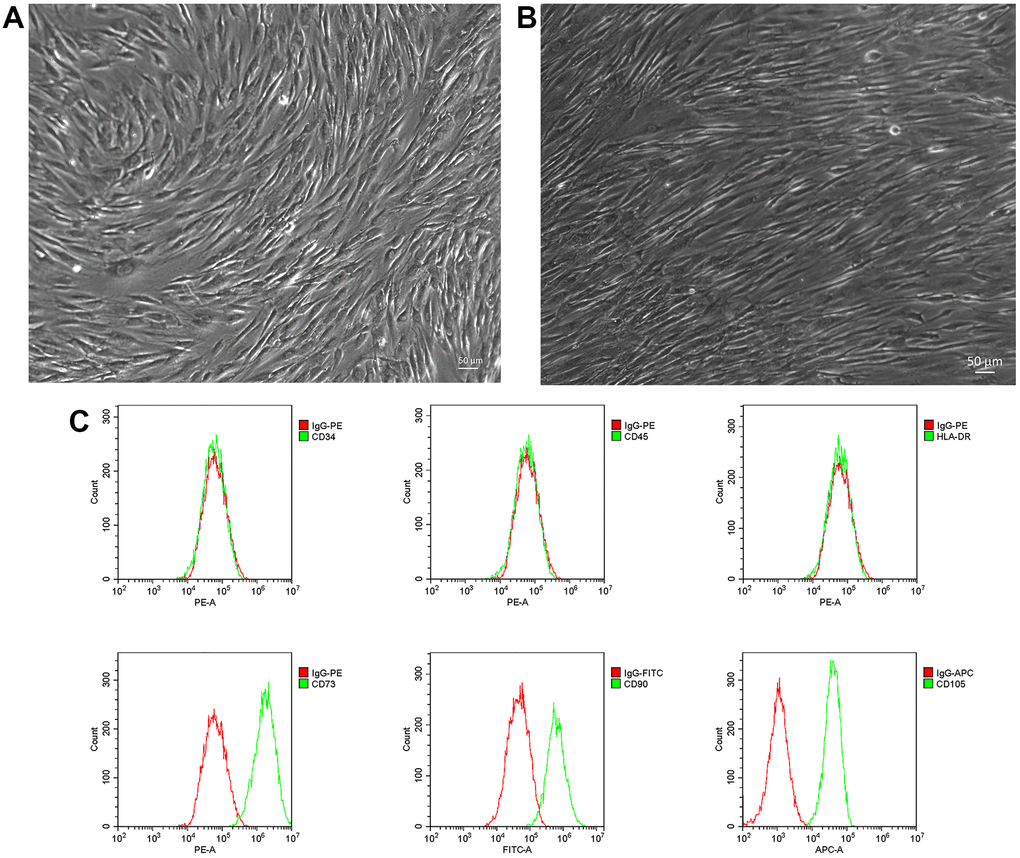
Figure 1. Isolation and phenotyping of in vitro cultured NP-MSCs. Representative phase-contrast images of passage 1 (A) and passage 2 (3 weeks) (bar = 50 μm) (B) NP-MSCs showing adherent cells with a long-spindle shape morphology. (C) Analyses of cell surface markers of NP-MSCs by flow cytometry, showing positive CD105, CD90, CD73 and negative CD34, CD45 and HLA-DR. All the samples were from case 5.
ASIC1 and ASIC3 are upregulated in human NP-MSCs with different degrees of degeneration
We investigated the expression of the ASIC isoforms, ASIC1, ASIC2, ASIC3 and ASIC4 in primary IVD isolates with differing degrees of degeneration. Western blotting analyses showed that the expression levels of ASIC1 and ASIC3 were progressively upregulated in NP-MSCs derived from mild (Pfirrmann grade II), moderate (Pfirrmann grade III) through to severely degenerated IVDs (Pfirrmann grade IV) compared with non-IVDD derived aspirate biopsy samples (Figure 2A, 2B and Supplementary Figure 1). Thus, there are specific effects on ASIC isoforms, a finding which is consistent with a prior report showing that ASIC1 and ASIC3 are most sensitive to H+ [21].
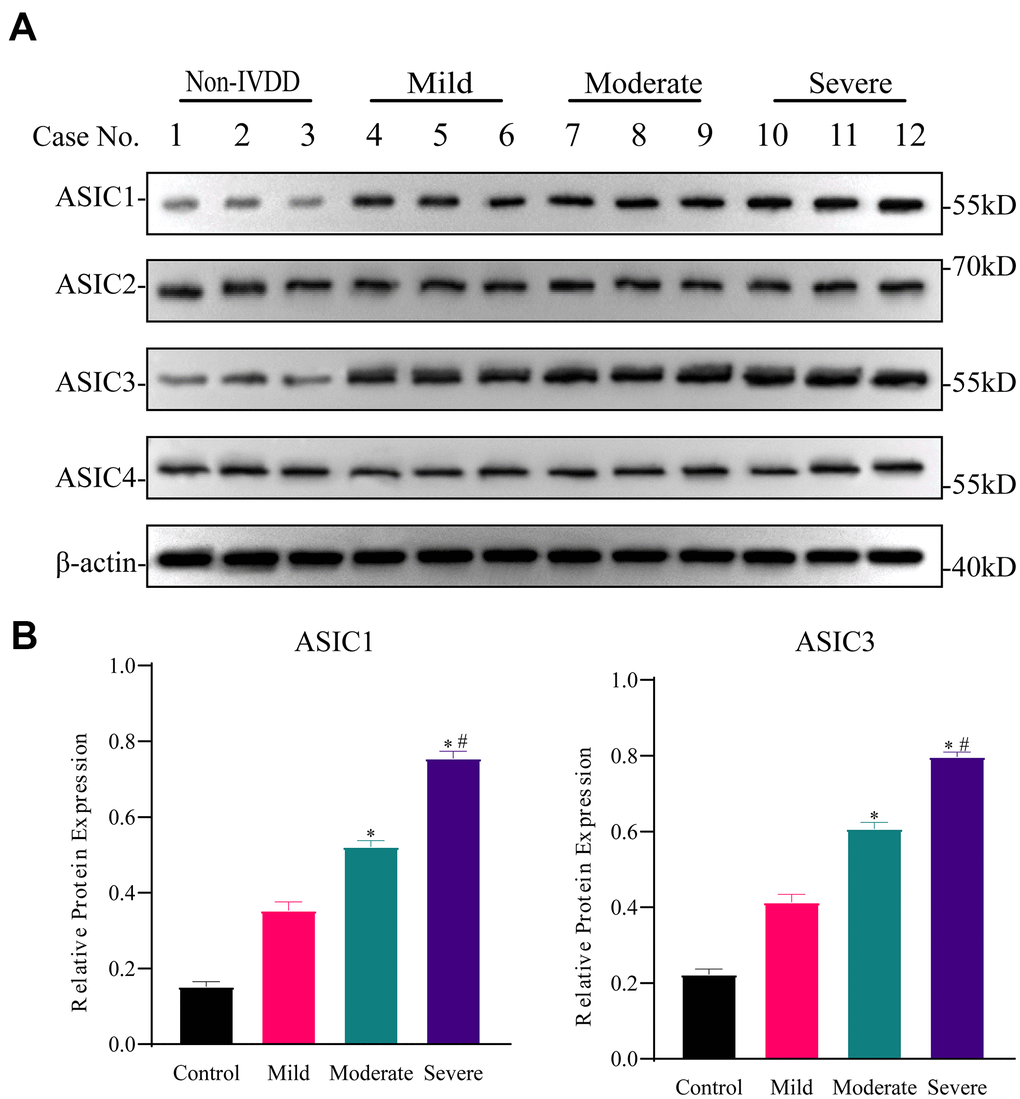
Figure 2. Differential expression of ASICs in freshly isolated human IVD tissues with different degrees of intervertebral disc degeneration (IVDD). (A) Representative Western blotting analyses of ASICs 1-4 in IVD tissues from non-IVDD and IVDD groups from mild to severe cases. (B) Analyses of the relative expression of ASIC1 and ASIC3 determined for all patient IVD tissues (n=3 for each group) using densitometric analysis of Western blotting as per (A). Data are mean ± SD. * indicates P≤0.05 compared to non-IVDD group; # indicates P≤0.05 compared to mild group.
Acidosis-induced upregulation of ASIC1 and ASIC3 can be reversed by ASIC inhibitors
To investigate the potential of acidic microenvironmental conditions to upregulate ASIC1 and ASIC3 in NP-MSCs, we cultured isolated cells under pH 6.6 conditions that mimic the degenerated intervertebral disc [17]. In concert we also evaluated the effects of the non-specific ASIC inhibitor Amiloride in addition to PcTx1 and APETx2 which are specific inhibitors of ASIC1 and ASIC3, respectively. Instructively, NP-MSCs grown under acidic conditions displayed increased expression of both ASIC1 and ASIC3 whereas treatment with Amiloride and PcTx1, and to a lesser extent APETx2, prevented acid-induced increases in their expression (Figure 3A, 3B and Supplementary Figures 2–4). These results support the notion that acidosis triggers upregulation of ASIC1 and ASIC3 in NP-MSCs and that this can be targeted using ASIC inhibitors.
