Introduction
Breast cancer ranks first among female malignant tumors [1]. According to the cancer statistics of China, the incidence of breast cancer in China is increasing at a rate of 3–4 % per year, and the age of onset is also gradually becoming younger [2]. Based on gene expression profiles, breast cancer can be divided into four subtypes, luminal A, luminal B, HER2 positive, and basal-like/triple-negative breast cancers (TNBC) [3, 4]. Among all the subtypes of breast cancer, TNBC is more aggressive and has higher rates of relapse and metastasis than other subtypes. In the past decades, hormone therapy and anti-HER2 targeted therapy have significantly improved the prognosis of ERα/PR+ and HER2+ breast cancers, respectively. However, TNBC does not have effective targeted therapies for lacking the expression of ERα, PR and HER2, chemotherapy remains the major option for TNBC patients [5]. Therefore, it is important to develop effective treatments for TNBC.
Mitochondria are cellular organelles that engage in aerobic respiration. In addition to producing ATP, they are also involved in apoptosis. Mitochondria are the central organelle in the intrinsic apoptotic pathway, acting through the release of cytochrome C, Smac/DIABLO (direct IAP binding protein) and apoptosis-inducing factor AIF, while Bcl-2 can inhibit apoptosis by inhibiting the release of cytochrome C and preventing the translocation of Smac/DIABLO and the apoptosis-inducing factor AIF [6, 7].
The tetrahydro-β-carboline (THβC) skeleton as one important class of natural indole alkaloids commonly was found in nature. It is a very important skeleton and is present in many pharmacologically active alkaloids such as reserpine, tadalafil, and tetrahydro-halamine [8]. In recent years, tetrahydro-β-carboline derivatives have also become popular in terms of cancer targeted therapy. The TGF-β signaling pathway [9], phosphodiesterase (PDE) [10, 11], and spindle kinesin (KSP) [12, 13] are potential anti-tumor targets of tetrahydro-β-carboline derivatives. To develop more potent anticancer reagents, we designed and synthesized 42 pyrrolo [3,4-b]-quinolin-9-amines by using THβC as the starting point through an oxidative rearrangement coupling reaction.
There are no reports on the anti-tumor activity of pyrrolo [3,4-b]-quinolin-9-amine compounds to date. In this study, we screened anti-cancer activities of the 42 compounds and identified FZU-0038-056 to be the most potent one. FZU-0038-056 significantly induced apoptosis in HCC1806 and HCC1937. We showed that FZU-0038-056 reduced the protein expression levels of Bcl-2, Mcl-1 and XIAP. Bcl-2 overexpression partially reduced the pro-apoptosis of FZU-0038-056. These findings suggest that FZU-0038-056 is a mitochondrial apoptotic pathway activator and may be an effective anticancer agent for the treatment of TNBC.
Results
FZU-0038-056 is the most potent anti-cancer compound among the 42 tested tetrahydro-β-carboline derivatives
The pyrrolo [3,4-b]-quinolin-9-amine compound is a novel scaffold derived from tetrahydro-β-carboline derivatives [8], and its anti-tumor roles has not been reported yet. We treated two human TNBC cell lines (HCC1806 and HCC1937) with 42 pyrrolo [3,4-b]-quinolin-9-amine compounds (10 μM) for 48 hours and the cell viability was measured by SRB assays. Among the 42 compounds, FZU-0038-056 and FZU-0038-058 showed the most potent cytotoxicity against both cancer cell lines (Figure 1A). Then, we examined the half maximal inhibitory concentrations (IC50) of FZU-0038-056 and FZU-0038-058 in four TNBC cell lines (HCC1937, HCC1806, MDA-MB-231 and MDA-MB-468), one ERα positive breast cancer cell line (MCF-7) and the human immortalized breast epithelial cell line 184B5. Both FZU-0038-056 and FZU-0038-058 showed stronger cytotoxic effects in TNBC cells than in ERα positive and immortalized cells (Figure 1B). Because the structure of FZU-0038-058 is similar to FZU-0038-056 (Figure 1C), while its inhibitory effect is weaker than that of FZU-0038-056 in breast cancer cells (Figure 1B), we selected compound FZU-0038-056 for further in vitro and in vivo anticancer studies. The IC50s of FZU-0038-056 in the HCC1806 and HCC1937 cells were 3–6 μM (Figure 1B).
