Introduction
Mitogen-activated protein kinase kinase kinase 5 (MAP3K5, also termed ASK1 [apoptosis signal-regulating kinase 1]) is a member of a family of enzymes involved in kinase signaling cascades in the cell. MAP3K5 plays an important role in cellular responses evoked by changes in environment. These include cell differentiation and survival, apoptosis, innate immune response, and oxidative stress response [1]. It mediates signal transduction of oxidative stress and receptor-mediated inflammatory signals, such as ones involving tumor necrosis factor and lipopolysaccharide. Its crucial role in the apoptosis signal transduction pathway is mediated by mitochondria-dependent caspase activation. MAP3K5 plays a role in the pathology of a wide range of diseases in which reactive oxygen species (ROS) and/or endoplasmic reticulum stress are causative factors. MAP3K5/ASK1 may influence in vivo insulin action and obesity, and MAP3K5 variants are associated with type 2 diabetes [2]. MAP3K5 may prevent stress-induced disorders and protect from bacterial and viral infection under physiological circumstances. On the contrary, it may exert adverse effects through excessive cellular apoptosis and increased inflammation under some pathological conditions, such as present in neurodegenerative disorders, cardiovascular diseases, inflammatory diseases, and chronic inflammation-induced carcinogenesis [3]. Once activated, MAP3K5/ASK1 acts as an upstream activator of the MKK/JNK and p38 MAPK signal transduction cascades through phosphorylation and activation of several MAP kinase kinases such as MAP2K4/SEK1, MAP2K3/MKK3, MAP2K6/MKK6 and MAP2K7/MKK7. These in turn activate p38 MAPKs and c-jun N-terminal kinases (JNKs). Both p38 MAPK and JNKs control the transcription factor activator protein-1 (AP-1) [4]. While the above pathways are critical for senescence, aging, and age-associated cardiovascular diseases MAP3K5 might not be the cause, but rather a response [5, 6].
In a study of 33 single nucleotide polymorphisms (SNPs) of MAP3K5 in our cohort of American men of Japanese ancestry (Supplementary Methods, Supplementary Tables 1, 2), alleles of two adjacent SNPs, and haplotypes of these alleles, were associated with longevity in men aged ≥ 95 years [7]. Bonferroni corrected p-values were 0.0043 for the SNP rs2076260 and 0.032 for the SNP rs6904753 in a heterozygous disadvantage model (Supplementary Table 3). Haplotype analysis of these yielded a p-value of 0.00004. Our rationale for studying MAP3K5 was because in mouse liver Map3k5 was differentially expressed in response to caloric restriction [8], a well-known longevity enhancer.
In the present study we tested the hypothesis that the longevity-associated alleles of MAP3K5 SNP rs2076260 mediate their effect on lifespan at least in part by protection against the detrimental effects of aging-related cardiometabolic diseases (CMD), namely diabetes and/or hypertension and/or coronary heart disease (CHD).
Results
Characteristics of subjects
Baseline (1991–1993) characteristics of men in the study for each genotype of rs2076260, adjusting, for age, genotype, and prevalence of medical conditions are shown in Supplementary Table 4. By December 31, 2019, 3,480 out of 3,516 subjects had died during the 29 years of follow-up. At baseline, among the 3,516 participants, 29% had been diagnosed with diabetes alone, 53% with hypertension alone, 21% with CHD alone, and 14% with cancer alone. Seventy percent had at least one CMD, and 5.2% had all three CMDs. Mean age at death was 88.6 ± 6.0 years for men with at least one CMD, and 89.4 ± 6.0 years for men who did not have any of the CMDs (p < 0.0001).
MAP3K5 genotype and survival in men with a CMD and men without a CMD
Survival curves for men without a CMD and those with a CMD showed that those who did not have a CMD lived longer (Kaplan-Meier Log-rank χ2 = 24.7, p = 1.9x10–6). Figure 1(A) shows survival curves for men with a CMD and men without a CMD according to whether they were major allele homozygotes (CC) of rs2076260 or minor allele carriers (CT/TT). Figure 1(B) shows survival curves for the men with a CMD and for men who did not have a CMD according to whether they were heterozygous (CT) or homozygous (CC+TT) for each allele. These curves were determined using a Cox proportional hazard model adding an interaction term of “MAP3K5 with CMD”. Only in men with CMD was the longevity-associated genotype associated with greater lifespan than the alternate genotype: p = 0.00004 for major allele homozygotes (CC) vs. minor allele carriers (CC/TT), and p = 0.000006 for heterozygotes (CT) vs. homozygotes (CC+TT). Overall, men without a CMD had the longest lifespans. Moreover, importantly, we noted that there was no statistical difference in lifespan for each genotype in men who did not have a CMD. Figure 2 shows forest plots of mortality risk for men with a CMD and men without a CMD for each genetic model.
