Introduction
Adult stem cells are responsible for the maintenance of tissue homeostasis during the life of the organism and this control is due to their specific characteristics of quiescence, self-renewal potential and capacity to differentiate to specialized cell lineages. Decline in stem cell function with age has been described in several compartments including muscle, brain, intestine, skin, and the hematopoietic system in mammals, and it is linked to decreased organ function and degenerative processes [1]. Stem cell aging is promoted by deregulation in different cell-intrinsic pathways, in cell-extrinsic signals that maintains microenvironment and niche homeostasis, as well as by systemic factors [1, 2]. The identification of critical underlying mechanisms responsible for the maintenance of the function of the adult stem cells or the delay of their exhaustion would allow delaying the tissue deterioration associated with age, an idea with great therapeutic potential.
p38 mitogen-activated protein kinase (p38MAPK) is a relevant sensor of multiple types of intrinsic and extrinsic stresses and controls key processes of cell homeostasis such as proliferation, death, self-renewal and differentiation [3]. The activation of p38MAPK signaling triggers the exhaustion of stem cells in different compartments, such as hematopoietic [4] lung [5] or muscle [6, 7]. In the brain, the activity of p38MAPK plays regulatory roles during embryo development and postnatal stage in neural stem/progenitor cells (NSC). However, the results obtained so far are disparate and show both positive and negative effects of p38MAPK signaling in NSC function [8–14].
NSCs reside in the subventricular zone (SVZ) of the lateral ventricle and the subgranular zone of the dentate gyrus (DG) in the hippocampus in the adult mammalian brain. NSCs give rise to intermediate progenitor cells, which divide generating immature neurons, that subsequently integrate into the neural networks within the neurons incorporated in the olfactory bulb or DG, respectively. The new neurons that incorporate into the neuronal circuitry are responsible for olfactory ability, memory, learning and behavior [15]. With age, there is a decline in functional NSCs and this correlates with the presence of lower neurogenesis, and the diminishment in the production of new neurons limit plasticity and repair of the brain and underlies age-related cognitive decline [16, 17]. In this work we characterized the impact of p38MAPK in NSCs in SVZ.
Results and Discussion
We first determined the activity of p38MAPK in young (2 month-old) and aged (≥ 2 year-old) SVZ from C57BL/6J mice. Immunofluorescence showed that p38MAPK phosphorylated in the activating residues (P-p38MAPK) was low in the cells along the SVZ in young mice and significantly increased in over 2 year-old animals (Figure 1A, 1B). Moreover, the expression of all p38MAPK family members (MAPK11 (β), MAPK12 (γ), MAPK13 (δ) and MAPK14 (α)) was elevated in ex vivo SVZ cells from aged compared to young mice (Figure 1C). A recent study detected diminishment of P-p38MAPK and total p38MAPK with age, particularly in the DG but also in SVZ [13]. In this study, younger animals (3 weeks vs 1.5 year-old mice) were analyzed than in ours (2 months vs over 2 year-old). Since it has been shown that the expression of total and phosphorylated p38MAPK decreases during embryo development and, at least, up to postnatal day14, where levels are undetectable [10], the differences between studies might be explained by the different age of the animals.
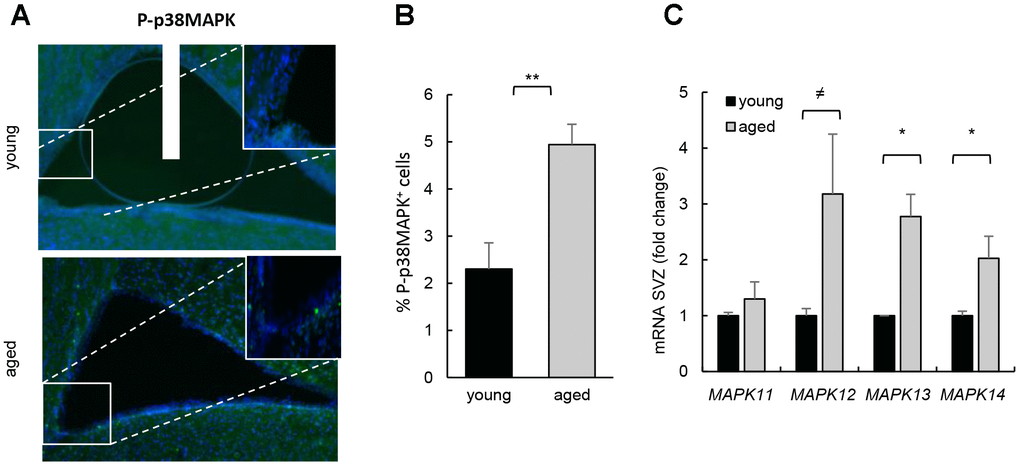
Figure 1. Increased p38MAPK activity in SVZ neurogenic niche with aging. (A) Representative immunofluorescence for P-p38MAPK in SVZ of young (2 month-old) and aged (over 24 month-old) C57BL/6J mice (n≥2). (B) Quantification of number of P-p38MAPK positive cells in this region. (C) MAPK11, MAPK12, MAPK13 and MAPK14 mRNA levels in SVZ of young (2 month-old) and aged (over 24 month-old) C57BL/6J mice (n≥4).
Next, we cultured neurospheres harvested from SVZ area of mouse of different ages (2 month-old vs 2 years) and observed, confirming previous studies, that aged cells presented decreased capability of neurosphere formation (Figure 2A), which correlated with lower levels of SOX2 stem cell regulator and higher p16Ink4a expression, gene related to cell cycle and senescence (Figure 2B). Interestingly, neurospheres derived from aged mice contained higher levels of P-p38MAPK (Figure 2B) and these cells also showed higher mRNA levels of all p38MAPK family members (Figure 2C). Together, our data show that the increase in p38MAPK activity coincides with the decline in the activity of NSCs in vitro and in vivo. On the contrary, the previous cited study detected decreased p38 mRNA expression, and lower p38α and P-p38MAPK immunoreactivity in SVZ neurospheres isolated from 6 month-old mice compared to 6 weeks old [13].
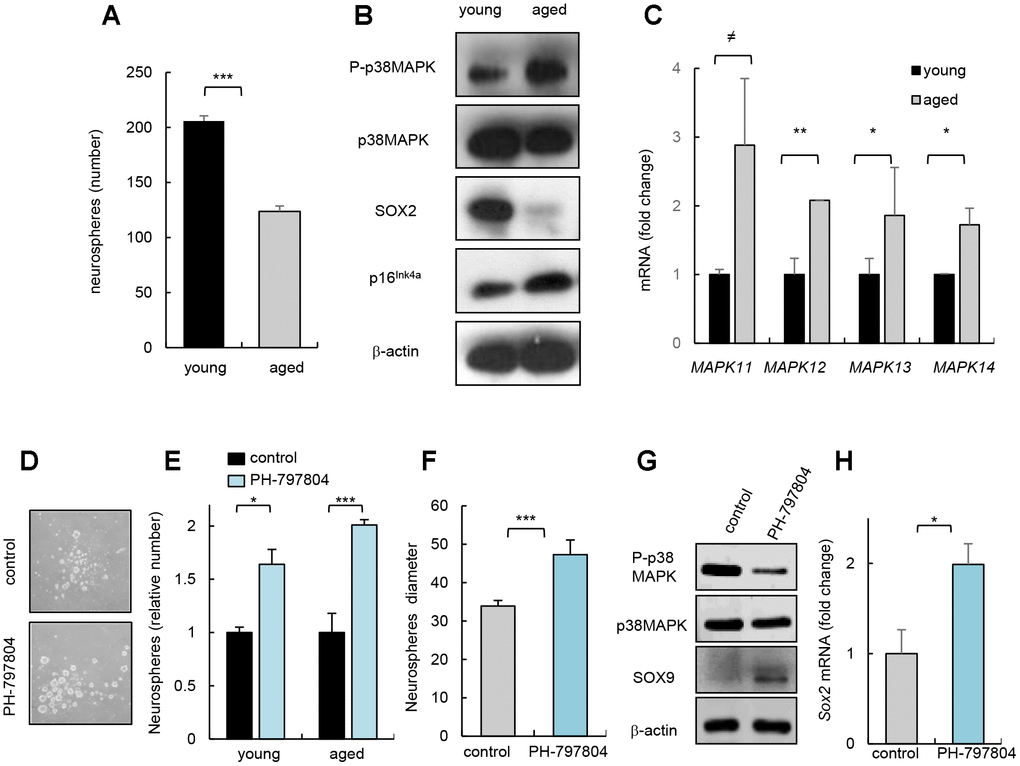
Figure 2. p38MAPK activity regulates NSC/progenitor aging in vitro. (A) Quantification of neurospheres from young (2 month-old) and aged (over 24 month-old) C57BL/6J mice (n=3). (B) P-p38MAPK, SOX2 and p16Ink4a expression in neurospheres derived from animals at the indicated ages (n=3). (C) Analysis of MAPK isoforms in neurospheres. (D) Representative image and (E) quantification of neurospheres derived from the SVZ of young and aged C57BL/6J mice treated with p38MAPK inhibitor (PH-797804) or control (DMSO) (n=4). (F) Quantification of the diameter of secondary neurospheres derived from aged mice treated with PH-797804 or control (n=4). (G) Representative western blot of P-p38MAPK, p38MAPK, SOX9 and ß-actin in 2ry neurospheres from aged mice (n=2). (H) Quantification of Sox2 mRNA levels in aged cells (n=3).
Previous studies observed that pharmacological inhibition of p38MAPK in NSC/progenitors derived from embryos or up to 4 month-old adult mice protects against apoptosis [8], increases proliferation [10], enhances self-renewal and differentiation potential [9, 18], and promotes migration [11] in vitro. In contrast, 6 month-old p38α conditional knockout mice under the control of the Nestin gene promoter formed lower and