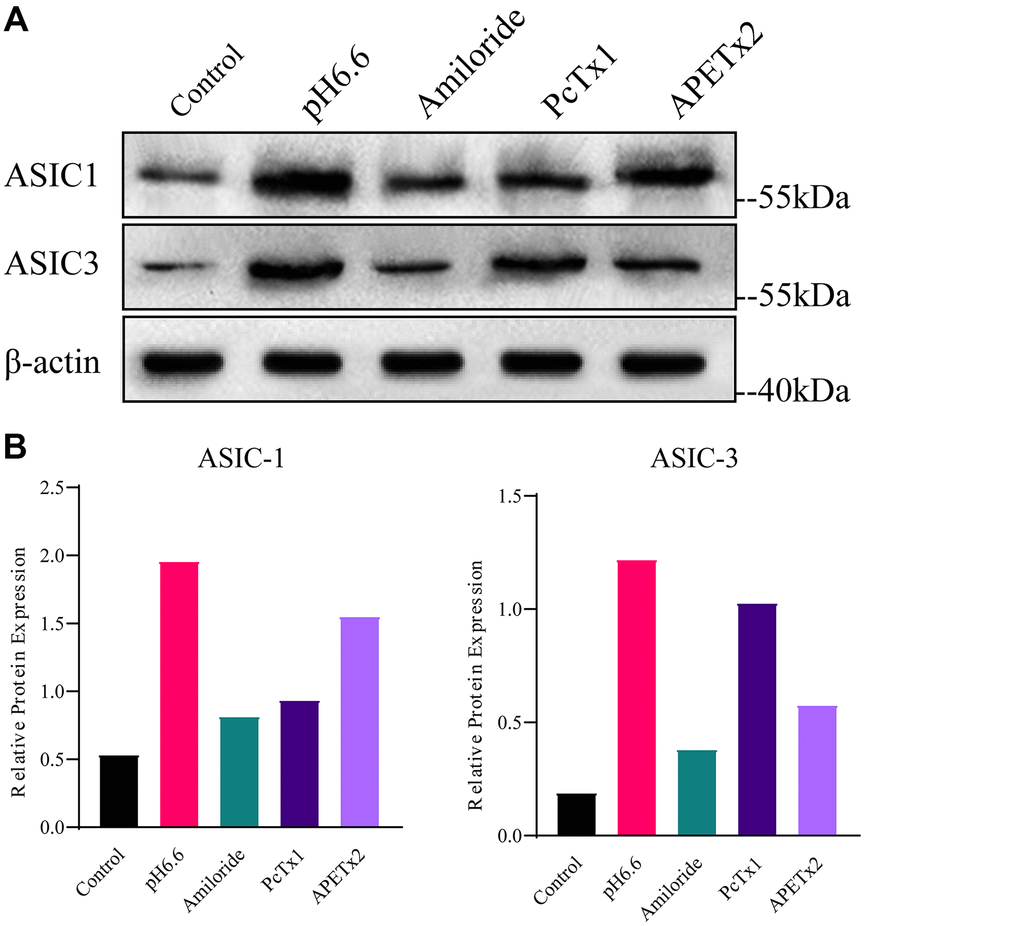
Figure 3. Analyses of ASIC1 and ASIC3 expression in primary cultured human NP-MSCs. (A) Western blotting analyses of ASIC1 and ASIC3 expression when exposed to extracellular acid (pH6.6) with or without treatment of inhibitor Amiloride, PcTx1 or APETx2. (B) The relative expression of ASIC1 and ASIC3 using densitometric analysis of specific bands revealed by Western blotting as per (A). The samples were from case 4. Data are mean ± SD. With * indicating P≤0.05.
ASIC inhibitors rescue the acidosis-induced decline in NP-MSC proliferation
Our previous work indicated that the proliferation rate of NP-MSCs decreases under acidic conditions [17]. Given the degeneration associated changes in ASIC1 and ASIC3 expression by NP-MSCs, in particular the increases triggered by acidic conditions, we considered that ASIC1 and ASIC3 may play a vital pH sensing role that affects NP-MSC proliferation. To test this notion, we took advantage of the ability of ASIC inhibitors to prevent upregulation of ASIC1 and ASIC3 triggered by acidosis. Measuring NP-MSC proliferation using CCK-8, an assay which uses cellular dehydrogenase activity as a proxy measure, showed that Pfirrmann grade II and grade IV isolates failed to grow at pH 6.6 in contrast to normal culture conditions (Figure 4A–4C). Importantly, treating the cells with ASIC inhibitors (Amiloride, PCTx1 and APETx2) served to restore the proliferation capacity of NP-MSCs to normal levels. Together these data suggest that inhibition of proliferation of NP-MSCs in the acidic microenvironment involves the functions of ASIC1 and ASIC3.
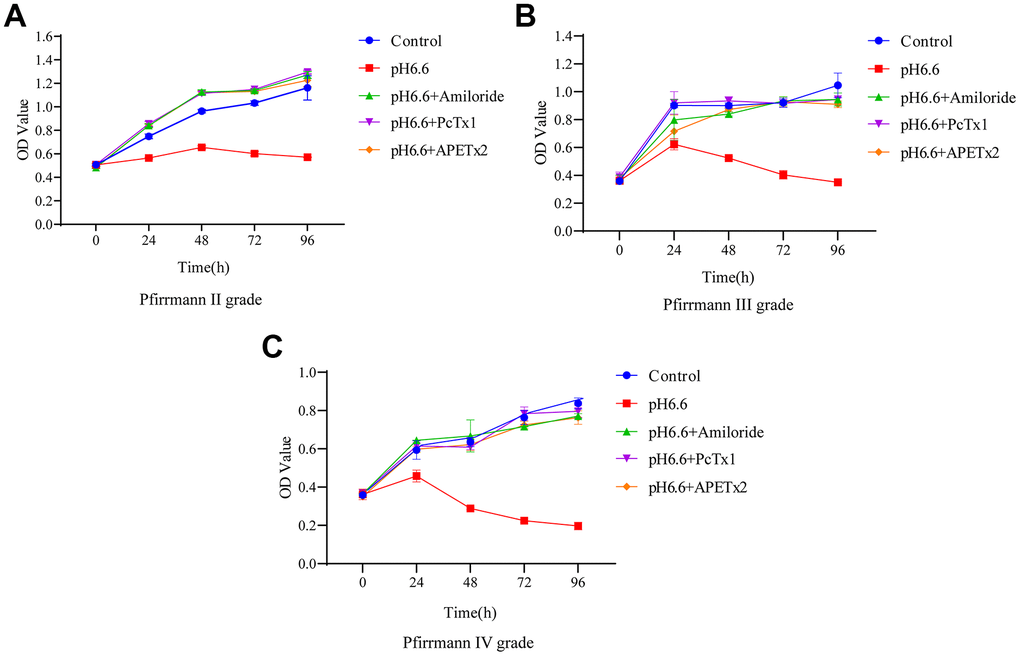
Figure 4. The effect of acidic culture conditions on the proliferation of human NP-MSCs. The proliferation ability of human NP-MSCs derived from patients with Pfirrmann grade II (A), III (B) and IV (C) degeneration was determined using CCK-8 assays under control conditions or after exposure to extracellular acid (pH6.6) alone and in combination with addition of the inhibitors Amiloride, PcTx1 or APETx2. n=3 for each group; data are mean ± SD.
Acidosis induces cell cycle arrest and senescence in NP-MSCs which can be prevented by ASIC inhibitors
To better understand why the population of NP-MSCs declined following culture in acid-conditioned media we sought to understand the processes involved. We considered the possibility that the cells underwent cell cycle arrest and subsequently became senescent. The impact of pH 6.6 culture on cell cycle phases was first evaluated by flow cytometry. Comparing NP-MSCs derived from Pfirrmann II grade IVDs under normal versus acidic conditions showed the numbers of proliferative cells (S+G2/M phases) substantially declined from 25.72 to 7.12% (Figure 5A). As anticipated from the proliferative assays, treatment of the NP-MSCs with the ASIC inhibitors, Amiloride, PcTx1 and APETx2 reversed the cell cycle phase changes with the numbers of proliferating cells determined as 29.27%, 27.79% and 25.56%, respectively. A comparable experiment conducted in NP-MSCs derived from a Pfirrmann IV grade case demonstrated similar findings albeit the basal proliferation rates of the cells was comparably less (Figure 5B). These data demonstrate that there is a significant population of cultured NP-MSCs that undergo cell cycle arrest.
