Introduction
The naked mole-rat (Heterocephalus glaber) is arguably unique among mammals in the extent of its social insect-like behaviour, first highlighted by Jarvis [1]. In more recent years it has also gained prominence as an important non-model organism for the study of longevity [2–5], and other extraordinary aspects of its biology that result from adaptations to the challenges of an extreme subterranean niche [6, 7]. Although little more than mouse-sized (mean body mass is around 35g), naked mole-rats are the longest-lived rodent with a maximum possible lifespan exceeding 31 years [5]. Furthermore, they resist all of the normal signs of ageing and are thus emerging as an important non-model organism for the study of longevity and healthspan. These unusual mammals uniquely do not show an increase in age-specific hazard of mortality, in defiance of Gompertz’s law [8]. They also show no decreased physiological capacity with age, maintaining vascular elasticity, cardiac function, gastrointestinal function, glucose tolerance, and reproductive capacity well into the third decade of life [2, 9, 10], and resist sarcopenia, the progressive loss of skeletal muscle with age [11]. Because of these traits, the naked mole-rat is of particular significance and interest in the study of biomarkers for ageing. Recently, breakthroughs in this field have indicated the presence of ‘epigenetic clocks’, mainly in studies of human and mouse tissues [12–15], but also in canids [16, 17], and humpback whales [18]. These are based on the temporal accumulation of DNA methylations at specific ageing-associated differentially methylated positions (aDMPs). Such CpG sites at which DNA methylation dynamics show significant correlations with age can potentially enable accurate age estimates for tissues across the life span of an individual, and it has been shown in humans that most tissues and organs from the same body exhibit broadly similar epigenetic ages [19].
In an analysis of aDMPs in six different mammals, including long and short-lived dog breeds, Lowe et al. [17] found a strong negative relationship between rate of change of methylation levels at aDMPs and lifespan. This study also identified 30 aDMPs in the naked mole-rat liver, that clustered in 12 different targeted aDMP regions, providing a potential molecular readout for aging in this species. A challenge for research on long-lived non-model organisms such as the naked mole-rat, especially when studying wild caught animals, is the determination of age (in the absence of birth and life history data). In some cases, this problem also applies to captive populations where pedigree data is not available. Crude categorical estimates are sometimes possible based on tooth wear, for example in other African mole-rats (Cryptomys and Fukomys; [20]), but these only differentiate relative age classes, rather than attempting to assign an absolute age estimate. Here, we aim to (i) consolidate and validate these initial results for the naked mole-rat, examining further samples across a wide age range in both liver and skin, and (ii) develop a method that will enable naked mole-rat aDMPs to be used to estimate age in animals of unknown provenance.
Results
To create a method for predicting the age of naked mole-rats based on changes in methylation, we initially sampled 24 naked mole-rat livers spanning an age range from 39 weeks to 1,144 weeks (approximately ten months to 22 years; Supplementary Table 1). We performed a targeted sequence-based method to determine the methylation of individual CpGs across the genome. These targeted regions were selected by mapping existing regions within the human genome, known to be associated with age, to the naked mole-rat genome (https://www.ncbi.nlm.nih.gov/pubmed/25172923). In total we selected 12 regions/primer pairs spanning a total of 51 different CpGs (Supplementary Table 2). Of these 51 different sites 23 (45%) were found to be associated with age (p-value < 0.05; Supplementary Table 3). The top hit (JH602136:8746439) showed a strong correlation (R=0.88) with age (Figure 1A) with a root mean square error of 541.95 weeks.