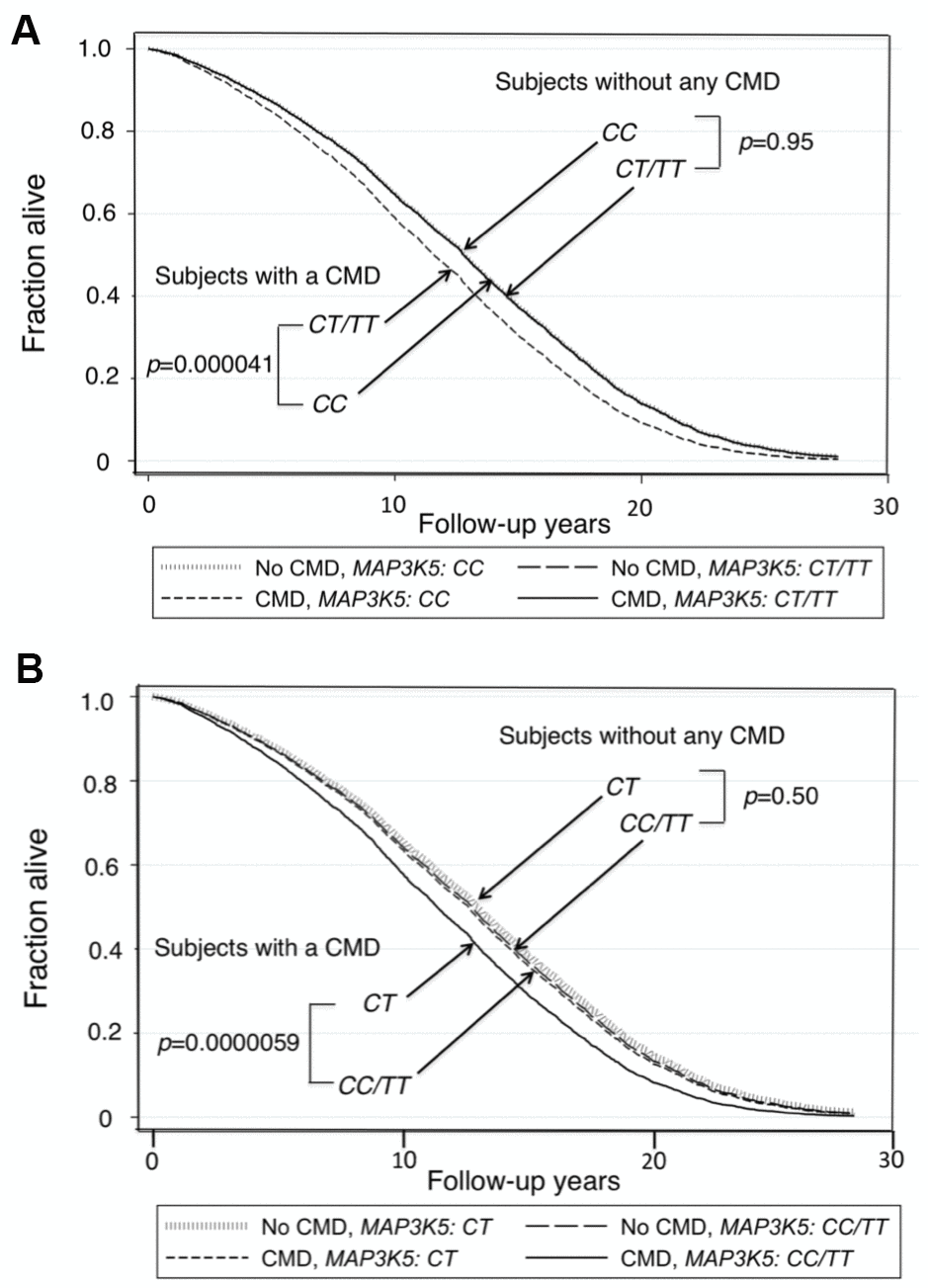
Figure 1. Survival curves spanning the period from baseline (1991–1993) to Dec 31, 2019 for men with and men without a CMD according to genotypes of MAP3K5 SNP rs2076260. The survival probabilities were estimated from the Cox proportional hazard model: h(t) = h(t0) * exp(β1*Age + β2*BMI + β3*Glucose + β4*CMD + β5*MAP3K5_xx + β6* (CMD*MAP3K5_xx)), where “xx” is genotype, by fixing age at 75 years, BMI at the mean, 23.5 kg/m2, and glucose at the mean, 113 mg/dL (where β6 is the effect of the interaction of CMD with MAP3K5 genotype on mortality, for CC vs CT/TT, i.e., a recessive model, giving p(β6) = 0.023). (A) Survival curves for men with a CMD vs. men without a CMD for major allele homozygote (CC) vs. minor allele carriers (CT+TT) (p=0.000041 and p=0.95, respectively). (B) Survival curves for men with a CMD vs. men without a CMD for heterozygote disadvantage model, CT vs. CC/TT (p=0.0000059 and p=0.50, respectively, giving p(β6) = 0.057).