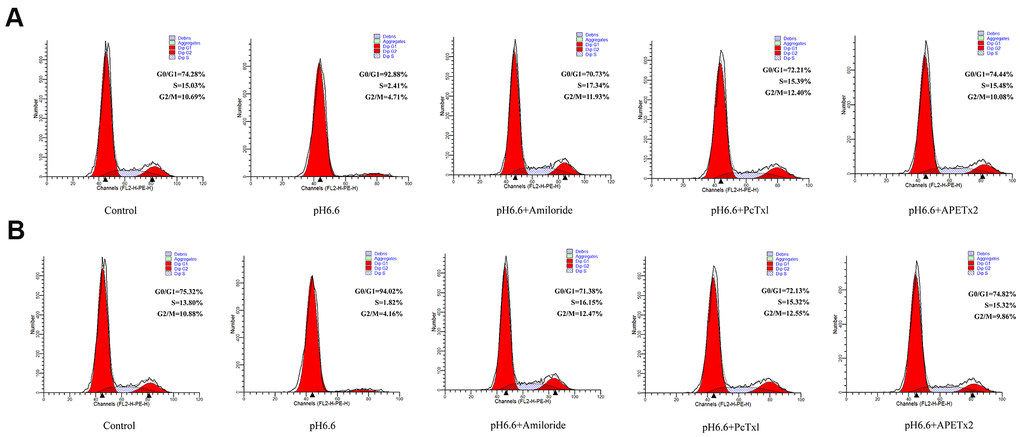
Figure 5. Acidic culture conditions inhibit cell cycle progression in human NP-MSCs which can be reversed using ASICs inhibitors. Flow cytometric DNA content profiles comparing cultured NP-MSCs derived from Pfirrmann grade II (A) and IV (B) cases cultured under control or acidic pH conditions in combination with or without treatment using Amiloride, PcTx1, APETx2, respectively. G0/G1, S and G2/M cell cycle phases were estimated as % totals using the ModFit software. The samples in (A) were from case 6 and that in (B) from case 11.
To ascertain if the arrested cells were entering senescence, we conducted senescence-associated β-galactosidase (SA-β-gal) staining of NP-MSCs cultures. Notably, acidic culture resulted in a time dependent increase in the percentage of senescence cells, reaching a striking ~80% of cells after 5 days (Figure 6A). Furthermore, comparing cultures of Pfirrmann stage III versus stage IV cultures showed the levels of senescent cells were double in the stage IV cells after 3 days of acidic culture (Figure 6B). Moreover, treating cells with ASIC inhibitors (Amiloride, PcTx1 or APETx2) resulted in significant reductions in the proportion of senescent cells induced by acidosis (Figure 6C).
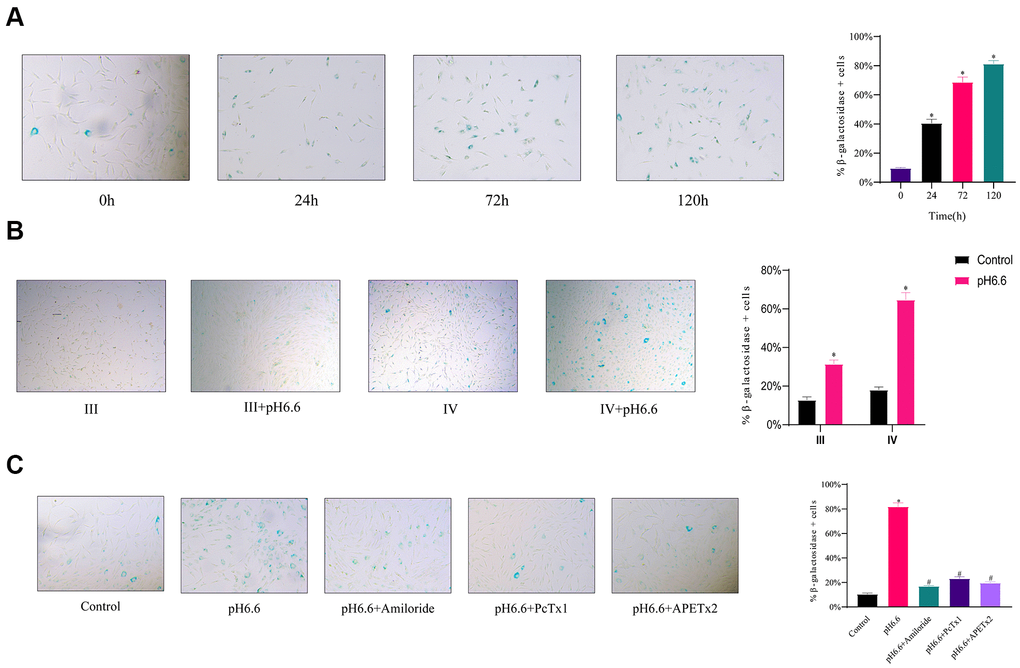
Figure 6. The induction of cellular senescence in human NP-MSCs exposed to acidic conditions can be reversed by ASICs inhibition. (A) Representative images (left) and the calculated percentage (right) of cultured Pfirrmann grade IV NP-MSCs undergoing senescence determined using SA-β-gal staining after exposure to acidic pH 6.6 conditions for 0, 24, 72 and 120 hours. n=3; data are mean ± SD, * P≤0.05 compared to 0 h group. (B) Comparative rates of senescence determined as per (A) in NP-MSCs derived from patients with Pfirrmann III and IV grades IVDD cultured under control or acidic pH conditions (pH 6.6) for 72 h. n=3; data are mean ± SD, * P≤0.05 compared to corresponding control group. (C) Effects of ASIC inhibitors on senescence induction in Pfirrmann grade II NP-MSCs cultured in control versus pH 6.6 for 120 h with or without treatment using Amiloride, PcTx1, APETx2 as indicated. The samples in (A) were from case 10, that in (B) of Pfirrmann III and IV group from case 7 and 11, respectively, and that in (C) from case 6. n=3; data are mean ±SD, * indicates P≤0.05 compared to control group; # indicates P≤0.05 compared to pH 6.6 group.
As confirmation that NP-MSCs exposed to acidic conditions were senescent, along with measuring the secreted levels of IL-6 and IL-8, inflammatory cytokines implicated in the senescence-associated secretory phenotype (SASP) [41, 42]. IL-6 and IL-8 are considered the most robust SASP markers and are involved in senescence-induced growth arrest through both autocrine and paracrine mechanisms [42, 43]. Examining the relative expression levels of IL-6 and IL-8 secreted in the NP-MSC culture medium by quantitative ELISA revealed significantly increased levels of secreted IL-6 and IL-8 by NP-MSCs under pH 6.6 culture conditions (Figure 7A, 7B). As anticipated, ASIC inhibitor treatment reversed the increases in cytokine expression.
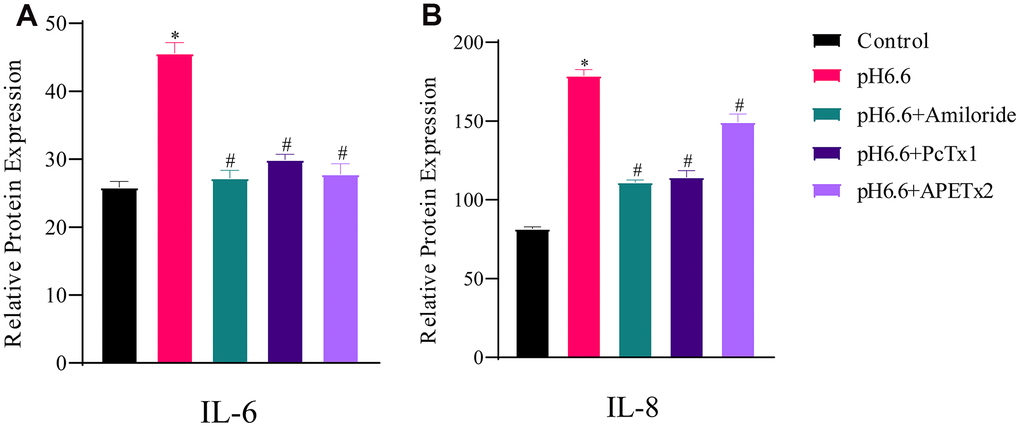
Figure 7. The production of secreted SASP cytokines by human NP-MSCs is increased by acidic culture and partially reversed by ASICs inhibition. ELISA assays measuring secreted IL-6 (A) and IL-8 (B) by human NP-MSCs after culture in control or acidic medium (pH 6.6) with or without treatment using the inhibitors Amiloride, PcTx1 or APETx2 as indicated. n=3; data are mean ±SD, * indicating P≤0.01 compared to control group at the same time points; # indicating P≤0.01 compared to pH 6.6 group at the same time points. The samples were from case 8.
Together these data indicate that acidosis inhibits NP-MSC proliferation through inducing cell cycle arrest and senescence, and that inhibiting ASIC1 and ASIC3 can prevent cells undergoing senescence.
Molecular profiling of protein expression in senescent NP-MSCs and the impact of ASICs
The entry of cells into senescence involves the upregulation and activation of transcription factors responsible for driving networks of genes involved in cell cycle arrest and senescence programming. These include p53 and its downstream targets p21 [44], p27 along with p16 and phosphorylated Rb1 [45, 46]. Other protein markers of interest to IVDD also include ECM components such as aggrecan and collagen II along with matrix metalloproteinases such as matrix metalloproteinase 3 and 9 (MMP3 and MMP9) which degrade multiple ECM components [47] To uncover which of these molecular mechanisms were relevant to the effects of acidosis on NP-MSCs we measured the expression of these proteins using Western blotting. Exposure of isolated NP-MSCs to acidic growth conditions resulted in relatively increased levels of p53, p21, p27, Rb1 and p16 proteins (Figure 8 and Supplementary Figures 5–7). Similarly, MMP3 and MMP9 levels also increased whereas aggrecan and collagen II levels decreased. The levels of an actin loading control protein were similar for each sample. Comparing cells treated with ASIC inhibitors showed their effectiveness at reversing the acid-induced changes to varying extents with Amiloride being the most efficient compared to the specific blockers PcTx1 and APETx2.
