Introduction
Breast cancer is the most common malignancy in women, causing significant morbidity and mortality. Progress in surgical treatment and adjuvant systemic therapies significantly improve survival of patients [1]. However, some cases are resistant to adjuvant therapies and therefore remain incurable [2]. Mechanisms mediating pathogenesis of breast cancer still need to be elucidated.
Tumor microenvironment, particularly in those with advanced stage diseases, plays an important role in supporting tumor growth, mediating drug resistance and promoting metastasis [3]. There is considerable interest in exploring the role of immune compartment of the tumor microenvironment, such as lymphocytes and innate immune cells which can be found in most solid tumors. These studies reported two main aspects of dysfunction regarding immune microenvironment: firstly, immune cell-derived growth factors promote tumor growth, such as vascular endothelial growth factor (VEGF) and epidermal growth factor (EGF); secondly, immunosuppressive effector molecules mediate immune escape of tumor cells, for example, transforming growth factor-β (TGF-β) and programmed death-ligand 1 (PD-L1) [4–6]. Thus, new strategies in controlling breast cancer progression had emerged accordingly.
Interleukin-22 (IL-22), an IL-10 family member, exerts an effect in regulating epithelial homeostasis [7]. IL-22 stimulates proliferation of epithelial cells in gut and thymus. We and others previously showed the primary cellular sources of IL-22 are innate lymphoid cell group 3 (ILC3), T-cell and macrophage [8, 9]. IL-22 shows an immune suppressive function in regulation of adaptive immune responses. The epithelial proliferative and immunosuppressive effects can be utilized by cancer cells to facilitate tumor growth [7, 10]. This hypothesis was proposed according to the association between IL-22 and several types of tumors [11–14]. Infiltration of ILC3 and increased IL-22 level were reported to correlate with progression of breast cancer [15, 16]. However, regulation of IL-22 in breast cancer still needs to be elucidated.
Based on these reports and our previous findings, we studied the role of IL-22 in tumor growth of breast cancer. We found IL-22 stimulated proliferation of breast cancer cells in a signal transducer and activator of transcription 3 (STAT3)-dependent manner. In 4T1 breast cancer model, ILC3 was a major producer of IL-22, which was increased by giving IL-1β and IL-23.
Results
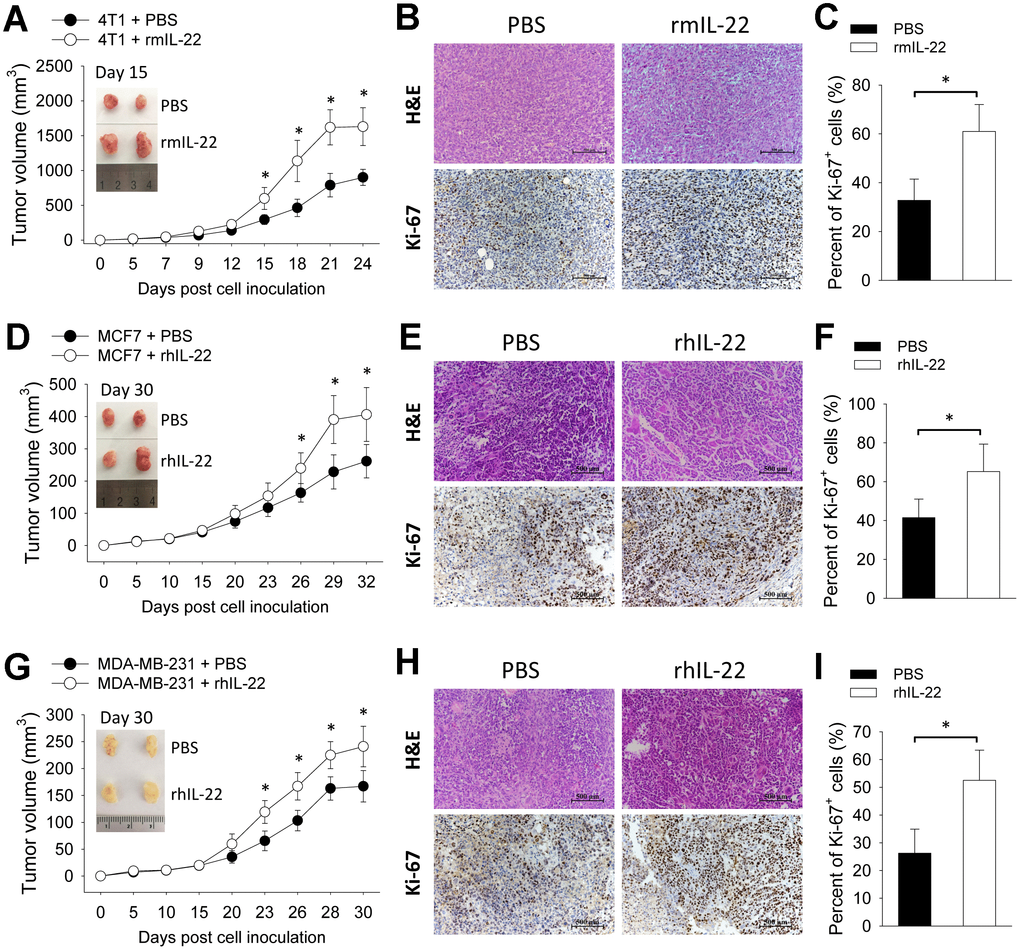
IL-22 promotes growth of breast cancer cells in vivo
To assess the impact of exogenous IL-22 on tumor growth, we applied three breast cancer models by transplanting 4T1 cells to wild type BALB/c mice and transplanting MCF7 cells and MDA-MB-231 cells to BALB/c Nude mice. In the 4T1 model, injection of IL-22 increased tumor size (Figure 1A) which was associated with increased proliferation of tumor cells as indicated by more Ki-67-positive cells (Figure 1B, 1C). This effect was also observed in MCF7 and MDA-MB-231 xenograft models (Figure 1D–1F). These results show that IL-22 promotes growth of breast cancer cells in vivo.