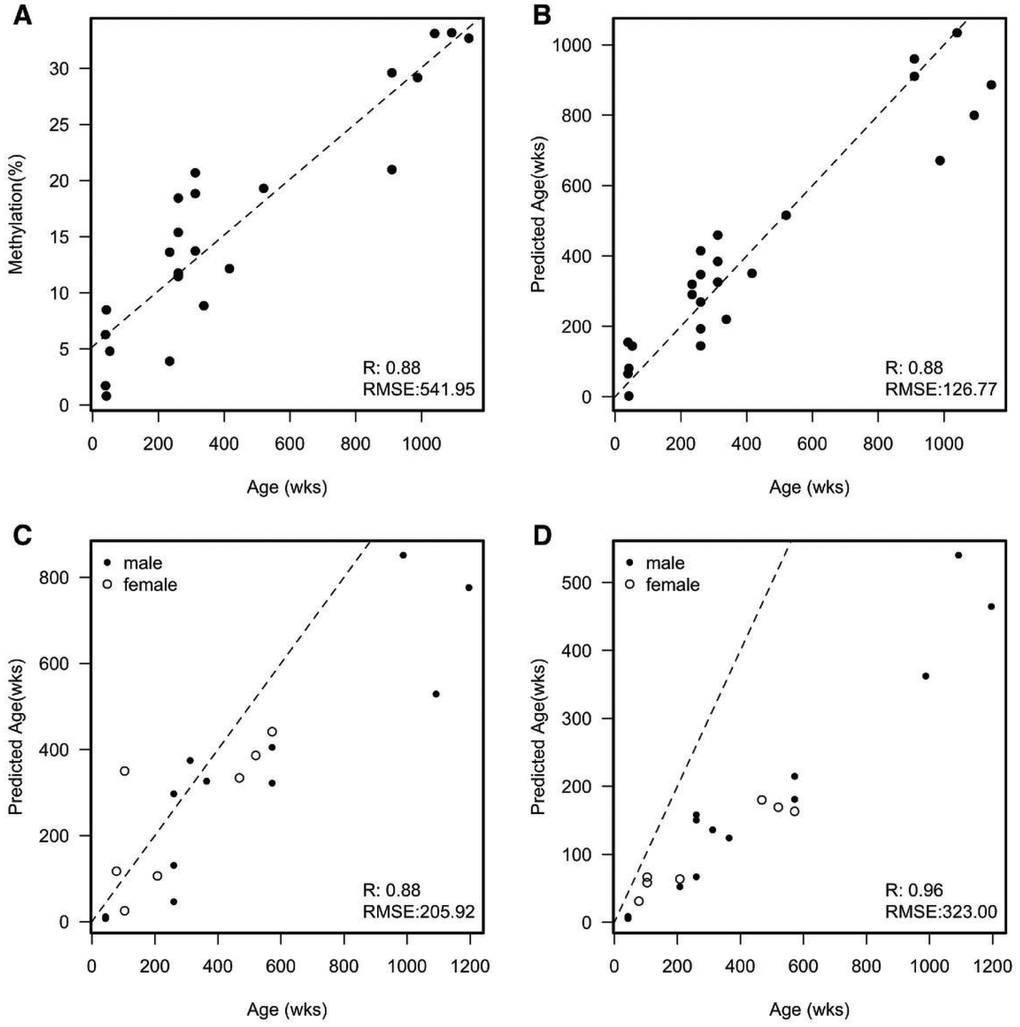
Figure 1. (A) Example of a single CpG that correlates with the age of each naked mole-rat. Dashed line is a fitted linear model; RMSE = Root Mean Square Error. Mean absolute deviation (MAD) = 412.282 and median absolute error (MAE) = 400.198; (B) A scatterplot of the predicted age of each naked mole-rat liver sample against the actual age in weeks from an initial sample set. The predicted age was calculated by removing the sample and fitting to the remaining samples. Dashed line represents y=x (e.g. perfect prediction). MAD = 96.882 and MAE = 120.840; (C) A scatter plot of the predicted age of a second set of naked mole-rat livers against the actual age in weeks. Dashed line represents y=x (e.g. perfect prediction). MAD = 118.941 and MAE = 126.883; (D) A scatter plot of the predicted age of skin samples against the actual age in weeks. Dashed line represents y=x (e.g. perfect prediction). MAD = 219.113 and MAE = 252.874.
