Introduction
Lung cancer is a highly aggressive neoplasm with an unfavorable survival [1]. Non-small cell lung cancer (NSCLC) is the most common histologic type of lung cancer, accounting for about 85% of all cases. Now, the 5-year survival rate is only 10-15% for NSCLC patients in advanced stage [2]. Lung squamous cell carcinoma (LUSC) and lung adenocarcinoma (LUAD) are two main pathological types of NSCLC. LUSC is closely correlated with a history of tobacco smoking. LUAD is currently the most common type of NSCLC in lifelong non-smokers, accounting for approximately 40% of NSCLC cases [1, 3]. However, the overall survival of patients with LUAD is poorer than LUSC patients, which may be the result of the different pathogenesis, tumor microenvironment or aberrant oncogenes [4].
Gamma-glutamyltransferase (GGT) is a membrane-bound extracellular enzyme involved in the hydrolysis of gamma-glutamyl bonds of glutathione (GSH) and other gamma-glutamyl compounds. In animals, GGT is highly expressed in tissues with secretory or absorptive functions, such as kidney, intestine, liver, prostate and gallbladder [5]. Moreover, the enzyme GGT as a new non-invasive marker can be detected in blood, which is used to indicate many diseases. Recent studies have demonstrated that increased serum GGT level predicted the advanced histological hepatic injury, insulin resistance and other chronic disease [6, 7]. Elevated serum GGT was associated with the ventricular repolarization abnormalities in the early stage of T2DM [8]. In addition, most studies showed that GGT expression was often significantly increased in human malignancies. For instance, overall survival for gallbladder cancer patients with elevated GGT was significantly worse than patients with the normal level of GGT [9]. Patients with primary metastatic breast cancer in the high risk GGT group had a poorer overall survival, compared to the low risk group [10]. Moreover, high pre-therapeutic GGT serum levels was associated with the advanced tumor stage and could serve as an independent prognostic marker for the worse survival in patients with epithelial ovarian cancer [11], pancreatic cancer [12] and endometrial cancer [13]. These studies just focus on the prognostic role of GGT in cancers, but its functions and mechanisms during cancer progression are not fully disclosed.
Gamma-glutamyltransferase 5 (GGT5), also known as GGT-like activity 1 (GGTLA1), is a member of the GGT protein family. Many tissue-local macrophages, such as Kupffer cells in the liver and alveolar macrophages, express GGT5 protein [14]. GGT5 is a cell membrane protein that hydrolyzes the gamma-glutamyl moiety of GSH and GSH S-conjugates [15]. It plays a critical role in redox regulation, drug metabolism, immune function and other processes within the body [16]. In addition, GGT5 is also able to convert leukotriene C4 (LTC4) to leukotriene D4, but appears to have distinct substrate specificity compared to GGT [17]. However, previous study suggested that GGT5 knockout mice did not show any phenotypic abnormalities unless under stress [18]. In zymosan-A-induced peritonitis model, GGT5-deficient mice cannot metabolize LTC4 in peritoneal fluid, which results in attenuated neutrophils infiltration into the peritoneum [19]. Moreover, during Aspergillus fumigatus induced asthma, GGT5 knockout mice have increased airway hyper-responsiveness [20]. These evidences suggest that GGT5 may promote the inflammatory response. However, the role of GGT5 in cancer progression have not yet been characterized.
In the present study, we first reported that high expression of protein GGT5 in cancer-associated fibroblasts (CAFs) predicted the poorer survival of LUAD patients. Moreover, our in vitro and in vivo functional studies indicated that CAFs-derived GGT5 promoted cell proliferation and drug resistance by reducing the reactive oxygen species (ROS) level in LUAD cells, suggesting that GGT5 may be a promising therapeutic target for patients with LUAD.
Results
High expression of GGT5 predicts the poor outcome of patients with LUAD
GGT5 is a member of the GGT family, but its role in cancer is undefined, especially in LUAD. First, Disease Ontology analysis of GGT5 in human using Coexpedia indicated that GGT5 was closely associated with malignant cancer development, including lung cancer (Supplementary Figure 1A). Moreover, gene expression data from The Cancer Genome Atlas (TCGA) database (Figure 1A) and Gene Expression Omnibus (GEO) cohort (GSE2514, Supplementary Figure 1B) demonstrated that GGT5 mRNA expression was substantially higher in LUAD tissues than that in normal lung tissues. Consistent with these biostatistics, IHC staining also showed the increased protein levels of GGT5 in clinical LUAD samples than that in the paired non-tumor tissues (Figure 1B, Supplementary Figure 1C).
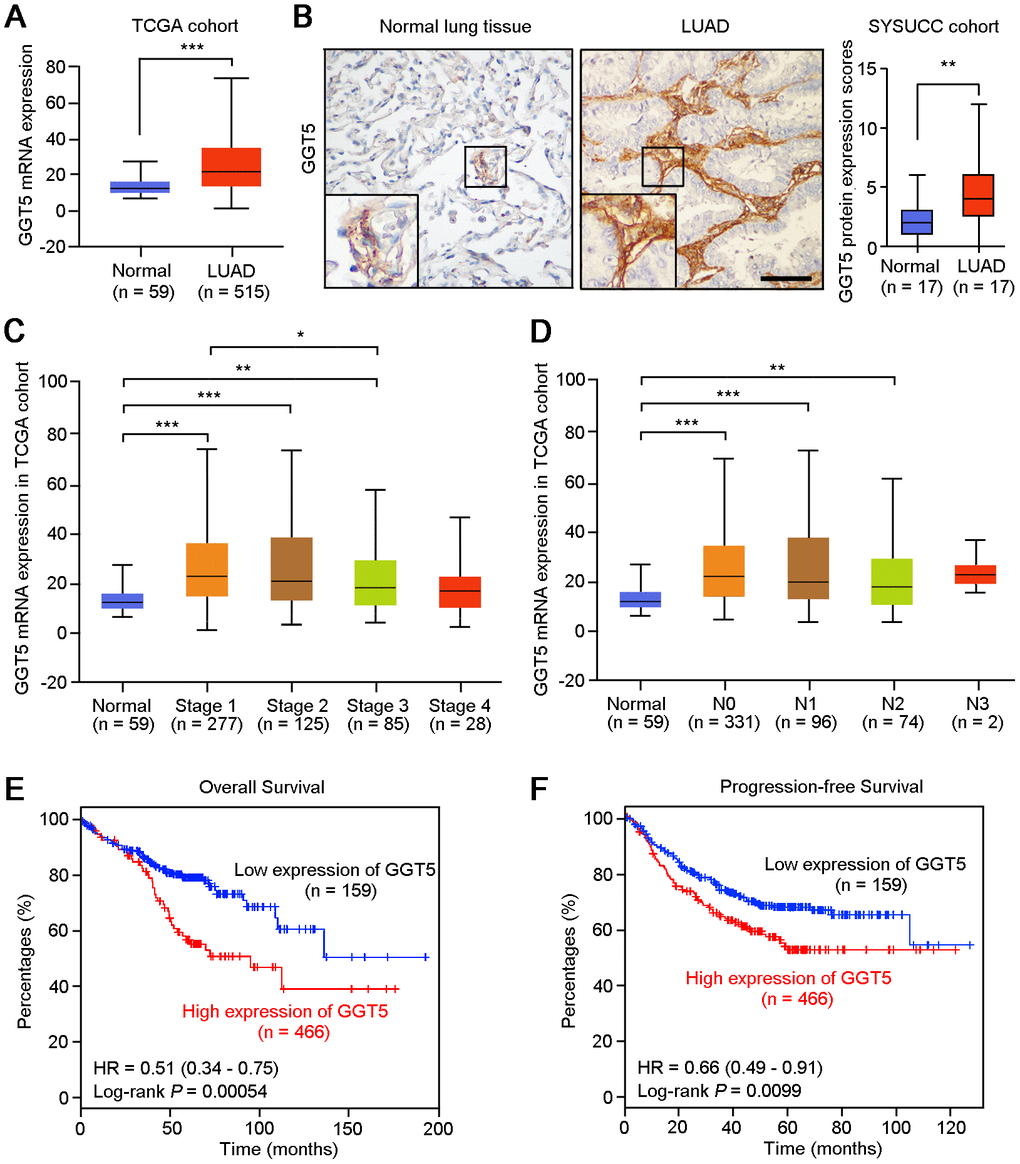
In addition, according to the clinical data from TCGA database, the mRNA expression of GGT5 was up-regulated in patients with early-stage of LUAD (Figure 1C) and lymph node metastasis (Figure 1D). Survival analysis with Kaplan-Meier Plotter (http://kmplot.com/analysis/) suggested that both overall survival (P = 0.00054) and progression-free survival (P = 0.0099) of LUAD patients with high GGT5 expression (≥ cutoff value 19) were significantly shorter than those with low levels of GGT5 expression (< cutoff value 19) (Figure 1E, 1F). Taken together, these evidences implicate an aggressive role of GGT5 in LUAD progression.
GGT5 is specifically expressed in CAFs, but not in tumor cells
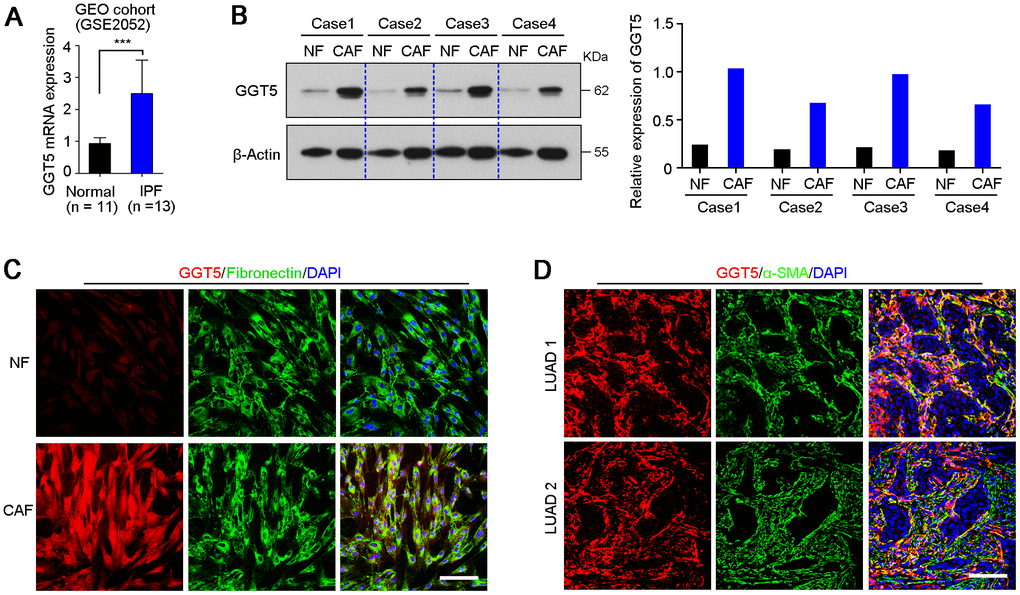
GEO public cohort analysis (GSE2052) indicated that gene GGT5 was high expressed in lung tissues from patients with idiopathic pulmonary fibrosis, a lethal disorder characterized by the aberrant accumulation of fibroblasts and myofibroblasts [21], than that in normal lung tissues (Figure 2A). Moreover, our previous data of IHC staining also showed that GGT5-positive cells was specifically deposited in tumor stroma in clinical LUAD tissue sections (Figure 1B). These evidences suggest that protein GGT5 may be expressed by CAFs in LUAD microenvironment. To confirm the hypothesis, the expression levels of GGT5 in primary normal lung fibroblasts (NFs) and paired CAFs from patients with LUAD were tested by western blotting (Figure 2B) and IF staining (Figure 2C). The results displayed the increased protein expression of GGT5 in CAFs that that in corresponding NFs (Figure 2B, 2C). In addition, IF double-staining with antibodies against GGT5 and α-SMA (a marker of CAFs) in LUAD tissues showed that GGT5 was specifically expressed in CAFs, but not in tumor cells (Figure 2D).
Interfering the expression of GGT5 in CAFs inhibits LUAD cell growth in vitro
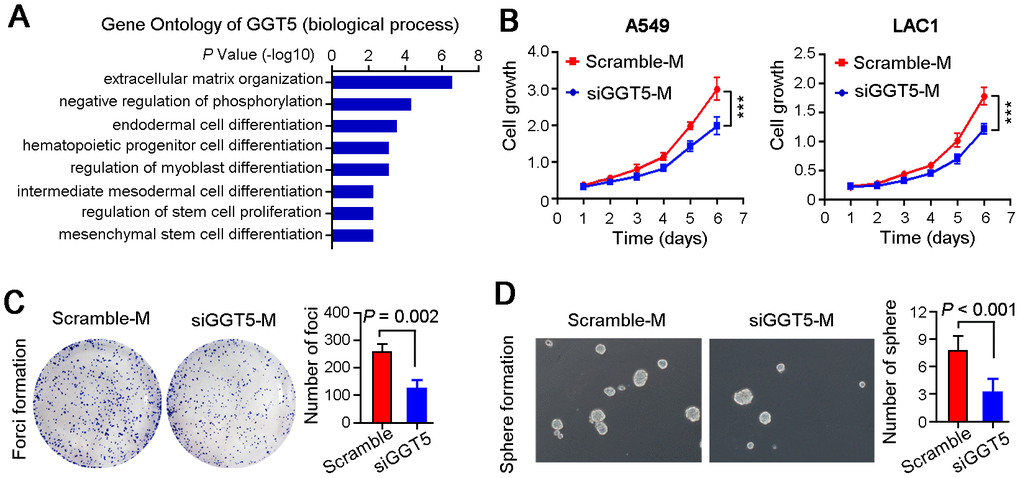
To investigate the role of GGT5 in LUAD progression, Gene Ontology Enrichment analysis (biological process) was performed using Coexpedia. Analysis results showed that GGT5 was significantly related to cell differentiation and cell cycle regulation, hinting that GGT5 might mediate the crosstalk between CAFs and tumor cells (Figure 3A). Hence, we analyzed the cell growth rate of two LUAD cell lines A549 and LAC1 treated with conditioned media from primary CAFs transfected with scramble (Scramble-M) or siRNA targeting GGT5 (siGGT5-M). First, the qPCR was used to confirm the knockdown of GGT5 by siRNA at mRNA level (Supplementary Figure 2A). Results indicated that down-regulation of GGT5 in CAFs attenuated the growth of tumor cell in vitro (Figure 3B). Moreover, foci formation in monolayer culture (Figure 3C) and spheres formation in soft agar (Figure 3D) analyses also suggested that interfering GGT5 expression in CAFs could inhibit tumor cell proliferation in vitro.
High expression of GGT5 in CAFs enhances the drug resistance of LUAD cells
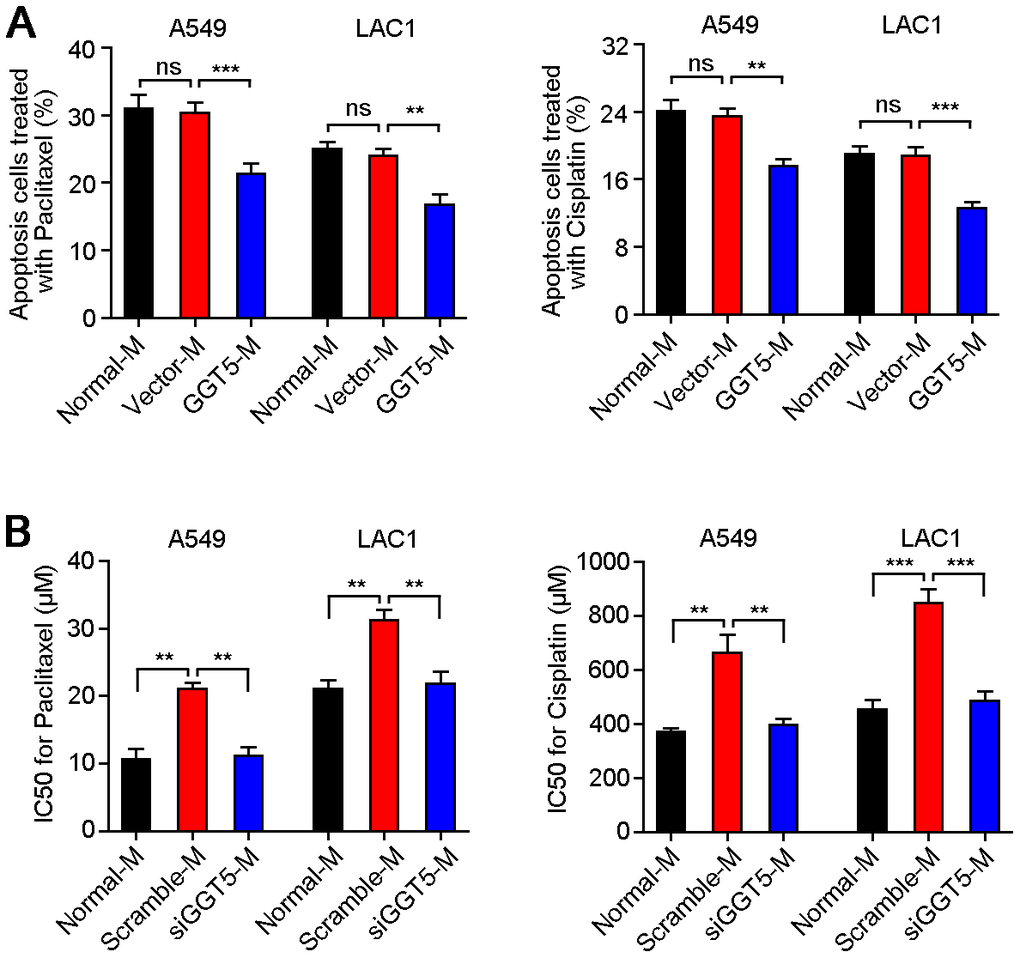
Since it has been proved that GGT1 as a member of GGT family was involved in drug resistance [22], but role of GGT5 during cancer chemotherapy is unclear. Therefore, exogenous overexpression of GGT5 in NFs was established (Supplementary Figure 2B), and the conditioned media from NFs transfected with vector (Vector-M) or GGT5 (GGT5-M) were used to culture A549 and LAC1 cells, respectively. Meanwhile, LUAD cells were treated with chemotherapeutic drugs Paclitaxel (5 μM) or Cisplatin (200 μM). Flow cytometry tests indicated that increased expression of GGT5 in NFs reduced the apoptotic rate of LUAD cells (Figure 4A). In addition, the chemosensitivity of LUAD cells regulated by CAFs with or without interference of GGT5 was also explored in vitro. Results showed that down-regulation of GGT5 in CAFs impaired the chemoresistance of LUAD cells (Figure 4B).