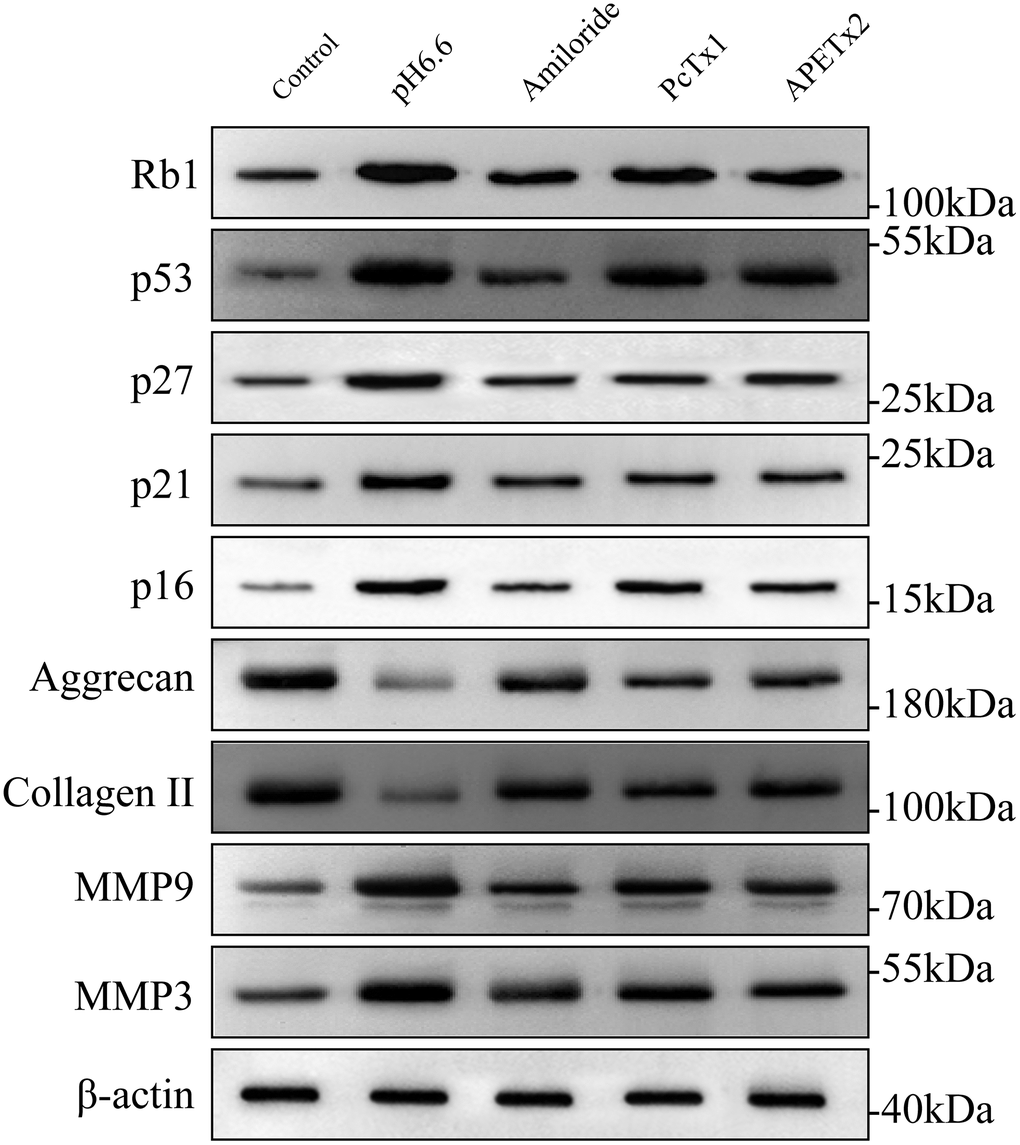
Figure 8. The effect of acidic culture conditions on senescence-related protein expression in primary cultured human NP-MSCs. Western blotting analyses were performed against Rb1, p53, p27, p21, p16, Aggrecan and Collagen II, MMP9 and MMP3 expression in Pfirrmann grade III NP-MSCs exposed to pH 6.6 culture conditions with or without treatment using the inhibitors Amiloride, PcTx1 or APETx2, respectively. The samples were from case 5, 9 and 12, respectively.
Discussion
In recent years, stem cell therapeutic approaches have become an attractive option for addressing the clinical problem of disc degeneration. The first reported isolation of MSC-like cells from degenerated IVDs by Risbud et al. [48] indicated the potential of employing stem cells for such therapies. Adding to the characterization of these cells, our previous research has established that IVD-isolated NP-MSCs have osteogenic, adipogenic and chondrogenic differentiation potential [17]. Other researchers including Fehlings, Huang and Zhang’s groups have all found that the NP-MSCs in intervertebral disc represent a readily accessible source for repair and regeneration [49–51]. Similarly, the means to activate or preserve endogenous NP-MSCs in the IVD represents a comparable strategy to stem cell replacement. Therefore, knowledge of the molecular mechanisms and downstream pathways affecting NP-MSCs during IVDD is crucially important in developing approaches that help maintain IVD homeostasis.
The pH in most human IVD decreases gradually with the age increase and degenerative processes [52]. The acidic environment causes physiological changes in intervertebral disc cells [5]. ASICs affect the biological activity of cells by regulating the flow of ions inside and outside of the cells [21] with a number of reports suggesting these play important roles in IVDD. Indeed, in vitro studies have shown that ASIC channels mediate an increase in intracellular Ca2+ of chondrocytes upon acidosis, and the abnormal increases in Ca2+ may result in cell death by apoptotic or pyroptotic mechanisms [32, 53]. In vitro animal experiments have observed that ASIC1 mediates cartilage endplate apoptosis and matrix metabolism under acidic conditions [54, 55]. Gilbert et al [12] also found that ASIC3 may be a potential therapeutic target for treatment of IVD degeneration. To better resolve which of these ASICs were implicated during IVDD we surveyed their expression.
Herein, we found there was progressively higher expression of ASIC1 and ASIC3, but not ASIC2 and ASIC4, in IVD tissues during IVDD ranging from mild to severe degeneration. According to our previous research, pH 6.6 culture conditions mimic the acidic microenvironment in the IVD without induction of cell death [56]. Applying this approach, we confirmed there was differential activation of ASIC1 and ASIC3 in IVD-isolated NP-MSCs. Collectively this supports the notion that acidosis was responsible for the changes in ASIC expression in vivo. This is the first report to definitively show that ASIC1 and ASIC3 are the key mediators of cellular senescence in human NP-MSCs during IVDD.
We had previously shown that exposing NP-MSCs to acidic conditions resulted in decreased cell proliferation [17] and we sought to expand on the underlying mechanisms, particularly the role played by ASICs. We showed cultured NP-MSCs exposed to an acidic microenvironment displayed a shift in the proportion of cells in the G0/G1 phase, indicative of cell cycle arrest which in turn is a prerequisite for entry of cells into senescence. Indeed, we showed that acidic culture conditions caused a remarkably high proportion of NP-MSCs to become senescent as scored by their expression of SA-β-gal activity and elevated expression of SASP cytokines. We then employed ASIC inhibitors to determine the impact of upregulated ASICs on the acidosis-induced changes. Prior experimental evidence has demonstrated that the ASIC1 specific blocker PcTx1, ASIC3 specific blocker APETx2, and the non-specific ASIC blocker Amiloride play roles in regulating the activity of NP cells [55, 57]. Here we showed that each of these ASIC inhibitors, to a lesser or greater extent, could reverse all of the acidosis-related changes in NP-MSCs. Amiloride, PcTx1 and APETx2 all reversed the decline in NP-MSC proliferative capacity and cell cycle arrest together with blocking cells from becoming senescent, providing direct evidence that targeting ASICs may be useful strategy in countering the effects of acidosis during IVDD.
Cellular senescence has been recently proposed to drive aging and age-related diseases including cancer [58]. Triggers for senescence include the constitutive DNA damage response (DDR) [58] with the downstream activation of senescence programming governed by a number of principal mediators [59] including cyclin-dependent kinase (CDK) inhibitors p21CIP1 (CDKN1A), p16INK4a (CDKN2A) and p27KIP1 (CDKN1B) [45, 60]. Previously, Human tissues and mouse models have both shown the correlation of cellular senescence with age-related disc degeneration [61–63]. Our results showed that compared to mild degenerated NP-MSCs, severe degenerated disc-derived human stem cells had higher ratio of SA-β-gal positive cells; likewise, with the increase of the induction time of acidosis, the ratio of positive cells increased. Here we showed that multiple mediators are implicated in the senescence of NP-MSCs induced by an acid microenvironment as indicated by the upregulation of the cell cycle inhibitors p27, p21 and p16. Moreover, the induction of senescence also coincided with the upregulation of p53 and Rb1, key tumor suppressor proteins associated with senescence pathways [63–68]. Instructively, the premature senescence of NP-MSCs was alleviated by ASIC inhibitors, suggesting these receptors decisively act upstream to activate senescence pathways including p53-p21/p27 and p16-Rb1 signaling.