IL-22 stimulates proliferation of breast cancer cells in vitro
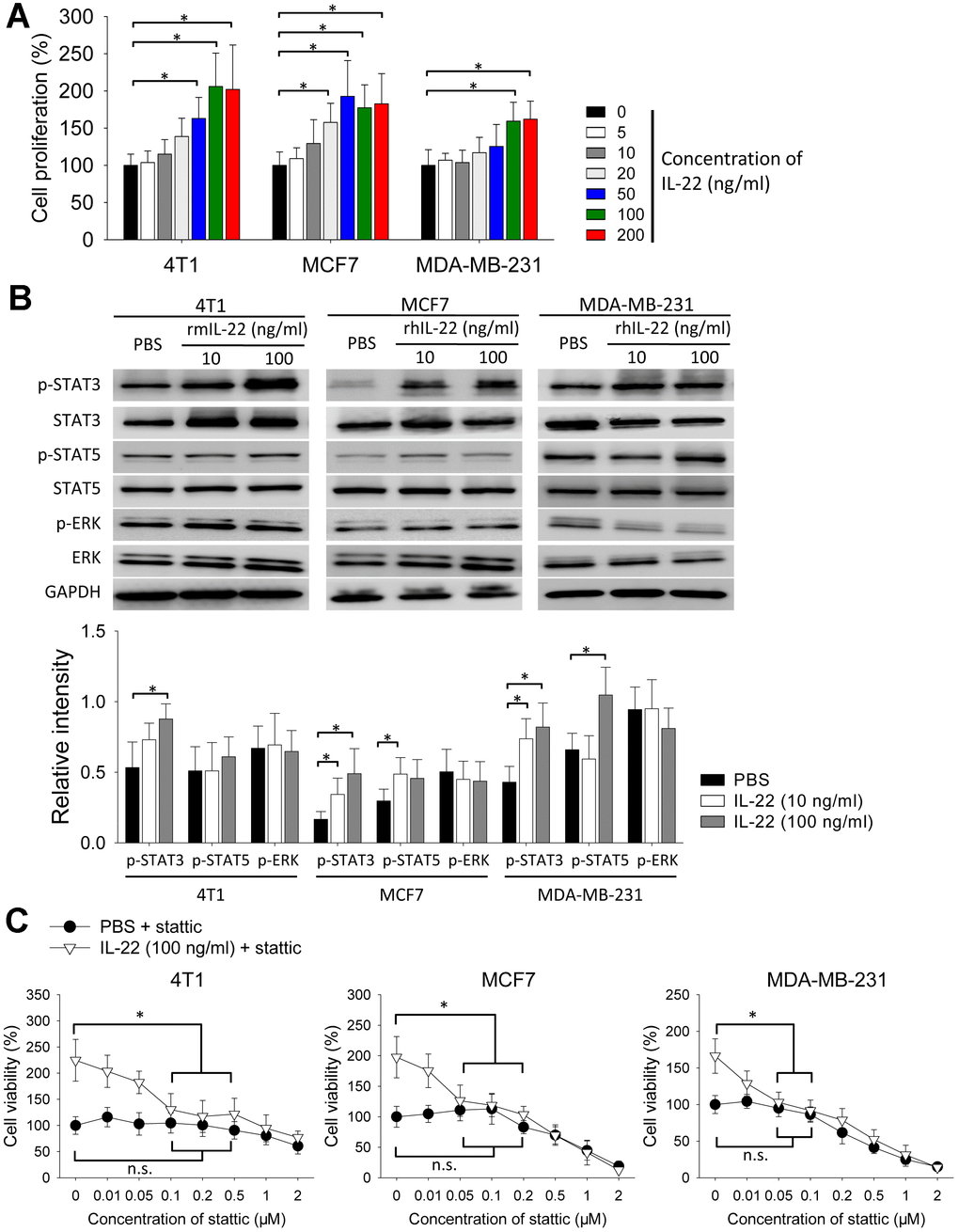
Next, we tested the effect of IL-22 in cell proliferation in vitro. IL-22 increased proliferation of 4T1, MCF7 and MDA-MB-231 cells (Figure 2A). Because IL-22 signal is transduced by STAT and extracellular signal-regulated kinase (ERK) pathways [7], we detected phosphorylation of these proteins. IL-22 increased levels of phosphorylated STAT3 in 4T1, MCF7 and MDA-MB-231 cells. IL-22 also increased phosphorylation of STAT5 in MCF7 and MDA-MB-231 cells but not in 4T1 cells. In contrast, IL-22 did not increase phosphorylation of ERK in these cells (Figure 2B). To test whether STAT3 is important for IL-22 mediated proliferative effect, we added STAT3-inhibitor stattic to cell cultures. IL-22-induced cell proliferation was reversed by stattic at the concentration ranging from 50 to 500 nM. In contrast, the same dose of stattic did not decrease proliferation of cells which were not treated with IL-22 (Figure 2C). These findings indicate IL-22 stimulates proliferation of breast cancer cells in a STAT3-dependent manner.