Increased GGT5 in CAFs reduces the ROS level in LUAD cells
To explore the mechanism of GGT5 in regulation of LUAD progression, we first tested the GGT activity of NFs and CAFs. Results showed that CAFs maintained the high activity of GGT than that in NFs (Figure 5A). GGT1 as a member of GGT family has been reported to promote lung cancer progression [23]. However, our IF staining showed that the expression of GGT1 in NFs and CAFs was negative (Supplementary Figure 3A). Next, we measured the GGT activity in NFs transfected with vector or GGT5 (Supplementary Figure 3B) and CAFs with or without siGGT5 (Supplementary Figure 3C). Results showed that GGT activity was increased in NFs after GGT5 overexpression and decreased in CAFs when GGT5 was silenced (Supplementary Figure 3B, 3C). Moreover, the increased GGT activity in NFs after GGT5 overexpression was inhibited by a GGT inhibitor GGsTop (Supplementary Figure 3D) [24]. These data suggested that up-regulated GGT5 was responsible for the high GGT activity of CAFs. It has been reported that GGT5 was capable of cleaving the gamma-glutamyl moiety of GSH [23]. Hence, GSH level in fibroblasts or conditioned media (CAFs-M and CAFs-M) were analyzed. CAFs synthesize more intracellular GSH compared to NFs, but less extracellular of GSH deposits in CAFs-M than that in CAFs-M, which may be due to that extracellular of GSH is cleaved into cysteine and glutamate by GGT5 (Figure 5B). Therefore, we next tested the extracellular cysteine or glutamate in NFs-M and CAFs-M, respectively. High levels of cysteine and glutamate in CAFs-M than that in NFs-M were confirmed (Figure 5C).
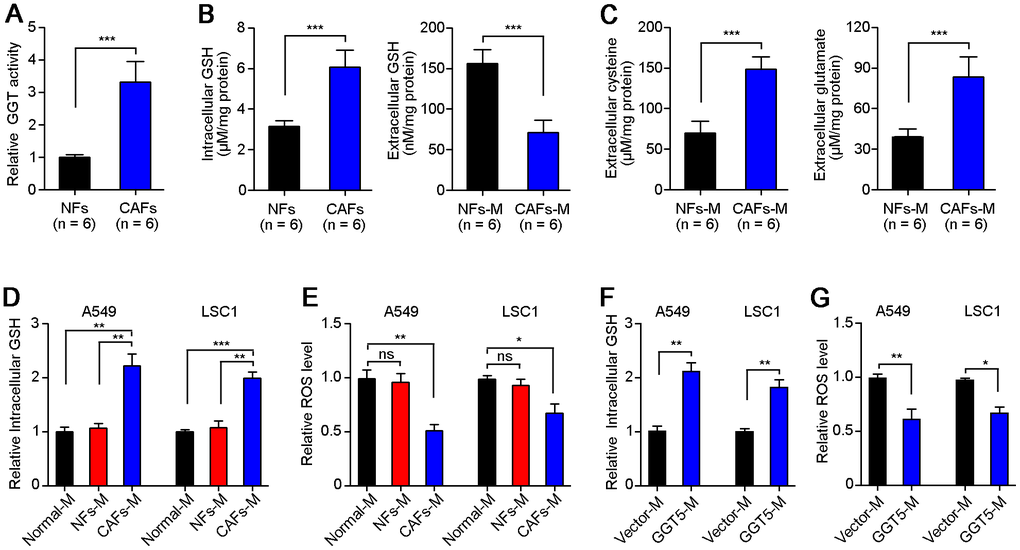
Next, we further analyzed the intracellular GSH (Figure 5D) and ROS (Figure 5E) in A549 and LAC1 cells treated with different cultures: Normal-M, NFs-M or CAFs-M. Unsurprisingly, CAFs-M treatment significantly increased the intracellular GSH in A549 and LAC1 cells (Figure 5D), and the intracellular ROS in LUAD cells was reduced after culture with CAFs-M, compared with NFs-M or Normal-M (Figure 5E). Most importantly, overexpression of GGT5 in NFs could also induce the high level of intracellular GSH and less intracellular ROS in A549 and LAC1 cells (Figure 5F). These in vitro data suggest that CAFs-derived GGT5 promotes the transfer of GSH into LUAD cells from tumor microenvironment, which further reduces the intracellular ROS level in LUAD cells.
Targeting GGT5 enhances the chemosensitivity of LUAD cells in vivo
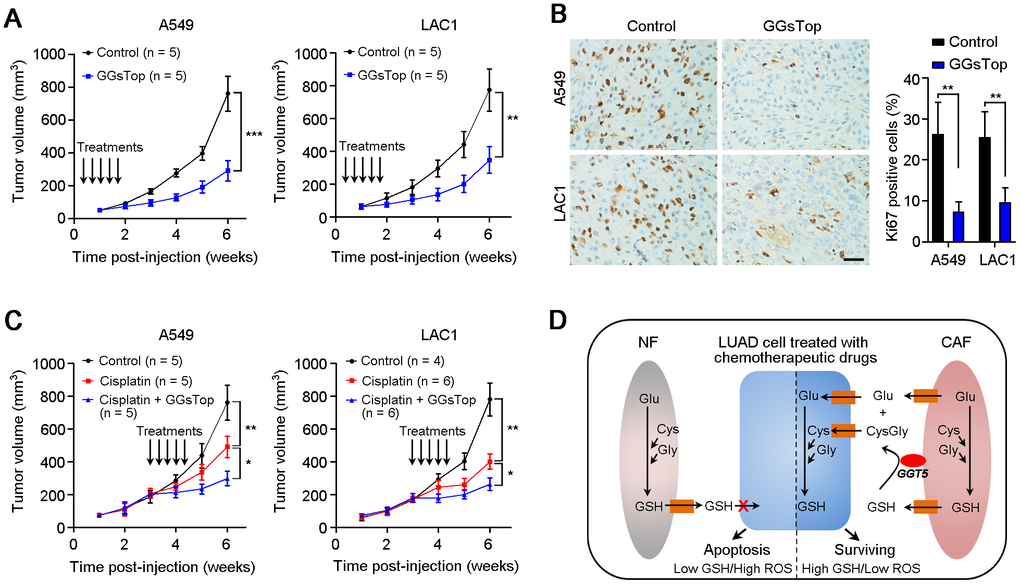
All the above data indicate that GGT5 may be a promising therapeutic target in LUAD. Hence, we further explored the influence of GGT5 blocking with GGsTop on tumor growth and chemosensitivity in mouse xenograft model. First, inhibiting GGT5 activity with GGsTop (i.p. injection, 1 mg/kg) markedly suppresses the tumor growth in nude mice xenograft model in A549 and LAC1 cells and does not affect the body weight of mice (Figure 6A, Supplementary Figure 4). Moreover, IHC staining with antibody against Ki67 (a marker of proliferative cell) in tissue sections from transplantation tumors showed that treatment with GGsTop inhibited the proliferation of A549 and LAC1 cells (Figure 6B). Furthermore, co-administration of Cisplatin (i.p. injection, 6 mg/kg) and GGsTop (i.p. injection, 1 mg/kg) significantly inhibited the tumor growth (Figure 6C). Taken together, our study illuminates that high level of GGT5 in CAFs contributes to tumor cell survival and drug resistance by increasing intracellular GSH and reducing the intracellular ROS level in LUAD cells.
Discussion
In this study, we analyzed the expression of GGT5 in LUAD and normal lung tissues for the first time, and found that GGT5 was highly expressed in LUAD, which predicted the poor prognosis of patients with LUAD. Interestingly, our study clearly confirmed that GGT5 specially expressed in CAFs, but not in tumor cells, which contributed to tumor cell proliferation and drug resistance by increasing intracellular GSH and reducing the intracellular ROS level in LUAD cells. Therefore, our data revealed a novel crosstalk mechanism mediated by GGT5 between tumor cells and CAFs during cancer progression and treatments.
Recently, accumulating evidences suggested that elevated serum GGT was involved in tumorigenesis and progression, such as gastric cancer [25], colorectal cancer [26], breast cancer [10] and cervical cancer [27]. However, the expression of GGT5 in primary tumor tissues has not been reported. Hence, based on the analysis of TCGA database and GEO public cohorts, we found the high expression of GGT5 in LUAD tissues than that in normal lung tissues. Moreover, high GGT5 in LUAD tissues predicts the adverse overall survival and progression-free survival for LUAD patients. These findings suggest the aggressive role of GGT5 in LUAD progression, but the function and mechanism are not clear. One recent study showed that GGT5 highly expressed in follicular dendritic cells promoting B cell confinement to germinal centers in lymphoid tissues [28]. Our further study proved that CAFs in LUAD microenvironment were GGT5-positive, which indicated that GGT5 might mediate the crosstalk between tumor cells and CAFs. Therefore, the conditioned medium from CAFs with siGTT5 was used to treat LUAD cell in vitro, and results showed that knockdown of GGT5 in CAFs could inhibit the growth of LUAD cells. These data suggest that CAFs remold the tumor microenvironment promoting tumor cell growth.
It has been proved that GGT was involved in drug resistance in tumor progression [29, 30]. Herein, we also test the regulation of CAFs-derived GGT5 on the chemosensitivity of LUAD cells. Results showed that down-regulation of GGT5 in CAFs impaired the chemoresistance of LUAD cells. These data indicate that GGT5 may be a potential therapeutic target for improving the treatment of LUAD patients. Oxidative stress is implicated in cancer development and progression, and increased serum GGT is associated with the high risk of cancers [25–27]. Anti-oxidation properties of GGT may underlie etiological mechanisms leading to these adverse outcomes for LUAD patients. Therefore, we firstly tested the intracellular and extracellular GSH for NFs and CAFs. Elevated intracellular GSH in CAFs and low level of GSH in CAFs-M were observed. Knockdown of GGT5 decreases the levels of cysteine and glutamate in CAFs-derived conditioned media. These substrates are necessary for synthesis of GSH. It is known that GSH as a vital regulator of the cellular redox state is involved in the proliferation and chemoresistance of cancer cells [31, 32]. Therefore, when the conditioned media from GGT5-silenced CAFs was used to treat LUAD cells, less cysteine and glutamate were provided for GSH synthesis in cancer cells, which accounted for the slower cell growth and higher drug-sensitivity of cancer cells after treatment the cultures. In tumor microenvironment, high levels of cysteine and glutamate in tumor microenvironment indicated that extracellular GSH was cleaved by CAFs-derived GGT5. These substrates will be further transported into tumor cells for GSH synthesis. Therefore, CAFs-derived GGT5 promotes cell survival and drug resistance by reducing the intracellular ROS level in LUAD cells (Figure 6D).
CAFs as a main component of tumor microenvironment have been proved to promote tumor progression, including proliferation promotion, angiogenesis, immunosuppression and drug resistance [32–34]. Therefore, targeting CAFs may be promising therapeutic strategy for cancer patients [35]. In this study, we confirmed that blocking GGT5 with a small-molecule inhibitor GGsTop could inhibit tumor growth and increase the drug-sensitivity of LUAD cells in mouse xenograft tumor model. In conclusion, our clinical data analysis and in vitro/in vivo functional studies demonstrated the aggressive role of GGT5 in LUAD, providing a novel diagnostic or therapeutic target for LUAD patients.
Materials and Methods
LUAD clinical samples and cell lines
Primary LUAD specimens and their corresponding non-tumor tissues were obtained with informed consent from patients at the Sun Yat-sen University Cancer Center (Guangzhou, China). All samples used in this study were approved by the Committees for Ethical Review of research involving human subjects at the Sun Yat-sen University Cancer Center (Guangzhou, China). LUAD cell line A549 was purchased from the American Type Culture Collection (ATCC, Manassas, Virginia, USA). The other LUAD cell line LAC1 (also known as ACC212102) were established in our laboratory [36, 37]. All cell lines were cultured in high-glucose DMEM (Gibco BRL, Grand Island, NY) supplemented with 10% fetal bovine serum (FBS, Gibco BRL, Grand Island, NY) at 37°C with 5% CO2.
Immunohistochemistry (IHC) and immunofluorescence (IF) staining
In brief, tissue slides with paraffin sections were deparaffinized by xylene, rehydrated using graded ethanol and rinsed with deionized water. Endogenous peroxidase activity was blocked with 3% hydrogen peroxide for 10 min at room temperature. For antigen retrieval, slides were high-pressure-treated and boiled in citrate buffer (pH 6.0, 10mM) for 15 min. Nonspecific binding was blocked with 5% normal goat serum for 30 min at room temperature. The slides were incubated with monoclonal antibodies against GGT5 (Santa Cruz Biotechnology, #sc-20640, 1:500 dilution), Ki67 (Abcam, #ab15580, 1:400 dilution), Fibronectin (Cell Signaling Technology, #26836, 1:200 dilution) or α-SMA (Cell Signaling Technology, #56856, 1:200 dilution) at 4°C overnight in a humidified chamber. For IHC staining, the slides were incubated with horseradish peroxidase-conjugated secondary antibody and stained with the DAB substrate. The representative figures of IHC staining were captured with light microscope (Olympus, Lake Success, NY). An immune-reactivity score system was applied as described previously [38]. The percentage of GGT5-positive cells was scored as 0, <5%; 1, 5%-25%; 2, 25%-50%; 3, 50%-75%; 4, 75%-100%. The intensity of GGT5-positive staining was scored as 0, negative; 1, weak; 2, moderate; 3, strong. The total score was determined by the following formula: Staining index = positive percentage × intensity. For IF staining, the slides were incubation with Alexa Fluor 594 or 488-conjugated secondary antibodies (Invitrogen). The cells on coverslips were counterstained with DAPI (Invitrogen) and imaged using a confocal laser-scanning microscope (Olympus, Lake Success, NY).
Isolation of fibroblasts
Primary NFs or CAFs were established in our laboratory after tumor resection. In detail, fresh LUAD tissue and paired non-tumorous lung tissue were cut into as small pieces as possible by surgical scissors in sterile culture dish, respectively. And then the tissue homogenate was transferred into sterile tube supplemented with 1ml Liberase (0.2 mg/ml, Roche, #5401119001) for enzymolysis at 37°C for 30 min. The suspension was filtered through a sterile Cell Strainer (40 μm, Corning) to collect a single-cell suspension. The filtrate was centrifuged at 1,500 rpm for 5 min at 4°C and washed twice with pure DMEM. The cell pellet was resuspended with 3 ml DMEM supplemented with 20% FBS and finally plated on 6-cm cell culture dishes. Based on the different adhesive capacity of fibroblasts and other cells, the unadherent cells (mainly tumor cells) were removed after culturing for 30 min at 37°C to obtain relative pure fibroblasts, because the adhesion time of fibroblasts is much shorter (~20-30 min) than tumor cells (usually > 1 h). Next, when the cell density reaches ~80% dish surface area, fibroblasts are further purified by short time digestion of trypsin (~30 s). The digested cells were cultured for further isolation of fibroblasts again. Finally, the pure fibroblasts are confirmed by immunostaining with antibody against Fibronectin (Cell Signaling Technology, #26836, 1:200 dilution).
Construction of GGT5 overexpression and knockdown fibroblasts
To study the function of GGT5 in NFs or CAFs, full-length human GGT5 complementary DNA was amplified by reverse transcription-PCR and was cloned into pLenti6.3/V5 expression vector (Invitrogen, Carlsbad, CA). For GGT5 interference assay, one small interfering RNA specifically targeting GGT5 (siGGT5, 5'-CGGGGACCAGCTGGGCAGATGAG-3') and the scramble control siRNA was purchased from GeneCopoeia (Rockville, MD). GGT5 expression vector and control plasmids, siGGT5 and scramble control siRNA were transfected into fibroblasts using the HilyMax transfection reagent (Dojindo Corp., Japan) according to the manufacturer’s instructions, respectively. The expression level of GGT5 was confirmed by quantitative real-time PCR (qPCR) using FastStart Universal SYBR Green Master (Roche) and a 96-well Real-Time PCR Detection System (Roche). ACTB was used as the internal control. The primers used were as follows: GGT5: 5'-GTCAGCCTAGTCCTGCTGG-3' (forward) and 5'-GGATGGCTCGTCCAATATCCG-3' (reverse); ACTB: 5'-CATGTACGTTGCTATCCAGGC-3'′ (forward) and 5'-CTCCTTAATGTCACGCACGAT-3' (reverse). The relative expression level (defined as fold change) of the target gene (2-ΔΔCt) was normalized to the endogenous ACTB reference (ΔCt).
Foci formation assay
Briefly, 1×103 A549 cells were seeded in each well of a 6-well plate with fibroblasts-conditioned media. After a week of culture, surviving colonies (>50 cells per colony) were counted with crystal violet (Sigma) staining. Triplicate-independent experiments were performed.
Cell proliferation assay
The Cell Counting Kit-8 (CCK-8) assay kit (Dojindo Corp., Japan) was applied to measure the cell growth rate. A suspension of 1×103 A549 or LAC1 cells were planted in each well of a 96-well plate, in which 10 μl CCK-8 was added to 90 μl of culture medium. And after 2 h incubation at 37 °C, the absorbance was measured at 450 nm. Triplicate independent experiments were performed.
Western blot analysis
Human paired NFs and CAFs were lysed with RIPA lysis buffer (Cell Signaling Technology, Beverly, MA) supplemented with cocktail protease inhibitor (Roche). After boiling for 5 min, protein lysates were separated by 10% SDS-PAGE and then transferred to PVDF membranes (Millipore, Billerica, MA, USA). The membranes were blocked with 5% non-fat milk in TBS/Tween20 (TBST, 0.05% v/v) for 1 hour at room temperature, and incubated with primary antibodies against GGT5 (Santa Cruz Biotechnology, #sc-20640, 1:2000 dilution) and β-Tubulin (Cell Signaling Technology, #2146, 1:2000 dilution) overnight at 4 °C. The membranes were washed three times with TBST buffer for 5 min per time, and then incubated with HRP-conjugated secondary antibodies for 2 hours at room temperature. The bands were visualized with the enhanced chemiluminescence (Thermo Scientific, NY, USA), and the signals were quantified by ImageJ software (http://rsb.info.nih.gov/ij).
GSH content and GGT activity assay
The GSH-Glo™ Glutathione Assay Kit (#V6912, Promega) was used to determine the reduced GSH content in the different medium samples. γ-Glutamyltransferase Activity Fluorometric Assay Kit (#MAK090-1KT, Sigma) was used to determine the GGT activity in fibroblasts or tumor cells. Experiments were performed with biological replicates.
In vivo xenograft assay
All animal experiments were approved by Animal Ethics Committee at Sun Yat-sen University Cancer Center. Five-week-old female BALB/c nude mice were purchased from the Guangdong Medical Laboratory Animal Center (Guangzhou China). Different numbers of LUAD cells (A549: 2×106 cells; LAC1: 4×106 cells) in 100μl phosphate buffered saline (PBS) were injected subcutaneously into nude mice. After tumor cells was injected, GGsTop (1 mg/kg, APExBIO, #B5602) or Cisplatin (6 mg/kg, Selleck, #S1166) was administered intraperitoneally every two days for total five times. The length (L) and width (W) of tumor were measured every week by calipers, and tumor volumes were calculated as volume (mm3) = L × W2 × 0.5.
Statistical analysis
SPSS version 17.0 (Chicago, IL) was used for all data analyses. An independent Student t test was used for continuous data. The mRNA expression of GGT5 in LUAD and normal lung tissues were obtained from TCGA database via a web server UALCAN (http://ualcan.path.uab.edu/analysis.html). Survival curves were obtained from Kaplan-Meier Plotter (http://kmplot.com/analysis/). Disease Ontology and Gene Ontology analyses were based on Coexpedia (http://www.coexpedia.org/). Results were considered statistically significant when P < 0.05.
Supplementary Materials
Author Contributions
Jia-Ru Wei: acquisition and analysis of data, and drafting of the manuscript; Jun Dong: material support and study design; Lei Li: study design and supervision.