A secondary but nonetheless important effect of acidosis concerned the effects on the cellular microenvironment. Indeed, the ECM plays a central role in the anatomical structure of the intervertebral disc and its loss during IVDD is a critical problem. Acidosis increased the expression of MMP3 and MMP9, enzymes which break down ECM components and accordingly, there were decreased levels of both collagen II and aggrecan recorded under acidic conditions. Again, Amiloride, PcTx1 and APETx2 suppressed these changes.
In summary, our work establishes that the acidic microenvironment promotes cellular senescence in human NP-MSCs, effects mediated by ASIC1 and ASIC3 which are implicated in pleiotropic effects ranging from the induction of multiple known senescence mediators as well as effects on the integrity of the extracellular microenvironment (Figure 9). Moreover, our in vitro system provides a means to evaluate potential therapies such as the application and evaluation of ASIC inhibitors. Nevertheless, there are still inherent limitations, particularly that the intervertebral disc microenvironment is more complicated, and other factors are involved including hypoxia, nutritional deficiencies and low cellularity [69–71]. However, some of these aspects could readily be replicated in vitro. Nor have we studied the ability of NP-MSCs to transform into nucleus pulposus-like cells under acidosis conditions. From the perspective of the role of ASICs in IVDD, the precise subunit composition still needs to be defined along with a better understanding of their signaling contributions which appear from our work to be extensive.
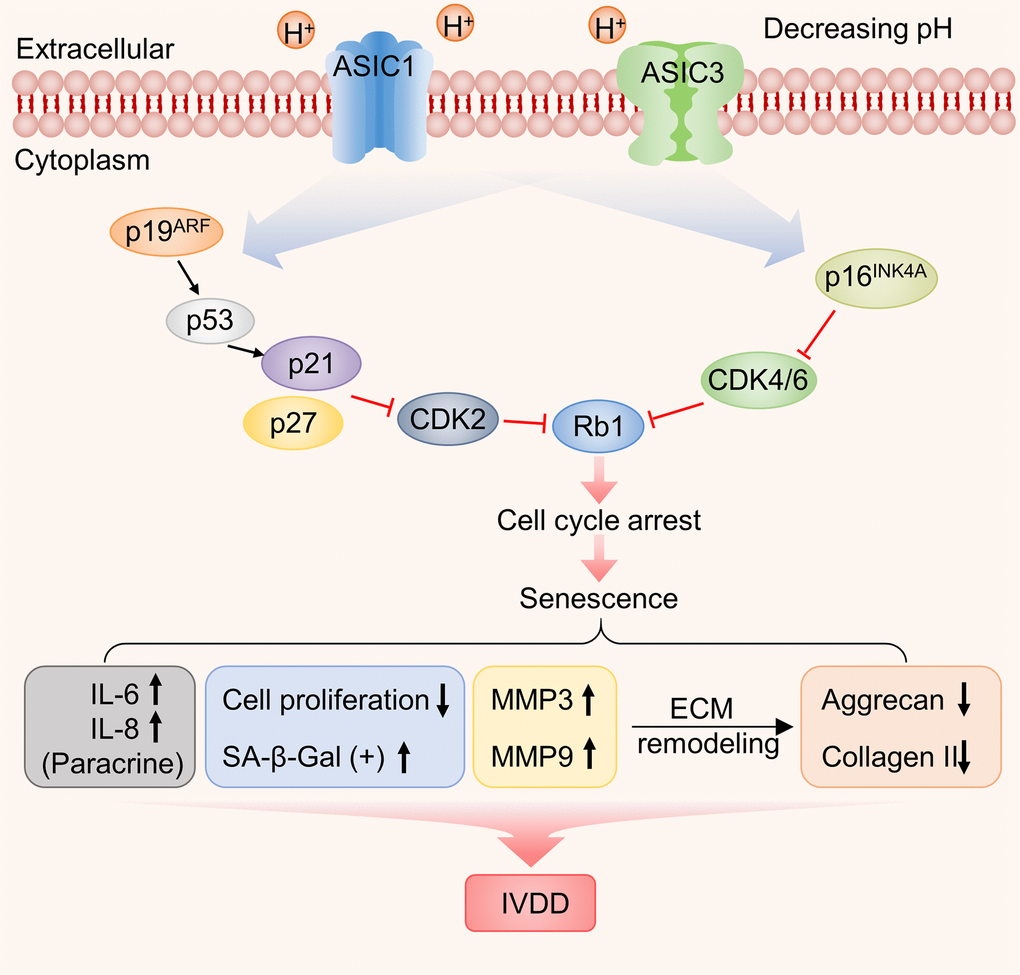
Figure 9. Working model illustrating the functional contribution of ASIC1/3 expression by NP-MSCs to the acidosis-associated development of IVDD.
Materials and Methods
Ethics statement
Human NP tissues represent non-IVDD group were collected from 3 patients who underwent idiopathic scoliosis. The degenerative human lumbar NP specimens were collected from 9 patients with a diagnosis of lumbar disc herniation during surgical procedures to remove IVD for treatment of low back pain at the Department of Spine Surgery, the First Affiliated Hospital of Anhui Medical University. Pfirrmann grading system was used to evaluate the degree of IDD. Table 1 provides detailed information about patients age, gender and Pfirrmann classification (n=12, 7 males and 5 females; ages range from 13 to 63 years; Pfirrmann grades I, II, III and IV, 3 patients each, respectively). All human tissues were obtained with specific written informed consent with a signature from patients or their guardians with this study approved by the institutional review board of Anhui Medical University (2017372) and carried out in accordance with the relevant guidelines and regulations.
Table 1. Patient demographic data and degree of disc degeneration.
| Case no. | Age (years) | Gender | Pfirrmann grade |
| Case 1 | 16 | Male | I |
| Case 2 | 15 | Male | I |
| Case 3 | 13 | Female | I |
| Case 4 | 19 | Male | II |
| Case 5 | 50 | Female | II |
| Case 6 | 49 | Female | II |
| Case 7 | 41 | Female | III |
| Case 8 | 42 | Male | III |
| Case 9 | 35 | Male | III |
| Case 10 | 26 | Female | IV |
| Case 11 | 56 | Male | IV |
| Case 12 | 63 | Male | IV |
Isolation of human NP-MSCs
Human NP tissue was isolated from peripheral annulus fibrous tissue and washed 3 times with sterile phosphate buffered saline (PBS, Sigma), minced carefully with aseptic ophthalmic scissors into ~0.5×0.5×0.5 mm pieces. A proportion of the minced pieces were reserved for the preparation of freshly isolated tissue homogenates while the rest were digested in 2 mg/ml type II collagenase solution (Sigma-Aldrich, USA) at 37° C for 4 hours. Following 1000×g centrifugation for 5 min, pellets (liberated cells and partially digested tissues) were collected and cultured with low-glucose Dulbecco’s modified Eagle’s medium (2 mM L-glutamine, HyClone) supplemented with 10% FBS, 1% penicillin/streptomycin in a humidified incubator at 37° C with 5% CO2. Culture medium was replaced twice a week with cells trypsinized with 0.25% Trypsin-EDTA (Biosharp) for further culture upon 80-90% confluence with subculture at 1:3 as passage 1 (P1). Cells at passage 2 were used for the in vitro experiments.
Immunophenotyping of NP-MSCs
NP-MSCs from patients’ IVDs were collected by trypsinization and resuspended at 1×106 cells/100 μl in PBS before immunophenotyping using flow cytometry analysis: Cells were incubated with fluorophore-conjugated monoclonal antibodies against CD45-PE, CD34-PE, HLA-DR-PE, CD73-PE, CD90-FITC and CD105-APC or isotype controls (all purchased from eBioscience, USA) at 25° C for 30 min in the dark as recommended by International Society for Cellular Therapy. The cells were washed twice with cold PBS and resuspended in 500 μl of PBS containing 1% paraformaldehyde before analysis by flow cytometry (Beckman, USA) according to standard procedures. The percentage of positive staining was calculated relative to that of the isotype control.
Preparation of acid-conditioned medium
One M of HCl was added to adjust medium to pH 6.6 as monitored using a microelectrode (Lazarlab). Medium was equilibrated overnight in a 5% CO2 incubator at 37° C and the pH was confirmed and sterilized using a 0.04 μm filter. All acid-conditioned media (ACM) were used within one week.