Cellular source of IL-22 in breast cancer
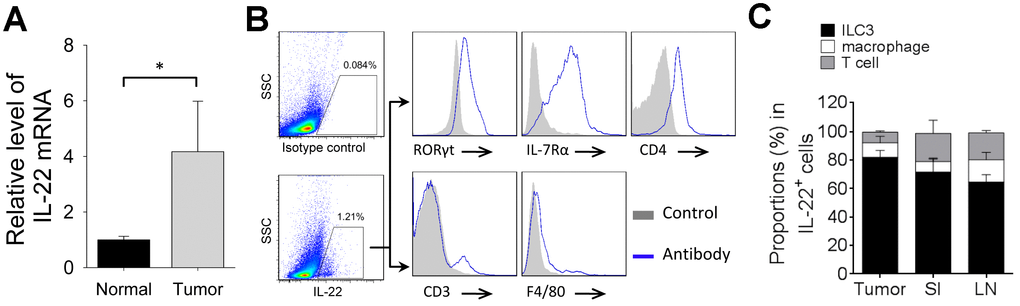
Next, we analyzed the endogenous IL-22 in 4T1 tumor model. Tumor tissue expressed higher level of IL-22 mRNA than that in normal mammary tissue (Figure 3A). IL-22-producing cells were detected in tumor tissue. Most IL-22-positive cells were ILC3s, whereas T-cell and macrophage took smaller proportions in IL-22-positive cells (Figure 3B, 3C). We also analyzed IL-22-producing cells in small intestine and lymph node. Percent of IL-22-positive ILC3 in tumor tissue was higher than that in lymph node (p < 0.05). Percent of IL-22-positive macrophage in lymph node was higher than that in small intestine (p < 0.05). Percent of IL-22-positive T-cell in small intestine and lymph node were higher than that in tumor tissue (p < 0.05) (Figure 3C). Thus, ILC3 is a predominant producer of IL-22 in tumor tissue of 4T1 model.
IL-1β and IL-23 increases intro-tumor level of IL-22 and promotes growth of breast cancer cells
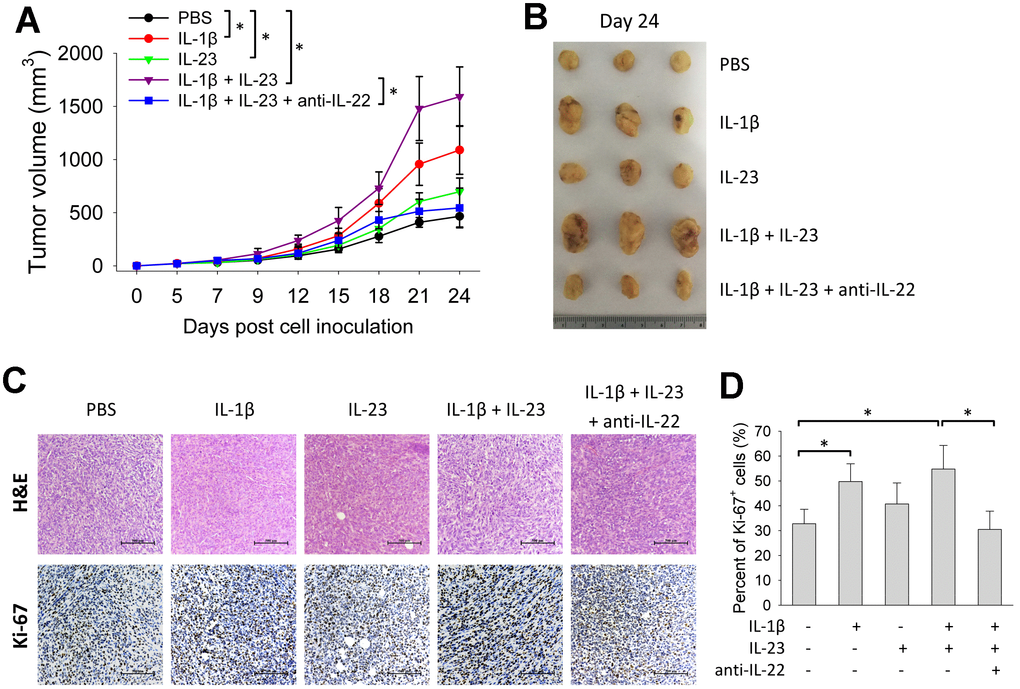
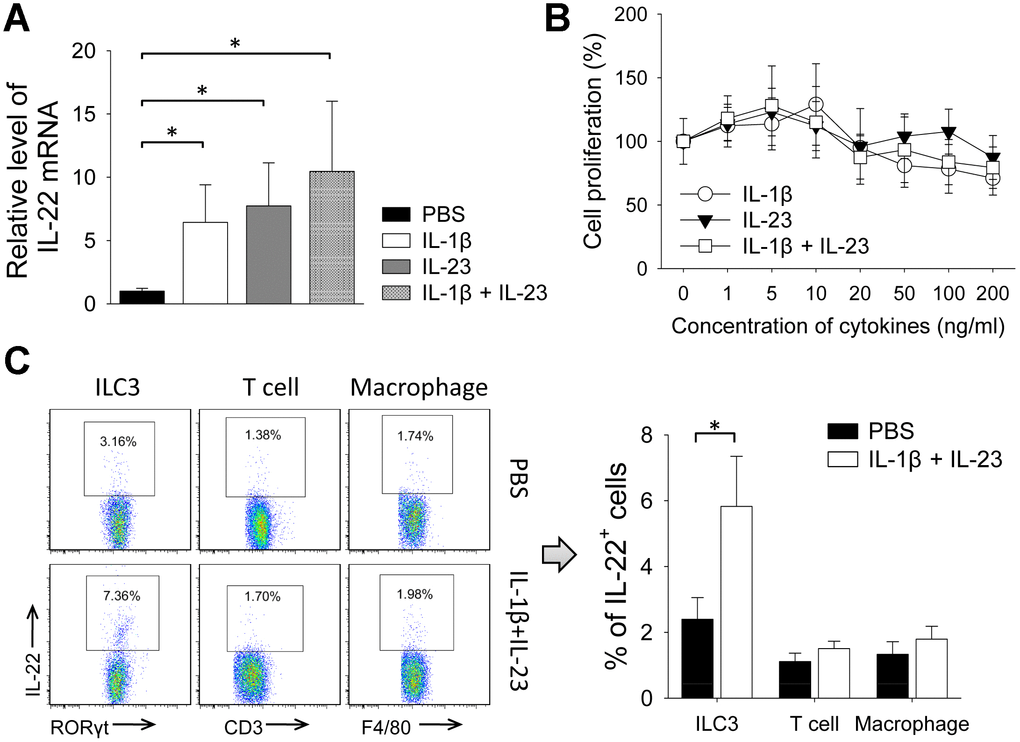
Because IL-1β and IL-23 are upstream cytokines in regulating production of IL-22 [17, 18], we tested the impact of IL-1β and IL-23 on tumor growth. Giving IL-1β or IL-23 increased size of 4T1 tumors. Tumor size was also increased by giving both IL-1β and IL-23, an effect reversed by concurrent use of an IL-22 neutralization antibody (Figure 4A, 4B). IL-1β with or without IL-23 increased percent of Ki-67-positive cells in the 4T1 tumors, which was decreased by concurrent use of the IL-22 antibody (Figure 4C, 4D). Giving IL-1β and/or IL-23 increased IL-22 mRNA levels in the 4T1 tumors (Figure 5A). IL-1β and IL-23 increased percent of IL-22-producing ILC3 in tumor tissue (Figure 5B). However, IL-1β and IL-23 did not show a direct proliferative effect on 4T1 cells in vitro (Figure 5C). These results indicate IL-22 might be important in mediating the tumor-promoting effect of IL-1β and IL-23.
Discussion
In this study, we showed IL-22 exerted a proliferative effect on breast cancer cells in a STAT3-dependent manner. In 4T1 tumor tissue, ILC3 is a major cellular source of IL-22 which can be increased by giving IL-1β and IL-23.