smaller number of neurospheres than controls in vitro and reduced the proliferation of progenitors in vivo [13]. Next, we tested whether inhibition of p38MAPK could prevent NSC/progenitor aging. For this, we cultured cells from SVZ of young and aged mice with PH-797804, a selective p38MAPK inhibitor [19]. We found that young and aged cells incubated with the p38MAPK inhibitor PH-797804 formed higher number of neurospheres in both ages (Figure 2D, 2E). The elevation in neurosphere formation ability of aged cells correlated with a larger size (Figure 2F), and a a reduction in P-p38MAPK as well as higher SOX9 and Sox2 levels (Figure 2G, 2H), supporting that decreased p38MAPK rejuvenates aged NSC/progenitor function.
Long term-cultured cells in vitro share multiple characteristics of physiological aging [20]. To further test the impact of p38MAPK inhibition in NSC/progenitor aging, we serially passaged neurosphere cultures. Cells after 7 passages (7ry) generated smaller and lower number of neurospheres than after passage 2 (2ry) (Figure 3A–3C). This correlated at molecular level with decreased SOX2 and increased p16Ink4a expression (Figure 3D). In this context, cells from 7ry passage also displayed increased P-p38MAPK (Figure 3D). Moreover, 7ry cells treated with p38MAPK inhibitor formed 3 times more neurospheres than non-treated control cells (Figure 3E). Treatment with the inhibitor also promoted a significant shift to larger size of neurospheres (Figure 3F) and elevation of Sox2 and Sox9 stem cell genes, and decrease of Mkp1/Dusp1 differentiation marker [21] (Figure 3G). These results further show that inhibition of intrinsic p38MAPK activity restores NSC activity and, together with the above-indicated studies, highlight the relevance of p38MAPK signaling in NSC homeostasis. In line with this idea, single cell transcriptomic analysis revealed that MAPK cascade related genes are altered in NSC/progenitor subpopulations with aging [22]. Moreover, our results also indicate that the role of p38MAPK in NSC/progenitor activity and aging, in contrast to stem cell populations in other tissues, might be more complex and may depend on a fine regulation of the timing and levels of its activity, the interactions within the neurogenic niche [14], and also with other signaling pathways. In this regard, Wnt pathway might be involved [13]. Moreover, EGF signaling might be associated with p38MAPK, since p38MAPK function is a crucial mediator of this pathway, and it is well known that EGF promotes NSC proliferation in vitro and in vivo [23] and decreased EGF receptor signaling associates with aging in the SVZ niche [24].