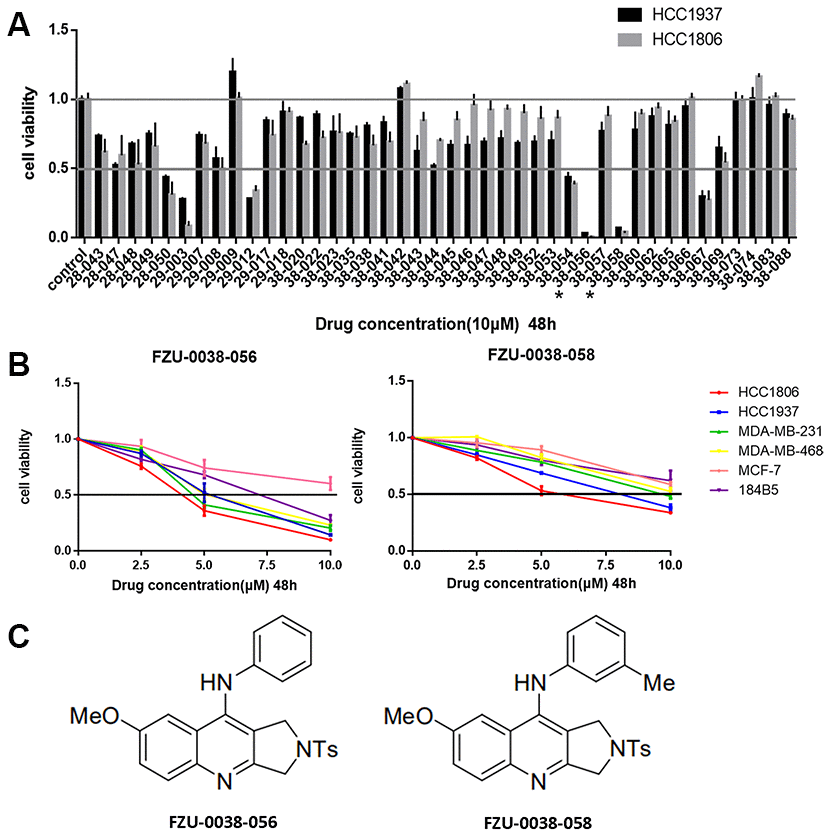
Figure 1. Identification of FZU-0038-056 as a potent anti-cancer compound in TNBC cell lines. (A) The HCC1806 and HCC1937 breast cancer cell lines were treated with 42 different compounds (10 μM) for 48 hours. DMSO was used as the negative control. Cell viability was measured using SRB assays. FZU-0038-056 and FZU-0038-058 labeled with an asterisk were selected for further studies. (B) Four different TNBC cell lines, two ER positive breast cancer cell lines and one human immortalized breast cell line were treated with DMSO control or FZU-0038-056/FZU-0038-058 at indicated concentrations (2.5, 5 and 10 μM, respectively) for 48 hours. Cell viability was measured using SRB assay. (C) The chemical structures of FZU-0038-056 and FZU-0038-058.
FZU-0038-056 induces apoptosis in HCC1806 and HCC1937 cells
Since both cell growth inhibition and apoptosis reduce cell viability, we investigated the effects of FZU-0038-056 on cell growth and apoptosis. After treating with FZU-0038-056 (10 μM) for 12 hours, HCC1806 and HCC1937 cells became round and detached (Figure 2A). To test whether FZU-0038-056 induced apoptosis, we further measured the apoptosis of HCC1806 and HCC1937 cells by Annexin V/PI staining using flow cytometry analysis. FZU-0038-056 (2.5–10 μM) induced apoptosis in both the HCC1806 and HCC1937 cell lines in dose-dependent manners (Figure 2B–2D). We also performed cell cycle analysis in HCC1806 and HCC1937 cells after FZU-0038-056 treatment. However, FZU-0038-056 did not affect cell cycle progression significantly in TNBC cells (Supplementary Figure 1).
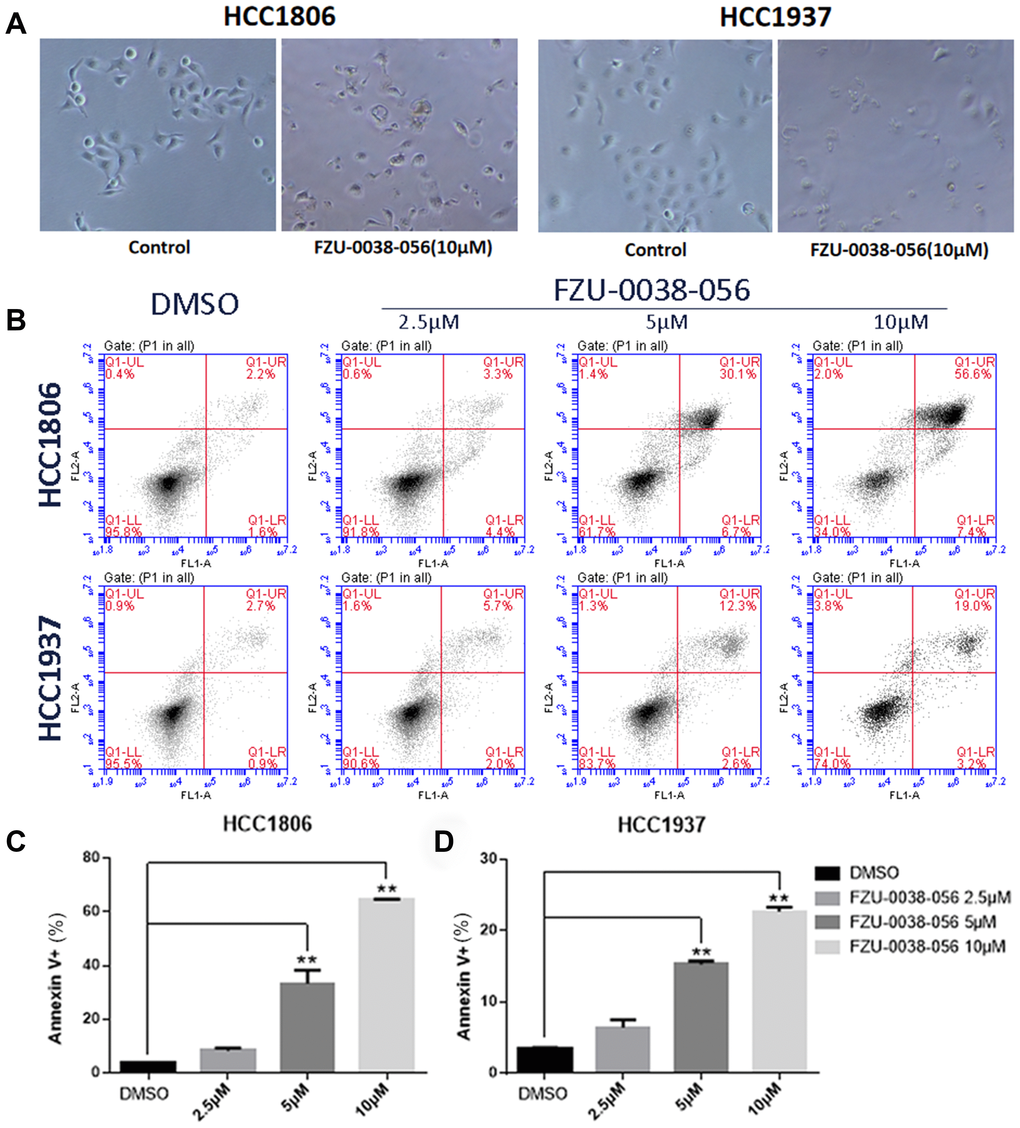
Figure 2. FZU-0038-056 induces apoptosis in HCC1806 and HCC1937 cells. (A) The cell morphology changes of HCC1806 and HCC1937 cells after the treatment of FZU-0038-056 (10μM) for 12 hours. (B) HCC1806 and HCC1937 cells were stained with Annexin V/PI and analyzed by flow cytometry analysis after the cells were treated with FZU-0038-056 (2.5, 5, 10 μM) for 24 hours. DMSO was added as the negative control. (C, D) The percentages of Annexin V-positive cells from panel B are shown. ** p < 0.01.
FZU-0038-056 does not induce TNBC apoptosis through activating p38
The p38 MAPK signaling pathway is well known to play important roles in various physiological processes, including apoptosis [14]. To test whether p38 activation leads to apoptosis, we knocked down p38 using two different siRNAs in HCC1806 and HCC1937 cells (Figure 4A, 4B). However, depletion of p38 did not attenuate the cell survival inhibition effects of FZU-0038-056 in either of the tested TNBC cell lines.
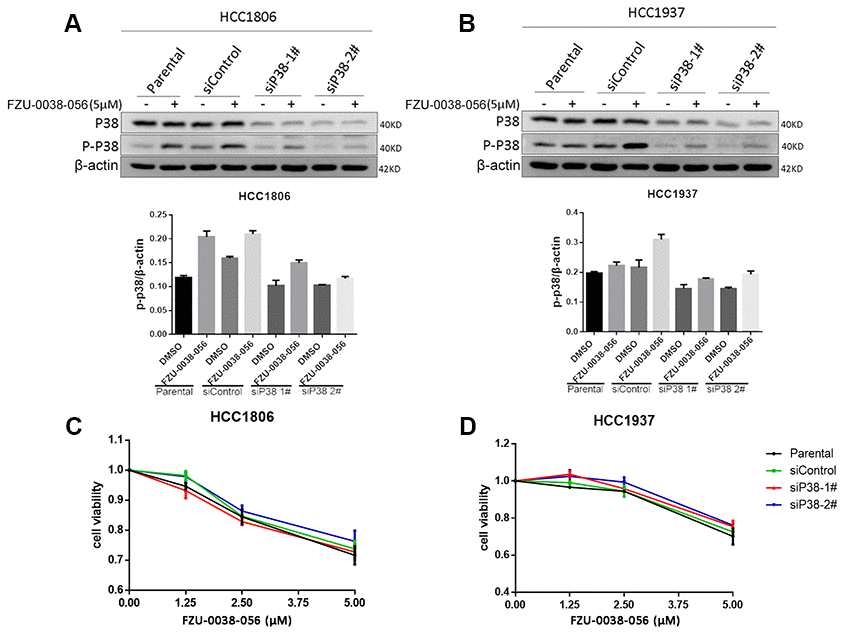
Figure 4. FZU-0038-056 induces TNBC apoptosis not through activating p38. (A, B) HCC1806 and HCC1937 cells were transfected with siRNAs targeting p38 for 36 hours, and treated with FZU-0038-056 (5 μM) or DMSO for 24 hours. Cell lysates were collected for WB assay. The quantification data of p-p38 protein expression was shown under the immunoblot images. (C, D) HCC1806 and HCC1937 cells were transfected with siRNA for 36 hours, then treated with FZU-0038-056 (1.25, 2.5, 5 μM) or DMSO for 48 hours. Cell viability was measured via the SRB assay.
FZU-0038-056 induces apoptosis partially through inhibiting the expression of Bcl-2
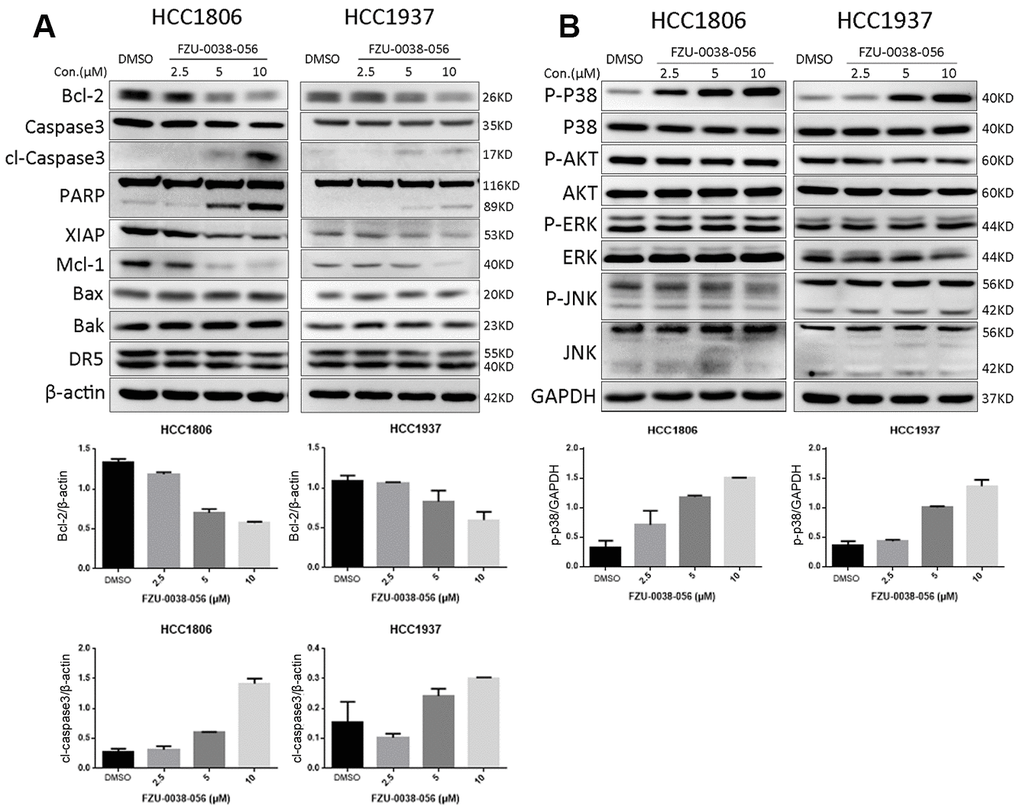
Since FZU-0038-056 downregulated the expression of Bcl-2, a key anti-apoptosis protein (Figure 3A), we further examined whether FZU-0038-056 facilitates apoptosis through Bcl-2. We overexpressed Bcl-2 according to published protocols [15] in HCC1806 cells (Figure 5A) and found that FZU-0038-056-induced cell viability loss was significantly rescued (Figure 5B). Moreover, we demonstrated that Bcl-2 overexpression significantly reduced FZU-0038-056-induced apoptosis, as measured by Annexin V staining (Figure 5C). Consistently, FZU-0038-056-induced cleavage of caspase-3 and PARP were also alleviated in Bcl-2 overexpressing HCC1806 cells (Figure 5D).
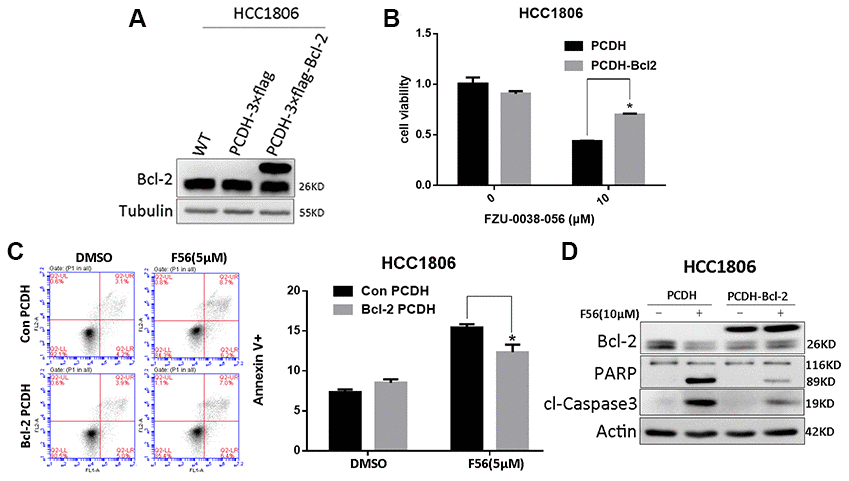
Figure 5. FZU-0038-056 induced apoptosis partially via inhibiting Bcl-2 in HCC1806 cells. (A) Bcl-2 was overexpressed in HCC1806 cells through a lentivirus system. (B) Overexpression of Bcl-2 reduced the cell viability inhibition triggered by FZU-0038-056. Cell viability was measured by the SRB assay. *p < 0.05. (C) FZU-0038-056-induced apoptosis were partially rescued by the overexpression of Bcl-2 compared with the control cells, as measured by Annexin V staining and flow cytometry analysis. Annexin V positive cell populations were quantified and shown on the right side. *p < 0.05. (D) FZU-0038-056-induced apoptosis were partially rescued by the overexpression of Bcl-2, as measured by Caspase-3 and PARP cleavage. The cells were treated with DMSO or FZU-0038-056 (10 μM) for 24 hours, and cell lysates were collected for WB analysis. β-actin was used as loading control.
FZU-0038-056 suppresses HCC1806 xenograft tumor growth in nude mice
To evaluate the antitumor activity of FZU-0038-056 in vivo, we established a HCC1806 xenograft model via subcutaneously injecting cancer cells into mammary fad pads of female nude mice. As the data show in Figure 6, tumor growth was significantly suppressed by FZU-0038-056 compared with the negative control. Compared with cisplatin (8mg/kg), FZU-0038-056 (15mg/kg) showed a similar anti-tumor effect but had less impact on the body weights of the mice (Figure 6A–6D).
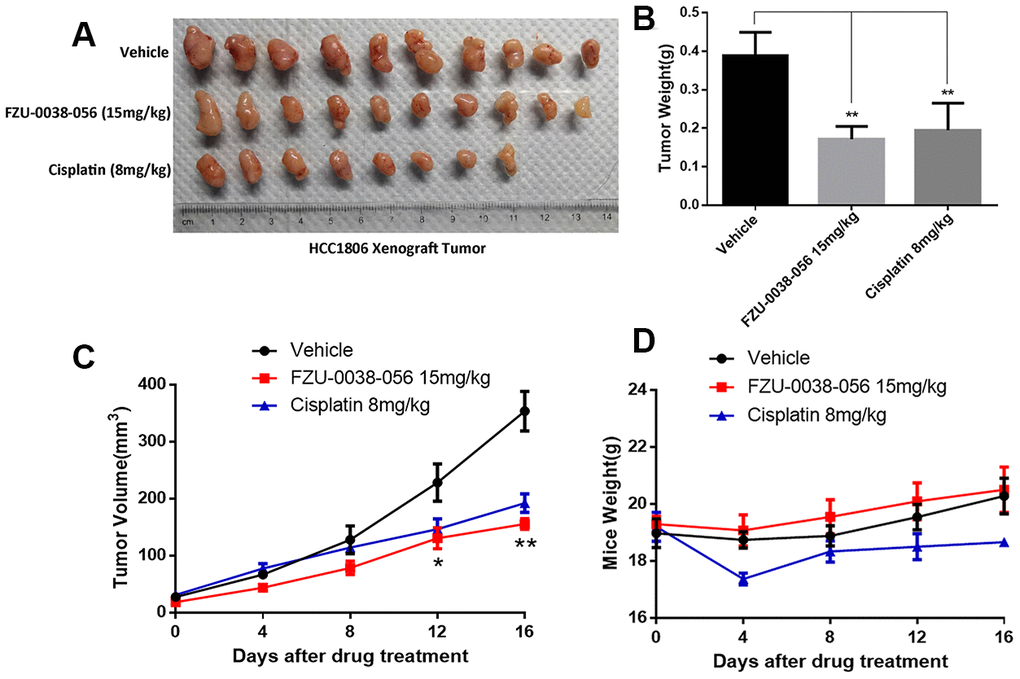
Figure 6. FZU-0038-056 suppresses HCC1806 xenograft tumor growth in nude mice. (A) FZU-0038-056 suppresses HCC1806 xenograft tumor growth in nude mice. Tumors were collected after the mice were treated with FZU-0038-056, vehicle, or cisplatin for 20 days. (B) FZU-0038-056 significantly reduced HCC1806 xenograft weights. Average tumor weights are graphed (n=5). **p < 0.01. (C) Tumor growth was significantly suppressed by FZU-0038-056 (15 mg/kg) and cisplatin (8 mg/kg) compared with vehicle control. **p < 0.01, *p < 0.05. (D) Compared to vehicle control, FZU-0038-056 (15 mg/kg) did not significantly reduce the mouse body weights although cisplatin (8 mg/kg) reduced the mouse body weights significantly.
Although FZU-0038-056 could efficiently kill cancer cells in our study, the dosage used is relatively high in vitro (Figure 1A, 1B) and in vivo (Figure 6A, 6C). Considering the side effects of cisplatin is severe, while FZU-0038-056 is much safer in vivo (Figure 6D), we tried to check whether FZU-0038-056 could strengthen the anti-tumor effect of cisplatin, thus cisplatin could be applied at lower dosages. As the data shown in Supplementary Figure 2, on one hand, cisplatin suppressed triple-negative breast cancer cell HCC1806 survival more efficiently when it was used together with FZU-0038-056. The IC50 of cisplatin was reduced from 6.5 μM to 2.7 μM (Supplementary Figure 2A, 2B) when 5 μM FZU-0038-056 was added. On the other hand, cisplatin also strengthened the anti-cancer activity of FZU-0038-056. The IC50 of FZU-0038-056 was reduced from 6.9 μM to 4.5 μM (Supplementary Figure 2C, 2D) when 5 μM cisplatin was added to treat HCC1806 cells. Taken together, our results show combination of FZU-0038-056 and cisplatin synergistically suppressed HCC1806 cell survival.
Discussion
In this study, we examined the cytotoxicity of 42 pyrrolo [3,4-b]-quinolin-9-amine compounds in two TNBC cell lines and selected the most potent anti-cancer compound FZU-0038-056 for further study. We demonstrated that FZU-0038-056 significantly inhibited TNBC cell survival. FZU-0038-056 induces apoptosis partially by inhibiting Bcl-2 expression. Finally, we demonstrated that FZU-0038-056 significantly suppressed HCC1806 xenograft tumor growth in vivo without affecting mouse body weight.
Apoptosis is one of the main mechanisms by which drugs inhibit tumor growth. There are three main pathways for apoptosis: the mitochondrial pathway, the death receptor pathway, and the endoplasmic reticulum pathway [16]. The mitochondrial programmed death pathway is a process in which multiple factors are interrelated and balance each other to promote cell death [17, 18]. When the apoptotic signal is transduced into mitochondria, the mitochondrial outer membrane forms poly channels, and mitochondrial contents such as cytochrome C (cytC), Smac and AIF are released [19]. Smac can relieve the inhibition of precursor caspase by IAPs. AIF induces fragmentation of DNA and concentration of chromosomes [19]. CytC can activate caspase-9 [20]. In this study, FZU-0038-056 downregulated the expression of Bcl-2 and Mcl-1 and induced the cleavage of caspase-3 and PARP (Figure 3). Meanwhile, the proapoptotic effect of FZU-0038-056 on TNBC cells can be significantly reduced by ectopically overexpressed Bcl-2 (Figure 5). Therefore, FZU-0038-056 activates the intrinsic mitochondrial apoptosis pathway partially by inhibiting the expression of Bcl-2. However, the exact mechanism by which FZU-0038-056 inhibits the expression of Bcl-2 is still unclear and requires further investigation. It would be interesting to identify direct binding molecules of FZU-0038-056.
Additionally, we found that FZU-0038-056 activated the p38 MAPK (Figures 3B and 4). However, when we depleted p38 expression using siRNAs, FZU-0038-056-induced apoptosis was not blocked (Figure 4), indicating that FZU-0038-056 does not induce apoptosis via activating the p38 pathway.
In summary, we demonstrated that FZU-0038-056 induces apoptosis of TNBC cells and inhibits TNBC tumor growth in nude mice. The mechanism by which FZU-0038-056 inhibits TNBC may involve the suppression of Bcl-2. Interestingly, we found combination application of FZU-0038-056 and cisplatin synergistically suppressed HCC1806 cell survival, which implicates possible application of FZU-0038-056 in combination with clinically used anti-tumor agents, including cisplatin. Therefore, FZU-0038-056 has potential as a novel anticancer agent for human TNBC.
Materials and Methods
Compounds and cell lines
FZU-0038-056 and other compounds were designed and synthesized from THβC as the lead compound [8]. FZU-0038-056 was dissolved in DMSO and diluted in the corresponding culture media for the experiments. All of the cell lines used in this study were purchased from the American Type Culture Collection (ATCC) and validated by STR analysis (Kunming Cell Bank, Kunming Institute of Zoology, Chinese Academy of Sciences). HCC1806 and HCC1937 were cultured in RPMI-1640 medium supplemented with 10 % fetal bovin serum (FBS). MDA-MB-231 and MDA-MB-468 were cultured in Dulbecco’s Modified Eagle’s Medium (DMEM) with 10 % FBS. MCF7 was cultured in Minimum Essential Medium (MEM) with 10 % FBS and 0.01 mg/ml human recombinant insulin. 184B5 cells were cultured in DMEM/F12 with 10 % FBS, 5 μg/ml insulin and 10 ng/ml cholera toxin. All cells were maintained at 37 °C with 5 % CO2 in a humidified atmosphere.
Antibodies
The anti- PARP, XIAP, Mcl-1, Bcl-2, Bax, Bak, DR5, p-JNK, JNK, p38, p-p38, ERK, p-ERK, AKT, p-AKT, and tubulin antibodies were purchased from Cell Signaling (Danvers, MA). The anti-caspase-3 and cleaved caspase-3 antibodies were purchased from Imagenex (San Diego, CA). The anti-β-actin antibody was purchased from Sigma (St. Louis, MO).
In vitro cytotoxicity assays
Normally cultured logarithmic growth phase cells were uniformly seeded at 30,000/well into 48-well plates. The day after plating, the cells were treated with the drugs at the indicated concentrations. Forty-eight hours later, the cells were fixed with 200 μl 10 % TCA (trichloroacetic acid) solution for 1 hour at room temperature, washed 5 times with deionized water, and then dried at room temperature. After drying, the cells were stained with 100 μl of 0.4 % (W/V) SRB in 1 % acetic acid for 5–15 min, followed by washing 5 times with 1 % glacial acetic acid and dried. Finally, 200 μl of 10 mM Tris base solution per well was added, and optical densities were measured at 530 nm in a spectrophotometric plate reader.
Apoptosis analysis
HCC1806 and HCC1937 were treated with different concentrations of FZU-0038-056 for 24 hours. DMSO was used as the negative control. The cells were stained with Annexin-V (BD, San Diego, CA) for 30 min and PI (BD, San Diego, CA) for 5 min in the dark at room temperature. Finally, the cells were analyzed by flow cytometry.
Western blotting (WB)
FZU-0038-056 or DMSO treated cancer cells were harvested using lysis buffer supplemented with a protease inhibitor cocktail (Roche Applied Science, Mannheim, Germany) for 30 min on ice. Proteins were collected and centrifuged at 4 °C, 13000 rpm for 10 min. Equal amounts of protein samples were then separated by SDS-PAGE electrophoresis and transferred to polyvinylidene fluoride (PVDF) membranes (Millipore, Bedford, MA). The membrane was blocked in 5 % skim milk for 1 hour, incubated with the primary antibody (1000 × dilution) overnight at 4 °C, washed 3 times with 1 × PBST for 10 min/time, and then incubated with secondary antibodies conjugated with horseradish peroxidase (HRP) (Jackson ImmunoResearch Laboratory, West Grove, PA) for 1 hour. They were then washed 3 times again with 1 × PBST. Finally, the membranes were incubated with Western Lighting Chemiluminescence Reagent Plus (PerkinElmer Life Sciences, Shelton, CT) and images were taken using an ImageQuant LAS4000 Biomolecular imager (GE Healthcare, UK).
Tumorigenicity assays
Eighteen six-week-old female nude mice were purchased from Hunan SJA Laboratory Animal Co., Ltd., (Changsha, Hunan, China) and were housed in an SPF animal facility. HCC1806 cells (1×106 cells per mouse, resuspended in 75 μl 1×PBS with 20 % Matrigel) were injected subcutaneously near the third mammary fat pad of the mice. When the tumor grew to 50 mm3, the mice were randomly divided into 3 groups (n=6). All mice were injected intraperitoneally (i.p.) with FZU-0038-056 solution or control every two days. The negative control group was treated with vehicle solution (5 % DMSO + 95 % saline), the experimental group was treated with FZU-0038-056 (15 mg/kg), and the positive control group was treated with cisplatin (8 mg/kg). The body weights and tumor volumes were measured every 4 days and the tumor volumes were calculated according to the formula V = 0.5 × L × W2, where L = length (mm) and W = width (mm). Animal studies were approved by the Institutional Ethics Committee of the Kunming Institute of Zoology, Chinese Academy of Sciences.
Statistical analysis
All data in this study were analyzed by SPSS 7.0 and are presented as the mean ± SD. Differences between groups were identified using Student’s t-test, and p<0.05 was considered statistically significant.
Supplementary Materials
Conflicts of Interest
These authors declare no conflicts of interest.
Funding
This study was supported in part by grants from Department of Science and Technology of Yunnan Province-Kunming Medical University (2019FE001(-063) to Wang, C and 2017FE467(-050) to Nie, Z), Yunnan Health Training Project of High Level Talents (D2018058 to Wang, C), the Project of Innovative Research Team of Yunnan Province (2019HC005 to Liu, R), National Key R&D Program of China (2018YFC2000400), the Shenzhen Municipal Government of China (KQTD20170810160226082), the National Nature Science Foundation of China (81830087, U1602221 and 31771516 to Chen, C, and 81772847 to Liu, R), the Natural Science Foundation of Fujian Province (2019J01202 to Chen, H).
References
- 1. Siegel RL, Miller KD, Jemal A. Cancer statistics, 2019. CA Cancer J Clin. 2019; 69:7–34. https://doi.org/10.3322/caac.21551 [PubMed]
- 2. Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F, Jemal A, Yu XQ, He J. Cancer statistics in China, 2015. CA Cancer J Clin. 2016; 66:115–32. https://doi.org/10.3322/caac.21338 [PubMed]
- 3. Perou CM, Sørlie T, Eisen MB, van de Rijn M, Jeffrey SS, Rees CA, Pollack JR, Ross DT, Johnsen H, Akslen LA, Fluge O, Pergamenschikov A, Williams C, et al. Molecular portraits of human breast tumours. Nature. 2000; 406:747–52. https://doi.org/10.1038/35021093 [PubMed]
- 4. Su Y, Zheng Y, Zheng W, Gu K, Chen Z, Li G, Cai Q, Lu W, Shu XO. Distinct distribution and prognostic significance of molecular subtypes of breast cancer in chinese women: a population-based cohort study. BMC Cancer. 2011; 11:292. https://doi.org/10.1186/1471-2407-11-292 [PubMed]
- 5. Fulford LG, Reis-Filho JS, Ryder K, Jones C, Gillett CE, Hanby A, Easton D, Lakhani SR. Basal-like grade III invasive ductal carcinoma of the breast: patterns of metastasis and long-term survival. Breast Cancer Res. 2007; 9:R4. https://doi.org/10.1186/bcr1636 [PubMed]
- 6. Lindsay J, Esposti MD, Gilmore AP. Bcl-2 proteins and Mitochondria—Specificity in membrane targeting for death. Biochim Biophys Acta. 2011; 1813:532–39. https://doi.org/10.1016/j.bbamcr.2010.10.017 [PubMed]
- 7. Goldstein JC, Waterhouse NJ, Juin P, Evan GI, Green DR. The coordinate release of cytochrome C during apoptosis is rapid, complete and kinetically invariant. Nat Cell Biol. 2000; 2:156–62. https://doi.org/10.1038/35004029 [PubMed]
- 8. Ye J, Wu J, Lv T, Wu G, Gao Y, Chen H. Oxidative rearrangement coupling reaction for the functionalization of tetrahydro-β-carbolines with aromatic amines. Angew Chem Int Ed Engl. 2017; 56:14968–72. https://doi.org/10.1002/anie.201708893 [PubMed]
- 9. Zheng C, Fang Y, Tong W, Li G, Wu H, Zhou W, Lin Q, Yang F, Yang Z, Wang P, Peng Y, Pang X, Yi Z, et al. Synthesis and biological evaluation of novel tetrahydro-β-carboline derivatives as antitumor growth and metastasis agents through inhibiting the transforming growth factor-β signaling pathway. J Med Chem. 2014; 57:600–12. https://doi.org/10.1021/jm401117t [PubMed]
- 10. Abdelwaly A, Salama I, Gomaa MS, Helal MA. Discovery of tetrahydro-ß-carboline derivatives as a new class of phosphodiesterase 4 inhibitors. Medicinal Chemistry Research. 2017; 26:3173–87. https://doi.org/10.1007/s00044-017-2011-x
- 11. Ahmed NS, Gary BD, Tinsley HN, Piazza GA, Laufer S, Abadi AH. Design, synthesis and structure-activity relationship of functionalized tetrahydro-β-carboline derivatives as novel PDE5 inhibitors. Arch Pharm (Weinheim). 2011; 344:149–57. https://doi.org/10.1002/ardp.201000236 [PubMed]
- 12. Ruan XQ, Chen M, Wu WT, You QD. [Synthesis and antitumor activity of novel tetrahydro-beta-carboline derivatives as KSP inhibitors]. Yao Xue Xue Bao. 2013; 48:1119–23. [PubMed]
- 13. Liu F, Yu LQ, Jiang C, Yang L, Wu WT, You QD. Discovery of tetrahydro-beta-carbolines as inhibitors of the mitotic kinesin KSP. Bioorg Med Chem. 2010; 18:4167–77. https://doi.org/10.1016/j.bmc.2010.05.024 [PubMed]
- 14. Kyriakis JM, Avruch J. MamMalian mitogen-activated protein kinase signal transduction pathways activated by stress and inflammation. Physiol Rev. 2001; 81:807–69. https://doi.org/10.1152/physrev.2001.81.2.807 [PubMed]
- 15. Rathmell JC, Thompson CB. Pathways of apoptosis in lymphocyte development, homeostasis, and disease. Cell. 2002; (Suppl 109):S97–107. https://doi.org/10.1016/s0092-8674(02)00704-3 [PubMed]
- 16. Brenner D, Mak TW. Mitochondrial cell death effectors. Curr Opin Cell Biol. 2009; 21:871–77. https://doi.org/10.1016/j.ceb.2009.09.004 [PubMed]
- 17. Chalah A, Khosravi-Far R. The mitochondrial death pathway. Adv Exp Med Biol. 2008; 615:25–45. https://doi.org/10.1007/978-1-4020-6554-5_3 [PubMed]
- 18. Doti N, Reuther C, Scognamiglio PL, Dolga AM, Plesnila N, Ruvo M, Culmsee C. Inhibition of the AIF/CypA complex protects against intrinsic death pathways induced by oxidative stress. Cell Death Dis. 2014; 5:e993. https://doi.org/10.1038/cddis.2013.518 [PubMed]
- 19. Pradelli LA, Bénéteau M, Ricci JE. Mitochondrial Control of Caspase-Dependent and -Independent Cell Death. Cell Mol Life Sci. 2010; 67:1589–97. https://doi.org/10.1007/s00018-010-0285-y [PubMed]
- 20. Zhou W, Fang H, Wu Q, Wang X, Liu R, Li F, Xiao J, Yuan L, Zhou Z, Ma J, Wang L, Zhao W, You H, et al. Ilamycin E, a natural product of marine actinomycete, inhibits triple-negative breast cancer partially through ER stress-CHOP-Bcl-2. Int J Biol Sci. 2019; 15:1723–32. https://doi.org/10.7150/ijbs.35284 [PubMed]