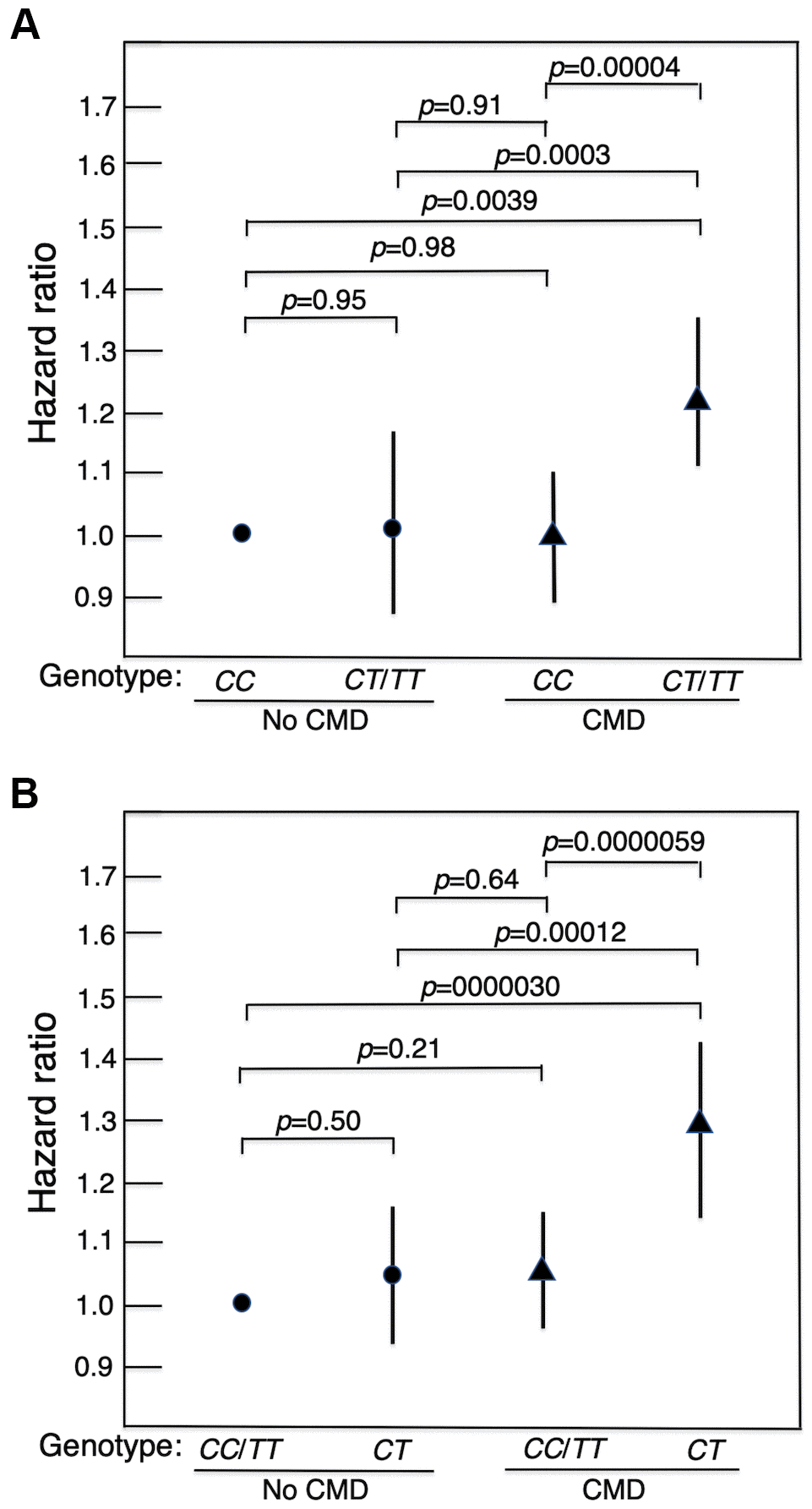
Figure 2. Forest plots of mortality risk (hazard ratio and 95% CI), adjusted for age, BMI and glucose at baseline, for men with a CMD and men without a CMD according to genotype of MAP3K5 SNP rs2076260 in each genetic model. (A) major allele homozygote (CC) vs. minor allele carriers (CT+TT). (B) heterozygote disadvantage model, CT vs. CC/TT. It can be seen that in men with a CMD who had the longevity-associated genotype, mortality risk was reduced to normal in that it did not differ significantly from the survival curve in men without a CMD. It should be noted that the HRs in Figure 2 differ slightly from those in Table 1. This is because the HRs in Table 1 were obtained from stratified analyses by diabetes, hypertension, CHD, and any CMD (i.e., were separately estimated by disease status). In Figure 2, we compared the HRs for the 4 groups by CMD and MAP3K5 genotype. The HRs and p-values for pairwise comparisons among the 4 groups were estimated in one Cox model.
Table 1. Hazard ratios (HR) by genotype of MAP3K5 SNP rs2076260 with total mortality in men with diabetes, CHD, hypertension, and any of these CMDs.
| Disease (n with, total) | Cox model* | Genetic model** | With a CMD | Without a CMD | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HR (95% CI) | p | HR (95% CI) | p | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diabetes | 1 | CT/TT vs. CC | 1.18 (1.03–1.36) | 0.016 | 1.11 (1.02–1.22) | 0.017 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| (990, 2478) | 2 | CT/TT vs. CC | 1.21 (1.05–1.41) | 0.011 | 1.08 (0.98–1.19) | 0.10 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hypertension | 1 | CT/TT vs. CC | 1.18 (1.07–1.30) | 0.0013 | 1.06 (0.95–1.19) | 0.27 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| (1877, 1639) | 2 | CT/TT vs. CC | 1.22 (1.09–1.36) | 0.00041 | 1.03 (0.92–1.16) | 0.60 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CHD | 1 | CT/TT vs. CC | 1.21 (1.02–1.42) | 0.026 | 1.11 (1.02–1.20) | 0.017 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| (724, 2792) | 2 | CT/TT vs. CC | 1.19 (0.99–1.43) | 0.059 | 1.11 (1.02–1.22) | 0.019 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Any CMD | 1 | CT/TT vs. CC | 1.20 (1.09–1.31) | 0.000075 | 0.99 (0.86–1.13) | 0.83 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| (2461, 1055) | 2 | CT/TT vs. CC | 1.23 (1.12–1.36) | 0.000023 | 0.90 (0.78–1.05) | 0.18 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Diabetes | 1 | CT vs. CC/TT | 1.25 (1.10–1.42) | 0.00054 | 1.14 (1.06–1.24) | 0.0010 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| (990, 2478) | 2 | CT vs. CC/TT | 1.26 (1.10–1.44) | 0.0010 | 1.10 (1.01–1.20) | 0.029 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hypertension | 1 | CT vs. CC/TT | 1.18 (1.08–1.29) | 0.00043 | 1.14 (1.04–1.26) | 0.0075 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| (1877, 1639) | 2 | CT vs. CC/TT | 1.21 (1.09–1.34) | 0.00021 | 1.08 (0.97–1.20) | 0.18 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CHD | 1 | CT vs. CC/TT | 1.34 (1.15–1.55) | 0.00012 | 1.14 (1.05–1.22) | 0.0009 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| (724, 2792) | 2 | CT vs. CC/TT | 1.33 (1.13–1.57) | 0.00058 | 1.11 (1.02–1.21) | 0.012 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Any CMD | 1 | CT vs. CC/TT | 1.21 (1.12–1.31) | 0.0000026 | 1.07 (0.95–1.21) | 0.25 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| (2461, 1055) | 2 | CT vs. CC/TT | 1.22 (1.12–1.33) | 0.0000081 | 0.98 (0.86–1.13) | 0.82 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| *Cox models: Model 1: Age-adjusted. Model 2: Covariate-adjusted, where covariates adjusted in Cox model were: age (years), BMI (kg/m2), glucose (mmol/l), insulin (mIU/dL), plasma fibrinogen (mg/dl), white blood count (103/μL), smoking (pack-years), alcohol intake (oz/mo), physical activity index, depression, cancer, and stroke. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| **Genetic models: Top half: Major allele homozygote (CC) model. Bottom half: Heterozygote disadvantage model. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Age-adjusted baseline variables of men with a CMD and men without a CMD according to genotype (CC, CT, TT) are shown in Supplementary Table 5. None of the variables showed a difference between genotypes. The variables included body mass index (BMI), wait-to-hip ratio, fasting plasma glucose, fasting plasma insulin, plasma fibrinogen, white blood cell count, smoking, alcohol intake and physical activity index. Analyses found no evidence of population stratification or admixture in the dataset (data not shown). In men with a CMD, prevalence of diabetes, hypertension and CHD was not statistically different between each genotype.
In men with a CMD, hazard ratios (HR) for association with mortality for just diabetes, just hypertension, just CHD, and for any of these CMDs were statistically significant in two genetic models (Table 1). These were for: (1) major allele homozygotes (CC) vs. minor allele carriers (CT/TT), and (2) heterozygotes (CT) vs. homozygotes of each allele (CC+TT). No significant difference was, however, found in two other genetic models, namely for minor allele homozygotes (TT) vs. major allele carriers (CC/CT), and for an additive model (Supplementary Table 6). Thus, in the 1st model, being homozygous for the major allele conferred strong protection against mortality in men with a CMD. In men without a CMD, however, lifespan was significantly longer irrespective of MAP3K5 genotype. In the 2nd model, heterozygotes were at a disadvantage in that men who had the CT genotype had a significantly higher risk of mortality than men who were homozygous for either the C or T allele.
Functional annotations
In an attempt to determine how MAP3K5 may influence disease resistance and help to identify biological pathways we examined the following: (1) MAP3K5 tissue expression, (2) transcription factors (TFs) that might be modified by our sentinel SNP (rs2076260), (3) SNPs in linkage disequilibrium (LD) that might modify transcription factor binding, (4) the expression patterns of these TFs, and (5) the location of any cis-regulatory elements that are physically linked with our sentinel SNP. Supplementary Figure 1 shows that MAP3K5 is expressed in most tissues, but notably at high levels in adrenal, ovary, and pituitary.
