Introduction
Mechanosensory hair cells (HCs) in the inner ear play a crucial role in sensing auditory and vestibular signals in the environment, and HC damage is a leading cause of hearing loss. Congenital hearing loss occurs in approximately 1/500 infants, and genetic factors account for at least 50% of cases [1]. Thus heritable hearing loss caused by developmental defect of HCs due to mutations in deafness genes is a significant contributor to reduced quality of life. The functions of deafness genes are mainly involved in cochlear homeostasis, cellular organization, coding for tectorial membrane-associated proteins, neuronal transmission, and cell growth, differentiation, and survival. To date a total of 121 non-syndromic hearing loss genes have been identified (https://hereditaryhearingloss.org, most recent update: 4/20/2020); however, there are likely to be many deafness genes waiting to be discovered. Therefore, identifying deafness genes is crucial for protecting HCs from injury and thus for preventing and treating hereditary hearing loss.
Ion homeostasis is essential for maintaining cell survival and proper cell function, and many hearing disorders are caused by inner ear HC ion homeostatic disruption. Therefore, membrane-associated ion channels, transporters, and exchangers are required for HC function, and loss of function of these proteins induces HC loss and thus leads to hearing deficits.
The solute carrier (SLC) group of membrane transport proteins consists of more than 60 families and over 400 members. Mutations of some SLC genes have been shown to lead to hearing impairments or HC defects, for example, human SLC44A4 [2] and zebrafish slc26a2 [3]. The HCO3−/Cl− anion exchangers (AEs) belong to the SLC4 family and include three primary members, namely AE1 (or SLC4A1), AE2 (or SLC4A2), and AE3 (or SLC4A3), each of which can exchange HCO3− for Cl− across the plasma membrane and thus play an important role in maintaining the intracellular pH (pHi), Cl− concentrations, and cell volume [4]. Among the three genes, the SLC4A2 gene appears to be more widely distributed than the other two, and it has been shown to be expressed in the inner and outer HCs and in the supporting cells of rat and guinea pig cochleae [5] and in the HCs, the supporting cells between the inner and outer sulcus cells, type I, II, III, and V fibrocytes of the spiral ligament, Reissner’s membrane cells, spiral limbus cells, and spiral ganglion neurons in the common marmoset cochlea [6]. SLC4A2 is required for proper osteoclast differentiation and function in mice [7], and mutations in bovine SLC4A2 are associated with osteopetrosis in Red Angus cattle [8]. SLC4A2-mediated HCO3−/Cl− exchange activity is essential for calpain-dependent regulation of the actin cytoskeleton in osteoclasts [9], and SLC4A2 is also involved in pHi homeostasis [10], spermatogenesis [11], gastric acid secretion [12], and immune responses [13, 10]. However, the role of SLC4A2 in hearing has not been explored.
In zebrafish, there are two unlinked genes that are equally closely related to the SLC4A2 genes in mammals, namely slc4a2a (also called ae2, ae2.1, and slc4a2) and slc4a2b (also called ae2.2) [14, 15]. slc4a2a mRNA can be detected in the prospective midbrain as early as the five-somite stage, then later in the pronephric primordia and in the developing pronephric duct, where it persists through 72 hpf (hours post fertilization), and it mediates both Na-independent electroneutral Cl−/Cl− exchange and Cl−/HCO3− exchange [14]. We demonstrate here that the slc4a2b gene is expressed in the otic vesicle and lateral line neuromasts in zebrafish. An in silico analysis showed that the slc4a2b gene is highly evolutionarily conserved, and we further show that loss of function of slc4a2b resulted in decreased numbers of posterior lateral line neuromasts, HC clusters, and HCs due to increased HC apoptosis. Taken together, our findings highlight a critical role for the slc4a2b gene in HC development, and thus slc4a2b might be a newly identified deafness-related gene.
Results
The slc4a2b gene is evolutionarily conserved and is expressed in the otic vesicle and the lateral line neuromasts in zebrafish
For a better understanding of the slc4a2b gene in zebrafish, we first analyzed the evolutionary features of the gene. As shown in Figure 1A, the zebrafish slc4a2b gene has significant amino acid sequence similarities to other species. Moreover, the phyloP8way conservation score of slc4a2b is greater than more than half of the zebrafish protein-coding genes (Figure 1B), and is ranked 7,365 among over 23,000 genes in zebrafish, which indicates its evolutionary conservation.
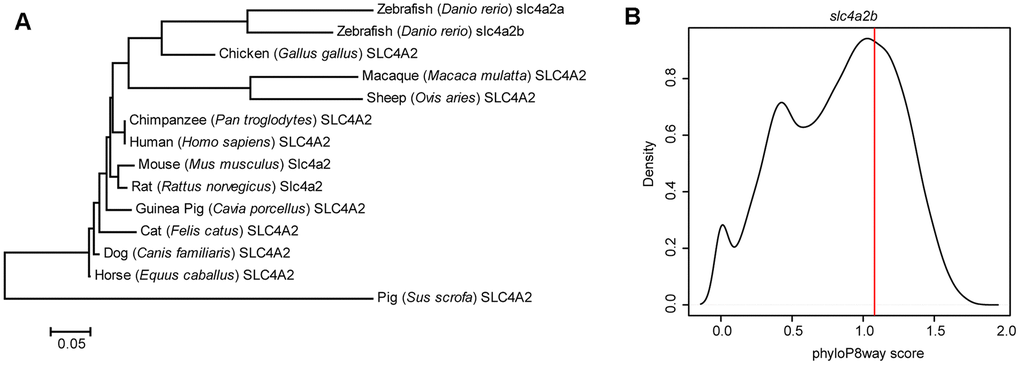
Figure 1. Evolutionary feature analysis of the slc4a2b gene. (A) Evolutionary relationships of SLC4A2 in different species. The evolutionary history was inferred using the Neighbor-Joining method, and the optimal tree with the sum of branch length = 1.66619414 is shown. The tree is drawn to scale, with branch lengths in the same units as those of the evolutionary distances used to infer the phylogenetic tree. The evolutionary distances were computed using the Poisson correction method and are in the units of the number of amino acid substitutions per site. The analysis involved 14 amino acid sequences. All positions containing gaps and missing data were eliminated. There were a total of 237 positions in the final dataset. The evolutionary analysis was conducted in MEGA6. (B) slc4a2b phyloP8way conservation score (vertical red line) and the zebrafish protein coding gene phyloP8way conservation score density distribution (black line). The x-axis is the average phyloP8way score for the coding sequence region of the gene, and the y-axis is the density of the number of genes in each conservation score range.
WISH was used to detect the expression of the slc4a2b gene in developing zebrafish embryos. As shown in Figure 2, slc4a2b mRNA was observed in the otic vesicle and caudal vein at 24 and 36 hpf (Figure 2A, 2B), while at 48 hpf the slc4a2b gene was not only expressed in otic vesicle, but also in posterior lateral line neuromasts (Figure 2C). With continued growth, the slc4a2b mRNA was found in the neuromasts on the head (Figure 2D). Together these results show that the slc4a2b gene is highly conserved and is expressed in lateral line neuromasts in zebrafish, which suggest its important role in neuromast development.