Utilising a similar methodology to Horvath [19] in producing his multi-tissue age prediction in humans, we built a predictor of age using the 23 sites that showed an association with age in the naked mole-rat. This uses elastic net regression which incorporates all 23 CpGs in a multivariate analysis. To validate this model, we performed a leave one out cross validation in which we remove a single sample to fit the parameters of our model and then predict the age of this single sample. We then repeated this until we had removed each sample and predicted its age. Utilising multiple CpGs in the model showed a slight improvement in correlation (0.89) but a large decrease in the RMSE (134.21) (Figure 1B). To further test whether this model could be useful, we sampled a further 19 livers with an age range 43 to 1,196 weeks (approximately ten months to 23 years; Supplementary Table 1). Using the model built from the initial 24 samples, we predicted the age of these new samples. As expected, given this was a different batch of samples both the R and RMSE dropped albeit only by a small amount (Figure 1C). Given the multi-tissue nature of some methylation changes within humans, we decided to test our model using a different set of tissue samples. We profiled a further 20 skin samples with the same age range of that of the 19 liver samples (19 of the 20 skin samples were from the same animals as the livers). Interestingly we found a striking correlation between the predicted age of these samples versus their actual age (R=0.93), however, this correlation was not along the identity line but showed a much lower predicted age than actual age suggesting that skin tissue ages slower than the liver. This change in rate accounts for the larger RMSE. To make this approach useable for other researchers we have provided the primer sequences that span the CpGs, and an online tool that can be downloaded as an NMR age predictor based on these aDMPs (https://github.com/ralowe/NMRAgePrediction; Supplementary Material).
Discussion
In an extensive study of naked mole-rats, we identify 23 CpGs in liver tissue that are significantly associated with age, and build a predictor of age model with a root mean squared error of 166 weeks, or approximately 10% of the published maximum possible lifespan (more than 31 years/1612 weeks; [5]). In profiling skin samples from the same individuals, we also found a striking correlation between the predicted age of these samples versus their actual age. Interestingly, when compared to the liver samples, this correlation showed a lower predicted age than actual age, suggesting that skin tissue ages more slowly than the liver in naked mole-rats. In humans, Horvath [19], found that most tissues from the same body exhibit broadly similar epigenetic ages. Other studies have showed that, while some age-associated changes in methylation are shared across tissues, others may be tissue-specific in humans [21, 22], mice (lung, liver, spleen and colon; [23]) and rats (liver and visceral adipose tissues; [24]). The relative proportions of age-associated epigenetic changes in the form of DNA methylations that are tissue-specific, compared to the amount that is general and non-specific remains a matter of debate. Zhu et al. [25] estimate that more than 70% is due to shared epigenetic drift across tissues, with the remainder down to tissue-specific and functionally important changes. The differences in ageing between liver and skin seen in the naked mole-rat may perhaps reflect the increased metabolic activity of the liver when compared to skin, and the fact that naked mole-rats are not exposed to UV radiation in captive or wild colonies. A variety of naked mole-rat tissues have been shown to resist the normal signs of senescence (see Lewis and Buffenstein [5] for review), including the liver where proteasome function increased or was maintained with age [26], and there was no accumulation of oxidative damage with age [27]. Although the ageing skin has not been investigated fully in naked mole-rats, their fibroblasts in culture are resistant to an array of toxins and stressors (compared with mice), including heavy metals, heat, and chemotherapeutic/DNA-damaging compounds [28, 29, 5]. Further, MacRae et al. [30] report that the naked mole-rat liver has higher expression of DNA repair genes, with significant upregulation of several DNA repair signaling pathways compared with the mouse. These observations of increased DNA repair in the naked mole-rat perhaps support the proposal of Field et al. [31], that long term maintenance of a steady state in dynamic chromatin (“chromostasis”) may slow the ticking of the epigenetic clock in long-lived species.
Horvath [19] suggests that understanding how and why the estimated epigenetic age differs across a group of individuals of the same chronological age could help to determine the impact of endogenous or exogenous stress factors on biological ageing. Humans suffering from Werner’s Syndrome, a condition that produces clinical signs of accelerated ageing also had associated epigenetic age acceleration and thus an increased DNA methylation age [32]. Other studies have shown that in humans, lifestyle factors including diet and physical activity, can have a positive association with epigenetic age acceleration i.e. a healthy lifestyle associates with a reduced epigenetic age [33]. Naked mole-rats are apparently unique among mammals in that they defy Gompertz’s Law in not showing increased risk of mortality with age [8]. Furthermore, there are no apparent sex or reproductive status differences in their maximum possible lifespan [3]. It is interesting that some variance in the percentage methylation is evident for liver tissue in Figure 1A, perhaps suggesting some differences in biological versus chronological age among these mole-rat samples.