Funding
This work was supported by grants from the National Science Foundation for Distinguished Young Scholars of China (81601581 and 81600606), the Basic and Applied Basic Research Foundation of Guangdong Province (2019A1515110660) and the National Science Foundation of Guangdong Province (2017A030313764).
Conflicts of Interest
The authors have no conflicts of interest to declare.
References
- 1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019; 69:7–34. https://doi.org/10.3322/caac.21551 [PubMed]
- 2. Bray F, Ferlay J, Soerjomataram I, Siegel RL, Torre LA, Jemal A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin. 2018; 68:394–424. https://doi.org/10.3322/caac.21492 [PubMed]
- 3. Zhou B, Wang GZ, Wen ZS, Zhou YC, Huang YC, Chen Y, Zhou GB. Somatic mutations and splicing variants of focal adhesion kinase in non-small cell lung cancer. J Natl Cancer Inst. 2018; 110:195–204. https://doi.org/10.1093/jnci/djx157 [PubMed]
- 4. Li L, Liu YD, Zhan YT, Zhu YH, Li Y, Xie D, Guan XY. High levels of CCL2 or CCL4 in the tumor microenvironment predict unfavorable survival in lung adenocarcinoma. Thorac Cancer. 2018; 9:775–84. https://doi.org/10.1111/1759-7714.12643 [PubMed]
- 5. Corti A, Dominici S, Piaggi S, Belcastro E, Chiu M, Taurino G, Pacini S, Bussolati O, Pompella A. Γ-glutamyltransferase enzyme activity of cancer cells modulates l-γ-glutamyl-p-nitroanilide (GPNA) cytotoxicity. Sci Rep. 2019; 9:891. https://doi.org/10.1038/s41598-018-37385-x [PubMed]
- 6. Yu Y, Fan Y, Yang Z, Lu Y, Xu Q, Chen X. Elevated serum gamma-glutamyltransferase predicts advanced histological liver damage in chronic hepatitis B. Discov Med. 2016; 21:7–14. [PubMed]
- 7. Mok Y, Son DK, Yun YD, Jee SH, Samet JM. Γ-glutamyltransferase and cancer risk: the korean cancer prevention study. Int J Cancer. 2016; 138:311–19. https://doi.org/10.1002/ijc.29659 [PubMed]
- 8. Wei D, Chen T, Gao Y, Tian H. Serum gamma-glutamyltransferase and ferritin are related to insulin resistance: a population-based study. Clin Lab. 2015; 61:1157–61. https://doi.org/10.7754/clin.lab.2015.150227 [PubMed]
- 9. Xu XS, Miao RC, Zhang LQ, Wang RT, Qu K, Pang Q, Liu C. Model based on alkaline phosphatase and gamma-glutamyltransferase for gallbladder cancer prognosis. Asian Pac J Cancer Prev. 2015; 16:6255–59. https://doi.org/10.7314/apjcp.2015.16.15.6255 [PubMed]
- 10. Staudigl C, Concin N, Grimm C, Pfeiler G, Nehoda R, Singer CF, Polterauer S. Prognostic relevance of pretherapeutic gamma-glutamyltransferase in patients with primary metastatic breast cancer. PLoS One. 2015; 10:e0125317. https://doi.org/10.1371/journal.pone.0125317 [PubMed]
- 11. Grimm C, Hofstetter G, Aust S, Mutz-Dehbalaie I, Bruch M, Heinze G, Rahhal-Schupp J, Reinthaller A, Concin N, Polterauer S. Association of gamma-glutamyltransferase with severity of disease at diagnosis and prognosis of ovarian cancer. Br J Cancer. 2013; 109:610–14. https://doi.org/10.1038/bjc.2013.323 [PubMed]
- 12. Xiao Y, Yang H, Lu J, Li D, Xu C, Risch HA. Serum gamma-glutamyltransferase and the overall survival of metastatic pancreatic cancer. BMC Cancer. 2019; 19:1020. https://doi.org/10.1186/s12885-019-6250-8 [PubMed]
- 13. Edlinger M, Concin N, Concin H, Nagel G, Ulmer H, Göbel G. Lifestyle-related biomarkers and endometrial cancer survival: elevated gamma-glutamyltransferase as an important risk factor. Cancer Epidemiol. 2013; 37:156–61. https://doi.org/10.1016/j.canep.2012.12.003 [PubMed]
- 14. Hanigan MH, Gillies EM, Wickham S, Wakeham N, Wirsig-Wiechmann CR. Immunolabeling of gamma-glutamyl transferase 5 in normal human tissues reveals that expression and localization differ from gamma-glutamyl transferase 1. Histochem Cell Biol. 2015; 143:505–15. https://doi.org/10.1007/s00418-014-1295-x [PubMed]
- 15. Wickham S, Regan N, West MB, Kumar VP, Thai J, Li PK, Cook PF, Hanigan MH. Divergent effects of compounds on the hydrolysis and transpeptidation reactions of γ-glutamyl transpeptidase. J Enzyme Inhib Med Chem. 2012; 27:476–89. https://doi.org/10.3109/14756366.2011.597748 [PubMed]
- 16. Wickham S, West MB, Cook PF, Hanigan MH. Gamma-glutamyl compounds: substrate specificity of gamma-glutamyl transpeptidase enzymes. Anal Biochem. 2011; 414:208–14. https://doi.org/10.1016/j.ab.2011.03.026 [PubMed]
- 17. Morris G, Anderson G, Dean O, Berk M, Galecki P, Martin-Subero M, Maes M. The glutathione system: a new drug target in neuroimmune disorders. Mol Neurobiol. 2014; 50:1059–84. https://doi.org/10.1007/s12035-014-8705-x [PubMed]
- 18. Carter BZ, Wiseman AL, Orkiszewski R, Ballard KD, Ou CN, Lieberman MW. Metabolism of leukotriene C4 in gamma-glutamyl transpeptidase-deficient mice. J Biol Chem. 1997; 272:12305–10. https://doi.org/10.1074/jbc.272.19.12305 [PubMed]
- 19. Shi ZZ, Han B, Habib GM, Matzuk MM, Lieberman MW. Disruption of gamma-glutamyl leukotrienase results in disruption of leukotriene D(4) synthesis in vivo and attenuation of the acute inflammatory response. Mol Cell Biol. 2001; 21:5389–95. https://doi.org/10.1128/MCB.21.16.5389-5395.2001 [PubMed]