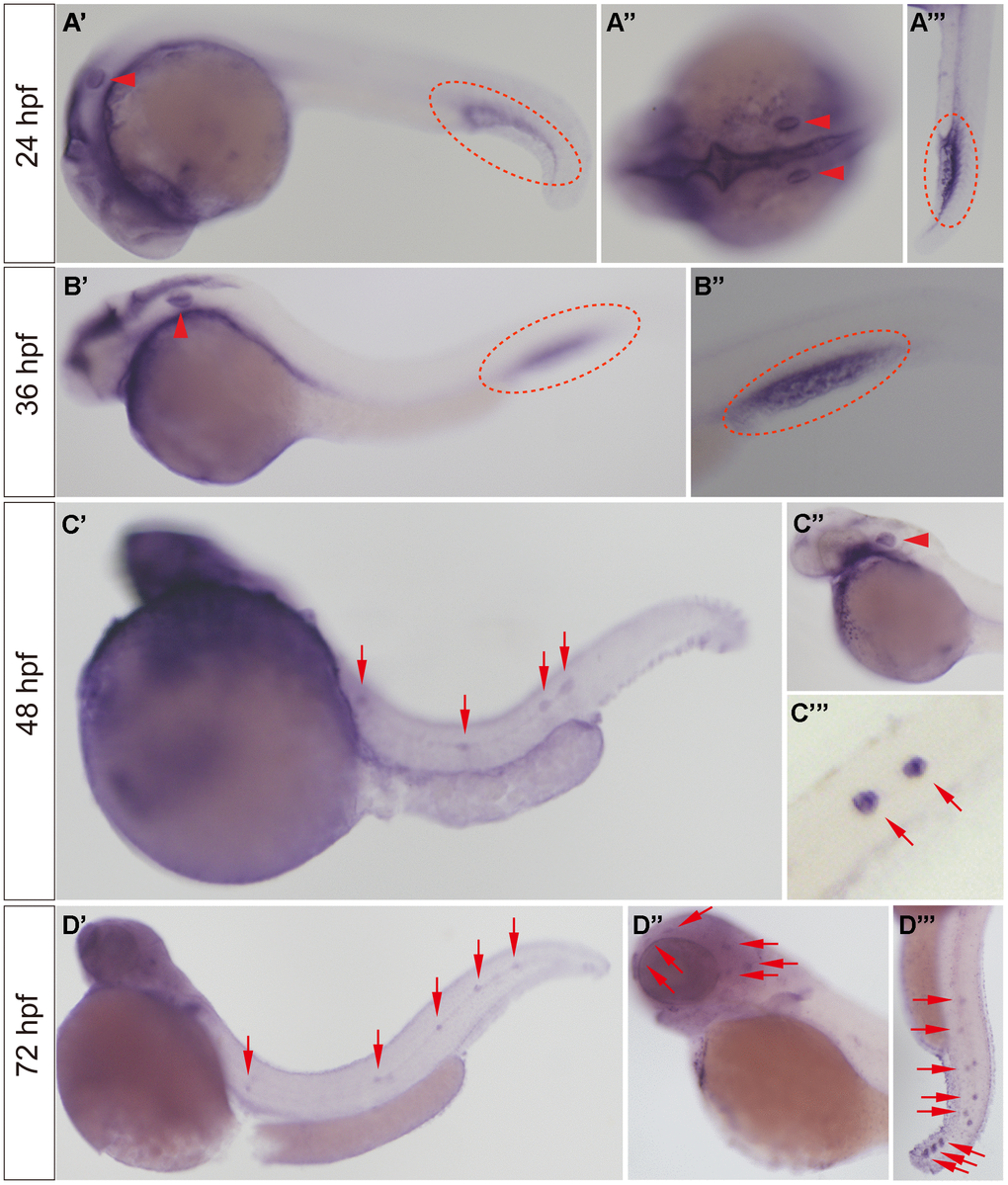
Figure 2. slc4a2b mRNA expression pattern detected by WISH. (A, B) The slc4a2b mRNA was mainly expressed in the otic vesicle (indicated by red arrowheads) and caudal vein (indicated by red dotted lines) at 24 hpf and 36 hpf. (A’’) shows the dorsal view of the otic vesicle, and (A’’’) and (B’’) are focused on the caudal vein. (C) At 48 hpf, slc4a2b mRNA was detected not only in the otic vesicle (indicated by the red arrowhead), but also in the lateral line neuromasts (indicated by red arrows). (C’’) and (C’’’) show the otic vesicle and the neuromasts at higher magnification, respectively. (D) slc4a2b mRNA was expressed in neuromasts at 72 hpf. (D’’) shows the neuromasts on the head (red arrowheads indicate the neuromast MI1, MI2, O2, et al.) and (D’’’) shows the posterior lateral line neuromasts.
Knockdown of the slc4a2b gene leads to decreased neuromasts and HCs
To determine the function of the slc4a2b gene, morpholino-mediated gene knockdown was used to downregulate its expression in the transgenic zebrafish line Tg(Brn3c:mGFP) [16] in which HCs are labeled by GFP. The slc4a2b-specific morpholino-injected larvae (Figure 3B’’, 3D’’, 3F’’, 3H’’) were morphologically similar at different stages to larvae injected with control morpholino (Figure 3A’’, 3C’’, 3E’’, 3G’’). However, fluorescence microscopic analysis showed that the HC clusters in the posterior lateral line of slc4a2b morphants (Figure 3B’, 3D’, 3F’, 3H’, 3I) decreased significantly at different stages in comparison to control larvae (Figure 3A’, 3C’, 3E’, 3G’, 3I).
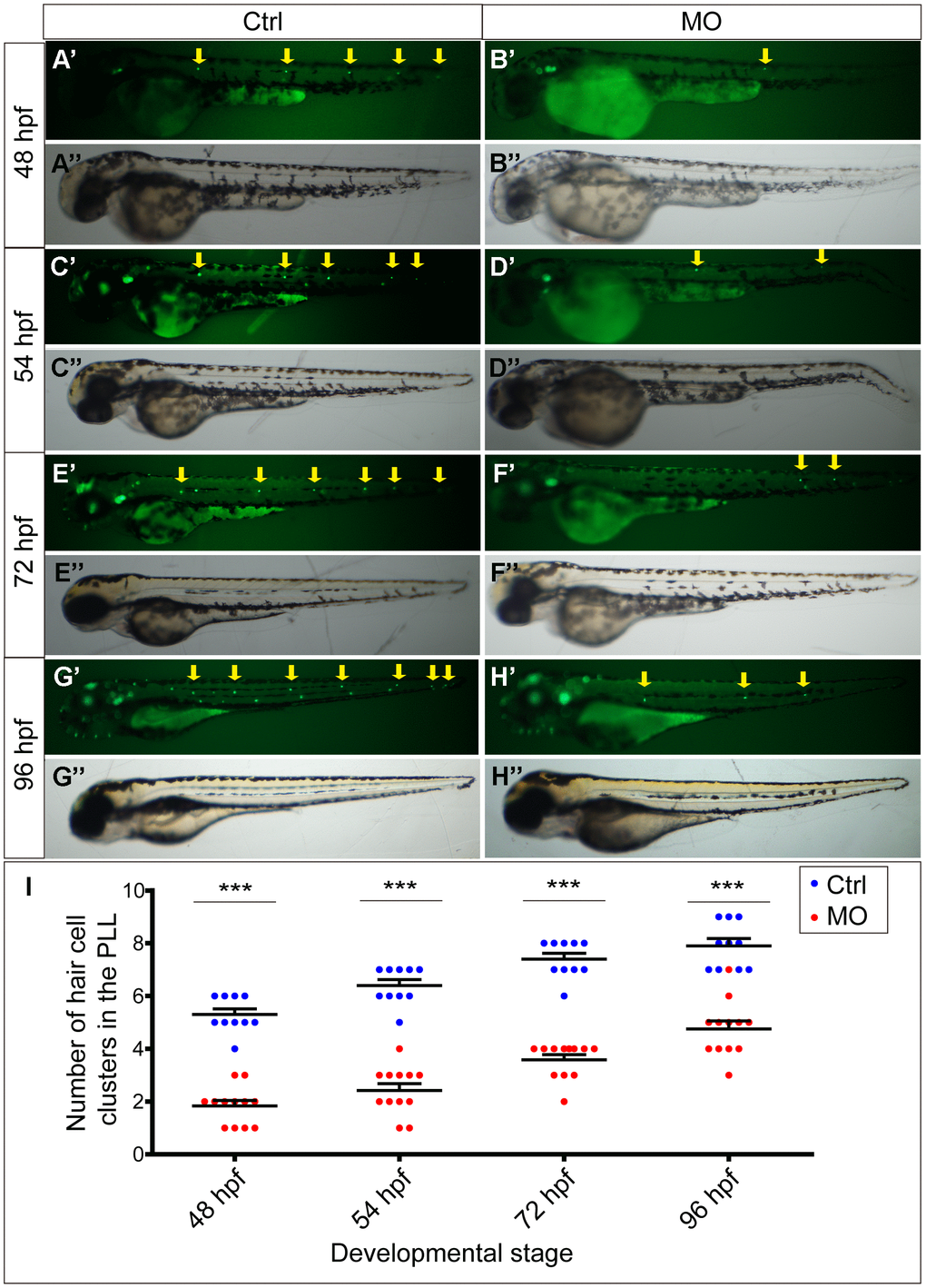
Figure 3. slc4a2b knockdown leads to decreased HC clusters in the posterior lateral line of zebrafish. (A–D) Zebrafish injected with slc4a2b-morpholino (MO) had normal morphology (B’’, D’’, F’’, H’’) but decreased HC clusters in the posterior lateral line (B’, D’, F’, H’) compared to the controls (Ctrl) (A’’, C’’, E’’, G’’ and A’, C’, E’, G’) at different developmental stages. The HC clusters in the posterior lateral line are indicated by yellow arrows. (I) Quantification of the number of HC clusters in the posterior lateral line in slc4a2b-morphants and controls at different developmental stages. ***P < 0.001.
The eya1 gene is a marker of lateral line neuromasts in zebrafish [17, 18], and the eya1-specific WISH experiment provided powerful evidence that slc4a2b gene knockdown led to reduced numbers of neuromasts in zebrafish (Figure 4).