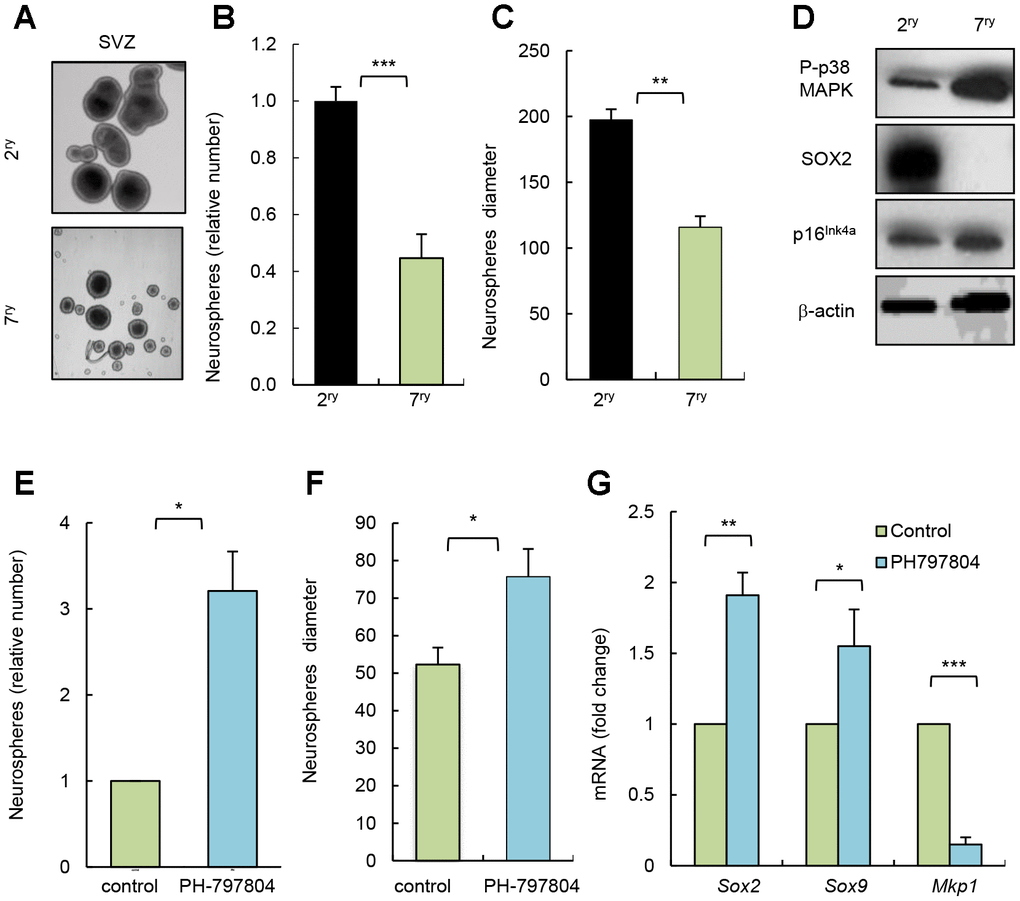
Figure 3. Pharmacological inhibition of p38MAPK rejuvenates NSC function in vitro. (A) Representative image and (B, C) quantification of number of neurospheres and their diameter at passage 2 (2ry) and 7 (7ry) from SVZ C57BL/6J mice (n=3). (D) Representative western blot of P-p38MAPK, SOX2, p16Ink4a and ß-actin at indicated conditions (n=3). (E) Relative number of neurospheres formed from passage 7 cells treated with p38MAPK inhibitor (PH-797804) or control (DMSO) (n=3). (F) Quantification of the diameter of neurospheres from 7ry passage treated with PH-797804 or control (n=3). (G) Sox2, Sox9 and Mkp1 expression in neurospheres maintained for passages (n=3).