Cell proliferation assay
NP-MSC proliferation was determined using the Cell Counting Kit-8 (CCK-8, Dojindo Laboratories, Kumamoto, Japan) according to the manufacturer’s protocol. NP-MSCs were seeded at 5×103 cells/well in triplicate in 96-well plates. Cells of experiment groups were cultured in ACM (pH 6.6) at 37° C with 5% CO2 except for that of the negative control group which were grown in normal media. As indicated, 100 μM Amiloride (Sigma), 20 nM PcTx1 (MCE) or 100 nM APETx2 (Abcam) was added, respectively. Cell proliferation was measured at different times by gently removing the culture media and replacing with 100 μl standard medium supplemented with 10 μl CCK-8 solution. After 4 h incubation at 37° C in the dark, the optical density (OD) values were measured at 450 nm using a SpectraMax microplate reader (Molecular Devices, USA).
Senescence-associated β-galactosidase (SA-β-gal) staining
SA-β-gal staining was performed according to the manufacturer’s instructions after culture in 6-well plates (Beyotime, China). Briefly, after washing with PBS, cells were fixed in 1 ml of SA-β-gal fixative solution for 15 min at room temperature, rinsed three times with PBS and incubated in SA-β-gal working solution (Reagents A, B, C, and X-Gal) overnight at 37° C under atmospheric conditions. The percentage of SA-β-gal-positive cells from three randomly selected fields was counted under microscopic examination with at least 1000 cells counted for each sample.
Cell cycle analysis
DNA content analysis was performed using the Cell Cycle Analysis Kit (BD Biosciences, USA) according to the manufacturer’s instructions. Cells were collected by trypsinization, washed twice with cold PBS and fixed with pre-chilled 75% ethanol solution overnight at -20° C. The next day, cells were pelleted by centrifugation at 1500×g for 3 min, washed twice with ice cold PBS and resuspended in 50 μl propidium iodide (PI) staining solution (BD Pharmingen, USA). After incubation for 15 min in the dark on ice, the samples were interrogated by flow cytometry using the 488 nm laser (Beckman) and the percentage of cells in the G0/G1, G2/M, and S phases determined using ModFit software (Verity Software House, Inc., Topsham, ME, USA).
Senescence-associated secretory phenotype (SASP) assays
Secreted IL-6 and IL-8 levels were measured using the human interleukin 6 (IL-6) and interleukin-8 (IL-8/CXCL8) ELISA kits (MM-0049H1 and MM-1558H1, Jiangsu Meimian Industrial Co., Ltd) according to the recommended protocols. Briefly, primary antibodies were used to coat microtiter plate wells before the addition of 1:5 diluted samples, followed by HRP-conjugates and TMB substrate solution. Reactions were terminated by sulphuric acid solution, sample ODs measured at 450 nm and the concentrations of IL-6 and IL-8 determined against a standard curve.
Western blotting analyses
Cell lysates were prepared using RIPA lysis butter (Beyotime P0013) and protein concentrations estimated using a Bradford protein assay (Sangon Biotech, China). Thirty μg of total protein was resolved by SDS-PAGE electrophoresis and then electro-transferred onto PVDF membranes (Bio-Rad, USA). Membranes were blocked with 5% skim milk in Tris-Buffered Saline-Tween-20 (TBST) for 1.5 h before incubating with diluted primary antibodies at 4° C overnight as follows: ASIC1 (Abcam ab240896, 1:1000), ASIC2 (Proteintech 17851-1-AP, 1:1000), ASIC3 (Alomone ASC018AN0702, 1:200), ASIC4 (Proteintech 12003-1-AP, 1:1000), p16 (Santa Cruz sc-166760, 1:500), p21 (Proteintech 10355-1-AP, 1:500), p53 (Proteintech 10442-1-AP, 1:1000), pRb1 (Proteintech 10048-2-Ig, 1:500), MMP3 (Proteintech 6338-1-Ig, 1:500), MMP9 (Proteintech 10375-2-AP, 1:500), Aggrecan (Proteintech 13880-1-AP, 1:500), Collagen II (Proteintech 15943-1-AP, 1:500) and β-actin (Proteintech 66009-1-lg, 1:10000). Antibodies were detected with the corresponding species-specific secondary antibodies at room temperature for 1 h and after washing with TBST, immunoreactive bands were observed by enhanced chemiluminescence (ECL) based detection (Thermo U1291095A). Densitometric analyses were performed using Image J with loading normalization performed against the β-actin control.
Statistical analyses
All experiments were performed at least in triplicate with data presented as the mean ± standard deviation (SD). Student’s t test or one-way analysis of variance (ANOVA) was used to compare between treatment groups as indicated using GraphPad Prism 7 (GraphPad Software, Inc, La Jolla, CA, USA). P values < 0.05 were considered statistically significant.
Supplementary Materials
Author Contributions
C.L.S, X.Y.L designed the research; J.Y.D, R.J.Z, H.M.L, Q.J and X.M.C performed the experiments; J.Y.D, H.M.L, Q.J analyzed the data; J.Y.D, X.Y.L, C.L.S, H.H and R.F.T wrote the manuscript. All authors reviewed and approved the submitted manuscript.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
This work was supported by the National Natural Science Foundation of China [Grant numbers 8177090864, 81772908].
References
- 1. Hart FD. Back, Pain in. French’s Index Differ Diagnosis. 1979; 85–91. https://doi.org/10.1016/B978-0-7236-0490-7.50029-X
- 2. James SL, Abate D, Abate KH, Abay SM, Abbafati C, Abbasi N, Abbastabar H, Abd-Allah F, Abdela J, Abdelalim A, Abdollahpour I, Abdulkader RS, Abebe Z, et al, and GBD 2017 Disease and Injury Incidence and Prevalence Collaborators. Global, regional, and national incidence, prevalence, and years lived with disability for 354 diseases and injuries for 195 countries and territories, 1990-2017: a systematic analysis for the global burden of disease study 2017. Lancet. 2018; 392:1789–858. https://doi.org/10.1016/S0140-6736(18)32279-7 [PubMed]
- 3. Vos T, Flaxman AD, Naghavi M, Lozano R, Michaud C, Ezzati M, Shibuya K, Salomon JA, Abdalla S, Aboyans V, Abraham J, Ackerman I, Aggarwal R, et al. Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990-2010: a systematic analysis for the global burden of disease study 2010. Lancet. 2012; 380:2163–96. https://doi.org/10.1016/S0140-6736(12)61729-2 [PubMed]
- 4. Raj PP. Intervertebral disc: anatomy-physiology-pathophysiology-treatment. Pain Pract. 2008; 8:18–44. https://doi.org/10.1111/j.1533-2500.2007.00171.x [PubMed]
- 5. Huang YC, Urban JP, Luk KD. Intervertebral disc regeneration: do nutrients lead the way? Nat Rev Rheumatol. 2014; 10:561–66. https://doi.org/10.1038/nrrheum.2014.91 [PubMed]
- 6. Weiss DJ, Bates JH, Gilbert T, Liles WC, Lutzko C, Rajagopal J, Prockop D. Stem cells and cell therapies in lung biology and diseases: conference report. Ann Am Thorac Soc. 2013; 10:S25–44. https://doi.org/10.1513/AnnalsATS.201304-089AW [PubMed]
- 7. Blanco JF, Graciani IF, Sanchez-Guijo FM, Muntión S, Hernandez-Campo P, Santamaria C, Carrancio S, Barbado MV, Cruz G, Gutierrez-Cosío S, Herrero C, San Miguel JF, Briñon JG, del Cañizo MC. Isolation and characterization of mesenchymal stromal cells from human degenerated nucleus pulposus: comparison with bone marrow mesenchymal stromal cells from the same subjects. Spine (Phila Pa 1976). 2010; 35:2259–65. https://doi.org/10.1097/BRS.0b013e3181cb8828 [PubMed]
- 8. Sakai D, Andersson GB. Stem cell therapy for intervertebral disc regeneration: obstacles and solutions. Nat Rev Rheumatol. 2015; 11:243–56. https://doi.org/10.1038/nrrheum.2015.13 [PubMed]