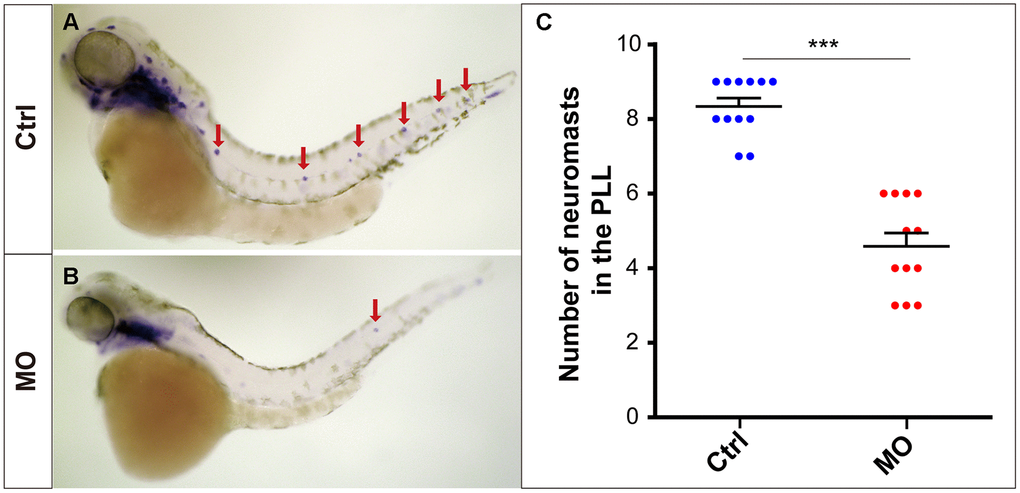
Figure 4. slc4a2b knockdown leads to decreased lateral line neuromasts in zebrafish. Zebrafish larvae at 72 hpf and the eya1 RNA probe were used in the WISH analysis. (A, B) slc4a2b-morphants (MO) (B) had fewer neuromasts (indicated by red arrows) compared to the controls (A). (C) Quantification of the number of the posterior lateral line neuromasts in slc4a2b-morphants and controls. ***P < 0.001.
Although slc4a2b gene knockdown led to decreased neuromasts and HC clusters, whether HCs in the remaining neuromasts were affected was not clear. To answer this, immunofluorescence experiments and confocal microscopic analysis were used to visualize the HCs. The HC number in the remaining neuromasts of the slc4a2b morphants was also reduced dramatically compared to controls (Figure 5A–5C), which further decreased the total HC number (Figure 5D). Taken together, these results suggest that slc4a2b gene knockdown has a negative effect on HC development in zebrafish.
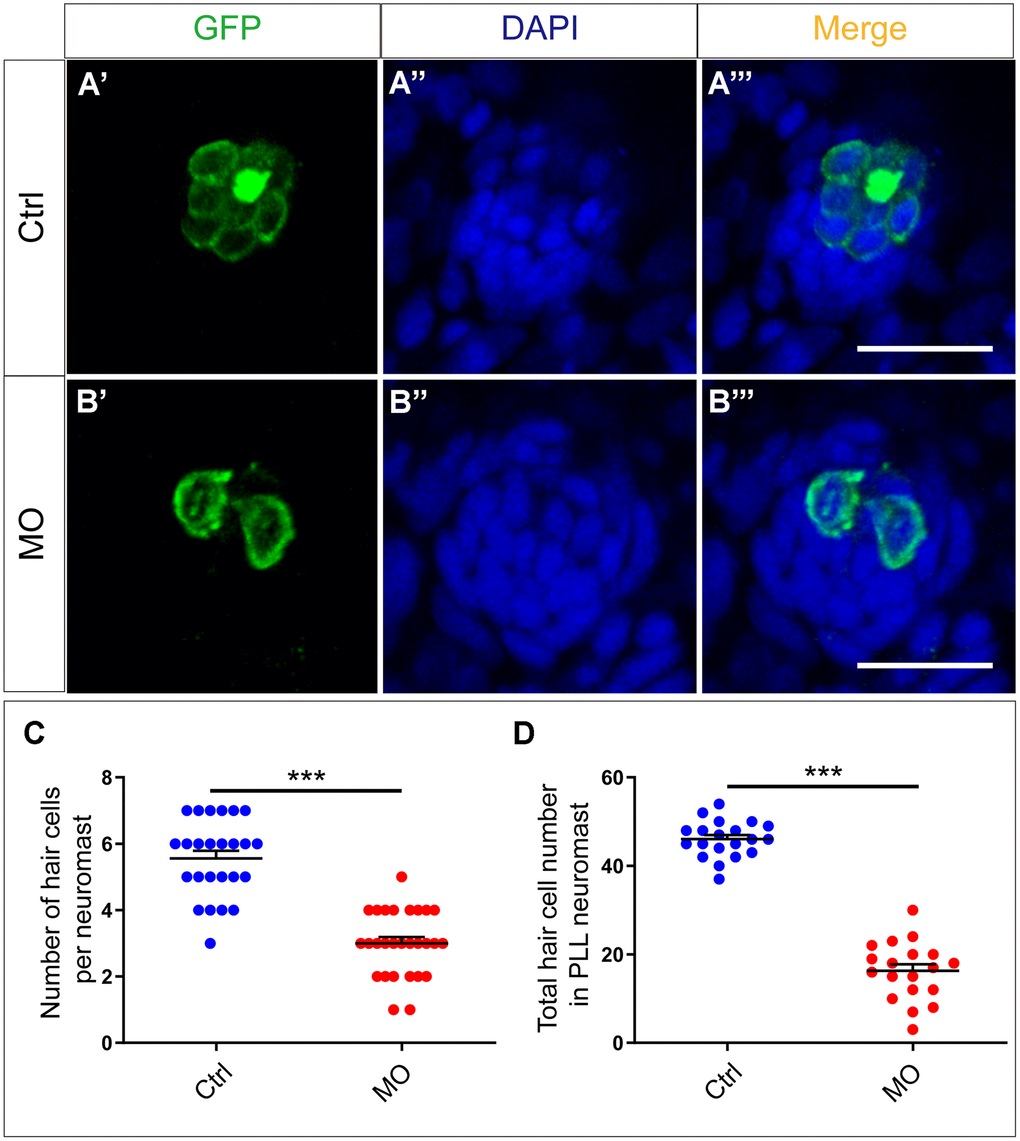
Figure 5. slc4a2b knockdown leads to decreased HCs in the posterior lateral line neuromast of zebrafish. (A, B) slc4a2b-morphants (MO) (B) had fewer HCs in each neuromast compared to controls (A). Tg(Brn3c:mGFP) transgenic zebrafish were used in the analysis, and the cells labeled by GFP represent HCs, while DAPI was used to stain the cell nuclei. (C, D) Quantification of the number of HCs in each neuromast and the total HCs in posterior lateral line neuromasts of the slc4a2b-morphants and controls. Scale bar = 20 μm, *** P < 0.001).
CRISPR/Cas9-mediated slc4a2b gene mutation disrupts HC development
To further identify the role of the slc4a2b gene in HC development, we generated slc4a2b mutant zebrafish (namely, Tg(Brn3c:mGFP);slc4a2bmut) using CRISPR/Cas9 gene editing technology with Tg(Brn3c:mGFP) transgenic zebrafish. As shown in Figure 6A, an sgRNA targeting the coding sequence in exon 2 of the slc4a2b gene was synthesized in vitro. After co-injection of slc4a2b-specific sgRNA and Cas9 mRNA, the zebrafish were grown and the genomic DNA was extracted for genotyping. The mutations were observed at the target site within the genome (Figure 6B).
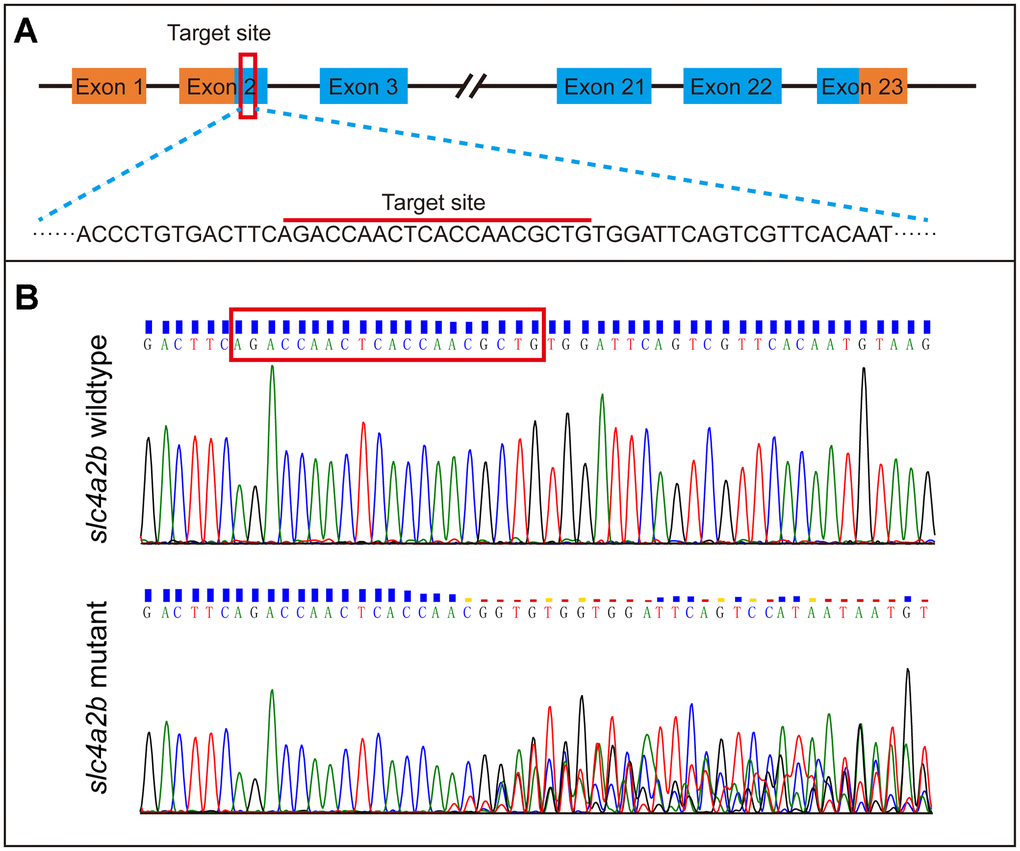
Figure 6. Generation of slc4a2b gene mutant zebrafish using CRISPR/Cas9 gene-editing technology. (A) The coding sequence in exon 2 of the slc4a2b gene was chosen to be the target of mutation. (B) Various mutations occurred in the target site of the slc4a2b gene in mutant zebrafish compared to the wild-type fish.