In summary, we show that p38MAPK activity is elevated in SVZ neurogenic niche of aged mice in vivo and in neurospheres derived from aged cells in vitro. Moreover, pharmacological inhibition of p38MAPK in aged neurospheres rejuvenates NSC/progenitor activity in vitro. Ultimately, our data postulate p38MAPK activity as an intrinsic regulator of NSC aging.
Materials and Methods
Mice handling and ethics statement
The C57BL/6J (Jackson Laboratory) mice were housed in specific pathogen-free barrier areas of the Biodonostia Health Research Institute. Mice were maintained and handled in compliance with the animal research regulations specified in the European Communities Directive [2010/63/EU]. All Animal studies were approved by the Biodonostia Health Research Institute Animal Care Committee.
Tissue immunofluorescence
Coronal serial sections of 50 μm were collected via SM2010 R Sliding Microtome (Leica), and selected brain sections were blocked with 10% donkey serum and incubated with anti-P-p38MAPK (1:200; rabbit, Cell Signalling) overnight at 4 °C. Nuclei were stained with DAPI (Sigma). Images were acquired with an inverted confocal light scanning microscope (CLSM 510Meta, Zeiss) with a 63x objective (NA 1.3, aprochromat) in sequential mode with 1024 x 1024 scan size. Processing and analysis was performed on the maximal intensity projection of the z-stack, and selection of the area was accomplished using the nucleus positives areas. Fiji public domain, open source software was used.
RNA analysis
Total RNA was extracted with Trizol (Life Technologies). Reverse transcription was performed using random priming and Superscript Reverse Transcriptase (Life Technologies), according to the manufacturer’s guidelines. Quantitative real-time PCR was performed using Absolute SYBR Green mix (Thermo Scientific) in an ABI PRISM 7500 thermocycler (Applied Biosystems).
Western blot analysis
Immunoblots were performed following standard procedures. Equal amounts of protein (20 μg) were separated on 15% SDS polyacrylamide gels and blotted onto nitrocellulose membranes (BioRad). Primary antibodies were SOX9 (Millipore), SOX2 (Millipore), P-p38MAPK (Cell Signalling), total p38MAPK (Santa Cruz), p16Ink4a (Santa Cruz) and β-actin (Sigma). Secondary antibodies were HRP-linked anti-mouse or rabbit (DAKO). Detection was performed by chemiluminescence using ECL (Amersham).
Neurosphere cultures
Isolation, culture, and assays of NSCs were carried out as previously described [25]. Briefly, NSCs were isolated from the mouse SVZ and grown for 10 days in DMEM/F12 growth medium (Sigma) in the presence of EGF (20 ng/mL, Sigma) and FGF-2 (20 ng/mL, Gibco). Primary neurospheres after being counted, were treated with accutase (Sigma) for 5 min, mechanically dissociated to a single-cell suspension and re-plated in growth medium containing EGF and FGF for 10 days (secondary neurospheres, 2ry). Serial passage experiment was done repeating this methodology up to seven passages (7ry neurospheres).
Statistics
Data are presented as mean values ± S.E.M., with the number of experiments (n) in parentheses. Unless otherwise indicated, statistical significance (p-values) was calculated using the Student’s t -test. Asterisks (*, **, and ***) indicate statistical significance (p < 0.05, p < 0.01, and p < 0.001, respectively).
Author Contributions
L.M-C and O.A performed all the in vitro and in vivo experiments. IL helped to perform and analyzed confocal images. A.M directed the project, contributed to data analysis and wrote the manuscript.
Acknowledgments
L.M-C and O.A were recipients of a predoctoral fellowship from the Department of Education, University and Research of the Basque Government.
Conflicts of Interest
The authors declare no competing financial interests.
Funding
This work is supported by grants from the Instituto Salud Carlos III and FEDER funds (CP16/00039, PI16/01580, CP19/00085) and Diputacion Foral Gipuzkoa.