- 9. Sakai D, Nakamura Y, Nakai T, Mishima T, Kato S, Grad S, Alini M, Risbud MV, Chan D, Cheah KS, Yamamura K, Masuda K, Okano H, et al. Exhaustion of nucleus pulposus progenitor cells with ageing and degeneration of the intervertebral disc. Nat Commun. 2012; 3:1264. https://doi.org/10.1038/ncomms2226 [PubMed]
- 10. Grunhagen T, Shirazi-Adl A, Fairbank JC, Urban JP. Intervertebral disk nutrition: a review of factors influencing concentrations of nutrients and metabolites. Orthop Clin North Am. 2011; 42:465–77. https://doi.org/10.1016/j.ocl.2011.07.010 [PubMed]
- 11. Roberts S, Urban JP, Evans H, Eisenstein SM. Transport properties of the human cartilage endplate in relation to its composition and calcification. Spine (Phila Pa 1976). 1996; 21:415–20. https://doi.org/10.1097/00007632-199602150-00003 [PubMed]
- 12. Bibby SR, Jones DA, Ripley RM, Urban JP. Metabolism of the intervertebral disc: effects of low levels of oxygen, glucose, and pH on rates of energy metabolism of bovine nucleus pulposus cells. Spine (Phila Pa 1976). 2005; 30:487–96. https://doi.org/10.1097/01.brs.0000154619.38122.47 [PubMed]
- 13. Ichimura K, Tsuji H, Matsui H, Makiyama N. Cell culture of the intervertebral disc of rats: factors influencing culture, proteoglycan, collagen, and deoxyribonucleic acid synthesis. J Spinal Disord. 1991; 4:428–36. https://doi.org/10.1097/00002517-199112000-00004 [PubMed]
- 14. Nachemson A. Intradiscal measurements of pH in patients with lumbar rhizopathies. Acta Orthop Scand. 1969; 40:23–42. https://doi.org/10.3109/17453676908989482 [PubMed]
- 15. Diamant B, Karlsson J, Nachemson A. Correlation between lactate levels and pH in discs of patients with lumbar rhizopathies. Experientia. 1968; 24:1195–96. https://doi.org/10.1007/BF02146615 [PubMed]
- 16. Sampara P, Banala RR, Vemuri SK, Av GR, Gpv S. Understanding the molecular biology of intervertebral disc degeneration and potential gene therapy strategies for regeneration: a review. Gene Ther. 2018; 25:67–82. https://doi.org/10.1038/s41434-018-0004-0 [PubMed]
- 17. Liu J, Tao H, Wang H, Dong F, Zhang R, Li J, Ge P, Song P, Zhang H, Xu P, Liu X, Shen C. Biological behavior of human nucleus pulposus mesenchymal stem cells in response to changes in the acidic environment during intervertebral disc degeneration. Stem Cells Dev. 2017; 26:901–11. https://doi.org/10.1089/scd.2016.0314 [PubMed]
- 18. Deval E, Gasull X, Noël J, Salinas M, Baron A, Diochot S, Lingueglia E. Acid-sensing ion channels (ASICs): pharmacology and implication in pain. Pharmacol Ther. 2010; 128:549–58. https://doi.org/10.1016/j.pharmthera.2010.08.006 [PubMed]
- 19. Leng T, Lin J, Cottrell JE, Xiong ZG. Subunit and frequency-dependent inhibition of acid sensing ion channels by local anesthetic tetracaine. Mol Pain. 2013; 9:27. https://doi.org/10.1186/1744-8069-9-27 [PubMed]
- 20. Li X, Fei J, Lei Z, Liu K, Wu J, Meng T, Yu J, Li J. Chloroquine impairs visual transduction via modulation of acid sensing ion channel 1a. Toxicol Lett. 2014; 228:200–06. https://doi.org/10.1016/j.toxlet.2014.05.008 [PubMed]
- 21. Zhou RP, Wu XS, Wang ZS, Xie YY, Ge JF, Chen FH. Novel insights into acid-sensing ion channels: implications for degenerative diseases. Aging Dis. 2015; 7:491–501. https://doi.org/10.14336/AD.2015.1213 [PubMed]
- 22. Zhou R, Wu X, Wang Z, Ge J, Chen F. Interleukin-6 enhances acid-induced apoptosis via upregulating acid-sensing ion channel 1a expression and function in rat articular chondrocytes. Int Immunopharmacol. 2015; 29:748–60. https://doi.org/10.1016/j.intimp.2015.08.044 [PubMed]
- 23. Jin C, Ye QH, Yuan FL, Gu YL, Li JP, Shi YH, Shen XM, Liu B, Lin ZH. Involvement of acid-sensing ion channel 1α in hepatic carcinoma cell migration and invasion. Tumour Biol. 2015; 36:4309–17. https://doi.org/10.1007/s13277-015-3070-6 [PubMed]
- 24. Uchiyama Y, Guttapalli A, Gajghate S, Mochida J, Shapiro IM, Risbud MV. SMAD3 functions as a transcriptional repressor of acid-sensing ion channel 3 (ASIC3) in nucleus pulposus cells of the intervertebral disc. J Bone Miner Res. 2008; 23:1619–28. https://doi.org/10.1359/jbmr.080502 [PubMed]
- 25. Sun X, Zhao D, Li YL, Sun Y, Lei XH, Zhang JN, Wu MM, Li RY, Zhao ZF, Zhang ZR, Jiang CL. Regulation of ASIC1 by Ca2+/calmodulin-dependent protein kinase II in human glioblastoma multiforme. Oncol Rep. 2013; 30:2852–58. https://doi.org/10.3892/or.2013.2777 [PubMed]
- 26. Lingueglia E. Acid-sensing ion channels in sensory perception. J Biol Chem. 2007; 282:17325–29. https://doi.org/10.1074/jbc.R700011200 [PubMed]
- 27. Wang YC, Li WZ, Wu Y, Yin YY, Dong LY, Chen ZW, Wu WN. Acid-sensing ion channel 1a contributes to the effect of extracellular acidosis on NLRP1 inflammasome activation in cortical neurons. J Neuroinflammation. 2015; 12:246. https://doi.org/10.1186/s12974-015-0465-7 [PubMed]
- 28. Chu XP, Grasing KA, Wang JQ. Acid-sensing ion channels contribute to neurotoxicity. Transl Stroke Res. 2014; 5:69–78. https://doi.org/10.1007/s12975-013-0305-y [PubMed]
- 29. Xiong ZG, Zhu XM, Chu XP, Minami M, Hey J, Wei WL, MacDonald JF, Wemmie JA, Price MP, Welsh MJ, Simon RP. Neuroprotection in ischemia: blocking calcium-permeable acid-sensing ion channels. Cell. 2004; 118:687–98. https://doi.org/10.1016/j.cell.2004.08.026 [PubMed]
- 30. Wemmie JA, Price MP, Welsh MJ. Acid-sensing ion channels: advances, questions and therapeutic opportunities. Trends Neurosci. 2006; 29:578–86. https://doi.org/10.1016/j.tins.2006.06.014 [PubMed]
- 31. Uchiyama Y, Cheng CC, Danielson KG, Mochida J, Albert TJ, Shapiro IM, Risbud MV. Expression of acid-sensing ion channel 3 (ASIC3) in nucleus pulposus cells of the intervertebral disc is regulated by p75NTR and ERK signaling. J Bone Miner Res. 2007; 22:1996–2006. https://doi.org/10.1359/jbmr.070805 [PubMed]
- 32. Li X, Wu FR, Xu RS, Hu W, Jiang DL, Ji C, Chen FH, Yuan FL. Acid-sensing ion channel 1a-mediated calcium influx regulates apoptosis of endplate chondrocytes in intervertebral discs. Expert Opin Ther Targets. 2014; 18:1–14. https://doi.org/10.1517/14728222.2014.859248 [PubMed]
- 33. Sherwood TW, Frey EN, Askwith CC. Structure and activity of the acid-sensing ion channels. Am J Physiol Cell Physiol. 2012; 303:C699–710. https://doi.org/10.1152/ajpcell.00188.2012 [PubMed]
- 34. Hu W, Chen FH, Yuan FL, Zhang TY, Wu FR, Rong C, Jiang S, Tang J, Zhang CC, Lin MY. Blockade of acid-sensing ion channels protects articular chondrocytes from acid-induced apoptotic injury. Inflamm Res. 2012; 61:327–35. https://doi.org/10.1007/s00011-011-0414-6 [PubMed]
- 35. Hwang HS, Kim HA. Chondrocyte apoptosis in the pathogenesis of osteoarthritis. Int J Mol Sci. 2015; 16:26035–54. https://doi.org/10.3390/ijms161125943 [PubMed]
- 36. Campisi J. Aging, cellular senescence, and cancer. Annu Rev Physiol. 2013; 75:685–705. https://doi.org/10.1146/annurev-physiol-030212-183653 [PubMed]
- 37. Muñoz-Espín D, Serrano M. Cellular senescence: from physiology to pathology. Nat Rev Mol Cell Biol. 2014; 15:482–96. https://doi.org/10.1038/nrm3823 [PubMed]
- 38. Burton DG, Krizhanovsky V. Physiological and pathological consequences of cellular senescence. Cell Mol Life Sci. 2014; 71:4373–86. https://doi.org/10.1007/s00018-014-1691-3 [PubMed]
- 39. Naylor RM, Baker DJ, van Deursen JM. Senescent cells: a novel therapeutic target for aging and age-related diseases. Clin Pharmacol Ther. 2013; 93:105–16. https://doi.org/10.1038/clpt.2012.193 [PubMed]
- 40. Liu S, Liang H, Lee SM, Li Z, Zhang J, Fei Q. Isolation and identification of stem cells from degenerated human intervertebral discs and their migration characteristics. Acta Biochim Biophys Sin (Shanghai). 2017; 49:101–49. https://doi.org/10.1093/abbs/gmw121 [PubMed]