IL-22 plays a role in regulating inflammatory diseases with a predominant anti-inflammatory effect. More recently, IL-22 was reported to be an important mediator of epithelial homeostasis [7]. These characters render a cancer-promoting function of IL-22 in epithelial cancers. This was implicated in the correlations between IL-22 and several types of carcinoma such as colon cancer and hepatocellular carcinoma [11–14]. Some recent studies indicated increased IL-22 level was associated with progression of breast cancer [16, 19]. We showed giving IL-22 increased proliferation of breast cancer cells including estrogen receptor-positive MCF7 cells and triple-negative 4T1 and MDA-MB-231 cells, which indicates IL-22-mediated proliferative effect might not depend on expression of estrogen receptor. Others reported expression of IL-22 in human breast cancer [16, 20]. However, the correlation between IL-22 level and receptor type of tumor cell is unclear in human breast cancer.
In the absence of exogenous IL-22, endogenous IL-22 level was also up-regulated in 4T1 model. Endogenous IL-22 might also exert proliferative effect in breast cancer, which can be confirmed by a test in IL-22 gene knock-out mice. Because MCF7 and MDA-MB-231 xenograft models were established on immune deficient mice, we analyzed IL-22 production in the 4T1 model which was performed with immune competent mice. IL-22 producing cells were enriched in T-cell, macrophage and ILC3, with ILC3 as the major producer. A recent study reported that ILC3 infiltration promoted lymphatic metastasis of breast cancer [15]. On the other hand, exhaustion of T-cell function can be found in breast cancer [21]. IL-22-mediated anti-inflammatory effect might further impair the anti-tumor function of T-cell. These findings suggest IL-22 is a cancer-promoting factor in breast cancer, and blocking IL-22 function might reduce tumor progression. IL-22 antibody was reported to reduce growth of other types of tumors [22, 23]. Nonetheless, results from human studies are needed to confirm the correlation between IL-22 and breast cancer.
Activation of STAT3 pathway was reported in a variety of malignancies including breast cancer [24]. Chronic inflammation was observed in pathogenesis of breast cancer [6]. IL-6 is a strong stimulator of STAT3 pathway which is important in regulating survival and growth of breast cancer cells [25]. We showed IL-22, an IL-10 family member, increased phosphorylation of STAT3 in 4T1, MCF7 and MDA-MB-231 cells. Inhibition of STAT3 reversed the proliferative effect of IL-22 on these cells. Moreover, STAT3 can be activated by other stimulators such as EGF and VEGF [26], which are important in formation of stromal compartment in breast cancer [24].