The potential for a forensic use for epigenetic clocks to determine the unknown age of a sample or individual has also been noted for humans by Wagner [34]. Our model and online tool will enable the prediction of age in wild caught naked mole-rats and captive animals of unknown age. Given the evidence for associations between epigenetic age acceleration and factors that may influence longevity in humans, and that most clocks have focussed on application in humans [14] our study will be invaluable for further mechanistic and functional studies of mammalian ageing in non-model organisms such as the naked mole-rat.
Materials and Methods
Naked mole-rats were maintained in the Biological Services Unit at Queen Mary University of London, and tissues obtained from post-mortem specimens from animals free from disease in compliance with national (Home Office) and institutional procedures and guidelines. Because sample collection was from post-mortem material, additional local ethical approval was not required for this study. Samples of abdominal skin and liver were snap frozen in liquid nitrogen and transferred for storage at -80°C. Full details of animals and samples are provided in Supplementary Table 1.
DNA extraction
DNA was extracted from tissues using the PureLink™ Genomic DNA kit (Thermo Fisher, Cat. K182002), as per the manufacturer’s instructions. Tissues were digested overnight at 55 °C using 180 μl PK buffer and 20 μl PK enzyme from the kit. DNA concentration was quantified using a High Sensitivity DNA Qubit® assay (Life Technologies, Cat. Q32851).
Bisulfite PCR sequencing (Bis-PCR-Seq)
DNA from tissues was diluted to a concentration of 11 ng/μl and 45 μl of each sample was used for generation of targeted bisulfite sequencing data by the Genome Centre Facility at the Blizard Institute, Queen Mary University of London. DNA was bisulfite converted in a 96 well plate format using the EZ-96 DNA Methylation™ Kit (Zymo, Cat. D5003). Target amplification was performed using the FastStart High Fidelity PCR System, dNTPack (Sigma-Aldrich, Cat. 4738284001) in the 48.48 layout on the Fluidigm® C1 system (Fluidigm®, USA), a microfluidics platform. Library preparation was performed using the same kit including 4 μl of Access Array Barcode Library Primer and 1 μl of PCR product diluted 1:100. Libraries were sequenced with Illumina MiSeq sequencing using v2 chemistry (150 bp, paired-end). Primers used for targeted bisulfite sequencing are listed in Supplementary Table 2.
Raw FASTQ files were mapped to the reference hetGla2 using BISMARK (v0.16.3) [35] and Bowtie2 (v2.2.8) [36]. Reads that mapped outside of the targeted regions were discarded from analyses, and methylated and unmethylated counts for each CpG were calculated using the custom C++ program (https://bitbucket.org/ lowelabqmul/methylation-extractor/src/master/). Those CpGs with a coverage < 50× were also discarded from analyses. Data created for this manuscript are available from GEO with accession number GSE86059 (sample set 1) and GSE137957 (sample set 2), and provided in Supplementary File 1.
Statistical analysis
The analysis model used elastic net regression incorporating all 23 CpGs in a multivariate analysis. Linear models and scatterplots were produced using R statistical software [37]. We have developed an online tool for analysis of similar NMR data (available at https://github.com/ralowe/NMRAgePrediction), with further information provided in the Supplementary Material).
Acknowledgments
We thank James Crowe at QMUL for IT help and support.
Conflicts of Interest
There are no conflicts of interest to be declared.
Funding
This research utilised Queen Mary’s Apocrita HPC facility, supported by QMUL Research-IT. https://doi.org/10.5281/zenodo.438045.