We screened SNPs in the block of LD containing rs2076260 (Supplementary Figure 2) for functional annotations using the HaploReg database. Seven additional SNPs were in near perfect LD with rs2076260 and are predicted to either modify transcription factor motifs and/or are predicted to alter promoter/enhancer histone marks or DNAse I sensitivity. The latter features are associated with access to regulatory proteins. All eight SNPs were examined in more detail (Supplementary Figure 3 and Supplementary Table 7). Five of those SNPs are predicted to modify TF binding and are shown in Supplementary Table 8. Also shown is whether the minor allele is predicted to create or abolish TF binding, the biological pathways, and the tissue expression patterns. While these TFs are expressed in most tissues, they are prominent in lung and thyroid. The biological pathways include: (1) activation of immunoglobulin heavy-chain transcription, (2) repression of GATA4 and GATA6 transcription, (3) unfolded protein response, (4) activation of many muscle-specific, growth factor-induced, and stressed-induced genes, and (5) expression of metallothionein proteins in response to exposure to heavy metals.
Since the MAP3K5 SNPs are in a non-coding region of the genome, they are presumed to affect transcription, whether directly or indirectly, as chromatin modifying units (i.e., cis-regulatory elements) (Supplementary Table 8). Screening of MAP3K5 for expression quantitative loci (eQTLs) using the GTEx portal identified 1,532 entries (not shown) and four splice-site sQTLs, which are variants associated with differential splicing (not shown). Most of the eQTLs in MAP3K5 are differentially expressed in artery (tibial), lung, breast, brain (cerebellar), esophagus (muscularis), spleen, and adipose (visceral) tissues, as defined in the GTEx database. As an aside, it should be noted that dbSNP misclassifies rs2076260 as a mis-sense mutation (https://www.ncbi.nlm.nih.gov/projects/SNP/snp_ref.cgi?do_not_redirect&rs=rs2076260). The base change is actually in the 3rd codon with no change in the encoded amino acid. We believe the confusion arose from sequencing of the opposite strand by some Human Genome Labs, where rs2076260 is a T → C change, whereas the complement is A → G.
The longevity-associated SNP rs2076260 and its nearest neighbor rs6906753 in LD (D’=0.99) demonstrated significant changes in binding capacities for TFs that included homeobox D10 (HOXD10), POU class 2 homeobox 2 (POU2F2), TATA box binding protein (TBP)-associated factor, RNA polymerase II (TATA), activating transcription factor 3 (ATF3), and Hes related family BHLH transcription factor with YRPW motif 1 (HEY1) (Supplementary Figure 3). The predicted effects of putative functional variants in the SNPs on TF binding and thus tissue expression patterns are shown in Supplementary Table 9.
In order to determine whether rs2076260 was near any additional regulatory sites and features, we examined the chromatin structures within and surrounding MAP3K5. Using the UCSC genome browser we identified 2 long noncoding RNAs (lncRNAs), MAP3K5-AS1 and LOC101928429, located within MAP3K5 (Supplementary Figure 4 and Supplementary Table 8). From the literature we identified a site, cg21506299, that is differentially methylated in individuals with increased BMI [9]. The above entities were mapped on to the LD heatmap (Supplementary Figure 2) using the LDhap Tool (https://analysistools.cancer.gov/LDlink/?tab=ldmatrix). The locations of these features (shown in Supplementary Figure 2 and summarized in Supplementary Table 8), indicate that the adjacent longevity SNPs (asterisked in Supplementary Table 2) overlap with MAP3K5-AS1, that is in LD. The differentially methylated site, cg21506299, overlaps with the lncRNA LOC101928429 near the promoter (Supplementary Figure 4).
A super-enhancer is a region of the mammalian genome comprising multiple enhancers collectively bound by an array of TFs to drive transcription of genes involved in cell identity. We identified super-enhancers overlapping the MAP3K5 promoter, using the dbSuper database (https://asntech.org/dbsuper/index.php). These enhancers were identified in various T- and B-cell lines. They are shown in Supplementary Figure 4 and listed in Supplementary Table 10.
We mapped additional potential regulatory sites using the WashU Genome Browser. Supplementary Figure 4 shows the locations of MAP3K5 relative to MAP3K5-AS1, rs2076260, cg21506299, LOC101928429, and super-enhancers. H3K4me3 indicates sites of tri-methylation of lysine 4 in histone H3 (green), locations of the H2 histone variant H2A.Z (blue), locations of RNA polymerase II (RNAPII) binding, and CTCF binding. H3K4me3 is associated with sites of open chromatin which are associated with activation of transcription of nearby genes [10]. H2A.Z is associated with regions of genome fluidity [10]. The MAP3K5 promoter overlaps with H3K4me3 sites as well as H2A.Z sites and is connected, through RNAPII binding/pausing to SNP rs2076260 as well as MAP3K5-AS1 and LOC101928429. Together these features are predicted to form a cis-regulatory unit. This is supported by the locations of CTCF binding sites that generally form insulator domains.
Discussion
The present study has found that the longevity-associated major allele of MAP3K5 SNP rs2076260 is associated with protection against mortality from CMD. As a result, individuals with either one, two or all of the disorders – diabetes, hypertension or CHD – live longer if they have the protective genotype. Our study showed in fact that lifespan of men with a CMD who had the longevity-associated allele was not only longer, but did not differ significantly from lifespan of men without CMD. This indicates that possession of the MAP3K5 longevity-associated genotype can mitigate the adverse effects on lifespan of having a CMD.
We also found an association with longevity when we tested the genetic data in a heterozygous disadvantage model. Heterozygote disadvantage is when a heterozygote has a lower overall fitness than either homozygote, and can be a potent driver of population genetic divergence [11].