References
- 1. Schultz MB, Sinclair DA. When stem cells grow old: phenotypes and mechanisms of stem cell aging. Development. 2016; 143:3–14. https://doi.org/10.1242/dev.130633 [PubMed]
- 2. DeCarolis NA, Kirby ED, Wyss-Coray T, Palmer TD. The Role of the Microenvironmental Niche in Declining Stem-Cell Functions Associated with Biological Aging. Cold Spring Harb Perspect Med. 2015; 5:a025874. https://doi.org/10.1101/cshperspect.a025874 [PubMed]
- 3. Cuadrado A, Nebreda AR. Mechanisms and functions of p38 MAPK signalling. Biochem J. 2010; 429:403–17. https://doi.org/10.1042/BJ20100323 [PubMed]
- 4. Ito K, Hirao A, Arai F, Takubo K, Matsuoka S, Miyamoto K, Ohmura M, Naka K, Hosokawa K, Ikeda Y, Suda T. Reactive oxygen species act through p38 MAPK to limit the lifespan of hematopoietic stem cells. Nat Med. 2006; 12:446–51. https://doi.org/10.1038/nm1388 [PubMed]
- 5. Ventura JJ, Tenbaum S, Perdiguero E, Huth M, Guerra C, Barbacid M, Pasparakis M, Nebreda AR. p38alpha MAP kinase is essential in lung stem and progenitor cell proliferation and differentiation. Nat Genet. 2007; 39:750–58. https://doi.org/10.1038/ng2037 [PubMed]
- 6. Bernet JD, Doles JD, Hall JK, Kelly Tanaka K, Carter TA, Olwin BB. p38 MAPK signaling underlies a cell-autonomous loss of stem cell self-renewal in skeletal muscle of aged mice. Nat Med. 2014; 20:265–71. https://doi.org/10.1038/nm.3465 [PubMed]
- 7. Cosgrove BD, Gilbert PM, Porpiglia E, Mourkioti F, Lee SP, Corbel SY, Llewellyn ME, Delp SL, Blau HM. Rejuvenation of the muscle stem cell population restores strength to injured aged muscles. Nat Med. 2014; 20:255–64. https://doi.org/10.1038/nm.3464 [PubMed]
- 8. Cheng A, Chan SL, Milhavet O, Wang S, Mattson MP. p38 MAP kinase mediates nitric oxide-induced apoptosis of neural progenitor cells. J Biol Chem. 2001; 276:43320–27. https://doi.org/10.1074/jbc.M107698200 [PubMed]
- 9. Yang SR, Kim SJ, Byun KH, Hutchinson B, Lee BH, Michikawa M, Lee YS, Kang KS. NPC1 gene deficiency leads to lack of neural stem cell self-renewal and abnormal differentiation through activation of p38 mitogen-activated protein kinase signaling. Stem Cells. 2006; 24:292–98. https://doi.org/10.1634/stemcells.2005-0221 [PubMed]
- 10. Sato K, Hamanoue M, Takamatsu K. Inhibitors of p38 mitogen-activated protein kinase enhance proliferation of mouse neural stem cells. J Neurosci Res. 2008; 86:2179–89. https://doi.org/10.1002/jnr.21668 [PubMed]
- 11. Hamanoue M, Morioka K, Ohsawa I, Ohsawa K, Kobayashi M, Tsuburaya K, Akasaka Y, Mikami T, Ogata T, Takamatsu K. Cell-permeable p38 MAP kinase promotes migration of adult neural stem/progenitor cells. Sci Rep. 2016; 6:24279. https://doi.org/10.1038/srep24279 [PubMed]
- 12. Cortez I, Bulavin DV, Wu P, McGrath EL, Cunningham KA, Wakamiya M, Papaconstantinou J, Dineley KT. Aged dominant negative p38α MAPK mice are resistant to age-dependent decline in adult-neurogenesis and context discrimination fear conditioning. Behav Brain Res. 2017; 322:212–222. https://doi.org/10.1016/j.bbr.2016.10.023 [PubMed]
- 13. Kase Y, Otsu K, Shimazaki T, Okano H. Involvement of p38 in Age-Related Decline in Adult Neurogenesis via Modulation of Wnt Signaling. Stem Cell Reports. 2019; 12:1313–28. https://doi.org/10.1016/j.stemcr.2019.04.010 [PubMed]