The phenotype caused by slc4a2b gene defects was analyzed at 3 dpf. First, by immunostaining we found that the number of HC clusters in the posterior lateral line of Tg(Brn3c:mGFP);slc4a2bmut zebrafish was decreased significantly compared to controls, which was similar to what was seen in the slc4a2b morphants (Figure 7A–7C). Here, an anti-GFP antibody was used to label the HCs. We further analyzed HC development in the neuromasts of the slc4a2b mutant zebrafish using both an anti-GFP antibody and FM1-43FX staining. Similarly, we found few GFP-positive or FM1-43FX-positive HCs in the neuromasts (Figure 7D–7H). Together these results suggest that loss of function of the slc4a2b gene disrupts HC development and function.
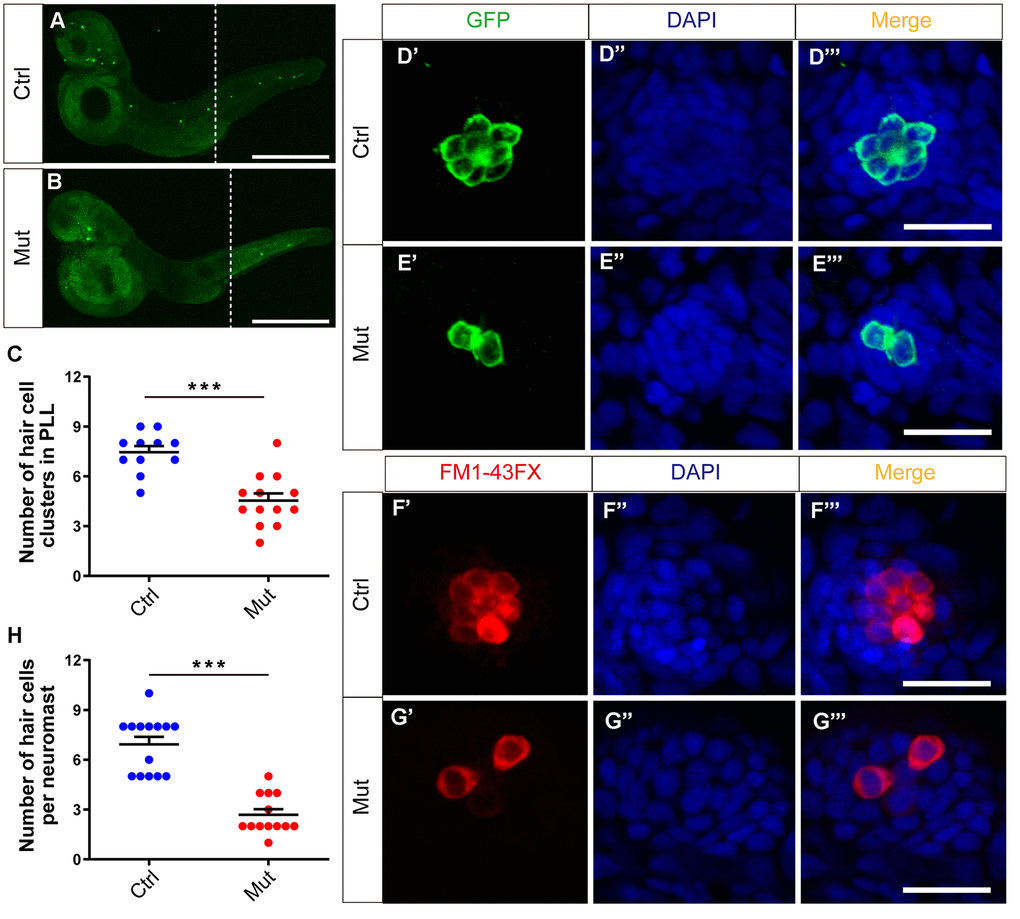
Figure 7. slc4a2b gene mutant zebrafish have fewer HC clusters and HCs in the posterior lateral line (PLL). (A, B) The Tg(Brn3c:mGFP);slc4a2bmut zebrafish had fewer HC clusters in the posterior lateral line compared to the controls. Here, the anti-GFP antibody was used to label the HCs. Scale bar = 500 μm. (C) Quantification of the number of HC clusters in the posterior lateral line of the slc4a2b gene mutant zebrafish and controls. ***P < 0.001. (D, E) The Tg(Brn3c:mGFP);slc4a2bmut zebrafish had fewer HCs in each posterior lateral line neuromast compared to controls. Here, anti-GFP antibody and DAPI were used to label the HCs and the nuclei, respectively. Scale bar = 20 μm. (F, G) The Tg(Brn3c:mGFP);slc4a2bmut zebrafish had fewer functional HCs in each posterior lateral line neuromast compared to controls. The successful staining with FM1-43FX dye was as a marker of functional HCs. Scale bar = 20 μm. (H) Quantification of the number of HCs in each neuromast of the slc4a2b mutant zebrafish and controls. *** P < 0.001.
slc4a2b mRNA injection can rescue the phenotype induced by slc4a2b gene deficiency
To further verify that the phenotype of slc4a2b mutant zebrafish was due to the loss of function of slc4a2b rather than to nonspecific effects, we performed a rescue experiment by injecting slc4a2b mRNA together with the slc4a2b sgRNA and Cas9 mRNA into 1-cell-stage Tg(Brn3c:mGFP) zebrafish embryos. Zebrafish injected with slc4a2b mRNA had significantly higher slc4a2b expression level (Supplementary Figure 2), and had more HC clusters and HCs in the posterior lateral line in comparison with those without slc4a2b mRNA injection (Figure 8). This significant difference between the two groups provided strong evidence that slc4a2b gene mutation affected the development of posterior lateral line HCs, and because slc4a2b mRNA injection could rescue this deficiency, this suggests that slc4a2b is required for HC development in zebrafish.
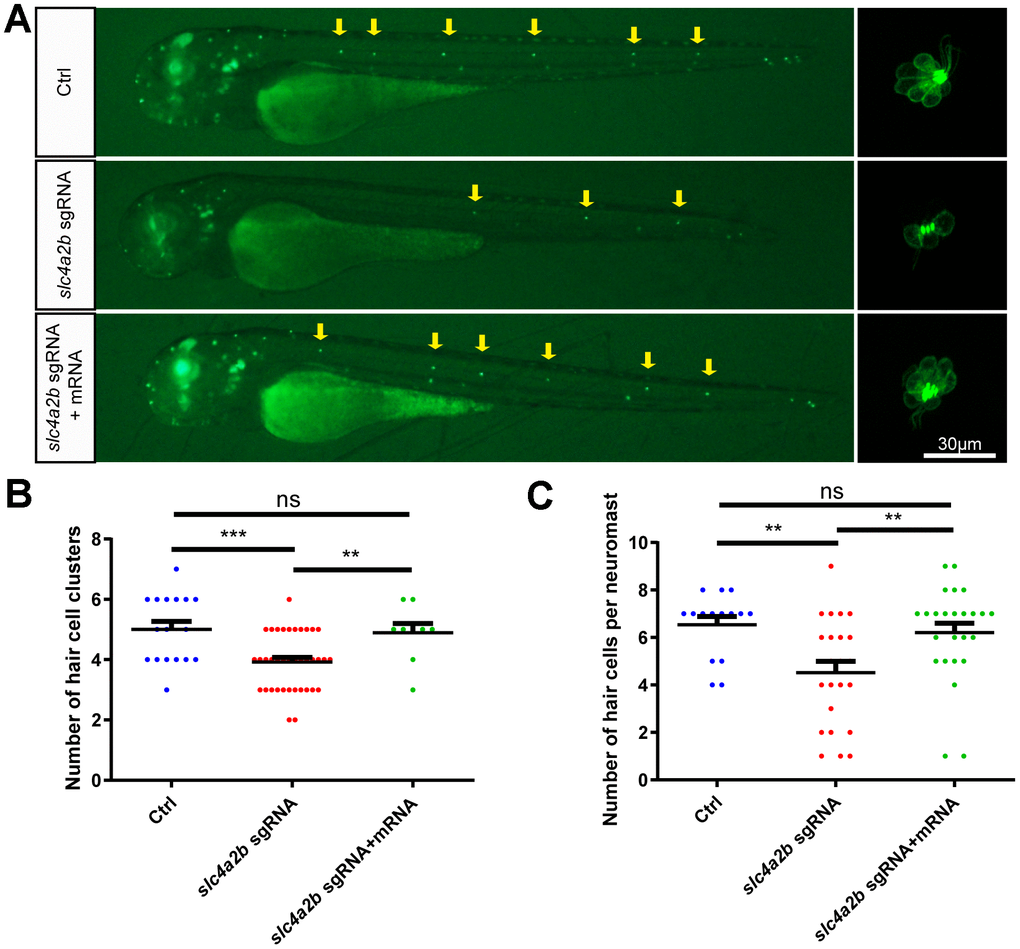
Figure 8. slc4a2b mRNA injection can rescue the phenotype induced by slc4a2b gene deficiency. The Tg(Brn3c:mGFP);slc4a2bmut zebrafish that were injected with Cas9 mRNA and slc4a2b sgRNA had fewer HC clusters (A, B) and fewer HCs in each posterior lateral line neuromast (A, C) compared to controls. However, slc4a2b mRNA injection could rescue the phenotype caused by slc4a2b mutation. *** P < 0.001; ** P < 0.01; ns, no significance.
Loss of function of the slc4a2b gene leads to HC apoptosis
To determine if apoptosis is the primary mechanism through which slc4a2b gene defects lead to decreased numbers of HCs, we used an antibody against cleaved caspase-3, which is a marker of apoptosis. As shown in Figure 9, we observed GFP and cleaved caspase-3 double-positive cells in the posterior lateral line neuromasts of Tg(Brn3c:mGFP);slc4a2bmut zebrafish indicating that loss of function of slc4a2b leads to HC apoptosis.
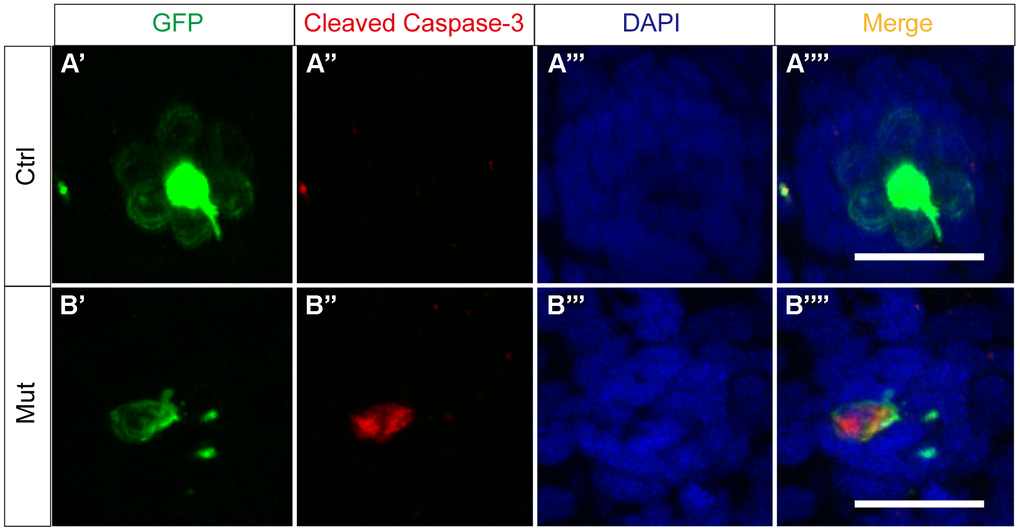
Figure 9. slc4a2b gene mutation leads to HC apoptosis. The control zebrafish, Tg(Brn3c:mGFP) (A), and the mutant zebrafish, Tg(Brn3c:mGFP);slc4a2bmut (B), were used for the apoptosis assay. Here, the anti-cleaved caspase-3 antibody was used to detect the apoptotic cells. Scale bar = 20 μm.
Discussion
Zebrafish is an excellent animal model owing to its optical transparency, high rate of reproduction, ectogenesis, and known genome sequence that has high homology with the human genome. Zebrafish also have many organs and systems that are similar to those of mammals and humans. For example, the zebrafish lateral line system, which consists of a number of neuromasts that enable the fish to perceive changes in its surroundings, consists of HCs, supporting cells and mantle cells. These HCs have similar morphologies and functions in the transmission of sensory information as mammalian HCs. The zebrafish lateral line neuromast is thus a very useful model to study the factors that affect HC development and function because this knowledge is likely to be applicable to our understanding of how dysfunction of inner ear HCs in mammals leads to hearing loss.
Recent decades have seen the identification of numerous genes that are responsible for hearing loss, and it is likely that there are still many deafness-related genes waiting to be discovered. Identifying such genes is a key step in a better understanding of the mechanisms of hearing loss and will form a fundamental basis for precise gene therapy in the future.
Ion homeostasis is essential for cell survival and function, and because HCs detect mechanical signals and transform them into electrical signals, ion homeostasis also plays an indispensable role in hearing. In HCs, some membrane-located transport proteins, for example, ion channels, transporters, and exchangers, play key roles, and dysfunction of these proteins leads to disruption of ion homeostasis and ultimately to cell death and subsequent hearing impairment.
The SLC protein family is made up of important solute carriers, and these proteins play vital roles in maintaining the physiology and function of numerous cell types. SLC4A2 is a sodium-independent anion exchanger of HCO3− and Cl−, and it ensures that the pHi is maintained within a narrow range. The zebrafish slc4a2b gene is a member of the slc4 gene family, but its role in zebrafish development has not been reported until now.
First, we analyzed the evolutionary conservation and expression pattern of the zebrafish slc4a2b gene. The phyloP8way conservation score analysis and the WISH experiment showed that the slc4a2b gene is highly conserved and is mainly expressed in the caudal vein, otic vesicle, and neuromasts at different developmental stages. We next examined the role of slc4a2b in zebrafish lateral line neuromast HC development using morpholino-mediated gene knockdown and CRISPR/Cas9-mediated gene editing methods. Loss of function analysis showed that the zebrafish slc4a2b gene is required for HC development. Further, we showed that the phenotype induced by slc4a2b gene deficiency can be rescued by slc4a2b-specific mRNA injection, and this verified the role of the slc4a2b gene. Finally, we sought to explain how slc4a2b gene defects can lead to decreased HCs. Staining for the apoptosis marker cleaved caspase-3 showed that slc4a2b deficiency results in HC apoptosis in the posterior lateral line neuromast.
Apoptosis is one of the main types of cell death and is characterized by chromatin condensation and DNA degradation, and it can be divided into an extrinsic (or death receptor) and an intrinsic (or mitochondrial) pathway [19, 20]. Apoptosis is considered an important mechanism in the sensory HC death induced by aminoglycosides, cisplatin, and noise exposure. Several factors, including cysteinyl aspartate-specific proteinases (caspases) [21–23], B-cell lymphoma-2 family members [24–26], reactive oxygen species [27–29], c-jun NH2-terminal kinase pathway molecules [30–32], the p53 tumor suppressor gene [33–36], and some microRNAs [37–40], are known to play crucial roles in HC apoptosis. Caspase-3 activation is a key step and has been seen in HC apoptosis caused by aminoglycosides [41–44], cisplatin [34], and acoustic trauma [45–47]. Here, we demonstrate that slc4a2b gene deficiency can induce HC apoptosis mediated by caspase-3 activation in zebrafish.
Intracellular pH has a critical role in the maintenance of normal cell function and cell survival. pH regulation is important for hearing as mutation in H+-ATPases are known to cause hearing loss in human deafness patients with distal tubular acidosis [48]. And it has also previously been shown to be important in hair cells of zebrafish as raising fish mutating gcm2, a global pH regulator, or raising fish in altered pHs have both been shown to decrease hair cell number and reduce hair cell function [49, 50].
Taken together, these results indicate that the slc4a2b gene is required for HC development in zebrafish and that slc4a2b gene deficiency leads to increased HC apoptosis. Given that SLC4A2 is involved in pHi regulation and that Slc4a2 deficiency alters pHi homeostasis in mice [9, 51, 52] and that disrupted pH homeostasis is an initiator of cell apoptosis [53, 54], we hypothesize that the HC apoptosis induced by the slc4a2b gene defect is due to changes in intracellular physiological parameters such as pHi, but this needs to be confirmed in future studies.