The role of IL-1β in breast cancer has been reported [27, 28]. Increased level of IL-23 was also observed in human breast cancer [29]. However, it is unclear whether these two cytokines mediate a direct effect on breast cancer cells. We showed IL-1β and IL-23 did not directly increase cell proliferation in cell culture, but rather increased tumor size in the transplant model in vivo. Interestingly, this effect was blocked by an IL-22 neutralization antibody. IL-1β and IL-23 increased IL-22 production of ILC3 in the tumor tissue. Thus, a possible mechanism might be IL-1β and IL-23 promoting breast cancer progression via IL-22. It has been well established that IL-1β and IL-23 stimulate production of IL-22 in immune cells [17, 18]. Macrophage, a major component of tumor immune microenvironment, is a potent producer of IL-1β and IL-23 [30, 31]. Pathogenic macrophages often present in human breast cancer tissues, and the macrophage-derived factors are involved in tumor progression [6, 32]. It can be deduced that pathogenic macrophages might up-regulate production of IL-22 in ILC3. In addition, we cannot rule out that IL-1β and IL-23 mediate breast cancer progression through immune-regulatory functions. Others reported breast cancer cell-derived IL-1α up-regulated IL-22 production of T cells in an AhR- and RORγt-dependent manner, which indicated a direct crosstalk between cancer cells and immune cells [16].
In conclusion, we confirm the proliferative effect of IL-22 in breast cancer. ILC3 is a primary cellular source of IL-22 in tumor tissue. Our findings suggest blocking IL-22 function might reduce IL-1β- and IL-23-induced tumor progression of breast cancer.
Materials and Methods
Mice
Female BALB/c mice and BALB/c Nude mice (18-20g) were purchased from Vital River (Charles River, Beijing, China). Mice were bred in a special pathogen free room. Mice were anesthetized by inhaling isoflurane before invasive operations. All procedures regarding animal care and experiments were approved by the Experimental Animal Care and Use Committee of Xuzhou Medical University.
Cell culture
MCF7 cells (ATCC HTB-22) were cultured in DMEM medium (Thermo Fisher Scientific, Waltham, MA, USA) supplied with 10% FBS (Gibco, Thermo Fisher Scientific). 4T1 (ATCC CRL-2539) cells were cultured in RPMI-1640 medium (Sigma-Aldrich, Shanghai, China) supplied with 10% FBS. MDA-MB-231 (ATCC HTB-26) cells were cultured in Leibovitz's L-15 medium (Thermo Fisher Scientific) containing 10% FBS in an incubator without CO2 equilibration.
Reagents
Recombinant murine IL-22 (rmIL-22), rmIL-1β and anti-murine IL-22 were purchased from PeproTech (Cat# 210-22, Cat# 211-11B and Cat# 500-P223, Rocky Hill, NJ, USA). rmIL-23 was purchased from BioLegend (Cat# 589002, San Diego, CA, USA). Recombinant human IL-22 (rhIL-22) was purchased from STEMCELL Technologies (Cat# 78038, Shanghai, China). Stattic was purchased from ApexBio (Cat# A2224, Houston, TX, USA).
Tumor models
4T1 cells (2×105) were injected into a single mammary fat pad of BALB/c mice. BALB/c Nude mice were inoculated subcutaneously with 2×106 MCF7 cells or 2×106 MDA-MB-231 cells. Mice were injected intraperitoneally (i.p.) with PBS, recombinant cytokines (20 μg/kg) or anti-murine IL-22 (5 mg/kg) thrice a week for up to 3 weeks after cell inoculation. Tumor size was measured continuously. Tumor volume was calculated as: Tumor volume (mm3) = length (mm) × width (mm) × width (mm) × 0.5.
H&E staining and immunohistochemistry
Tissue slides of tumors were prepared from samples collected from in vivo experiments. H&E staining was performed as described previously [33].
To detect Ki-67-expressing cells, immunohistochemistry was performed on tissue slides. Ki-67 antibodies were purchased from ZSGB-BIO (Cat# ZM-0166, Beijing, China) and Affinity Biosciences (Cat# AF0198, Cincinnati, OH, USA). Second antibody was peroxidase-conjugated goat anti-rabbit IgG (ZSGB-BIO, Cat# PV-6000). DAB substrate (ZSGB-BIO) was used for visualization of positive cells.
Cell proliferation
Cell proliferation was measured with a Cell Counting Kit-8 (CCK-8) from DOJINDO (CK04, Tokyo, Japan). Cells were cultured with CCK-8 reagent for 1 hour followed by detection of optical density at 450 nm.
Western blotting
Proteins were extracted from cells using Cell Lysis Buffer (Cell Signaling Technology, Danvers, MA, USA). Western blot analysis was performed to detect protein levels with the following antibodies: Phospho-STAT3 (Tyr705), STAT3 (4904), Phospho-STAT5 (Tyr694), STAT5 (94205), Phospho-ERK (T202/Y204), ERK (4695) and GAPDH (D16H11) from Cell Signaling Technology.