- 14. Moreno-Cugnon L, Revuelta M, Arrizabalaga O, Colie S, Moreno-Valladares M, Jimenez-Blasco D, Gil-Bea F, Llarena I, Bolaños JP, Nebreda AR, Matheu A. Neuronal p38α mediates age-associated neural stem cell exhaustion and cognitive decline. Aging Cell. 2019; 18:e13044. https://doi.org/10.1111/acel.13044 [PubMed]
- 15. Bond AM, Ming GL, Song H. Adult Mammalian Neural Stem Cells and Neurogenesis: Five Decades Later. Cell Stem Cell. 2015; 17:385–95. https://doi.org/10.1016/j.stem.2015.09.003 [PubMed]
- 16. Encinas JM, Sierra A. Neural stem cell deforestation as the main force driving the age-related decline in adult hippocampal neurogenesis. Behav Brain Res. 2012; 227:433–39. https://doi.org/10.1016/j.bbr.2011.10.010 [PubMed]
- 17. Capilla-Gonzalez V, Herranz-Pérez V, García-Verdugo JM. The aged brain: genesis and fate of residual progenitor cells in the subventricular zone. Front Cell Neurosci. 2015; 9:365. https://doi.org/10.3389/fncel.2015.00365 [PubMed]
- 18. Yoshioka K, Namiki K, Sudo T, Kasuya Y. p38α controls self-renewal and fate decision of neurosphere-forming cells in adult hippocampus. FEBS Open Bio. 2015; 5:437–44. https://doi.org/10.1016/j.fob.2015.05.001 [PubMed]
- 19. Colié S, Sarroca S, Palenzuela R, Garcia I, Matheu A, Corpas R, Dotti CG, Esteban JA, Sanfeliu C, Nebreda AR. Neuronal p38α mediates synaptic and cognitive dysfunction in an Alzheimer’s mouse model by controlling β-amyloid production. Sci Rep. 2017; 7:45306. https://doi.org/10.1038/srep45306 [PubMed]
- 20. Sherr CJ, DePinho RA. Cellular senescence: mitotic clock or culture shock? Cell. 2000; 102:407–10. https://doi.org/10.1016/S0092-8674(00)00046-5 [PubMed]
- 21. Arrizabalaga O, Moreno-Cugnon L, Auzmendi-Iriarte J, Aldaz P, Ibanez de Caceres I, Garros-Regulez L, Moncho-Amor V, Torres-Bayona S, Pernía O, Pintado-Berninches L, Carrasco-Ramirez P, Cortes-Sempere M, Rosas R, et al. High expression of MKP1/DUSP1 counteracts glioma stem cell activity and mediates HDAC inhibitor response. Oncogenesis. 2017; 6:401. https://doi.org/10.1038/s41389-017-0003-9 [PubMed]
- 22. Shi Z, Geng Y, Liu J, Zhang H, Zhou L, Lin Q, Yu J, Zhang K, Liu J, Gao X, Zhang C, Yao Y, Zhang C, Sun YE. Single-cell transcriptomics reveals gene signatures and alterations associated with aging in distinct neural stem/progenitor cell subpopulations. Protein Cell. 2018; 9:351–64. [PubMed]
- 23. Reynolds BA, Weiss S. Clonal and population analyses demonstrate that an EGF-responsive mammalian embryonic CNS precursor is a stem cell. Dev Biol. 1996; 175:1–13. https://doi.org/10.1006/dbio.1996.0090 [PubMed]
- 24. Enwere E, Shingo T, Gregg C, Fujikawa H, Ohta S, Weiss S. Aging results in reduced epidermal growth factor receptor signaling, diminished olfactory neurogenesis, and deficits in fine olfactory discrimination. J Neurosci. 2004; 24:8354–65. https://doi.org/10.1523/JNEUROSCI.2751-04.2004 [PubMed]
- 25. Carrasco-Garcia E, Arrizabalaga O, Serrano M, Lovell-Badge R, Matheu A. Increased gene dosage of Ink4/Arf and p53 delays age-associated central nervous system functional decline. Aging Cell. 2015; 14:710–14. https://doi.org/10.1111/acel.12343 [PubMed]