References
- 1. Jarvis JU. Eusociality in a mammal: cooperative breeding in naked mole-rat colonies. Science. 1981; 212:571–73. https://doi.org/10.1126/science.7209555 [PubMed]
- 2. Buffenstein R, Jarvis JU. The naked mole-rat—a new record for the oldest living rodent. Sci Aging Knowledge Environ. 2002; 2002:pe7. https://doi.org/10.1126/sageke.2002.21.pe7 [PubMed]
- 3. Buffenstein R. Negligible senescence in the longest living rodent, the naked mole-rat: insights from a successfully aging species. J Comp Physiol B. 2008; 178:439–45. https://doi.org/10.1007/s00360-007-0237-5 [PubMed]
- 4. Edrey YH, Hanes M, Pinto M, Mele J, Buffenstein R. Successful aging and sustained good health in the naked mole rat: a long-lived mammalian model for biogerontology and biomedical research. ILAR J. 2011; 52:41–53. https://doi.org/10.1093/ilar.52.1.41 [PubMed]
- 5. Lewis KN, Buffenstein R. The Naked Mole-Rat. In: Kaeberlein MR, Martin GM, editors. Handbook of the Biology of Aging. ed. New York: Academic Press; 2016. pp. 179–204. https://doi.org/10.1016/B978-0-12-411596-5.00006-X
- 6. Park TJ, Lu Y, Jüttner R, Smith ES, Hu J, Brand A, Wetzel C, Milenkovic N, Erdmann B, Heppenstall PA, Laurito CE, Wilson SP, Lewin GR. Selective inflammatory pain insensitivity in the African naked mole-rat (Heterocephalus glaber). PLoS Biol. 2008; 6:e13. https://doi.org/10.1371/journal.pbio.0060013 [PubMed]
- 7. Park TJ, Reznick J, Peterson BL, Blass G, Omerbašić D, Bennett NC, Kuich PH, Zasada C, Browe BM, Hamann W, Applegate DT, Radke MH, Kosten T, et al. Fructose-driven glycolysis supports anoxia resistance in the naked mole-rat. Science. 2017; 356:307–11. https://doi.org/10.1126/science.aab3896 [PubMed]
- 8. Ruby JG, Smith M, Buffenstein R. Naked Mole-Rat mortality rates defy gompertzian laws by not increasing with age. eLife. 2018; 7:e31157. https://doi.org/10.7554/eLife.31157 [PubMed]
- 9. Buffenstein R, Kang J, Biney A. Glucose tolerance and insulin sensitivity in an extremely long-living rodent, the naked mole-rat. FASEB J. 2007; 21.
- 10. Csiszar A, Labinskyy N, Orosz Z, Xiangmin Z, Buffenstein R, Ungvari Z. Vascular aging in the longest-living rodent, the naked mole rat. Am J Physiol Heart Circ Physiol. 2007; 293:H919–27. https://doi.org/10.1152/ajpheart.01287.2006 [PubMed]
- 11. Stoll EA, Karapavlovic N, Rosa H, Woodmass M, Rygiel K, White K, Turnbull DM, Faulkes CG. Naked mole-rats maintain healthy skeletal muscle and Complex IV mitochondrial enzyme function into old age. Aging (Albany NY). 2016; 8:3468–85. https://doi.org/10.18632/aging.101140 [PubMed]
- 12. Horvath S, Raj K. DNA methylation-based biomarkers and the epigenetic clock theory of ageing. Nat Rev Genet. 2018; 19:371–84. https://doi.org/10.1038/s41576-018-0004-3 [PubMed]
- 13. Thompson MJ, Chwiałkowska K, Rubbi L, Lusis AJ, Davis RC, Srivastava A, Korstanje R, Churchill GA, Horvath S, Pellegrini M. A multi-tissue full lifespan epigenetic clock for mice. Aging (Albany NY). 2018; 10:2832–54. https://doi.org/10.18632/aging.101590 [PubMed]
- 14. Ecker S, Beck S. The epigenetic clock: a molecular crystal ball for human aging? Aging (Albany NY). 2019; 11:833–35. https://doi.org/10.18632/aging.101712 [PubMed]
- 15. Lee Y, Choufani S, Weksberg R, Wilson SL, Yuan V, Burt A, Marsit C, Lu AT, Ritz B, Bohlin J, Gjessing HK, Harris JR, Magnus P, et al. Placental epigenetic clocks: estimating gestational age using placental DNA methylation levels. Aging (Albany NY). 2019; 11:4238–53. https://doi.org/10.18632/aging.102049 [PubMed]
- 16. Thompson MJ, vonHoldt B, Horvath S, Pellegrini M. An epigenetic aging clock for dogs and wolves. Aging (Albany NY). 2017; 9:1055–68. https://doi.org/10.18632/aging.101211 [PubMed]
- 17. Lowe R, Barton C, Jenkins CA, Ernst C, Forman O, Fernández-Twinn DS, