Introduction
Patterns of energy consumption are a powerful regulator of mortality and longevity. In addition to caloric restriction, which has been extensively shown to extend maximum lifespan across species [1–7], time restricted feeding (TRF; also referred to as meal feeding or intermittent fasting) may also improve morbidity and mortality rates [8]. Intermittent periods of eating inevitably lead to episodes of ketogenesis, in which fat is oxidized into ketone bodies, reducing the need for glycolysis [8–10].
Without fasting, ketosis can also be achieved with high fat, low carbohydrate ketogenic diets (KD). Importantly, long-term ketosis has also been shown to prevent cognitive and physical declines associated with aging [11–18]. It remains to be determined, however, whether TRF alone can produce sufficient levels of ketosis to elicit similar health benefits and the extent to which TRF with a KD may have additive benefits on the healthspan of older adults and other animals.
The process of switching from glycolysis to a reliance on fat-derived ketone bodies as a primary energy source is termed keto-adaptation [19, 20]. While nutritional ketosis has been proposed as an intervention for a number of age-related metabolic and neurological diseases [21–23], aged animals take longer than young to produce high levels of circulating ketone bodies [14, 15]. Understanding the factors responsible for delayed keto-adaptation in old animals, and how to overcome this, are critical as brain utilization of ketone bodies directly correlates with circulating levels in the periphery [24–26]. Thus, in order to design precision ketogenic diet therapy [27] that can improve cognitive functioning and physical health in older adults, it is imperative to understand ketogenesis across the lifespan and the metabolic variables that can delay or promote metabolic switching.
The prevailing theory regarding the initiation of ketogenesis is that ketone body production is triggered once the body depletes its glycogen stores. This glycogen depletion hypothesis likely stems from the body’s natural metabolic response to vigorous exercise or longer fasts [28, 29]. Following 1 hour of exercise, there is an inverse relationship between glycogen content and ketone body levels [30]. Given the presumed relationship between keto-adaptation and glycogen levels, it is conceivable that aged rats take longer to achieve stable nutritional ketosis due to higher levels of glycogen or altered glycogen dynamics. There are differences in body mass with age [15], as well as evidence that aged rats have reduced glycogenolytic capacity [31]. Specifically, the hepatic release of glycogen [32], which increases blood glucose in response to epinephrine [33], is decreased in aged compared to young rats [34]. Thus, old animals may be deficient at converting glycogen to glucose on demand, which would decrease the rate of glycogen depletion. An alternative to the glycogen depletion hypothesis is that old animals have impaired metabolic switching, herein defined as the ability to utilize either carbohydrates or fat as a fuel source when appropriate. In line with this idea, insulin insensitivity occurs in advanced age [35–37], and fasting insulin levels are often elevated. Since insulin inhibits fat oxidation, higher insulin levels could also delay keto-adaptation. The current study tested the extent to which altered keto-adaptation in old age was related to impaired glycogen dynamics or metabolic dysfunction in rats that were placed on TRF (fed once per day) with a standard diet (SD) or TRF in combination with an isocaloric ketogenic diet (KD). For some comparisons, a third group of rats fed ad libitum (also referred to as free feeding) standard rodent chow was included.
Results
Previous ketogenic diet experience enhances keto-adaptation beyond time-restricted feeding alone
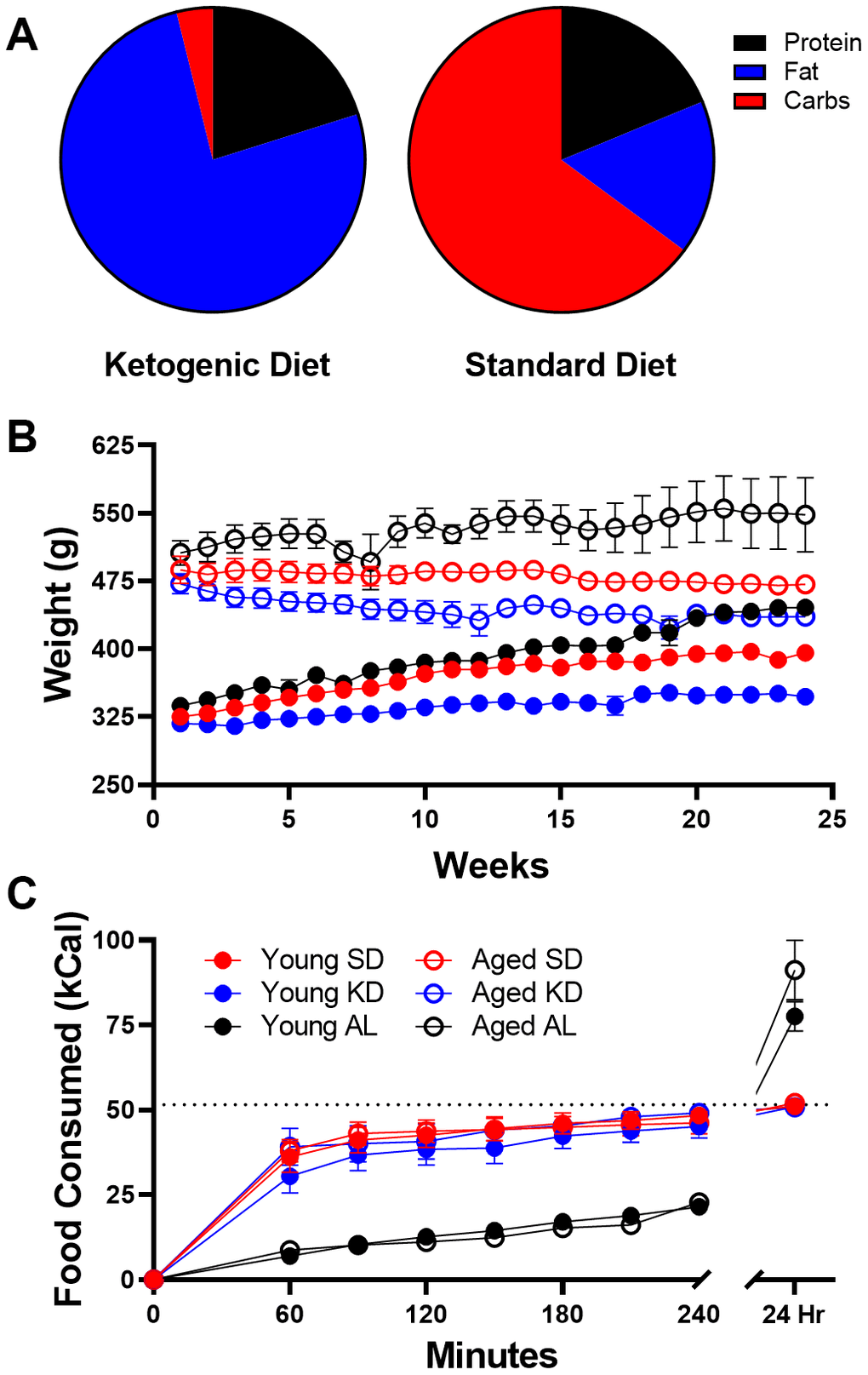
The TRF implemented in the current experiments was associated with a ~21 hour fast, as both young and aged rats consumed close to 100% of the available calories within 3 hours (Figure 1C). Because intermittent fasting [43] and the KD [44] have both been reported to improve insulin receptor sensitivity, as well as other markers of metabolic function, it is possible that either manipulation could enhance keto-adaptation in young and aged rats. To test this, young and aged rats were divided equally into groups A and B, with both groups receiving once daily TRF of 51 kcal as described previously with either the KD or SD for 6 weeks. Group A was placed on the KD for 4 consecutive weeks and then cycled onto the SD for 1 week, followed by a final week of the KD. Group B was placed on the SD for 4 weeks, followed by 1 week of the KD and a final week of the SD (Figure 7A). Blood glucose and BHB levels were collected every 3 days to calculate the GKI across the different diet cycles. Figure 7B shows the GKI values for groups A and B during KD initiation across the different cycles. During the cycles on the SD, all GKI values rapidly returned to high baseline levels on the first day and this did not vary between age groups (F[1,14] = 1.35; p = 0.26; Figure 7C).
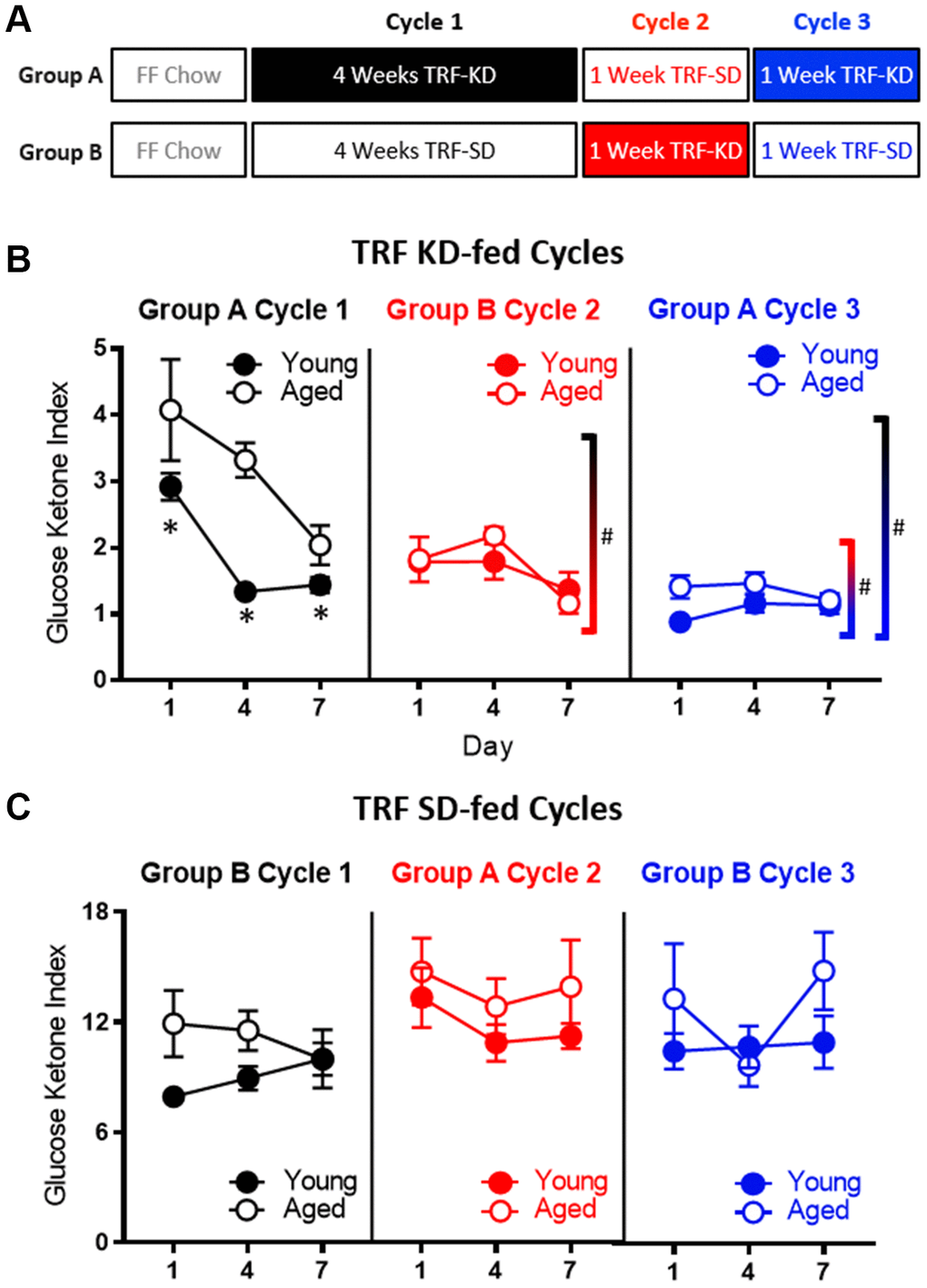
Figure 7. Age-related impairments in keto-adaptation are ameliorated with TRF and a previous keto-adaptation cycle. (A) Timeline of dietary interventions used for groups (A, B). Solid blocks indicate TRF-KD, empty blocks indicate TRF-SD. (B) GKI during 1 week of keto-adaptation is significantly lower in TRF-KD-fed rats that transitioned from TRF (red) relative to ad libitum-fed animals (black). However, rats that transitioned from TRF-SD to TRF-KD with a previous cycle of TRF-KD (blue) showed the lowest GKI. (C) Consumption of the TRF-SD was associated with high baseline GKI levels immediately after the cessation of carbohydrate restriction. Data are represented as group mean ± 1 SEM, * indicates p < 0.05 for individual time points, whereas # indicates significant difference across cycles, irrespective of day. For all panels n = 10 young, n = 8 aged.
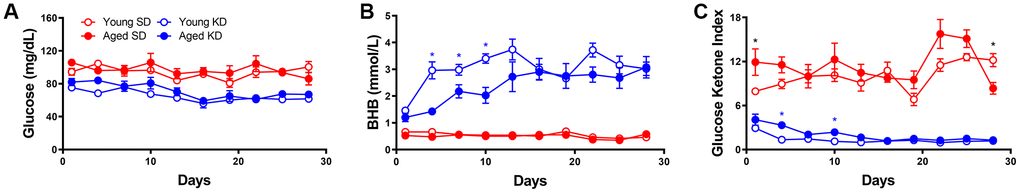
As in the previous cohorts of rats, when dietary ketosis with TRF was initiated following ad libitum feeding, aged rats showed significant delays in keto-adaptation (F[1,21] = 21.05; p < 0.001). Keto-adaptation was compared between rats that were naïve to dietary ketosis prior to Day 1 that cycled into ketosis following ad libitum feeding (Group A cycle 1) to rats that were naïve to dietary ketosis prior to Day following 4 weeks of TRF (Group B cycle 2). Overall, across days 1-7 on the KD, the GKI significantly decreased as a function of days on the diet (F[2,28] = 22.89; p < 0.001). The GKI, however, was significantly lower in Group B during cycle 2, compared to Group A during cycle 1 (F[1,14] = 16.74; p = 0.001), indicating that 4 weeks of fasting through TRF facilitated the ability to flexibly switch from glycolysis to ketosis compared to consuming food ad libitum, presumably by decreasing insulin levels. Across both cycles of ketosis initiation, the significant main effect of age persisted (F[1,14] = 10.44; p = 0.001), with aged rats having higher GKI values relative to young animals. Interestingly, there was a significant interaction effect between age and cycle (F[1,14] = 8.20; p = 0.01), indicating that the fasting through TRF expedited keto-adaptation in aged rats to a greater extent than young. This interaction is likely due to metabolic impairments and hyperinsulinemia of aged rats while being fed ad libitum, which was at least partially being overcome by TRF.
To examine the impact of previous experience on the KD to the rate of keto-adaptation in young and aged animals, we then compared GKI values from rats that were naïve to dietary ketosis prior to Day 1 (Group A cycle 1) to their GKI values during their second cycle of ketosis (Group A cycle 3). GKI levels were significantly lower during the second keto-adaptation cycle than the first (F[1,14] = 52.12; p < 0.001). Moreover, cycle significantly interacted with age (F[1,14] = 6.66; p = 0.02), indicating that the age-associated delay in keto-adaptation was mitigated by previous experience on the KD.
Because TRF with the SD was sufficient to influence keto-adaptation, the degree to which previous experience in ketosis influenced successive cycles of keto-adaptation was also examined. Specifically, Group A cycle 3 (TRF with prior KD experience) was compared to Group B cycle 2 (TRF and KD naïve prior to Day 1). Critically, both groups had 1 month of TRF prior to transitioning to the KD, with these groups differing only in terms of previous experience on the KD. Importantly, the GKI was significantly lower in Group A cycle 3 compared to Group B cycle 2 (F[1,14] = 15.89; p = 0.001). This observation indicates that a previous cycle of keto-adaptation enhanced the rate of metabolic switching to promote higher rates of ketogenesis in a shorter amount of time following carbohydrate restriction. In other words, the increased rate of keto-adaptation induced by 4 weeks of TRF was further augmented by previous experience with keto-adaptation. In contrast to Cycle 1 in Group A (first cycle of keto-adaptation following ad libitum feeding), there were no significant effects of age (F[1,14] = 2.43; p = 0.14) across the two later cycles.
Keto-adaptation in young and aged rats is decoupled from glycogen content
One hypothesis regarding the initiation of ketosis is that fat oxidation is promoted when glycogen content in the liver and muscles is depleted. The age-related delay in keto-adaptation observed in aged animals could therefore be due to increased glycogen levels in liver or muscle. Thus, to determine how glycogen dynamics interact with ketosis, we measured the glycogen content in the liver and muscle of young and aged rats on the SD or KD for at least 12 weeks prior to sacrifice. Most rats were fasted for at least 15 hours prior to sacrifice, however a small subset of rats was fed 1 hour prior to sacrifice. While there were no differences in liver glycogen content between age groups (F[1,50] = 2.50; p = 0.12; Figure 8A), there was a significant decrease in liver glycogen in KD-fed rats (F[1,50] = 6.60; p = 0.01). Additionally, for the SD-fed rats only, fasted rats had significantly less glycogen in their liver than fed rats (F[1,50] = 4.88; p = 0.03). This difference between fasted and unfasted animals was particularly evident in the aged rats and was not observed in the KD group, indicating that ketosis prevents fasting-induced glycogen depletion. These data suggest that long-term ketosis reduced liver glycogen content, but these levels were not affected while fasting. In contrast, aged rats on a SD that were fasted quickly lost liver glycogen, which was not observed in young rats. There were no differences in glycogen content within the muscle across age (F[1,50] = 0.47; p = 0.50), diet (F[1,50] = 0.04; p = 0.85) or fasted versus fed groups (F[1,50] = 1.31; p = 0.26; Figure 8B). Furthermore, there were no significant interactions between any of these variables (p > 0.14 for all).
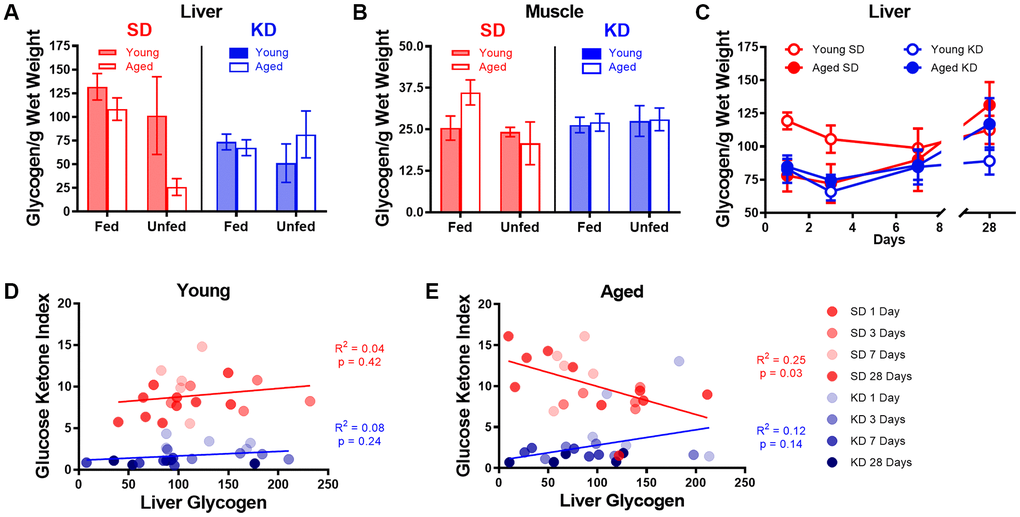
Figure 8. Keto-adaptation does not rely on glycogen depletion. (A) Glycogen levels in the liver while fasted or fed in young and aged rats on the SD or KD. SD-fed aged rats were not able to maintain liver glycogen when fasted, whereas KD-fed aged rats were. (B) Muscle glycogen while fasted or fed in young and aged rats on the SD or KD did not change across age, diet or feeding status groups. (C) Young SD-fed rats had significantly higher liver glycogen than all aged rats and KD-fed young rats during the first 3 days of the diet, but levels were similar in all groups on day 7 and beyond. (D) The GKI did not correlate with the amount of glycogen in the liver during keto-adaptation for young animals on the SD or KD. (E) The GKI did not correlate with the amount of glycogen in the liver during keto-adaptation for aged animals on the KD, but did significantly negatively correlate during this time period in SD-fed rats. Data are represented as group mean ± 1 SEM. In panels A-B: TRF standard diet n = 14 young, n = 15 aged; TRF ketogenic diet n = 14 young, n = 15 aged. For panels C-E: n = 20 per group for all groups.
To determine if the glycogen content within the liver changed during the process of keto-adaptation, 40 young and 40 aged rats were split into groups 1-4, which were fed 51 kcal once daily of the KD or SD for 1, 3, 7 or 28 days and then sacrificed. Because age differences in liver glycogen were only detected in fasted animals, all rats were sacrificed 18-22 hours after eating for quantification of liver glycogen content. Consistent with our previous data, following 1 and 3 days of consuming the SD, liver glycogen was significantly lower in aged rats relative to young (t[8] = 3.00; p = 0.02 and t[8] = 3.23; p = 0.01, respectively; Figure 8C). In the KD group at days 1 and 3, there was no age difference in liver glycogen (t[8] = 0.51; p = 0.62 and t[8] = 0.16; p = 0.88, respectively). By days 7 and 28, there was no longer a significant difference in liver glycogen content across age or diet groups nor did these two factors significantly interact (p > 0.26 for all comparisons). When time point was considered as a factor, there was a significant change in glycogen levels by day (F[3,54] = 3.18; p = 0.03), as well by diet (F[1,54] = 6.01; p = 0.02). While there was no significant effect of age group (F[1,54] = 1.8; p = 0.17), this factor did significantly interact with length of time on the diet (F[1,54] = 5.26; p = 0.03). This could be due to the tendency for glycogen levels to increase after 3 days on the diet in KD-fed rats.
Surprisingly, in young rats, liver glycogen content did not correlate with the GKI during the initiation of TRF in either KD-fed rats (R2 = 0.08; p = 0.24) or SD-fed rats (R2 = 0.04; p = 0.42; Figure 8D). Furthermore, there was no correlation between liver glycogen and GKI in aged KD-fed rats during the initiation of ketosis (R2 = 0.12; p = 0.14; Figure 8E). However, in the SD-fed rats that switched from ad libitum feeding to TRF, liver glycogen in aged SD-fed rats significantly negatively correlated with GKI (R2 = 0.25; p = 0.03). These data demonstrate that it is not critical for liver glycogen to be depleted to initiate dietary ketosis in either aged or young animals. Moreover, in aged rats that are transitioning from ad libitum feeding to TRF of a SD, more glycogen is associated with lower GKI values. This suggests that the aged rats that adapt to being fasted by decreasing blood glucose and increasing BHB level spare liver glycogen content.
Metabolic responses to a physiological stressor are enhanced by ketosis, but not time-restricted feeding
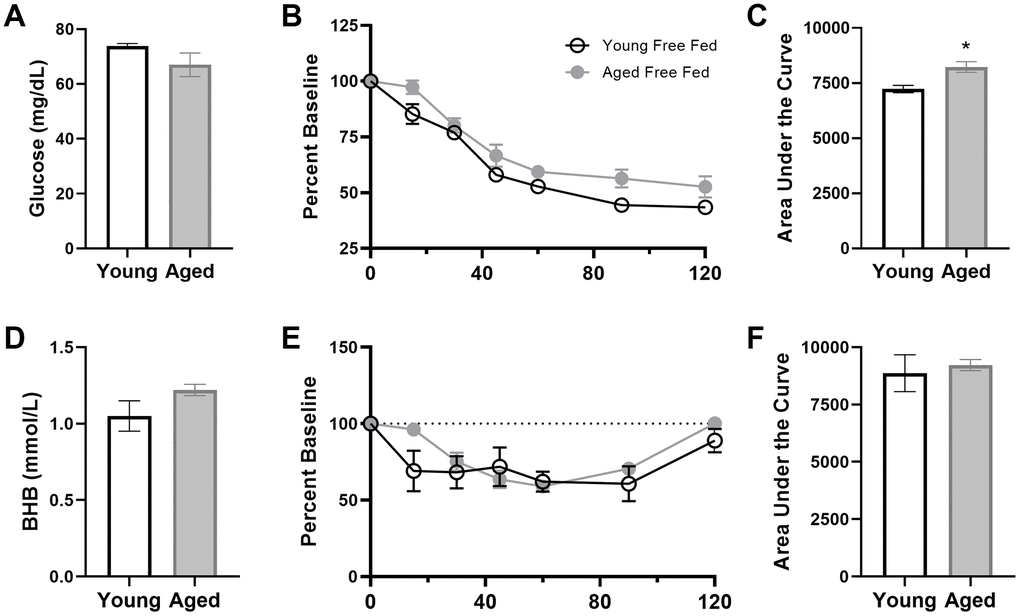
To investigate whether liver glycogen could still be utilized in ketotic animals, the metabolic response to stress, which involves a rapid hepatic conversion of glycogen to glucose [45], was pharmacologically induced. Importantly, this evolutionarily conserved metabolic response to a stressor can be essential for properly adapting to activities of daily living, especially during strenuous circumstances. To investigate this, serum levels of glucose and BHB following epinephrine injections were quantified in rats fed ad libitum with traditional rodent chow (Envigo, Teklad 2918). These same animals were then placed on the KD or SD for 12 weeks. At both time points, rats were given an intraperitoneal injection of either saline or 0.1mg/kg epinephrine 30 minutes postprandially, and blood glucose and BHB levels were quantified 30 minutes post-injection. Injections were separated by a 48-hour washout period, with the order counterbalanced across animals. The post-injection values of blood glucose, BHB and the GKI for saline and epinephrine were then used to calculate the percent change between saline and epinephrine injections for blood levels of glucose (Figure 9A), BHB (Figure 9B) and the GKI (Figure 9C).
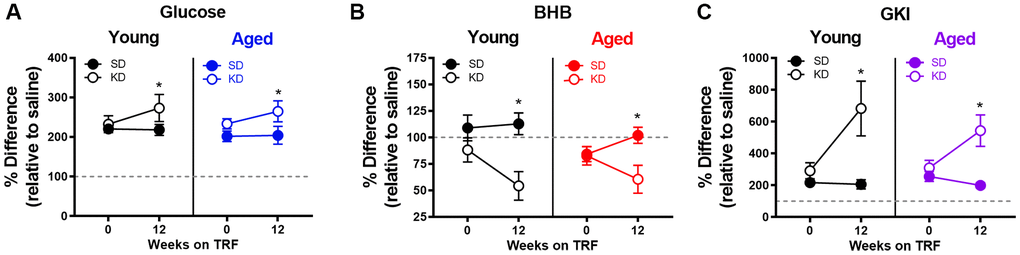
Figure 9. Keto-adaptation enhances physiological metabolic response to stress. The percent difference in blood (A) glucose, (B) BHB and (C) GKI between saline and epinephrine injections during ad libitum feeding and TRF with SD or KD. Data are represented as group mean ± 1 SEM, * = p < 0.05. For all panels: TRF standard diet n = 6 young, n = 8 aged; TRF ketogenic diet n = 6 young, n = 7 aged.
All rats, including those on the KD, showed a marked increase in blood glucose in response to epinephrine (Figure 9). In fact, there was a significant effect of diet on epinephrine-induced increases in blood glucose, with KD-fed rats having a larger change relative to the baseline saline condition (F[1,23] = 5.21; p = 0.032). This observation indicates a conservation of the stress-induced glucose response while in ketosis. The enhanced ability to mobilize glucose in response to a stressor was not due to the TRF alone, as the SD-fed rats did not show this same enhancement between ad libitum feeding and 12 weeks of the SD (F[1,13] = 0.29; p = 0.599).
In line with these data, blood levels of the pancreatic hormone glucagon, which is required for the breakdown of glycogen to glucose, was not altered in KD-fed rats (F[1,13] = 0.77; p = 0.394; Figure 10A). While we did not observe an age difference in our SD-fed rats, when older groups of animals were included there was a significant difference between young and old animals (Figure 10B, 10C), replicating previously published work [46, 47].
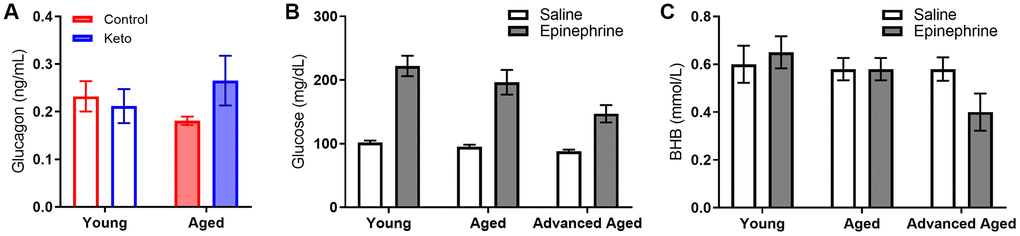
Figure 10. Glucagon, glucose and BHB response to epinephrine change as a function of advancing age. (A) Glucagon levels were not influenced by KD implementation. In rats of this strain there is not an age-related impairment in the glucose response to epinephrine at 24 months, but there is as 33 months in both (B) glucose and (C) BHB following epinephrine injections. Data are represented as group mean ± 1 SEM, * = p < 0.05. In panel A: TRF standard diet n = 4 young, n = 4 aged; TRF ketogenic diet n = 5 young, n = 4 aged. In panels B-C: ad libitum n = 6 young, n = 10 aged, n = 5 advanced age.
Although BHB levels were unaffected by epinephrine during ad libitum feeding with chow or during TRF with the SD, epinephrine decreased blood BHB levels in KD-fed rats. Moreover, in rats that went from ad libitum feeding to TRF with the KD, there was a significant change in the percent decrease in BHB induced by epinephrine (F[1,23] = 8.90; p = 0.007). In KD-fed rats, the glucose increase and concurrent decrease in BHB, was associated with a significant epinephrine-induced increase in GKI that did not occur during ad libitum feeding (F[1,23] = 15.39; p = 0.001). Despite the isocaloric 51 kcal per day across both diet groups, the abrupt increase in the post-epinephrine GKI experienced by the KD-fed rats was not observed in the SD-fed group, indicating that TRF alone did not enhance the metabolic response to stress in either young or aged rats. The present observations suggest that long-term nutritional ketosis does not deplete glycogen stores. Moreover, hepatic glycogen can be mobilized in response to a physiological stressor to a greater extent when in ketosis relative to glycolysis alone.
Discussion
Here we show that age-related metabolic dysfunction is mitigated by changing an ad libitum energy consumption pattern to time-restricted feeding (TRF; once daily feeding) of 51 kcal. These benefits in metabolic health were further bolstered by a high fat, low carbohydrate ketogenic diet (KD). We replicated previous data [14, 15] demonstrating an age-related impairment in keto-adaptation, and extended these observations to show that keto-adaptation induced by diet does not correlate with liver glycogen depletion. While a lack of correlation between liver glycogen and level of ketosis does not in itself mandate a decoupling of these two processes, taken together with the rest of the data presented here, it is clear that ketosis can be decoupled from glycogen depletion. Surprisingly, when fasted, rats in long-term ketosis had levels of liver and muscle glycogen that were comparable to young rats on the standard diet (SD) and considerably higher than aged SD-fed rats. Moreover, the KD appeared to enhance glycogen dynamics, with KD-fed rats showing an enhanced increase in blood glucose in response to epinephrine. Thus, age-related declines in metabolic switching do not appear to result from differences in glycogen storage and dynamics, but rather emerge from metabolic dysfunction associated with hyperinsulinemia.
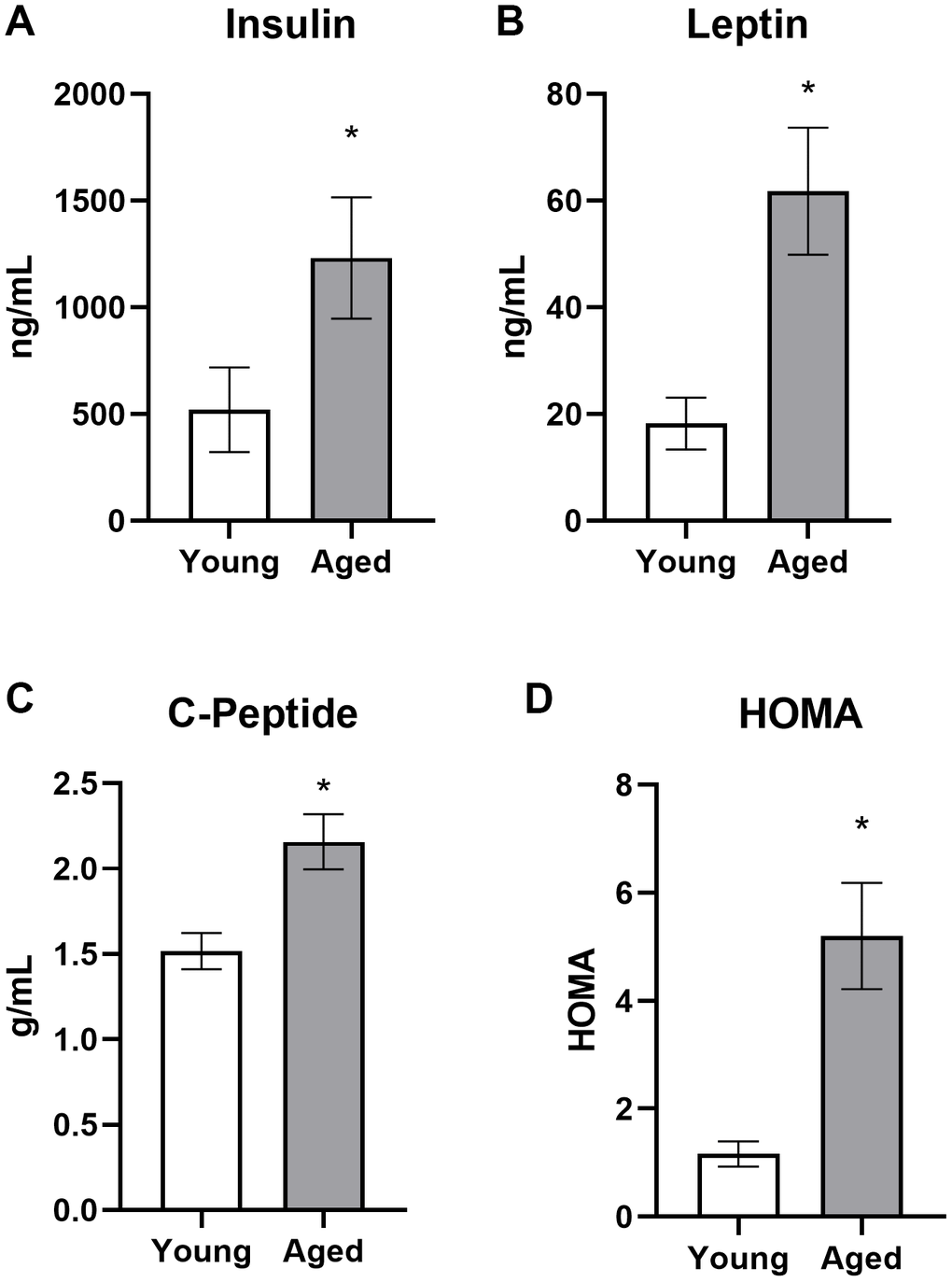
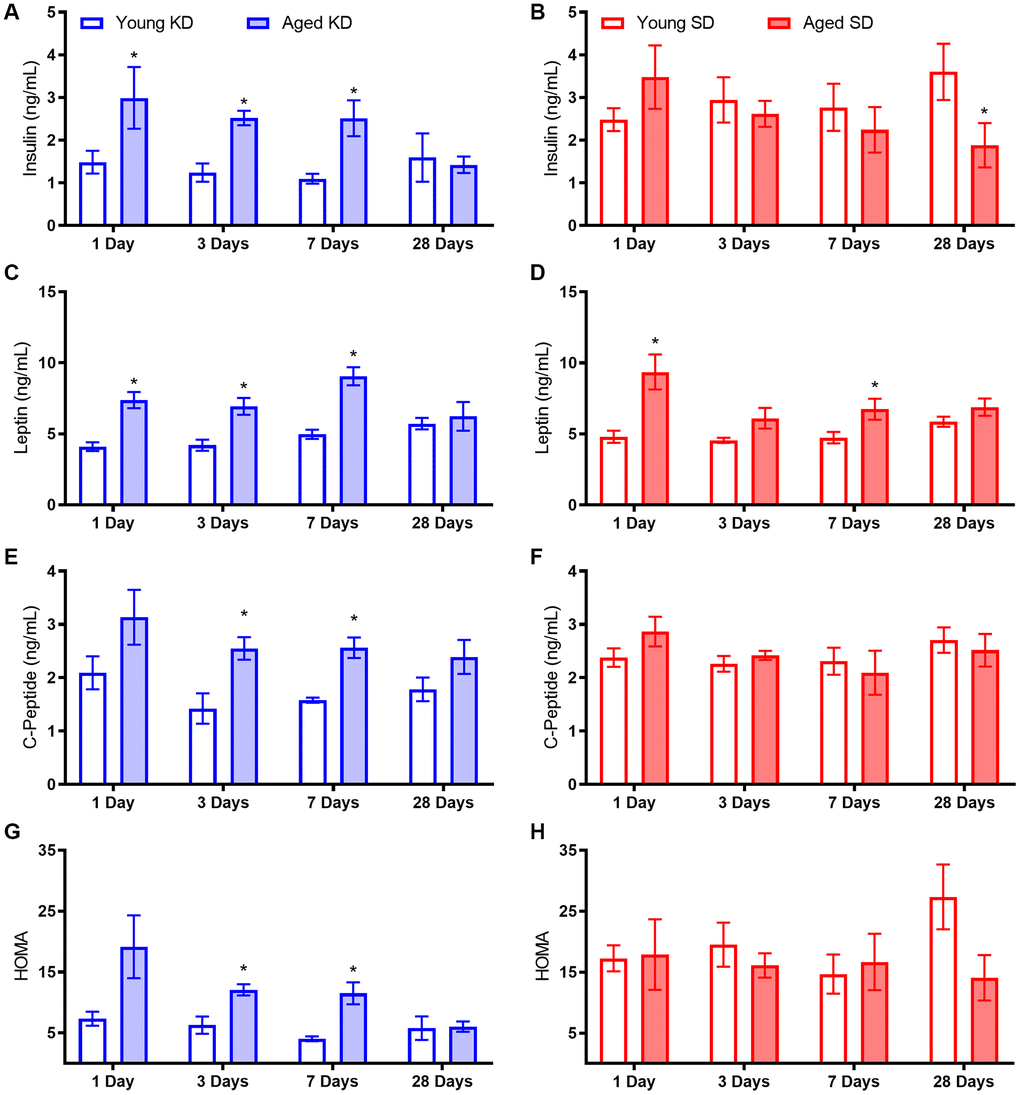
Aged rats had elevated fasting and postprandial insulin levels while ad libitum feeding with standard rodent chow, as well as during the initial stages of keto-adaptation. In young rats, fasting insulin levels decreased significantly following the first 24 hours of a KD. This decrease in fasting insulin was not observed in aged rats during the first week of a KD. In addition to insulin deficits, aged animals fed ad libitum with chow had significantly elevated leptin, c-peptide, and HOMA. While some metabolic factors were significantly improved following TRF with the SD, such as the insulin tolerance test, combining TRF with a KD was most efficacious at restoring metabolic markers in aged rats to levels comparable to young animals, as well as enhancing the epinephrine-induced increase in peripheral glucose.
If metabolic switching is defined as being able to switch bi-directionally between periods of carbohydrate/glucose usage and fatty acid/ketone body usage [48], then the KD enhanced metabolic switching beyond what was observed with TRF alone. While TRF did increase the rate at which aged rats could transition from glycolysis to ketosis, enhanced keto-adaptation was further augmented by TRF+KD. These data concur with previous studies demonstrating the additive benefits of food restriction with a KD at increasing seizure threshold [49]. Surprisingly, young and aged rats in our study consuming the KD still showed an enhanced glucose response to an acute increase in epinephrine, beyond what was observed in rats fed TRF+SD. This observation further supports the notion that TRF+KD may confer the greatest improvement in metabolic switching. Enhancing metabolic switching may be particularly important in aged populations. While epinephrine is able to improve memory performance in young rats through hepatic glucogenesis, aged rats show an attenuated elevation in blood glucose in response to epinephrine, and no cognitive benefits. Thus, old animals do not cognitively benefit from this physiological stress response that is an evolutionary conserved mechanism to promote survival [50].
In the short term, consumption of a large quantity of fatty acids, and relatively little glucose, initiates an inhibition of glycogen production via the glucose fatty acid cycle [51]. These effects are due to changes in enzymatic activity in response to dietary composition [52]. However, the glucose fatty acid cycle is dysfunctional in metabolically impaired individuals, such as those with type 2 diabetes. Thus, it is conceivable that the aged rats in the current study, which had elevated fasting insulin and impaired insulin sensitivity, may have metabolic deficits that produce impairments in the glucose fatty cycle. How this specifically interacts with long-term versus cyclical dietary ketosis is an avenue of investigation worth pursuit. Additionally, weight differences across age groups may also be an influencing factor on keto-adaptation, as obesity increases the incidence of insulin resistance and other metabolic issues, and could be addressed in future studies through the implementation of weight control measures prior to KD intervention.
Although TRF alone can enhance metabolic switching, and improve cognitive health across the lifespan [8, 9, 48, 53–55], the current data suggest that additional benefits can be obtained through nutritional ketosis. It is also notable that the benefits of TRF for cognitive function might be due to comparisons with ad libitum fed rodents that are metabolically morbid [56]. In fact, it is well documented that excessive energy consumption can lead to cognitive impairments [57–59]. A strong indicator that TRF alone is not sufficient for extending the cognitive healthspan is the wealth of studies demonstrating age-related cognitive decline in rodents while utilizing tasks requiring appetitive motivation. Many tests of cognitive function require animals to be restricted to <85% of their body weight to motivate participation by feeding once daily, making a large portion of investigations on the neurobiology of cognitive aging into incidental experiments of caloric restriction with TRF. While incentive motivation for food rewards does not change with age [60], robust age-related deficits are observed on working memory [61–63], other executive functions [64–66], as well as behaviors that require the prefrontal cortex and medial temporal lobe [14, 67–69].
Importantly, there may be pertinent differences in metabolism, brain aging, and responses to dietary ketosis between males and females [70]. Unfortunately, the current lack of availability of female Fischer 344 x Brown Norway hybrid rats from maintained rodent colonies (https://www.nia.nih.gov/research/dab/aged-rodent-colonies-handbook) did not permit the consideration of sex as a variable in the current experiments. Potential interactions of sex and dietary ketosis will need to be examined in future studies.
We’ve shown that combining TRF with a KD provides a more robust restoration of metabolic health than TRF alone. Furthermore, as the number of individuals living with metabolic disease (e.g. diabetes, metabolic syndrome, insulin resistance, etc.) continues to increase, these data demonstrate the translational potential of TRF+KD for alleviating many of the health burdens facing these patients. In addition to the metabolic factors measured here, a previous study has shown that this feeding pattern can enhance cognitive function and decrease anxiety-like behaviors [14]. Moreover, TRF+KD alters transporter protein expression within the prefrontal cortex and hippocampus [14, 15], increases BDNF [71, 72], decreases pain and inflammation [73], modulates KATP channels [72, 74], modifies neurotransmitter levels [75, 76] and lessens amyloid and tau pathologies [77]. Each of these biochemical alterations induced by ketosis are likely to confer resilience to cognitive decline.
In addition to peripheral health, memory deficits and other types of cognitive impairment have a profound contribution to decreasing quality of life in individuals and their caregivers. Although the increased mean life expectancy has had a commensurate increase in the number of aged people living without physical limitations [78], the same cannot be said for cognitive deficits. In fact, the proportion of individuals experiencing mild or severe memory impairments will reach 1 in 3 by the 8th decade of life [79], and disrupted brain bioenergetics is a common feature of aging [80, 81]. Improving metabolic function in the aged population can have profound impacts on cognitive outcomes, as the brain disproportionately uses metabolic resources [80] Furthermore, cerebral glucose metabolism decreases with age, most notably within the hippocampus and prefrontal cortex [82], both of which are critical for higher cognitive function [83–85]. Importantly, this decline correlates with worse spatial memory performance [82]. Thus, developing metabolic-based interventions for improving peripheral metabolic health will likely have profound therapeutic potential for increasing the cognitive healthspan.
Materials and Methods
Subjects and handling
Young (4 months; n = 112; mean weight 337.59g) and aged (20 months: n = 112; mean weight 526.04) male Fischer 344 x Brown Norway F1 Hybrid rats from the NIA colony at Charles River were used in this study (see Table 1 for sample sizes per experiment). Rats were housed individually and maintained on a reverse 12-hr light/dark cycle, and all feeding, metabolic and biochemical assays were performed in the dark phase. Rats were given one week to acclimate to the facility prior to any intervention. All experimental procedures were performed in accordance with National Institutes of Health guidelines and were approved by the Institutional Animal Care and Use Committee at the University of Florida.
Table 1. Sample size per dietary group per experiment.
| Figure | Ad libitum chow-fed rats | TRF standard diet-fed rats | TRF ketogenic diet-fed rats |
| Figure 1: Feeding pattern detection | Young n = 6 Aged n = 5 | Young n = 7 Aged n = 5 | Young n = 7 Aged n = 4 |
| Figure 2: Keto-adaptation rate | Young n = 5 Aged n = 4 | Young n = 5 Aged n = 4 | |
| Figure 3-4: Age-related metabolic impairment | Young n = 6-27 Aged n = 5-24 | ||
| Figure 5: Insulin during keto-adaptation | Young n = 20 Aged n = 20 | Young n = 20 Aged n = 20 | |
| Figure 6: Insulin tolerance testing | Young n = 6 Aged n = 5 | Young n = 7 Aged n = 7 | Young n = 7 Aged n = 6 |
| Figure 7: Naïve vs experienced keto-adaptation | Young n = 10; Aged n = 8 All rats alternated dietary regiments, see text and Figure 7 for details. | ||
| Figure 8: Glycogen content in liver and muscle during keto-adaptation | Young n = 20 Aged n = 20 | Young n = 20 Aged n = 20 | |
| Figure 9: Epinephrine-induced stress response | Young n = 6 Aged n = 8 | Young n = 6 Aged n = 7 | |
| Figure 10A: Systemic glucagon | Young n = 4 Aged n = 4 | Young n = 5 Aged n = 4 | |
| Figure 10B, 10C: Epinephrine-induced stress response in advanced age | Young n = 6 Aged n = 10 Advanced Age n = 5 | ||
| Supplementary Figure 1: Glucose tolerance testing | Young n = 6 Aged n = 5 | Young n = 7 Aged n = 7 | Young n = 7 Aged n = 6 |
Dietary interventions
For all experiments, the same ketogenic diet (KD) and standard diet (SD) were used as published previously [14, 15, 68]. An additional group of rats were fed ad libitum with standard laboratory chow (Envigo, Teklad 2918). The KD was a high fat/low carbohydrate diet (Lab Supply; 5722, Fort Worth, Texas) mixed with MCT oil (Neobee 895, Stephan, Northfield, Illinois) with a macronutrient profile of 76% fat, 4% carbohydrates, and 20% protein (see Figure 1A). The micronutrient-matched SD (Lab Supply; 1810727, Fort Worth, Texas) had a macronutrient profile of 16% fat, 65% carbohydrates, and 19% protein (see Figure 1A). The KD and SD were made fresh weekly and kept at 4°C until use each day. For additional micronutrient details for both diets, see [15]. The Teklad standard laboratory chow had a macronutrient composition of 5% fat, 75% carbohydrates and 20% protein. The SD and KD were micronutrient matched, and rats were fed the same number of calories each day (~51kcal) during the dark phase between 14:00 and 17:00 hours (lights on at 20:00) while on TRF, regardless of diet. Access to water was ad libitum for all rats on all diets, and rats were weighed daily when fed. These different diets are reported to not alter water consumption patterns or induce a prolonged stress response [15].
Blood collection, testing, and metabolic data
All blood samples collected prior to sacrifice were collected from a small nick in the tail. Rats were briefly restrained, tails were cleaned with an alcohol swab, and then a clean scalpel blade was used to make a superficial cut approximately 1 inch from the end of the tail. Following blood collection, wounds were wiped clean and Wonder Dust (Farnam Companies, Phoenix, AZ) was applied to promote wound healing. Blood collected on the day of sacrifice was collected directly from the trunk immediately following decapitation.
For blood glucose and ketone body (β-hydroxybutyrate; BHB) determination, one drop per test was collected directly onto the appropriate test strip (Abbott Diabetes Care, Inc, Alameda, CA; glucose SKU#: 9972865 and ketone SKU#: 7074565) in a Precision Xtra blood monitoring system (Abbott Diabetes Care, Inc, Alameda, CA; SKU#: 9881465).
For the insulin tolerance test (ITT) and glucose tolerance test (GTT), rats were fasted overnight and injected intraperitoneally with either 0.75 U/kg of human insulin (Hanna Pharmaceutical Supply Co #NC0769896) or 1 g/kg glucose (Fisher Scientific #D16-500). Blood glucose and ketone body levels were measured as described above at 0, 15, 30, 45, 60, and 120 minutes post injection.
For metabolic biomarker quantification, approximately 750 μL of blood was collected from the tail nick at the time of glucose testing. Collected blood was left at room temperature for at least 25 minutes prior to centrifugation at 13,000g for 10 minutes at 4°C. 0.35 μL of supernatant was collected into a tube containing 0.4 μL DPP-IV inhibitor (EMD Millipore, NC9010908), 0.4 μL protease inhibitor cocktail (Millipore Sigma, S8820-2TAB) and 0.32 μL 0.9% PBS. Samples were kept frozen at -80°C prior to use in the Rat Metabolic Array (MRDMET; EVE Technologies, Calgary, AB). This multiplex immunoassay (BioPlex 200) quantified levels of insulin, leptin, C-peptide 2 and glucagon. Each sample was analyzed in duplicate. All values outside of the liner range were excluded.
Epinephrine administration
Prior to starting any dietary intervention, half of the rats were given 0.1mg/kg epinephrine (diluted in 0.9% sterile saline solution) and half were given the equivalent volume of saline delivered via intraperitoneal injection. After 30 minutes, glucose and BHB were determined as described above. Groups were counterbalanced for drug across two days of testing with one day off in between. That is, following a 48-hour washout period, the groups were reversed and the process was repeated. This entire 2-day process was repeated after 12 weeks on the TRF+SD and TRF+KD fed rats, with epinephrine injections occurring 30 minutes postprandially.