Until our earlier study [7], MAP3K5 had not previously been linked directly to human aging and longevity. Most studies have, however, involved European populations [10]. According to dbSNP, the frequency of the C allele of rs2076260 in the 1000 Genomes East Asian super-population is 0.55, whereas it is only 0.17 in the European super-population (https://www.ncbi.nlm.nih.gov/snp/?term=rs2076260), thus diminishing the chance of detection of an association with longevity in genetic association studies of Europeans. Various genetic factors have been identified that are associated with aging-related risk factors and diseases [12]. In Japanese people, healthy traditional dietary, lifestyle and cultural factors, by reducing risk of CMDs, are thought to contribute to their longer average lifespans [13]. In extreme old age genetic factors become more important than environmental factors in lifespan determination [10]. Our study used a homogeneous population of American men of Japanese ancestry in which there was no evidence of population stratification or admixture [14]. Thus, various favorable factors appear to have contributed to our previous positive genetic finding of an association of SNPs in MAP3K5 with longevity [7].
MAP3K5 is involved in stress response, inflammation, and apoptosis [1]. Disruption of Map3k5 in mice attenuates left ventricular remodelling [15]. Prolonged activation of p38 or JNK by MAP3K5 results in long-term cellular damage. MAP3K5 inhibition may serve as a therapeutic target, as summarized in studies that have tested the effects of MAP3K5/ASK1 inhibition in cell and animal disease models, as well as in human clinical trials for a variety of diseases [16].
MAP3K5 is expressed in a large number of tissues, most notably adipose tissue, artery, Epstein barr virus-transformed lymphocytes, ovary, and pituitary (as documented in the GTEx database: https://gtexportal.org/home/gene/MAP3K5). Moreover, MAP3K5 has several isoforms that are differentially expressed in various tissues (see GTEx database). It is likely that the longevity-associated MAP3K5 genotype is in LD with functional allele(s) of regulatory variant(s) of MAP3K5 that have a stronger effect on gene expression than the alternative genotypes, so boosting the levels of the encoded MAP3K5 protein and leading to higher beneficial biological effects, such as apoptosis. Two longevity-associated SNPs, rs2076260 and its near neighbor rs6904753, in LD with each other, are predicted to influence binding capacities for six transcription factors. The common allele of rs2076260 is predicted to create/enhance binding of factors HOXD10, POU2F2, and TATA, thus increasing MAP3K5 expression, but reduce binding of DMRT1, while the common allele of rs6904753 is predicted to abolish/reduce binding of factors ATF3 and HEY1, which are generally repressive, and thus increase MAP3K5 expression. The additional SNPs in LD may also influence TF binding. Since these TFs are differentially expressed, their specific effect would depend on the tissue.
Data on the tissue expression distributions for the lncRNAs were not available. The lncRNA, MAP3K5-AS1, located within MAP3K5 close to rs2076260, may influence MAP3K5 expression, either negatively by recruiting RNAPII on the negative strand or positively by recruiting other transcription enhancers and/or splicing factors on the positive strand. Since MAP3K5-AS1 is transcribed in the opposite direction as MAP3K5, it operates in effect as an anti-sense RNA, interfering with MAP3K5 transcription, as above. The possibility of a role for MAP3K5-AS1 in the genotypic effect we have observed is therefore of interest. The cg21506299 site differentially methylated in subjects with increased BMI [9], and located in the MAP3K5 promoter, may be of interest in mediating the contrasting genotypic effect found in our men who had a CMD. This is because high BMI is associated with diabetes, hypertension and CHD. Another lncRNA, LOC101928429, at this site may influence transcription as well. Notable too was that the promoter and both lncRNAs overlap with peaks of H3K4me3 tri-methylation and of peaks of the histone variant H2A.Z, which is associated with regions of genome fluidity in cells [10].
In conclusion, in two closely related genetic models, we show that longevity-associated genotypes of MAP3K5 are associated with virtually complete mitigation of the lifespan shortening effect of having a CMD. Since lifespan was the same irrespective of genotype in men without a CMD, the overall association of genetic variation in MAP3K5 with longevity is contributed entirely by allele-dependent amelioration of the increased mortality risk conferred by CMD. This finding adds to our recent findings of a similar explanation for the longevity association conferred by particular SNPs in FOXO3 [17] and PIK3R5 [18]. The effect does not, however, explain the longevity association we have found for most other genes in our cohort of elderly men [7, 19] (unpublished).
Materials and Methods
Study participants
Genotyping
Genotyping methods were as described previously [7] (Supplementary Methods).
Variant search
Variants surrounding rs2076260 were screened on the RegulomeDB site, which includes known and predicted regulatory elements in the intergenic regions, as well as regions of DNAase hypersensitivity, binding sites for transcription factors, and promoter regions. Sources of these data included public datasets from GEO, the ENCODE project, and published literature [20]. Chromosome 6 locations used the GRCh37.p13 genome build (http://www.gencodegenes.org/releases/19.html).
We also screened the variants using HaploReg, which is a tool for exploring annotations of the noncoding genome at variants on haplotype blocks, such as candidate regulatory SNPs at disease-associated loci [21]. Using LD information from the 1000 Genomes Project, linked SNPs can be visualized along with chromatin state and protein binding annotation from the Roadmap Epigenomics and ENCODE projects, the effect of SNPs on regulatory motifs, and the effect of SNPs on expression from QTL studies. We searched HaploReg version 4.1 for the region between SNPs in LD, rs9494547 and rs9494552, Nov 5, 2015 build, hg38.
Statistical analyses
General linear models were used to compare age-adjusted indirect measurements between groups, and logistic models were used to compare the age-adjusted direct measurements. Cox proportional models were used to assess the association of MAP3K5 for various genetic models on mortality stratified by disease status, such as by diabetes, by hypertension, by CHD, and by any of CHD, diabetes, or hypertension. The Cox proportional hazard assumption was tested for each Cox model. The effect of interaction of disease with MAP3K5 genotype on mortality was tested in the Cox model. All statistical analyses were performed using the Statistical Analysis System version 9.4 [22]. Figures were generated using STATA 12 Graphics [23].
Author Contributions
B.J.M., R.C., T.A.D., K.H.M., D.C.W., R.C.A., B.J.W. contributed to the study concept and design; R.C. carried out the statistical analyses; K.H.M. supervised recruitment and data collection; T.A.D. supervised the genotyping; B.J.M. drafted the manuscript; R.C., B.J.M., T.A.D., K.H.M., R.C.A., D.C.W. and B.J.W. provided critical input into data interpretation and manuscript preparation.
Acknowledgments
The authors thank all study participants and their families for their cooperation and the Hawaii State Department of Health for its help. The authors wish to acknowledge Dr. Alvin T. Onaka, Brian Horiuchi, and Caryn Tottori of the Hawaii State Department of Health for providing death certificate data on cause of death for the KHHP participants, Ms. Ayako Elliott and Ms. Eva Ardo for assistance with genotyping, and Ms. Hiromi Nakada and Ms. Ka-on Fong for monitoring the vital status of KHHP participants.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
Research reported in this publication was supported by the Kuakini Medical Center, the US National Institutes of Health (contract N01-AG-4-2149, Grants 5 U01 AG019349-05, 5R01AG027060 [Kuakini Hawaii Lifespan Study], 5R01AG038707 [Kuakini Hawaii Healthspan Study], 1P20GM125526-01A1 [Kuakini Center of Biomedical Research Excellence for Clinical and Translational Research on Aging]), and contract N01-HC-05102 from the National Heart, Lung, and Blood Institute.
References
- 1. Ichijo H, Nishida E, Irie K, ten Dijke P, Saitoh M, Moriguchi T, Takagi M, Matsumoto K, Miyazono K, Gotoh Y. Induction of apoptosis by ASK1, a mammalian MAPKKK that activates SAPK/JNK and p38 signaling pathways. Science. 1997; 275:90–94. https://doi.org/10.1126/science.275.5296.90 [PubMed]
- 2. Bian L, Hanson RL, Ossowski V, Wiedrich K, Mason CC, Traurig M, Muller YL, Kobes S, Knowler WC, Baier LJ, Bogardus C. Variants in ASK1 are associated with skeletal muscle ASK1 expression, in vivo insulin resistance, and type 2 diabetes in pima Indians. Diabetes. 2010; 59:1276–82. https://doi.org/10.2337/db09-1700 [PubMed]
- 3. Hayakawa R, Hayakawa T, Takeda K, Ichijo H. Therapeutic targets in the ASK1-dependent stress signaling pathways. Proc Jpn Acad Ser B Phys Biol Sci. 2012; 88:434–53. https://doi.org/10.2183/pjab.88.434 [PubMed]
- 4. Papaconstantinou J. The role of signaling pathways of inflammation and oxidative stress in development of senescence and aging phenotypes in cardiovascular disease. Cells. 2019; 8:1383. https://doi.org/10.3390/cells8111383 [PubMed]
- 5. Pepin E, Higa A, Schuster-Klein C, Bernard C, Sulpice T, Guardiola B, Chevet E, Alquier T. Deletion of apoptosis signal-regulating kinase 1 (ASK1) protects pancreatic beta-cells from stress-induced death but not from glucose homeostasis alterations under pro-inflammatory conditions. PLoS One. 2014; 9:e112714. https://doi.org/10.1371/journal.pone.0112714 [PubMed]
- 6. Fukumoto J, Cox R
Jr , Fukumoto I, Cho Y, Parthasarathy PT, Galam L, Lockey RF, Kolliputi N. Deletion of ASK1 protects against hyperoxia-induced acute lung injury. PLoS One. 2016; 11:e0147652. https://doi.org/10.1371/journal.pone.0147652 [PubMed] - 7. Donlon TA, Morris BJ, Chen R, Masaki KH, Allsopp RC, Willcox DC, Tiirikainen M, Willcox BJ. Analysis of polymorphisms in 59 potential candidate genes for association with human longevity. J Gerontol A Biol Sci Med Sci. 2018; 73:1459–64. https://doi.org/10.1093/gerona/glx247 [PubMed]
- 8. Estep PW 3rd, Warner JB, Bulyk ML. Short-term calorie restriction in male mice feminizes gene expression and alters key regulators of conserved aging regulatory pathways. PLoS One. 2009; 4:e5242. https://doi.org/10.1371/journal.pone.0005242 [PubMed]
- 9. Dhana K, Braun KV, Nano J, Voortman T, Demerath EW, Guan W, Fornage M, van Meurs JB, Uitterlinden AG, Hofman A, Franco OH, Dehghan A. An epigenome-wide association study of obesity-related traits. Am J Epidemiol. 2018; 187:1662–69. https://doi.org/10.1093/aje/kwy025 [PubMed]
- 10. Morris BJ, Willcox BJ, Donlon TA. Genetic and epigenetic regulation of human aging and longevity. Biochim Biophys Acta Mol Basis Dis. 2019; 1865:1718–44. https://doi.org/10.1016/j.bbadis.2018.08.039 [PubMed]
- 11. Láruson ÁJ, Reed FA. Stability of underdominant genetic polymorphisms in population networks. J Theor Biol. 2016; 390:156–63. https://doi.org/10.1016/j.jtbi.2015.11.023 [PubMed]
- 12. Pilling LC, Atkins JL, Bowman K, Jones SE, Tyrrell J, Beaumont RN, Ruth KS, Tuke MA, Yaghootkar H, Wood AR, Freathy RM, Murray A, Weedon MN, et al. Human longevity is influenced by many genetic variants: evidence from 75,000 UK biobank participants. Aging (Albany NY). 2016; 8:547–60. https://doi.org/10.18632/aging.100930 [PubMed]
- 13. Arai Y, Sasaki T, Hirose N. Demographic, phenotypic, and genetic characteristics of centenarians in Okinawa and Honshu, Japan: part 2 Honshu, Japan. Mech Ageing Dev. 2017; 165:80–85. https://doi.org/10.1016/j.mad.2017.02.005 [PubMed]
- 14. Willcox BJ, Donlon TA, He Q, Chen R, Grove JS, Yano K, Masaki KH, Willcox DC, Rodriguez B, Curb JD. FOXO3A genotype is strongly associated with human longevity. Proc Natl Acad Sci USA. 2008; 105:13987–92. https://doi.org/10.1073/pnas.0801030105 [PubMed]
- 15. Yamaguchi O, Higuchi Y, Hirotani S, Kashiwase K, Nakayama H, Hikoso S, Takeda T, Watanabe T, Asahi M, Taniike M, Matsumura Y, Tsujimoto I, Hongo K, et al. Targeted deletion of apoptosis signal-regulating kinase 1 attenuates left ventricular remodeling. Proc Natl Acad Sci USA. 2003; 100:15883–88. https://doi.org/10.1073/pnas.2136717100 [PubMed]
- 16. Ogier JM, Nayagam BA, Lockhart PJ. ASK1 inhibition: A therapeutic strategy with multi-system benefits. J Mol Med (Berl). 2020; 98:335–48. https://doi.org/10.1007/s00109-020-01878-y [PubMed]
- 17. Chen R, Morris BJ, Donlon TA, Masaki KH, Willcox DC, Davy PM, Allsopp RC, Willcox BJ. FOXO3 longevity genotype mitigates the increased mortality risk in men with a cardiometabolic disease. Aging (Albany NY). 2020; 12:23509–24. https://doi.org/10.18632/aging.202175 [PubMed]
- 18. Donlon TA, Chen R, Masaki KH, Willcox BJ, Morris BJ. Association with longevity of phosphatidylinositol 3-kinase regulatory subunit 1 gene (PIK3R1) variants stems from protection against mortality risk in men with cardiovascular disease. Gerontology. 2021. [Epub ahead of print].
- 19. Donlon TA, Morris BJ, He Q, Chen R, Masaki KH, Allsopp RC, Willcox DC, Tranah GJ, Parimi N, Evans DS, Flachsbart F, Nebel A, Kim DH, et al. Association of polymorphisms in connective tissue growth factor and epidermal growth factor receptor genes with human longevity. J Gerontol A Biol Sci Med Sci. 2017; 72:1038–44. https://doi.org/10.1093/gerona/glw116 [PubMed]
- 20. Boyle AP, Hong EL, Hariharan M, Cheng Y, Schaub MA, Kasowski M, Karczewski KJ, Park J, Hitz BC, Weng S, Cherry JM, Snyder M. Annotation of functional variation in personal genomes using RegulomeDB. Genome Res. 2012; 22:1790–97. https://doi.org/10.1101/gr.137323.112 [PubMed]
- 21. Ward LD, Kellis M. HaploReg: a resource for exploring chromatin states, conservation, and regulatory motif alterations within sets of genetically linked variants. Nucleic Acids Res. 2012; 40:D930–34. https://doi.org/10.1093/nar/gkr917 [PubMed]
- 22. Statistical Analysis System (SAS) version 9.4. SAS Institute, Cary, NC, USA. https://libguides.library.kent.edu/statconsulting/SAS. 2020.
- 23. StataCorp L. Stata Statistical Software: Release 12. College Station, TX: StataCorp LP 2011.