Quantitative polymerase chain reaction
Quantitative polymerase chain reaction (qPCR) was performed to detect IL-22 mRNA as described previously [34]. RNA was isolated using TRIzol (Invitrogen, Thermo Fisher Scientific). cDNA was synthesized using PrimeScript cDNA Synthesis Kit (Takara Bio Inc., Shiga, Japan). cDNA concentrations were analyzed by qPCR with LightCycler 480 SYBR Green I Master qRT-PCR kit (Roche, Mannheim, Germany). GAPDH mRNA level was used for normalization. qPCR was performed on an LC480 cycler (Roche) with the following primers (Thermo Fisher Scientific, Shanghai, China): IL-22 (TCGCCTTGATCTCTCCACTC and GCTCAGCTCCTGTCACATCA) and GAPDH (TTGATGGCAACAATCTCCAC and CGTCCCGTAGACAAAATGGT). Relative mRNA levels are expressed as fold change calculated from –ΔΔCT values.
Isolation of mononuclear cells from tissues
To isolate mononuclear cells from lymph node, samples were mechanically dissociated in RPMI-1640 medium containing 2% FBS. Cells were recovered by centrifugation in Lymphocyte Separation Medium (DAKEWE, Shanghai, China).
To isolate mononuclear cells from small intestine, samples were obtained and Peyer’s patches were removed. Samples were cut into small pieces and incubated with 5 mM EDTA, followed by a digestion with collagenase type I (1 mg/ml), collagenase type IV (0.5 mg/ml) and DNase I (1 mg/ml). Remained tissues were digested with dispase (0.5 mg/ml) and collagenase type I (1 mg/ml). Released cells were collected from every digestion. Cells were recovered by centrifugation in Percoll buffer (GE Healthcare Bio-Sciences, Pittsburgh, PA, USA).
To isolate mononuclear cells from tumor, samples were twice digested with collagenase type I (1 mg/ml), collagenase type IV (0.5 mg/ml) and DNase I (1 mg/ml). Released cells were centrifuged in Percoll buffer. Enzymes were purchased from Sigma-Aldrich (Shanghai, China).
Flow cytometry
For analyzing IL-22-producing cells, isolated mononuclear cells were stimulated with PMA (Sigma-Aldrich), ionomycin (Sigma-Aldrich) and Brefeldin A (Thermo Fisher Scientific) for 4 h, followed by treatment with Transcription Factor Staining Buffer (eBioscience, Thermo Fisher Scientific). Cells were stained with the following antibodies: anti-CD45 (30-F11), anti-IL-22 (Poly5164), anti-CD3 (145-2C11), anti-CD4 (RM4-5), anti-F4/80 (T45-2342), anti-RORγt (Q31-378) and anti-IL-7Rα (SB/199). T cells are defined as CD3+. Macrophages are defined as F4/80+. CD3-CD4+IL-7Rα+RORγt+ cells were considered as ILC3. Antibodies were from BioLegend (San Diego, CA, USA), eBioscience (Thermo Fisher Scientific) or BD Biosciences (San Jose, CA, USA). Cells were acquired and analyzed on an LSRFortessa flow cytometer (BD Biosciences).
Statistics
Group size (n) refers to independent values. Data are presented as mean ± standard deviation (SD). Comparison of means was performed with unpaired Student t test or one-way ANOVA test followed by Bonferroni correction. P-value < 0.05 was considered statistically significant.
Author Contributions
B.P. designed experiments, performed experiments, analyzed data and wrote the manuscript. S.Y. contributed to the concept, analyzed data and revised the manuscript. Y.Z., C.L. and J.G. performed experiments and analyzed data. S.S. and Y.C. performed experiments. Y.Z., C.L. and J.G. contributed equally to this study.
Conflicts of Interest
The authors declare no conflicts of interest.
Funding
This study is supported by National Natural Science Foundation of China (81970159, 81930005 and 81671584) and Postgraduate Research and Practice Innovation Program of Jiangsu Province (KYCX18-2177 and KYCX19-2248).
References
- 1. Harbeck N, Gnant M. Breast cancer. Lancet. 2017; 389:1134–50. https://doi.org/10.1016/S0140-6736(16)31891-8 [PubMed]
- 2. Harbeck N, Penault-Llorca F, Cortes J, Gnant M, Houssami N, Poortmans P, Ruddy K, Tsang J, Cardoso F. Breast cancer. Nat Rev Dis Primers. 2019; 5:66. https://doi.org/10.1038/s41572-019-0111-2 [PubMed]
- 3. Nienhuis HH, Gaykema SB, Timmer-Bosscha H, Jalving M, Brouwers AH, Lub-de Hooge MN, van der Vegt B, Overmoyer B, de Vries EG, Schröder CP. Targeting breast cancer through its microenvironment: current status of preclinical and clinical research in finding relevant targets. Pharmacol Ther. 2015; 147:63–79. https://doi.org/10.1016/j.pharmthera.2014.11.004 [PubMed]