Tissue collection and glycogen quantification
On the day of sacrifice, a subset of rats was fed their respective diets one hour prior to being anesthetized. All other rats were sacrificed without feeding since the prior day (approximately 15-22 hours fasted). All rats were rapidly sedated in a jar containing concentrated isoflurane and then decapitated once the righting reflex had ceased. The liver and tibialis anterior muscle were isolated, rinsed in cold PBS and immediately frozen on dry ice. Tissue was stored at -80°C until use.
Glycogen content in the liver and muscle tissue were determined per mg wet tissue weight, modeled after previously published protocols [86–88]. Approximately 50 mg frozen tissue was placed in a pre-weighed tube containing 660 μL NaOH at 100°C and tissue weight was determined. Tubes were boiled at 100°C for 15 minutes, vortexed and then brought to neutral pH using 320 μL 2M HCl. Samples were vortexed again and 150 μL of muscle, or 20 μL of liver with 130 μL of ddH2O, were placed into two tubes each. A sodium acetate solution was added to each tube, one with and one without the addition of amyloglucosidase, and samples were incubated at 37°C for 2 hours. Following a 20-minute centrifuge at 14,000 RPM, 50 μL of supernatant was plated in triplicate into a 96 well plate. 100 μL Tris buffer was added to each well, which was then read on a Synergy HT Microplate Spectrophotometer (BioTek) at 340 nm. 50 μL hexokinase solution was added to each well, and the plate was incubated at room temperature on a shaker, away from light before reading again at 340 nm. The optical density reading before the addition of hexokinase was subtracted from the final optical density reading to correct for free glucose values. Final glycogen content was extrapolated using a standard glycogen curve run on each plate and normalized to tissue weight.
Quantification and statistical analysis
All data are expressed as group means ± standard error of the mean (SEM) unless otherwise reported. The glucose ketone index (GKI) was calculated as described in a previous publication [39]. Briefly, the glucose (mg/dL) was divided by the molecular weight of glucose (expressed as (mol/(g x dL))), and this value was then divided by the BHB value (mM). Differences across age and diet groups were analyzed using a two-factor ANOVA with the between subjects factors of age (2 levels: young and aged) and diet (2 levels: SD and KD or 3 levels: SD, KD and ad libitum). When time was a factor, an ANOVA-RM was used with the same factors. Post hoc analyses were done utilizing independent-samples t-tests where appropriate. Null hypotheses were rejected at the level of p > 0.05. All analyses were performed with the Statistical Package for the Social Sciences v25 (IBM, Armonk, NY) or GraphPad Prism version 7.03 for Windows (GraphPad Software, La Jolla, California USA).
Author Contributions
ARH designed the study, contributed to data acquisition, analysis and interpretation, and prepared the manuscript. LMT, QPF, KTC, BMM, NF & MH contributed to data acquisition and analysis. DD contributed to interpretation and prepared the manuscript. SNB designed the study, contributed to data analysis, interpretation and prepared the manuscript.
Conflicts of Interest
No authors have a conflicts of interest to report.
Funding
This work was supported by the National Institutes of Health, National Institute on Aging (RF1AG060977; 1F31AG058455).
References
- 1. Masoro EJ. Overview of caloric restriction and ageing. Mech Ageing Dev. 2005; 126:913–22. https://doi.org/10.1016/j.mad.2005.03.012 [PubMed]
- 2. Fontana L, Klein S. Aging, adiposity, and calorie restriction. JAMA. 2007; 297:986–94. https://doi.org/10.1001/jama.297.9.986 [PubMed]
- 3. Fontana L, Partridge L, Longo VD. Extending healthy life span—from yeast to humans. Science. 2010; 328:321–26. https://doi.org/10.1126/science.1172539 [PubMed]
- 4. de Cabo R, Carmona-Gutierrez D, Bernier M, Hall MN, Madeo F. The search for antiaging interventions: from elixirs to fasting regimens. Cell. 2014; 157:1515–26. https://doi.org/10.1016/j.cell.2014.05.031 [PubMed]
- 5. Longo VD, Mattson MP. Fasting: molecular mechanisms and clinical applications. Cell Metab. 2014; 19:181–92. https://doi.org/10.1016/j.cmet.2013.12.008 [PubMed]
- 6. Mattson MP, Allison DB, Fontana L, Harvie M, Longo VD, Malaisse WJ, Mosley M, Notterpek L, Ravussin E, Scheer FA, Seyfried TN, Varady KA, Panda S. Meal frequency and timing in health and disease. Proc Natl Acad Sci USA. 2014; 111:16647–53. https://doi.org/10.1073/pnas.1413965111 [PubMed]
- 7. Longo VD, Panda S. Fasting, Circadian Rhythms, and Time-Restricted Feeding in Healthy Lifespan. Cell Metab. 2016; 23:1048–59. https://doi.org/10.1016/j.cmet.2016.06.001 [PubMed]
- 8. Mitchell SJ, Bernier M, Mattison JA, Aon MA, Kaiser TA, Anson RM, Ikeno Y, Anderson RM, Ingram DK, de Cabo R. Daily Fasting Improves Health and Survival in Male Mice Independent of Diet Composition and Calories. Cell Metab. 2019; 29:221–228.e3. https://doi.org/10.1016/j.cmet.2018.08.011 [PubMed]
- 9. Anson RM, Guo Z, de Cabo R, Iyun T, Rios M, Hagepanos A, Ingram DK, Lane MA, Mattson MP. Intermittent fasting dissociates beneficial effects of dietary restriction on glucose metabolism and neuronal resistance to injury from calorie intake. Proc Natl Acad Sci USA. 2003; 100:6216–20. https://doi.org/10.1073/pnas.1035720100 [PubMed]
- 10. Mitchell SJ, Madrigal-Matute J, Scheibye-Knudsen M, Fang E, Aon M, González-Reyes JA, Cortassa S, Kaushik S, Gonzalez-Freire M, Patel B, Wahl D, Ali A, Calvo-Rubio M, et al. Effects of Sex, Strain, and Energy Intake on Hallmarks of Aging in Mice. Cell Metab. 2016; 23:1093–112. https://doi.org/10.1016/j.cmet.2016.05.027 [PubMed]
- 11. Bredesen DE. Reversal of cognitive decline: a novel therapeutic program. Aging (Albany NY). 2014; 6:707–17. https://doi.org/10.18632/aging.100690 [PubMed]
- 12. Choi HR, Kim J, Lim H, Park YK. Two-Week Exclusive Supplementation of Modified Ketogenic Nutrition Drink Reserves Lean Body Mass and Improves Blood Lipid Profile in Obese Adults: A Randomized Clinical Trial. Nutrients. 2018; 10:10. https://doi.org/10.3390/nu10121895 [PubMed]
- 13. Greco T, Glenn TC, Hovda DA, Prins ML. Ketogenic diet decreases oxidative stress and improves mitochondrial respiratory complex activity. J Cereb Blood Flow Metab. 2016; 36:1603–13. https://doi.org/10.1177/0271678X15610584 [PubMed]
- 14. Hernandez AR, Hernandez CM, Campos K, Truckenbrod L, Federico Q, Moon B, McQuail JA, Maurer AP, Bizon JL, Burke SN. A Ketogenic Diet Improves Cognition and Has Biochemical Effects in Prefrontal Cortex That Are Dissociable From Hippocampus. Front Aging Neurosci. 2018; 10:391. https://doi.org/10.3389/fnagi.2018.00391 [PubMed]
- 15. Hernandez AR, Hernandez CM, Campos KT, Truckenbrod LM, Sakarya Y, McQuail JA, Carter CS, Bizon JL, Maurer AP, Burke SN. The Antiepileptic Ketogenic Diet Alters Hippocampal Transporter Levels and Reduces Adiposity in Aged Rats. J Gerontol A Biol Sci Med Sci. 2018; 73:450–58. https://doi.org/10.1093/gerona/glx193 [PubMed]
- 16. Hussain TA, Mathew TC, Dashti AA, Asfar S, Al-Zaid N, Dashti HM. Effect of low-calorie versus low-carbohydrate ketogenic diet in type 2 diabetes. Nutrition. 2012; 28:1016–21. https://doi.org/10.1016/j.nut.2012.01.016 [PubMed]
- 17. Kephart WC, Mumford PW, Mao X, Romero MA, Hyatt HW, Zhang Y, Mobley CB, Quindry JC, Young KC, Beck DT, Martin JS, McCullough DJ, D’Agostino DP, et al. The 1-Week and 8-Month Effects of a Ketogenic Diet or Ketone Salt Supplementation on Multi-Organ Markers of Oxidative Stress and Mitochondrial Function in Rats. Nutrients. 2017; 9:1019. https://doi.org/10.3390/nu9091019 [PubMed]
- 18. Westman EC, Yancy WS
Jr , Mavropoulos JC, Marquart M, McDuffie JR. The effect of a low-carbohydrate, ketogenic diet versus a low-glycemic index diet on glycemic control in type 2 diabetes mellitus. Nutr Metab (Lond). 2008; 5:36. https://doi.org/10.1186/1743-7075-5-36 [PubMed] - 19. Phinney SD. Ketogenic diets and physical performance. Nutr Metab (Lond). 2004; 1:2. https://doi.org/10.1186/1743-7075-1-2 [PubMed]
- 20. Volek JS, Freidenreich DJ, Saenz C, Kunces LJ, Creighton BC, Bartley JM, Davitt PM, Munoz CX, Anderson JM, Maresh CM, Lee EC, Schuenke MD, Aerni G, et al. Metabolic characteristics of keto-adapted ultra-endurance runners. Metabolism. 2016; 65:100–10. https://doi.org/10.1016/j.metabol.2015.10.028 [PubMed]
- 21. Gasior M, Rogawski MA, Hartman AL. Neuroprotective and disease-modifying effects of the ketogenic diet. Behav Pharmacol. 2006; 17:431–39. https://doi.org/10.1097/00008877-200609000-00009 [PubMed]
- 22. Hartman AL. Neuroprotection in metabolism-based therapy. Epilepsy Res. 2012; 100:286–94. https://doi.org/10.1016/j.eplepsyres.2011.04.016 [PubMed]
- 23. Paoli A, Rubini A, Volek JS, Grimaldi KA. Beyond weight loss: a review of the therapeutic uses of very-low-carbohydrate (ketogenic) diets. Eur J Clin Nutr. 2013; 67:789–96. https://doi.org/10.1038/ejcn.2013.116 [PubMed]
- 24. Bentourkia M, Tremblay S, Pifferi F, Rousseau J, Lecomte R, Cunnane S. PET study of 11C-acetoacetate kinetics in rat brain during dietary treatments affecting ketosis. Am J Physiol Endocrinol Metab. 2009; 296:E796–801. https://doi.org/10.1152/ajpendo.90644.2008 [PubMed]
- 25. Daniel PM, Love ER, Moorehouse SR, Pratt OE, Wilson P. Factors influencing utilisation of ketone-bodies by brain in normal rats and rats with ketoacidosis. Lancet. 1971; 2:637–38. https://doi.org/10.1016/S0140-6736(71)80073-9 [PubMed]
- 26. Hawkins RA, Mans AM, Davis DW. Regional ketone body utilization by rat brain in starvation and diabetes. Am J Physiol. 1986; 250:E169–78. https://doi.org/10.1152/ajpendo.1986.250.2.E169 [PubMed]
- 27. Lukosaityte J, Allen HJ, Lennon DJ, Borum PR. BMI Z-Score Improves after 24 Months of Precision Ketogenic Therapy in Patients with Abnormal Baseline BMI Z-Score. FASEB J. 2017; 31:963.3–963.3.
- 28. Dohm GL, Tapscott EB, Barakat HA, Kasperek GJ. Influence of fasting on glycogen depletion in rats during exercise. J Appl Physiol Respir Environ Exerc Physiol. 1983; 55:830–33. https://doi.org/10.1152/jappl.1983.55.3.830 [PubMed]
- 29. Ivy JL. Muscle glycogen synthesis before and after exercise. Sports Med. 1991; 11:6–19. https://doi.org/10.2165/00007256-199111010-00002 [PubMed]
- 30. Adams JH, Koeslag JH. Carbohydrate homeostasis and post-exercise ketosis in trained and untrained rats. J Physiol. 1988; 407:453–61. https://doi.org/10.1113/jphysiol.1988.sp017425 [PubMed]
- 31. Morris KA, Chang Q, Mohler EG, Gold PE. Age-related memory impairments due to reduced blood glucose responses to epinephrine. Neurobiol Aging. 2010; 31:2136–45. https://doi.org/10.1016/j.neurobiolaging.2008.12.003 [PubMed]
- 32. Talley CE, Kahn S, Alexander LJ, Gold PE. Epinephrine fails to enhance performance of food-deprived rats on a delayed spontaneous alternation task. Neurobiol Learn Mem. 2000; 73:79–86. https://doi.org/10.1006/nlme.1999.3920 [PubMed]
- 33. McNay EC, Gold PE. Age-related differences in hippocampal extracellular fluid glucose concentration during behavioral testing and following systemic glucose administration. J Gerontol A Biol Sci Med Sci. 2001; 56:B66–71. https://doi.org/10.1093/gerona/56.2.B66 [PubMed]
- 34. Morris KA, Gold PE. Epinephrine and glucose modulate training-related CREB phosphorylation in old rats: relationships to age-related memory impairments. Exp Gerontol. 2013; 48:115–27. https://doi.org/10.1016/j.exger.2012.11.010 [PubMed]
- 35. Fink RI, Kolterman OG, Griffin J, Olefsky JM. Mechanisms of insulin resistance in aging. J Clin Invest. 1983; 71:1523–35. https://doi.org/10.1172/JCI110908 [PubMed]
- 36. Ryan AS. Insulin resistance with aging: effects of diet and exercise. Sports Med. 2000; 30:327–46. https://doi.org/10.2165/00007256-200030050-00002 [PubMed]
- 37. Refaie MR, Sayed-Ahmed NA, Bakr AM, Abdel Aziz MY, El Kannishi MH, Abdel-Gawad SS. Aging is an Inevitable Risk Factor for Insulin Resistance. J Taibah Univ Med Sci. 2006; 1:30–41. https://doi.org/10.1016/S1658-3612(06)70005-1
- 38. Volek JS, Noakes T, Phinney SD. Rethinking fat as a fuel for endurance exercise. Eur J Sport Sci. 2015; 15:13–20. https://doi.org/10.1080/17461391.2014.959564 [PubMed]
- 39. Meidenbauer JJ, Mukherjee P, Seyfried TN. The glucose ketone index calculator: a simple tool to monitor therapeutic efficacy for metabolic management of brain cancer. Nutr Metab (Lond). 2015; 12:12. https://doi.org/10.1186/s12986-015-0009-2 [PubMed]
- 40. Fukao T, Lopaschuk GD, Mitchell GA. Pathways and control of ketone body metabolism: on the fringe of lipid biochemistry. Prostaglandins Leukot Essent Fatty Acids. 2004; 70:243–51. https://doi.org/10.1016/j.plefa.2003.11.001 [PubMed]
- 41. Meigs JB, Rutter MK, Sullivan LM, Fox CS, D’Agostino RB
Sr , Wilson PW. Impact of insulin resistance on risk of type 2 diabetes and cardiovascular disease in people with metabolic syndrome. Diabetes Care. 2007; 30:1219–25. https://doi.org/10.2337/dc06-2484 [PubMed] - 42. Bonora E, Formentini G, Calcaterra F, Lombardi S, Marini F, Zenari L, Saggiani F, Poli M, Perbellini S, Raffaelli A, Cacciatori V, Santi L, Targher G, et al. HOMA-estimated insulin resistance is an independent predictor of cardiovascular disease in type 2 diabetic subjects: prospective data from the Verona Diabetes Complications Study. Diabetes Care. 2002; 25:1135–41. https://doi.org/10.2337/diacare.25.7.1135 [PubMed]
- 43. Mattson MP, Wan R. Beneficial effects of intermittent fasting and caloric restriction on the cardiovascular and cerebrovascular systems. J Nutr Biochem. 2005; 16:129–37. https://doi.org/10.1016/j.jnutbio.2004.12.007 [PubMed]
- 44. Ballard KD, Quann EE, Kupchak BR, Volk BM, Kawiecki DM, Fernandez ML, Seip RL, Maresh CM, Kraemer WJ, Volek JS. Dietary carbohydrate restriction improves insulin sensitivity, blood pressure, microvascular function, and cellular adhesion markers in individuals taking statins. Nutr Res. 2013; 33:905–12. https://doi.org/10.1016/j.nutres.2013.07.022 [PubMed]
- 45. Sherwin RS, Saccà L. Effect of epinephrine on glucose metabolism in humans: contribution of the liver. Am J Physiol. 1984; 247:E157–65. https://doi.org/10.1152/ajpendo.1984.247.2.E157 [PubMed]
- 46. Gold PE. Glucose and age-related changes in memory. Neurobiol Aging. 2005 (Suppl 1); 26:60–64. https://doi.org/10.1016/j.neurobiolaging.2005.09.002 [PubMed]
- 47. Korol DL. Enhancing cognitive function across the life span. Ann N Y Acad Sci. 2002; 959:167–79. https://doi.org/10.1111/j.1749-6632.2002.tb02091.x [PubMed]
- 48. Mattson MP, Moehl K, Ghena N, Schmaedick M, Cheng A. Intermittent metabolic switching, neuroplasticity and brain health. Nat Rev Neurosci. 2018; 19:63–80. https://doi.org/10.1038/nrn.2017.156 [PubMed]
- 49. Bough KJ, Valiyil R, Han FT, Eagles DA. Seizure resistance is dependent upon age and calorie restriction in rats fed a ketogenic diet. Epilepsy Res. 1999; 35:21–28. https://doi.org/10.1016/S0920-1211(98)00125-9 [PubMed]
- 50. Gold PE, Korol DL. Forgetfulness during aging: an integrated biology. Neurobiol Learn Mem. 2014; 112:130–38. https://doi.org/10.1016/j.nlm.2014.03.005 [PubMed]
- 51. Randle PJ, Garland PB, Hales CN, Newsholme EA. The glucose fatty-acid cycle. Its role in insulin sensitivity and the metabolic disturbances of diabetes mellitus. Lancet. 1963; 1:785–89. https://doi.org/10.1016/S0140-6736(63)91500-9 [PubMed]
- 52. Hue L, Taegtmeyer H. The Randle cycle revisited: a new head for an old hat. Am J Physiol Endocrinol Metab. 2009; 297:E578–91. https://doi.org/10.1152/ajpendo.00093.2009 [PubMed]
- 53. Harvie M, Wright C, Pegington M, McMullan D, Mitchell E, Martin B, Cutler RG, Evans G, Whiteside S, Maudsley S, Camandola S, Wang R, Carlson OD, et al. The effect of intermittent energy and carbohydrate restriction v. daily energy restriction on weight loss and metabolic disease risk markers in overweight women. Br J Nutr. 2013; 110:1534–47. https://doi.org/10.1017/S0007114513000792 [PubMed]
- 54. Harvie MN, Pegington M, Mattson MP, Frystyk J, Dillon B, Evans G, Cuzick J, Jebb SA, Martin B, Cutler RG, Son TG, Maudsley S, Carlson OD, et al. The effects of intermittent or continuous energy restriction on weight loss and metabolic disease risk markers: a randomized trial in young overweight women. Int J Obes. 2011; 35:714–27. https://doi.org/10.1038/ijo.2010.171 [PubMed]
- 55. Hatori M, Vollmers C, Zarrinpar A, DiTacchio L, Bushong EA, Gill S, Leblanc M, Chaix A, Joens M, Fitzpatrick JA, Ellisman MH, Panda S. Time-restricted feeding without reducing caloric intake prevents metabolic diseases in mice fed a high-fat diet. Cell Metab. 2012; 15:848–60. https://doi.org/10.1016/j.cmet.2012.04.019 [PubMed]
- 56. Martin B, Ji S, Maudsley S, Mattson MP. “Control” laboratory rodents are metabolically morbid: why it matters. Proc Natl Acad Sci USA. 2010; 107:6127–33. https://doi.org/10.1073/pnas.0912955107 [PubMed]
- 57. Bocarsly ME, Fasolino M, Kane GA, LaMarca EA, Kirschen GW, Karatsoreos IN, McEwen BS, Gould E. Obesity diminishes synaptic markers, alters microglial morphology, and impairs cognitive function. Proc Natl Acad Sci USA. 2015; 112:15731–36. https://doi.org/10.1073/pnas.1511593112 [PubMed]
- 58. Kanoski SE, Davidson TL. Western diet consumption and cognitive impairment: links to hippocampal dysfunction and obesity. Physiol Behav. 2011; 103:59–68. https://doi.org/10.1016/j.physbeh.2010.12.003 [PubMed]
- 59. Stranahan AM, Norman ED, Lee K, Cutler RG, Telljohann RS, Egan JM, Mattson MP. Diet-induced insulin resistance impairs hippocampal synaptic plasticity and cognition in middle-aged rats. Hippocampus. 2008; 18:1085–88. https://doi.org/10.1002/hipo.20470 [PubMed]
- 60. Simon NW, LaSarge CL, Montgomery KS, Williams MT, Mendez IA, Setlow B, Bizon JL. Good things come to those who wait: attenuated discounting of delayed rewards in aged Fischer 344 rats. Neurobiol Aging. 2010; 31:853–62. https://doi.org/10.1016/j.neurobiolaging.2008.06.004 [PubMed]
- 61. Bañuelos C, Beas BS, McQuail JA, Gilbert RJ, Frazier CJ, Setlow B, Bizon JL. Prefrontal cortical GABAergic dysfunction contributes to age-related working memory impairment. J Neurosci. 2014; 34:3457–66. https://doi.org/10.1523/JNEUROSCI.5192-13.2014 [PubMed]
- 62. Beas BS, McQuail JA, Banuelos C, Setlow B, Bizon JL. Prefrontal cortical GABAergic signaling and impaired behavioral flexibility in aged F344 rats. Neuroscience. 2017; 345:274–86. https://doi.org/10.1016/j.neuroscience.2016.02.014 [PubMed]
- 63. Hernandez CM, McQuail JA, Schwabe MR, Burke SN, Setlow B, Bizon JL. Age-Related Declines in Prefrontal Cortical Expression of Metabotropic Glutamate Receptors that Support Working Memory. eNeuro. 2018; 5:5. https://doi.org/10.1523/ENEURO.0164-18.2018 [PubMed]
- 64. Barense MD, Fox MT, Baxter MG. Aged rats are impaired on an attentional set-shifting task sensitive to medial frontal cortex damage in young rats. Learn Mem. 2002; 9:191–201. https://doi.org/10.1101/lm.48602 [PubMed]
- 65. Hernandez CM, Vetere LM, Orsini CA, McQuail JA, Maurer AP, Burke SN, Setlow B, Bizon JL. Decline of prefrontal cortical-mediated executive functions but attenuated delay discounting in aged Fischer 344 × brown Norway hybrid rats. Neurobiol Aging. 2017; 60:141–52. https://doi.org/10.1016/j.neurobiolaging.2017.08.025 [PubMed]
- 66. Samson RD, Venkatesh A, Patel DH, Lipa P, Barnes CA. Enhanced performance of aged rats in contingency degradation and instrumental extinction tasks. Behav Neurosci. 2014; 128:122–33. https://doi.org/10.1037/a0035986 [PubMed]
- 67. Hernandez AR, Maurer AP, Reasor JE, Turner SM, Barthle SE, Johnson SA, Burke SN. Age-related impairments in object-place associations are not due to hippocampal dysfunction. Behav Neurosci. 2015; 129:599–610. https://doi.org/10.1037/bne0000093 [PubMed]
- 68. Hernandez AR, Reasor JE, Truckenbrod LM, Campos KT, Federico QP, Fertal KE, Lubke KN, Johnson SA, Clark BJ, Maurer AP, Burke SN. Dissociable effects of advanced age on prefrontal cortical and medial temporal lobe ensemble activity. Neurobiol Aging. 2018; 70:217–32. https://doi.org/10.1016/j.neurobiolaging.2018.06.028 [PubMed]
- 69. Johnson SA, Turner SM, Santacroce LA, Carty KN, Shafiq L, Bizon JL, Maurer AP, Burke SN. Rodent age-related impairments in discriminating perceptually similar objects parallel those observed in humans. Hippocampus. 2017; 27:759–76. https://doi.org/10.1002/hipo.22729 [PubMed]
- 70. Zhao L, Mao Z, Woody SK, Brinton RD. Sex differences in metabolic aging of the brain: insights into female susceptibility to Alzheimer’s disease. Neurobiol Aging. 2016; 42:69–79. https://doi.org/10.1016/j.neurobiolaging.2016.02.011 [PubMed]
- 71. Garriga-Canut M, Schoenike B, Qazi R, Bergendahl K, Daley TJ, Pfender RM, Morrison JF, Ockuly J, Stafstrom C, Sutula T, Roopra A. 2-Deoxy-D-glucose reduces epilepsy progression by NRSF-CtBP-dependent metabolic regulation of chromatin structure. Nat Neurosci. 2006; 9:1382–87. https://doi.org/10.1038/nn1791 [PubMed]
- 72. Masino SA, Rho JM. Mechanisms of Ketogenic Diet Action. In: Noebels JL, Avoli M, Rogawski MA, Olsen RW, Delgado-Escueta AV, editors. Jasper’s Basic Mechanisms of the Epilepsies. ed. Bethesda (MD): National Center for Biotechnology Information; 2012. https://doi.org/10.1093/med/9780199746545.003.0078 http://www.ncbi.nlm.nih.gov/books/NBK98219/.
- 73. Ruskin DN, Kawamura M, Masino SA. Reduced pain and inflammation in juvenile and adult rats fed a ketogenic diet. PLoS One. 2009; 4:e8349. https://doi.org/10.1371/journal.pone.0008349 [PubMed]
- 74. Ma W, Berg J, Yellen G. Ketogenic diet metabolites reduce firing in central neurons by opening K(ATP) channels. J Neurosci. 2007; 27:3618–25. https://doi.org/10.1523/JNEUROSCI.0132-07.2007 [PubMed]
- 75. Yudkoff M, Daikhin Y, Nissim I, Horyn O, Lazarow A, Luhovyy B, Wehrli S, Nissim I. Response of brain amino acid metabolism to ketosis. Neurochem Int. 2005; 47:119–28. https://doi.org/10.1016/j.neuint.2005.04.014 [PubMed]
- 76. Yudkoff M, Daikhin Y, Horyn O, Nissim I, Nissim I. Ketosis and brain handling of glutamate, glutamine, and GABA. Epilepsia. 2008 (Suppl 8); 49:73–75. https://doi.org/10.1111/j.1528-1167.2008.01841.x [PubMed]
- 77. Kashiwaya Y, Bergman C, Lee JH, Wan R, King MT, Mughal MR, Okun E, Clarke K, Mattson MP, Veech RL. A ketone ester diet exhibits anxiolytic and cognition-sparing properties, and lessens amyloid and tau pathologies in a mouse model of Alzheimer’s disease. Neurobiol Aging. 2013; 34:1530–39. https://doi.org/10.1016/j.neurobiolaging.2012.11.023 [PubMed]
- 78. Christensen K, Doblhammer G, Rau R, Vaupel JW. Ageing populations: the challenges ahead. Lancet. 2009; 374:1196–208. https://doi.org/10.1016/S0140-6736(09)61460-4 [PubMed]
- 79. Blazer D, Yaffe K, Liverman C. Committee on the Public Health Dimensions of Cognitive Aging; Board on Health Sciences Policy; Institute of Medicine. Cognitive Aging: Progress in Understanding and Opportunities for Action. Washington (DC): National Academies Press; 2015. https://www.ncbi.nlm.nih.gov/books/NBK316202/.
- 80. Cunnane S, Nugent S, Roy M, Courchesne-Loyer A, Croteau E, Tremblay S, Castellano A, Pifferi F, Bocti C, Paquet N, Begdouri H, Bentourkia M, Turcotte E, et al. Brain fuel metabolism, aging, and Alzheimer’s disease. Nutrition. 2011; 27:3–20. https://doi.org/10.1016/j.nut.2010.07.021 [PubMed]
- 81. Mattson MP, Arumugam TV. Hallmarks of Brain Aging: Adaptive and Pathological Modification by Metabolic States. Cell Metab. 2018; 27:1176–99. https://doi.org/10.1016/j.cmet.2018.05.011 [PubMed]
- 82. Gage FH, Kelly PA, Björklund A. Regional changes in brain glucose metabolism reflect cognitive impairments in aged rats. J Neurosci. 1984; 4:2856–65. https://doi.org/10.1523/JNEUROSCI.04-11-02856.1984 [PubMed]
- 83. Frith C, Dolan R. The role of the prefrontal cortex in higher cognitive functions. Brain Res Cogn Brain Res. 1996; 5:175–81. https://doi.org/10.1016/S0926-6410(96)00054-7 [PubMed]
- 84. Morris RG, Garrud P, Rawlins JN, O’Keefe J. Place navigation impaired in rats with hippocampal lesions. Nature. 1982; 297:681–83. https://doi.org/10.1038/297681a0 [PubMed]
- 85. Sloan HL, Good M, Dunnett SB. Double dissociation between hippocampal and prefrontal lesions on an operant delayed matching task and a water maze reference memory task. Behav Brain Res. 2006; 171:116–26. https://doi.org/10.1016/j.bbr.2006.03.030 [PubMed]
- 86. Passonneau JV, Gatfield PD, Schulz DW, Lowry OH. An enzymic method for measurement of glycogen. Anal Biochem. 1967; 19:315–26. https://doi.org/10.1016/0003-2697(67)90167-4 [PubMed]
- 87. Passonneau JV, Lauderdale VR. A comparison of three methods of glycogen measurement in tissues. Anal Biochem. 1974; 60:405–12. https://doi.org/10.1016/0003-2697(74)90248-6 [PubMed]
- 88. Shetty PK, Sadgrove MP, Galeffi F, Turner DA. Pyruvate incubation enhances glycogen stores and sustains neuronal function during subsequent glucose deprivation. Neurobiol Dis. 2012; 45:177–87. https://doi.org/10.1016/j.nbd.2011.08.002 [PubMed]