Zebrafish share approximately 70% of their genes with humans, and more than 80% of known human disease-related genes have a zebrafish counterpart [55]. Based on the evidence presented here, we propose that the human SLC4A2 gene, the homologue of the zebrafish slc4a2b gene, is a potential deafness gene, and this should be confirmed in clinical studies.
Materials and Methods
Zebrafish strains and maintenance
Two zebrafish lines were used in this study, the wild-type AB line and the transgenic line Tg(Brn3c:mGFP). In the latter, membrane-localized green fluorescent protein (GFP) is expressed specifically in the HCs [16]. All of the zebrafish were raised at 28.5°C.
The phyloP8way conservation score analysis
The phyloP8way conservation score file for zebrafish (genome reference version: danRer7) was downloaded from the UCSC Browser (http://hgdownload.soe.ucsc.edu/goldenPath/danRer7/phyloP8way/vertebrate.phyloP8way.bw), where the 8 species used for the conservation score calculation are Homo sapiens (hg19), Mus musculus (mm9), Xenopus tropicalis (xenTro2), Tetraodon nigroviridis (tetNig2), Takifugu rubripes (fr2), Gasterosteus aculeatus (gasAcu1), Oryzias latipes (oryLat2), and Danio rerio (danRer7). The corresponding gene annotation gtf file was downloaded from Ensembl (ftp://ftp.ensembl.org/pub/release-74/gtf/danio_rerio/Danio_rerio.Zv9.74.gtf.gz). The gtf annotation file was converted into bed format using a homemade python script. The bigWigAverageOverBed software from the UCSC browser was used to calculate the average phyloP8way conservation score for each protein-coding gene (only considering the coding sequence regions). The phyloP8way conservation score density plot was drawn using an R script.
Whole-mount in situ hybridization
The whole-mount in situ hybridization (WISH) was performed according to standard procedures. First, a part of coding sequence of slc4a2b gene, amplified from zebrafish cDNA using the primers (5’-CAG CAT GGA TGA AGT GAC GG-3’ and 5’-CAG AAC CCT TGA CCA GCA TG-3’), was subcloned into the pGEM-T Easy vector, and then a gene-specific digoxigenin-labeled RNA probe was transcribed in vitro using the DIG RNA Labeling Kit (SP6&.T7) (Roche, #11175025910) following the manufacturer’s instructions. The pre-fixed embryos were incubated with the probe overnight at 4°C. Second, an alkaline phosphatase (AP)-conjugated antibody against digoxigenin (Roche, #11093274910) was used to detect the digoxigenin-labeled RNA probe. Finally, the AP-substrate NBT/BCIP solution (Roche, #11681451001) was added to the reaction system, and the development of color allowed the expression of the gene of interest to be visualized.
Morpholino-mediated gene knockdown of slc4a2b in zebrafish
The oligo sequence of the slc4a2b gene-specific morpholino was 5’-TCA GGA CAC TGT GAA CCC GCT GAA C-3’, and it was obtained from Gene Tools, LLC. In this study, 2–3 nL of 0.3 mM morpholino oligo was microinjected into the embryos at the 1-cell stage. The morpholino blocks the translation of the Slc4a2b protein when the oligo binds to the slc4a2b mRNA.
CRISPR/Cas9-mediated gene editing of slc4a2b in zebrafish
To generate the slc4a2b gene mutant zebrafish, as described in our previous work [56], 2–3 nL of a solution containing specific single guide RNA (sgRNA), which targeted exon 2 of the slc4a2b gene, and Cas9 mRNA was microinjected into each zebrafish embryo at the 1–2 cell stage. For the sgRNA synthesis, a forward primer (5’- TAA TAC GAC TCA CTA TAA GAC CAA CTC ACC AAC GCT GGT TTT AGA GCT AGA AAT AGC-3’), which contained a T7 promoter region and a slc4a2b gene-targeting region, and a universal reverse primer (5’-AAA AAA AGC ACC GAC TCG GTG CCA C-3’) were used in the PCR reaction with pT7 plasmid (Supplementary Figure 1) as the template to obtain the sgDNA that was then transcribed into sgRNA in vitro using the MAXIscript® Kit (Ambion, #AM1308). For the Cas9 mRNA synthesis, a linearized pXT7-Cas9 plasmid was used as the template for transcription into Cas9 mRNA in vitro using the mMESSAGE mMACHINE® Kit (Ambion, #AM1340).
mRNA rescue experiment
In the rescue experiment, 2–3 nL of 50 ng/μL slc4a2b mRNA was co-injected into the embryos with the slc4a2b sgRNA. The mRNA was transcribed in vitro using the mMESSAGE mMACHINE® Kit (Ambion, #AM1340). Briefly, approximately 3700 bp DNA fragments were synthesized by PCR using the primers slc4a2b-mRNA-EcoRI-F (5’-CGG AAT TCC GTG AGG TTA TGC TGC CCG TAA-3’) and slc4a2b-mRNA- XbaI-R (5’-GCT CTA GAG CTG ATA GCA GCT CAA ACG CTC-3’). The DNA fragments were subcloned into the pCS2+ vector, and the recombinant plasmid was linearized using the restriction endonuclease Not I and then transcribed into mRNA in vitro.
FM1-43FX staining
The vital dye FM1-43FX (Molecular Probe, #F35355) was used to specifically label functional HCs in the neuromasts. First, the live zebrafish were anesthetized using MS-222 and then immersed into the labeling solution for 45 s at room temperature in the dark. Afterwards, the fish were rinsed three times using the egg water and then fixed for 2 hours at room temperature in 4% paraformaldehyde. After washing three times with PBS-T (0.1% Triton X-100 in PBS), 4',6-diamidino-2-phenylindole (DAPI) was added to label the nuclei.
Immunofluorescence
For immunofluorescence, the fish were anesthetized and then fixed using 4% paraformaldehyde. After washing three times with PBS-T, the fish were incubated in the antigen retrieval solution (Beyotime Biotechnology, China, #P0088) for 15 min at 98°C. Nonspecific binding was then blocked with 10% donkey serum in PBS-T. Next, specific primary antibodies against GFP (Abcam, #ab13970) and cleaved caspase-3 (CST, #9664) were added, and secondary antibodies were used to detect the primary antibodies.
Statistical analysis
All data were analyzed using GraphPad Prism 8.0.2. Two-tailed, unpaired Student’s t-tests were used to determine statistical significance when comparing two groups. A value of P < 0.05 was considered statistically significant. All data are presented as means with SEM, and all experiments were repeated at least three times.
Ethical approval
All animal procedures were performed according to protocols approved by the Animal Care and Use Committee of Southeast University and Nantong University and were consistent with the National Institutes of Health Guide for the Care and Use of Laboratory Animals. All efforts were made to minimize the number of animals used and to prevent their suffering.
Supplementary Materials
Author Contributions
Fuping Qian, Huijun Yuan, Dong Liu and Renjie Chai conceived and designed the experiments. Fuping Qian, Xin Wang, and Zhenhua Yin performed the experiments. Fuping Qian, Gangcai Xie, Dong Liu and Renjie Chai analyzed the data. Fuping Qian, Dong Liu, and Renjie Chai wrote the paper. All authors read and approved the final manuscript.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
Funding
This work was supported by grants from Key Project of National Natural Science Foundation of China (81530031), Strategic Priority Research Program of the Chinese Academy of Sciences (XDA16010303), National Key R&D Program of China (2017YFA0103903), National Natural Science Foundation of China (81970882), Natural Science Foundation from Jiangsu Province (BE2019711, BK20180048, BRA2019278, 17KJA180008), Boehringer Ingelheim Pharma GmbH, K. C. Wong Education Foundation and Open Research Fund of State Key Laboratory of Genetic Engineering, Fudan University (No. SKLGE1809), Shenzhen Fundamental Research Program (JCYJ20190814093401920).