- 4. Speiser DE, Ho PC, Verdeil G. Regulatory circuits of T cell function in cancer. Nat Rev Immunol. 2016; 16:599–611. https://doi.org/10.1038/nri.2016.80 [PubMed]
- 5. DeNardo DG, Ruffell B. Macrophages as regulators of tumour immunity and immunotherapy. Nat Rev Immunol. 2019; 19:369–82. https://doi.org/10.1038/s41577-019-0127-6 [PubMed]
- 6. Qiu SQ, Waaijer SJ, Zwager MC, de Vries EG, van der Vegt B, Schröder CP. Tumor-associated macrophages in breast cancer: innocent bystander or important player? Cancer Treat Rev. 2018; 70:178–89. https://doi.org/10.1016/j.ctrv.2018.08.010 [PubMed]
- 7. Dudakov JA, Hanash AM, van den Brink MR. Interleukin-22: immunobiology and pathology. Annu Rev Immunol. 2015; 33:747–85. https://doi.org/10.1146/annurev-immunol-032414-112123 [PubMed]
- 8. Pan B, Wang D, Li L, Shang L, Xia F, Zhang F, Zhang Y, Gale RP, Xu M, Li Z, Xu K. IL-22 accelerates thymus regeneration via Stat3/Mcl-1 and decreases chronic graft-versus-host disease in mice after allotransplants. Biol Blood Marrow Transplant. 2019; 25:1911–19. https://doi.org/10.1016/j.bbmt.2019.06.002 [PubMed]
- 9. Dudakov JA, Hanash AM, Jenq RR, Young LF, Ghosh A, Singer NV, West ML, Smith OM, Holland AM, Tsai JJ, Boyd RL, van den Brink MR. Interleukin-22 drives endogenous thymic regeneration in mice. Science. 2012; 336:91–95. https://doi.org/10.1126/science.1218004 [PubMed]
- 10. Markota A, Endres S, Kobold S. Targeting interleukin-22 for cancer therapy. Hum Vaccin Immunother. 2018; 14:2012–15. https://doi.org/10.1080/21645515.2018.1461300 [PubMed]
- 11. Kryczek I, Lin Y, Nagarsheth N, Peng D, Zhao L, Zhao E, Vatan L, Szeliga W, Dou Y, Owens S, Zgodzinski W, Majewski M, Wallner G, et al. IL-22(+)CD4(+) T cells promote colorectal cancer stemness via STAT3 transcription factor activation and induction of the methyltransferase DOT1L. Immunity. 2014; 40:772–84. https://doi.org/10.1016/j.immuni.2014.03.010 [PubMed]
- 12. Fukui H, Zhang X, Sun C, Hara K, Kikuchi S, Yamasaki T, Kondo T, Tomita T, Oshima T, Watari J, Imura J, Fujimori T, Sasako M, Miwa H. IL-22 produced by cancer-associated fibroblasts promotes gastric cancer cell invasion via STAT3 and ERK signaling. Br J Cancer. 2014; 111:763–71. https://doi.org/10.1038/bjc.2014.336 [PubMed]
- 13. Khosravi N, Caetano MS, Cumpian AM, Unver N, De la Garza Ramos C, Noble O, Daliri S, Hernandez BJ, Gutierrez BA, Evans SE, Hanash S, Alekseev AM, Yang Y, et al. IL22 promotes Kras-mutant lung cancer by induction of a protumor immune response and protection of stemness properties. Cancer Immunol Res. 2018; 6:788–97. https://doi.org/10.1158/2326-6066.CIR-17-0655 [PubMed]
- 14. Zhao D, Long XD, Lu TF, Wang T, Zhang WW, Liu YX, Cui XL, Dai HJ, Xue F, Xia Q. Metformin decreases IL-22 secretion to suppress tumor growth in an orthotopic mouse model of hepatocellular carcinoma. Int J Cancer. 2015; 136:2556–65. https://doi.org/10.1002/ijc.29305 [PubMed]
- 15. Irshad S, Flores-Borja F, Lawler K, Monypenny J, Evans R, Male V, Gordon P, Cheung A, Gazinska P, Noor F, Wong F, Grigoriadis A, Fruhwirth GO, et al. RORγt+ innate lymphoid cells promote lymph node metastasis of breast cancers. Cancer Res. 2017; 77:1083–96. https://doi.org/10.1158/0008-5472.CAN-16-0598 [PubMed]
- 16. Voigt C, May P, Gottschlich A, Markota A, Wenk D, Gerlach I, Voigt S, Stathopoulos GT, Arendt KA, Heise C, Rataj F, Janssen KP, Königshoff M, et al. Cancer cells induce interleukin-22 production from memory CD4+ T cells via interleukin-1 to promote tumor growth. Proc Natl Acad Sci USA. 2017; 114:12994–99. https://doi.org/10.1073/pnas.1705165114 [PubMed]
- 17. Ebbo M, Crinier A, Vély F, Vivier E. Innate lymphoid cells: major players in inflammatory diseases. Nat Rev Immunol. 2017; 17:665–78. https://doi.org/10.1038/nri.2017.86 [PubMed]
- 18. Fatkhullina AR, Peshkova IO, Dzutsev A, Aghayev T, McCulloch JA, Thovarai V, Badger JH, Vats R, Sundd P, Tang HY, Kossenkov AV, Hazen SL, Trinchieri G, et al. An interleukin-23-interleukin-22 axis regulates intestinal microbial homeostasis to protect from diet-induced atherosclerosis. Immunity. 2018; 49:943–57.e9. https://doi.org/10.1016/j.immuni.2018.09.011 [PubMed]
- 19. Wang S, Yao Y, Yao M, Fu P, Wang W. Interleukin-22 promotes triple negative breast cancer cells migration and paclitaxel resistance through JAK-STAT3/MAPKs/AKT signaling pathways. Biochem Biophys Res Commun. 2018; 503:1605–09. https://doi.org/10.1016/j.bbrc.2018.07.088 [PubMed]
- 20. Niakan A, Faghih Z, Talei AR, Ghaderi A. Cytokine profile of CD4+ CD25- FoxP3+ T cells in tumor-draining lymph nodes from patients with breast cancer. Mol Immunol. 2019; 116:90–97. https://doi.org/10.1016/j.molimm.2019.10.007 [PubMed]
- 21. Canale FP, Ramello MC, Núñez N, Araujo Furlan CL, Bossio SN, Gorosito Serrán M, Tosello Boari J, Del Castillo A, Ledesma M, Sedlik C, Piaggio E, Gruppi A, Acosta Rodríguez EA, Montes CL. CD39 expression defines cell exhaustion in tumor-infiltrating CD8+ T cells. Cancer Res. 2018; 78:115–28. https://doi.org/10.1158/0008-5472.CAN-16-2684 [PubMed]
- 22. Abikhair M, Mitsui H, Yanofsky V, Roudiani N, Ovits C, Bryan T, Oberyszyn TM, Tober KL, Gonzalez J, Krueger JG, Felsen D, Carucci JA. Cyclosporine a immunosuppression drives catastrophic squamous cell carcinoma through IL-22. JCI Insight. 2016; 1:e86434. https://doi.org/10.1172/jci.insight.86434 [PubMed]
- 23. Protopsaltis NJ, Liang W, Nudleman E, Ferrara N. Interleukin-22 promotes tumor angiogenesis. Angiogenesis. 2019; 22:311–23. https://doi.org/10.1007/s10456-018-9658-x [PubMed]
- 24. Banerjee K, Resat H. Constitutive activation of STAT3 in breast cancer cells: a review. Int J Cancer. 2016; 138:2570–78. https://doi.org/10.1002/ijc.29923 [PubMed]
- 25. Jin S, Mutvei AP, Chivukula IV, Andersson ER, Ramsköld D, Sandberg R, Lee KL, Kronqvist P, Mamaeva V, Ostling P, Mpindi JP, Kallioniemi O, Screpanti I, et al. Non-canonical notch signaling activates IL-6/JAK/STAT signaling in breast tumor cells and is controlled by p53 and IKKα/IKKβ. Oncogene. 2013; 32:4892–902. https://doi.org/10.1038/onc.2012.517 [PubMed]
- 26. Wu S, Fu J, Dong Y, Yi Q, Lu D, Wang W, Qi Y, Yu R, Zhou X. GOLPH3 promotes glioma progression via facilitating JAK2-STAT3 pathway activation. J Neurooncol. 2018; 139:269–79. https://doi.org/10.1007/s11060-018-2884-7 [PubMed]
- 27. Kaplanov I, Carmi Y, Kornetsky R, Shemesh A, Shurin GV, Shurin MR, Dinarello CA, Voronov E, Apte RN. Blocking IL-1β reverses the immunosuppression in mouse breast cancer and synergizes with anti-PD-1 for tumor abrogation. Proc Natl Acad Sci USA. 2019; 116:1361–69. https://doi.org/10.1073/pnas.1812266115 [PubMed]
- 28. Wu TC, Xu K, Martinek J, Young RR, Banchereau R, George J, Turner J, Kim KI, Zurawski S, Wang X, Blankenship D, Brookes HM, Marches F, et al. IL1 receptor antagonist controls transcriptional signature of inflammation in patients with metastatic breast cancer. Cancer Res. 2018; 78:5243–58. https://doi.org/10.1158/0008-5472.CAN-18-0413 [PubMed]
- 29. Sheng S, Zhang J, Ai J, Hao X, Luan R. Aberrant expression of IL-23/IL-23R in patients with breast cancer and its clinical significance. Mol Med Rep. 2018; 17:4639–44. https://doi.org/10.3892/mmr.2018.8427 [PubMed]
- 30. Mangan MS, Olhava EJ, Roush WR, Seidel HM, Glick GD, Latz E. Targeting the NLRP3 inflammasome in inflammatory diseases. Nat Rev Drug Discov. 2018; 17:588–606. https://doi.org/10.1038/nrd.2018.97 [PubMed]
- 31. Shapouri-Moghaddam A, Mohammadian S, Vazini H, Taghadosi M, Esmaeili SA, Mardani F, Seifi B, Mohammadi A, Afshari JT, Sahebkar A. Macrophage plasticity, polarization, and function in health and disease. J Cell Physiol. 2018; 233:6425–40. https://doi.org/10.1002/jcp.26429 [PubMed]
- 32. Song W, Thakor P, Vesey DA, Gobe GC, Morais C. Conditioned medium from stimulated macrophages inhibits growth but induces an inflammatory phenotype in breast cancer cells. Biomed Pharmacother. 2018; 106:247–54. https://doi.org/10.1016/j.biopha.2018.06.126 [PubMed]
- 33. Zhang Y, Xia F, Zhang F, Cui Y, Wang Q, Liu H, Wu Y. miR-135b-5p enhances doxorubicin-sensitivity of breast cancer cells through targeting anterior gradient 2. J Exp Clin Cancer Res. 2019; 38:26. https://doi.org/10.1186/s13046-019-1024-3 [PubMed]
- 34. Pan B, Zhang F, Lu Z, Li L, Shang L, Xia F, Fu R, Xu M, Zeng L, Xu K. Donor t-cell-derived interleukin-22 promotes thymus regeneration and alleviates chronic graft-versus-host disease in murine allogeneic hematopoietic cell transplant. Int Immunopharmacol. 2019; 67:194–201. https://doi.org/10.1016/j.intimp.2018.12.023 [PubMed]