- 20. Han B, Luo G, Shi ZZ, Barrios R, Atwood D, Liu W, Habib GM, Sifers RN, Corry DB, Lieberman MW. Gamma-glutamyl leukotrienase, a novel endothelial membrane protein, is specifically responsible for leukotriene D(4) formation in vivo. Am J Pathol. 2002; 161:481–90. https://doi.org/10.1016/s0002-9440(10)64204-6 [PubMed]
- 21. Pardo A, Selman M. Lung fibroblasts, aging, and idiopathic pulmonary fibrosis. Ann Am Thorac Soc. 2016 (Suppl 5); 13:S417–21. https://doi.org/10.1513/AnnalsATS.201605-341AW [PubMed]
- 22. Bansal A, Sanchez DJ, Nimgaonkar V, Sanchez D, Riscal R, Skuli N, Simon MC. Gamma-glutamyltransferase 1 promotes clear cell renal cell carcinoma initiation and progression. Mol Cancer Res. 2019; 17:1881–92. https://doi.org/10.1158/1541-7786.MCR-18-1204 [PubMed]
- 23. Lukic A, Ji J, Idborg H, Samuelsson B, Palmberg L, Gabrielsson S, Rådmark O. Pulmonary epithelial cancer cells and their exosomes metabolize myeloid cell-derived leukotriene C4 to leukotriene D4. J Lipid Res. 2016; 57:1659–69. https://doi.org/10.1194/jlr.M066910 [PubMed]
- 24. Shimamura Y, Takeuchi I, Terada H, Makino K. Therapeutic effect of GGsTop, selective gamma-glutamyl transpeptidase inhibitor, on a mouse model of 5-fluorouracil-induced oral mucositis. Anticancer Res. 2019; 39:201–06. https://doi.org/10.21873/anticanres.13098 [PubMed]
- 25. Wang Q, Shu X, Dong Y, Zhou J, Teng R, Shen J, Chen Y, Dong M, Zhang W, Huang Y, Xie S, Wei Q, Zhao W, et al. Tumor and serum gamma-glutamyl transpeptidase, new prognostic and molecular interpretation of an old biomarker in gastric cancer. Oncotarget. 2017; 8:36171–84. https://doi.org/10.18632/oncotarget.15609 [PubMed]
- 26. He WZ, Guo GF, Yin CX, Jiang C, Wang F, Qiu HJ, Chen XX, Rong RM, Zhang B, Xia LP. Gamma-glutamyl transpeptidase level is a novel adverse prognostic indicator in human metastatic colorectal cancer. Colorectal Dis. 2013; 15:e443–52. https://doi.org/10.1111/codi.12258 [PubMed]
- 27. Polterauer S, Hofstetter G, Grimm C, Rahhal J, Mailath-Pokorny M, Kohl M, Concin N, Tempfer C, Marth C, Reinthaller A. Relevance of gamma-glutamyltransferase—a marker for apoptotic balance—in predicting tumor stage and prognosis in cervical cancer. Gynecol Oncol. 2011; 122:590–94. https://doi.org/10.1016/j.ygyno.2011.05.027 [PubMed]
- 28. Lu E, Wolfreys FD, Muppidi JR, Xu Y, Cyster JG. S-geranylgeranyl-L-glutathione is a ligand for human B cell-confinement receptor P2RY8. Nature. 2019; 567:244–48. https://doi.org/10.1038/s41586-019-1003-z [PubMed]
- 29. Pompella A, Corti A, Paolicchi A, Giommarelli C, Zunino F. Gamma-glutamyltransferase, redox regulation and cancer drug resistance. Curr Opin Pharmacol. 2007; 7:360–66. https://doi.org/10.1016/j.coph.2007.04.004 [PubMed]
- 30. Corti A, Franzini M, Paolicchi A, Pompella A. Gamma-glutamyltransferase of cancer cells at the crossroads of tumor progression, drug resistance and drug targeting. Anticancer Res. 2010; 30:1169–81. [PubMed]
- 31. Park WH, Kim SH. MAPK inhibitors augment gallic acid-induced A549 lung cancer cell death through the enhancement of glutathione depletion. Oncol Rep. 2013; 30:513–19. https://doi.org/10.3892/or.2013.2447 [PubMed]
- 32. Wang W, Kryczek I, Dostál L, Lin H, Tan L, Zhao L, Lu F, Wei S, Maj T, Peng D, He G, Vatan L, Szeliga W, et al. Effector T cells abrogate stroma-mediated chemoresistance in ovarian cancer. Cell. 2016; 165:1092–105. https://doi.org/10.1016/j.cell.2016.04.009 [PubMed]
- 33. Ma J, Song X, Xu X, Mou Y. Cancer-associated fibroblasts promote the chemo-resistance in gastric cancer through secreting IL-11 targeting JAK/STAT3/Bcl2 pathway. Cancer Res Treat. 2019; 51:194–210. https://doi.org/10.4143/crt.2018.031 [PubMed]
- 34. Wang JW, Wu XF, Gu XJ, Jiang XH. Exosomal miR-1228 from cancer-associated fibroblasts promotes cell migration and invasion of osteosarcoma by directly targeting SCAI. Oncol Res. 2019; 27:979–86. https://doi.org/10.3727/096504018X15336368805108 [PubMed]
- 35. Chen X, Song E. Turning foes to friends: targeting cancer-associated fibroblasts. Nat Rev Drug Discov. 2019; 18:99–115. https://doi.org/10.1038/s41573-018-0004-1 [PubMed]
- 36. Li J, Yang H, Chen L, Li Y, Zhu Y, Dai Y, Chen K, Ai J, Zeng T, Mao X, Liu L, Li X, Guan XY. Establishment and characterization of human non-small cell lung cancer cell lines. Mol Med Rep. 2012; 5:114–17. https://doi.org/10.3892/mmr.2011.613 [PubMed]
- 37. Li L, Li JC, Yang H, Zhang X, Liu LL, Li Y, Zeng TT, Zhu YH, Li XD, Li Y, Xie D, Fu L, Guan XY. Expansion of cancer stem cell pool initiates lung cancer recurrence before angiogenesis. Proc Natl Acad Sci USA. 2018; 115:E8948–57. https://doi.org/10.1073/pnas.1806219115 [PubMed]
- 38. Li L, Zheng YL, Jiang C, Fang S, Zeng TT, Zhu YH, Li Y, Xie D, Guan XY. HN1L-mediated transcriptional axis AP-2γ/METTL13/TCF3-ZEB1 drives tumor growth and metastasis in hepatocellular carcinoma. Cell Death Differ. 2019; 26:2268–83. https://doi.org/10.1038/s41418-019-0301-1 [PubMed]