References
- 1. Morton CC, Nance WE. Newborn hearing screening—a silent revolution. N Engl J Med. 2006; 354:2151–64. https://doi.org/10.1056/NEJMra050700 [PubMed]
- 2. Ma Z, Xia W, Liu F, Ma J, Sun S, Zhang J, Jiang N, Wang X, Hu J, Ma D. SLC44A4 mutation causes autosomal dominant hereditary postlingual non-syndromic mid-frequency hearing loss. Hum Mol Genet. 2017; 26:3234. https://doi.org/10.1093/hmg/ddx232 [PubMed]
- 3. Liu F, Xia W, Hu J, Wang Y, Yang F, Sun S, Zhang J, Jiang N, Wang H, Tian W, Wang X, Ma D. Solute carrier family 26 member a2 (slc26a2) regulates otic development and hair cell survival in zebrafish. PLoS One. 2015; 10:e0136832. https://doi.org/10.1371/journal.pone.0136832 [PubMed]
- 4. Alper SL. The band 3-related anion exchanger (AE) gene family. Annu Rev Physiol. 1991; 53:549–64. https://doi.org/10.1146/annurev.ph.53.030191.003001 [PubMed]
- 5. Stanković KM, Brown D, Alper SL, Adams JC. Localization of pH regulating proteins H+ATPase and cl-/HCO3- exchanger in the Guinea pig inner ear. Hear Res. 1997; 114:21–34. https://doi.org/10.1016/s0378-5955(97)00072-5 [PubMed]
- 6. Hosoya M, Fujioka M, Kobayashi R, Okano H, Ogawa K. Overlapping expression of anion exchangers in the cochlea of a non-human primate suggests functional compensation. Neurosci Res. 2016; 110:1–10. https://doi.org/10.1016/j.neures.2016.04.002 [PubMed]
- 7. Wu J, Glimcher LH, Aliprantis AO. HCO3-/Cl- anion exchanger SLC4A2 is required for proper osteoclast differentiation and function. Proc Natl Acad Sci USA. 2008; 105:16934–39. https://doi.org/10.1073/pnas.0808763105 [PubMed]
- 8. Meyers SN, McDaneld TG, Swist SL, Marron BM, Steffen DJ, O’Toole D, O’Connell JR, Beever JE, Sonstegard TS, Smith TP. A deletion mutation in bovine SLC4A2 is associated with osteopetrosis in red angus cattle. BMC Genomics. 2010; 11:337. https://doi.org/10.1186/1471-2164-11-337 [PubMed]
- 9. Coury F, Zenger S, Stewart AK, Stephens S, Neff L, Tsang K, Shull GE, Alper SL, Baron R, Aliprantis AO. SLC4A2-mediated cl-/HCO3- exchange activity is essential for calpain-dependent regulation of the actin cytoskeleton in osteoclasts. Proc Natl Acad Sci USA. 2013; 110:2163–68. https://doi.org/10.1073/pnas.1206392110 [PubMed]
- 10. Concepcion AR, Salas JT, Sarvide S, Sáez E, Ferrer A, López M, Portu A, Banales JM, Hervás-Stubbs S, Oude Elferink RP, Prieto J, Medina JF. Anion exchanger 2 is critical for CD8+ T cells to maintain pHi homeostasis and modulate immune responses. Eur J Immunol. 2014; 44:1341–51. https://doi.org/10.1002/eji.201344218 [PubMed]
- 11. Medina JF, Recalde S, Prieto J, Lecanda J, Saez E, Funk CD, Vecino P, van Roon MA, Ottenhoff R, Bosma PJ, Bakker CT, Elferink RP. Anion exchanger 2 is essential for spermiogenesis in mice. Proc Natl Acad Sci USA. 2003; 100:15847–52. https://doi.org/10.1073/pnas.2536127100 [PubMed]
- 12. Recalde S, Muruzábal F, Looije N, Kunne C, Burrell MA, Sáez E, Martínez-Ansó E, Salas JT, Mardones P, Prieto J, Medina JF, Elferink RP. Inefficient chronic activation of parietal cells in Ae2a,b-/- mice. Am J Pathol. 2006; 169:165–76. https://doi.org/10.2353/ajpath.2006.051096 [PubMed]
- 13. Concepcion AR, Salas JT, Sáez E, Sarvide S, Ferrer A, Portu A, Uriarte I, Hervás-Stubbs S, Oude Elferink RP, Prieto J, Medina JF. CD8+ T cells undergo activation and programmed death-1 repression in the liver of aged Ae2a,b-/- mice favoring autoimmune cholangitis. Oncotarget. 2015; 6:28588–606. https://doi.org/10.18632/oncotarget.5665 [PubMed]
- 14. Shmukler BE, Kurschat CE, Ackermann GE, Jiang L, Zhou Y, Barut B, Stuart-Tilley AK, Zhao J, Zon LI, Drummond IA, Vandorpe DH, Paw BH, Alper SL. Zebrafish slc4a2/ae2 anion exchanger: cDNA cloning, mapping, functional characterization, and localization. Am J Physiol Renal Physiol. 2005; 289:F835–49. https://doi.org/10.1152/ajprenal.00122.2005 [PubMed]
- 15. Shmukler BE, Clark JS, Hsu A, Vandorpe DH, Stewart AK, Kurschat CE, Choe SK, Zhou Y, Amigo J, Paw BH, Alper SL. Zebrafish ae2.2 encodes a second slc4a2 anion exchanger. Am J Physiol Regul Integr Comp Physiol. 2008; 294:R1081–91. https://doi.org/10.1152/ajpregu.00690.2007 [PubMed]
- 16. Xiao T, Roeser T, Staub W, Baier H. A GFP-based genetic screen reveals mutations that disrupt the architecture of the zebrafish retinotectal projection. Development. 2005; 132:2955–67. https://doi.org/10.1242/dev.01861 [PubMed]
- 17. Kozlowski DJ, Whitfield TT, Hukriede NA, Lam WK, Weinberg ES. The zebrafish dog-eared mutation disrupts eya1, a gene required for cell survival and differentiation in the inner ear and lateral line. Dev Biol. 2005; 277:27–41. https://doi.org/10.1016/j.ydbio.2004.08.033 [PubMed]
- 18. Sahly I, Andermann P, Petit C. The zebrafish eya1 gene and its expression pattern during embryogenesis. Dev Genes Evol. 1999; 209:399–410. https://doi.org/10.1007/s004270050270 [PubMed]
- 19. Furness DN. Molecular basis of hair cell loss. Cell Tissue Res. 2015; 361:387–99. https://doi.org/10.1007/s00441-015-2113-z [PubMed]
- 20. Morrill S, He DZ. Apoptosis in inner ear sensory hair cells. J Otol. 2017; 12:151–64. https://doi.org/10.1016/j.joto.2017.08.001 [PubMed]
- 21. Liu L, Chen Y, Qi J, Zhang Y, He Y, Ni W, Li W, Zhang S, Sun S, Taketo MM, Wang L, Chai R, Li H. Wnt activation protects against neomycin-induced hair cell damage in the mouse cochlea. Cell Death Dis. 2016; 7:e2136. https://doi.org/10.1038/cddis.2016.35 [PubMed]
- 22. Han X, Ge R, Xie G, Li P, Zhao X, Gao L, Zhang H, Wang O, Huang F, Han F. Caspase-mediated apoptosis in the cochleae contributes to the early onset of hearing loss in A/J mice. ASN Neuro. 2015; 7:1759091415573985. https://doi.org/10.1177/1759091415573985 [PubMed]
- 23. Wang J, Ladrech S, Pujol R, Brabet P, Van De Water TR, Puel JL. Caspase inhibitors, but not c-jun NH2-terminal kinase inhibitor treatment, prevent cisplatin-induced hearing loss. Cancer Res. 2004; 64:9217–24. https://doi.org/10.1158/0008-5472.CAN-04-1581 [PubMed]
- 24. Alam SA, Ikeda K, Oshima T, Suzuki M, Kawase T, Kikuchi T, Takasaka T. Cisplatin-induced apoptotic cell death in Mongolian gerbil cochlea. Hear Res. 2000; 141:28–38. https://doi.org/10.1016/s0378-5955(99)00211-7 [PubMed]
- 25. Alam SA, Oshima T, Suzuki M, Kawase T, Takasaka T, Ikeda K. The expression of apoptosis-related proteins in the aged cochlea of Mongolian gerbils. Laryngoscope. 2001; 111:528–34. https://doi.org/10.1097/00005537-200103000-00026 [PubMed]
- 26. Someya S, Xu J, Kondo K, Ding D, Salvi RJ, Yamasoba T, Rabinovitch PS, Weindruch R, Leeuwenburgh C, Tanokura M, Prolla TA. Age-related hearing loss in C57BL/6J mice is mediated by bak-dependent mitochondrial apoptosis. Proc Natl Acad Sci USA. 2009; 106:19432–37. https://doi.org/10.1073/pnas.0908786106 [PubMed]
- 27. He Z, Sun S, Waqas M, Zhang X, Qian F, Cheng C, Zhang M, Zhang S, Wang Y, Tang M, Li H, Chai R. Reduced TRMU expression increases the sensitivity of hair-cell-like HEI-OC-1 cells to neomycin damage in vitro. Sci Rep. 2016; 6:29621. https://doi.org/10.1038/srep29621 [PubMed]