- 41. Krtolica A, Parrinello S, Lockett S, Desprez PY, Campisi J. Senescent fibroblasts promote epithelial cell growth and tumorigenesis: a link between cancer and aging. Proc Natl Acad Sci USA. 2001; 98:12072–77. https://doi.org/10.1073/pnas.211053698 [PubMed]
- 42. Coppé JP, Patil CK, Rodier F, Sun Y, Muñoz DP, Goldstein J, Nelson PS, Desprez PY, Campisi J. Senescence-associated secretory phenotypes reveal cell-nonautonomous functions of oncogenic RAS and the p53 tumor suppressor. PLoS Biol. 2008; 6:2853–68. https://doi.org/10.1371/journal.pbio.0060301 [PubMed]
- 43. Rodier F, Coppé JP, Patil CK, Hoeijmakers WA, Muñoz DP, Raza SR, Freund A, Campeau E, Davalos AR, Campisi J. Persistent DNA damage signalling triggers senescence-associated inflammatory cytokine secretion. Nat Cell Biol. 2009; 11:973–79. https://doi.org/10.1038/ncb1909 [PubMed]
- 44. Jackson JG, Pereira-Smith OM. P53 is preferentially recruited to the promoters of growth arrest genes p21 and GADD45 during replicative senescence of normal human fibroblasts. Cancer Res. 2006; 66:8356–60. https://doi.org/10.1158/0008-5472.CAN-06-1752 [PubMed]
- 45. Calcinotto A, Kohli J, Zagato E, Pellegrini L, Demaria M, Alimonti A. Cellular senescence: aging, cancer, and injury. Physiol Rev. 2019; 99:1047–78. https://doi.org/10.1152/physrev.00020.2018 [PubMed]
- 46. Flores JM, Martín-Caballero J, García-Fernández RA. P21 and p27 a shared senescence history. Cell Cycle. 2014; 13:1655–56. https://doi.org/10.4161/cc.29147 [PubMed]
- 47. Pittayapruek P, Meephansan J, Prapapan O, Komine M, Ohtsuki M. Role of matrix metalloproteinases in photoaging and photocarcinogenesis. Int J Mol Sci. 2016; 17:868. https://doi.org/10.3390/ijms17060868 [PubMed]
- 48. Risbud MV, Guttapalli A, Tsai TT, Lee JY, Danielson KG, Vaccaro AR, Albert TJ, Gazit Z, Gazit D, Shapiro IM. Evidence for skeletal progenitor cells in the degenerate human intervertebral disc. Spine (Phila Pa 1976). 2007; 32:2537–44. https://doi.org/10.1097/BRS.0b013e318158dea6 [PubMed]
- 49. Sang C, Cao X, Chen F, Yang X, Zhang Y. Differential characterization of two kinds of stem cells isolated from rabbit nucleus pulposus and annulus fibrosus. Stem Cells Int. 2016; 2016:8283257. https://doi.org/10.1155/2016/8283257 [PubMed]
- 50. Erwin WM, Islam D, Eftekarpour E, Inman RD, Karim MZ, Fehlings MG. Intervertebral disc-derived stem cells: implications for regenerative medicine and neural repair. Spine (Phila Pa 1976). 2013; 38:211–16. https://doi.org/10.1097/BRS.0b013e318266a80d [PubMed]
- 51. Wang H, Zhou Y, Chu TW, Li CQ, Wang J, Zhang ZF, Huang B. Distinguishing characteristics of stem cells derived from different anatomical regions of human degenerated intervertebral discs. Eur Spine J. 2016; 25:2691–704. https://doi.org/10.1007/s00586-016-4522-4 [PubMed]
- 52. Zhou G, Dai L, Jiang X, Ma Z, Ping J, Li J, Li X. Effects of human midkine on spontaneous resorption of herniated intervertebral discs. Int Orthop. 2010; 34:103–08. https://doi.org/10.1007/s00264-009-0740-2 [PubMed]
- 53. Wu X, Ren G, Zhou R, Ge J, Chen FH. The role of Ca2+ in acid-sensing ion channel 1a-mediated chondrocyte pyroptosis in rat adjuvant arthritis. Lab Invest. 2019; 99:499–513. https://doi.org/10.1038/s41374-018-0135-3 [PubMed]
- 54. Yuan FL, Zhao MD, Jiang DL, Jin C, Liu HF, Xu MH, Hu W, Li X. Involvement of acid-sensing ion channel 1a in matrix metabolism of endplate chondrocytes under extracellular acidic conditions through NF-κB transcriptional activity. Cell Stress Chaperones. 2016; 21:97–104. https://doi.org/10.1007/s12192-015-0643-7 [PubMed]
- 55. Xie ZY, Chen L, Zhang C, Liu L, Wang F, Cai F, Wang XH, Shi R, Sinkemani A, Yu HM, Hong X, Wu XT. Acid-Sensing Ion Channel 1a Regulates Fate of Rat Nucleus Pulposus Cells in Acid Stimulus Through Endoplasmic Reticulum Stress. Biores Open Access. 2018; 7:2–9. https://doi.org/10.1089/biores.2017.0049 [PubMed]
- 56. Han B, Wang HC, Li H, Tao YQ, Liang CZ, Li FC, Chen G, Chen QX. Nucleus pulposus mesenchymal stem cells in acidic conditions mimicking degenerative intervertebral discs give better performance than adipose tissue-derived mesenchymal stem cells. Cells Tissues Organs. 2014; 199:342–52. https://doi.org/10.1159/000369452 [PubMed]
- 57. Gilbert HT, Hodson N, Baird P, Richardson SM, Hoyland JA. Acidic pH promotes intervertebral disc degeneration: acid-sensing ion channel -3 as a potential therapeutic target. Sci Rep. 2016; 6:37360. https://doi.org/10.1038/srep37360 [PubMed]
- 58. Childs BG, Durik M, Baker DJ, van Deursen JM. Cellular senescence in aging and age-related disease: from mechanisms to therapy. Nat Med. 2015; 21:1424–35. https://doi.org/10.1038/nm.4000 [PubMed]
- 59. Calcinotto A, Alimonti A. Aging tumour cells to cure cancer: "pro-senescence" therapy for cancer. Swiss Med Wkly. 2017; 147:w14367. [PubMed]
- 60. Roy A, Banerjee S. P27 and leukemia: cell cycle and beyond. J Cell Physiol. 2015; 230:504–09. https://doi.org/10.1002/jcp.24819 [PubMed]
- 61. Vo N, Seo HY, Robinson A, Sowa G, Bentley D, Taylor L, Studer R, Usas A, Huard J, Alber S, Watkins SC, Lee J, Coehlo P, et al. Accelerated aging of intervertebral discs in a mouse model of progeria. J Orthop Res. 2010; 28:1600–07. https://doi.org/10.1002/jor.21153 [PubMed]
- 62. Le Maitre CL, Freemont AJ, Hoyland JA. Accelerated cellular senescence in degenerate intervertebral discs: a possible role in the pathogenesis of intervertebral disc degeneration. Arthritis Res Ther. 2007; 9:R45. https://doi.org/10.1186/ar2198 [PubMed]
- 63. Carnero A, Blanco C, Blanco F, Castro ME, Guijarro MV, Fominaya J, Link W, Martínez JF, Moneo V, Renner O, Romero L, Rosado A. Exploring cellular senescence as a tumor suppressor mechanism. Clin Transl Oncol. 2003; 5:249–65.
- 64. Beauséjour CM, Krtolica A, Galimi F, Narita M, Lowe SW, Yaswen P, Campisi J. Reversal of human cellular senescence: roles of the p53 and p16 pathways. EMBO J. 2003; 22:4212–22. https://doi.org/10.1093/emboj/cdg417 [PubMed]
- 65. Campisi J. Senescent cells, tumor suppression, and organismal aging: good citizens, bad neighbors. Cell. 2005; 120:513–22. https://doi.org/10.1016/j.cell.2005.02.003 [PubMed]
- 66. Ben-Porath I, Weinberg RA. The signals and pathways activating cellular senescence. Int J Biochem Cell Biol. 2005; 37:961–76. https://doi.org/10.1016/j.biocel.2004.10.013 [PubMed]
- 67. Ben-Porath I, Weinberg RA. When cells get stressed: an integrative view of cellular senescence. J Clin Invest. 2004; 113:8–13. https://doi.org/10.1172/JCI20663 [PubMed]
- 68. Feng C, Yang M, Zhang Y, Lan M, Huang B, Liu H, Zhou Y. Cyclic mechanical tension reinforces DNA damage and activates the p53-p21-Rb pathway to induce premature senescence of nucleus pulposus cells. Int J Mol Med. 2018; 41:3316–26. https://doi.org/10.3892/ijmm.2018.3522 [PubMed]
- 69. Sakai D, Grad S. Advancing the cellular and molecular therapy for intervertebral disc disease. Adv Drug Deliv Rev. 2015; 84:159–71. https://doi.org/10.1016/j.addr.2014.06.009 [PubMed]
- 70. Yurube T, Ito M, Kakiuchi Y, Kuroda R, Kakutani K. Autophagy and mTOR signaling during intervertebral disc aging and degeneration. JOR Spine. 2020; 3:e1082. https://doi.org/10.1002/jsp2.1082 [PubMed]
- 71. Kwon WK, Moon HJ, Kwon TH, Park YK, Kim JH. The role of hypoxia in angiogenesis and extracellular matrix regulation of intervertebral disc cells during inflammatory reactions. Neurosurgery. 2017; 81:867–75. https://doi.org/10.1093/neuros/nyx149 [PubMed]