- 28. Hirose K, Hockenbery DM, Rubel EW. Reactive oxygen species in chick hair cells after gentamicin exposure in vitro. Hear Res. 1997; 104:1–14. https://doi.org/10.1016/s0378-5955(96)00169-4 [PubMed]
- 29. Ohlemiller KK, Wright JS, Dugan LL. Early elevation of cochlear reactive oxygen species following noise exposure. Audiol Neurootol. 1999; 4:229–36. https://doi.org/10.1159/000013846 [PubMed]
- 30. Cai C, Lin J, Sun S, He Y. JNK inhibition inhibits lateral line neuromast hair cell development. Front Cell Neurosci. 2016; 10:19. https://doi.org/10.3389/fncel.2016.00019 [PubMed]
- 31. Matsui JI, Gale JE, Warchol ME. Critical signaling events during the aminoglycoside-induced death of sensory hair cells in vitro. J Neurobiol. 2004; 61:250–66. https://doi.org/10.1002/neu.20054 [PubMed]
- 32. Pirvola U, Xing-Qun L, Virkkala J, Saarma M, Murakata C, Camoratto AM, Walton KM, Ylikoski J. Rescue of hearing, auditory hair cells, and neurons by CEP-1347/KT7515, an inhibitor of c-jun n-terminal kinase activation. J Neurosci. 2000; 20:43–50. https://doi.org/10.1523/JNEUROSCI.20-01-00043.2000 [PubMed]
- 33. Devarajan P, Savoca M, Castaneda MP, Park MS, Esteban-Cruciani N, Kalinec G, Kalinec F. Cisplatin-induced apoptosis in auditory cells: role of death receptor and mitochondrial pathways. Hear Res. 2002; 174:45–54. https://doi.org/10.1016/s0378-5955(02)00634-2 [PubMed]
- 34. Zhang M, Liu W, Ding D, Salvi R. Pifithrin-alpha suppresses p53 and protects cochlear and vestibular hair cells from cisplatin-induced apoptosis. Neuroscience. 2003; 120:191–205. https://doi.org/10.1016/s0306-4522(03)00286-0 [PubMed]
- 35. Wu X, Li X, Song Y, Li H, Bai X, Liu W, Han Y, Xu L, Li J, Zhang D, Wang H, Fan Z. Allicin protects auditory hair cells and spiral ganglion neurons from cisplatin - induced apoptosis. Neuropharmacology. 2017; 116:429–40. https://doi.org/10.1016/j.neuropharm.2017.01.001 [PubMed]
- 36. Yang Q, Zhou Y, Yin H, Li H, Zhou M, Sun G, Cao Z, Man R, Wang H, Li J. PINK1 protects against gentamicin-induced sensory hair cell damage: possible relation to induction of autophagy and inhibition of p53 signal pathway. Front Mol Neurosci. 2018; 11:403. https://doi.org/10.3389/fnmol.2018.00403 [PubMed]
- 37. Li Y, Li A, Wu J, He Y, Yu H, Chai R, Li H. MiR-182-5p protects inner ear hair cells from cisplatin-induced apoptosis by inhibiting FOXO3a. Cell Death Dis. 2016; 7:e2362. https://doi.org/10.1038/cddis.2016.246 [PubMed]
- 38. Lin Y, Shen J, Li D, Ming J, Liu X, Zhang N, Lai J, Shi M, Ji Q, Xing Y. MiR-34a contributes to diabetes-related cochlear hair cell apoptosis via SIRT1/HIF-1α signaling. Gen Comp Endocrinol. 2017; 246:63–70. https://doi.org/10.1016/j.ygcen.2017.02.017 [PubMed]
- 39. Tan PX, Du SS, Ren C, Yao QW, Zheng R, Li R, Yuan YW. MicroRNA-207 enhances radiation-induced apoptosis by directly targeting Akt3 in cochlea hair cells. Cell Death Dis. 2014; 5:e1433. https://doi.org/10.1038/cddis.2014.407 [PubMed]
- 40. Xue T, Wei L, Zha DJ, Qiu JH, Chen FQ, Qiao L, Qiu Y. miR-29b overexpression induces cochlear hair cell apoptosis through the regulation of SIRT1/PGC-1α signaling: implications for age-related hearing loss. Int J Mol Med. 2016; 38:1387–94. https://doi.org/10.3892/ijmm.2016.2735 [PubMed]
- 41. Cheng AG, Cunningham LL, Rubel EW. Hair cell death in the avian basilar papilla: characterization of the in vitro model and caspase activation. J Assoc Res Otolaryngol. 2003; 4:91–105. https://doi.org/10.1007/s10162-002-3016-8 [PubMed]
- 42. Cunningham LL, Cheng AG, Rubel EW. Caspase activation in hair cells of the mouse utricle exposed to neomycin. J Neurosci. 2002; 22:8532–40. https://doi.org/10.1523/JNEUROSCI.22-19-08532.2002 [PubMed]
- 43. Lee JE, Nakagawa T, Kim TS, Iguchi F, Endo T, Kita T, Murai N, Naito Y, Lee SH, Ito J. Signaling pathway for apoptosis of vestibular hair cells of mice due to aminoglycosides. Acta Otolaryngol Suppl. 2004; 69–74. https://doi.org/10.1080/03655230310016799 [PubMed]
- 44. Mangiardi DA, McLaughlin-Williamson K, May KE, Messana EP, Mountain DC, Cotanche DA. Progression of hair cell ejection and molecular markers of apoptosis in the avian cochlea following gentamicin treatment. J Comp Neurol. 2004; 475:1–18. https://doi.org/10.1002/cne.20129 [PubMed]
- 45. Hu BH, Henderson D, Nicotera TM. Involvement of apoptosis in progression of cochlear lesion following exposure to intense noise. Hear Res. 2002; 166:62–71. https://doi.org/10.1016/s0378-5955(02)00286-1 [PubMed]
- 46. Hu BH, Henderson D, Nicotera TM. F-actin cleavage in apoptotic outer hair cells in chinchilla cochleas exposed to intense noise. Hear Res. 2002; 172:1–9. https://doi.org/10.1016/s0378-5955(01)00361-6 [PubMed]
- 47. Nicotera TM, Hu BH, Henderson D. The caspase pathway in noise-induced apoptosis of the chinchilla cochlea. J Assoc Res Otolaryngol. 2003; 4:466–77. https://doi.org/10.1007/s10162-002-3038-2 [PubMed]
- 48. Karet FE, Finberg KE, Nelson RD, Nayir A, Mocan H, Sanjad SA, Rodriguez-Soriano J, Santos F, Cremers CW, Di Pietro A, Hoffbrand BI, Winiarski J, Bakkaloglu A, et al. Mutations in the gene encoding B1 subunit of H+-ATPase cause renal tubular acidosis with sensorineural deafness. Nat Genet. 1999; 21:84–90. https://doi.org/10.1038/5022 [PubMed]
- 49. Lin LY, Hung GY, Yeh YH, Chen SW, Horng JL. Acidified water impairs the lateral line system of zebrafish embryos. Aquat Toxicol. 2019; 217:105351. https://doi.org/10.1016/j.aquatox.2019.105351 [PubMed]
- 50. Stawicki TM, Owens KN, Linbo T, Reinhart KE, Rubel EW, Raible DW. The zebrafish merovingian mutant reveals a role for pH regulation in hair cell toxicity and function. Dis Model Mech. 2014; 7:847–56. https://doi.org/10.1242/dmm.016576 [PubMed]
- 51. Fickert P, Trauner M. When lightning strikes twice: the plot thickens for a dual role of the anion exchanger 2 (AE2/SLC4A2) in the pathogenesis and treatment of primary biliary cirrhosis. J Hepatol. 2009; 50:633–35. https://doi.org/10.1016/j.jhep.2008.12.006 [PubMed]
- 52. Mardones P, Medina JF, Elferink RP. Activation of cyclic AMP signaling in Ae2-deficient mouse fibroblasts. J Biol Chem. 2008; 283:12146–53. https://doi.org/10.1074/jbc.M710590200 [PubMed]
- 53. Zhao Y, Li W, Li M, Hu Y, Zhang H, Song G, Yang L, Cai K, Luo Z. Targeted inhibition of MCT4 disrupts intracellular pH homeostasis and confers self-regulated apoptosis on hepatocellular carcinoma. Exp Cell Res. 2019; 384:111591. https://doi.org/10.1016/j.yexcr.2019.111591 [PubMed]
- 54. Yu XH, Peng CC, Sun XX, Chen WH. Synthesis, anionophoric activity and apoptosis-inducing bioactivity of benzimidazolyl-based transmembrane anion transporters. Eur J Med Chem. 2018; 152:115–25. https://doi.org/10.1016/j.ejmech.2018.04.036 [PubMed]
- 55. Das S, Rajanikant GK. Huntington disease: can a zebrafish trail leave more than a ripple? Neurosci Biobehav Rev. 2014; 45:258–61. https://doi.org/10.1016/j.neubiorev.2014.06.013 [PubMed]
- 56. Gong J, Wang X, Zhu C, Dong X, Zhang Q, Wang X, Duan X, Qian F, Shi Y, Gao Y, Zhao Q, Chai R, Liu D. Insm1a regulates motor neuron development in zebrafish. Front Mol Neurosci. 2017; 10:274. https://doi.org/10.3389/fnmol.2017.00274 [PubMed]



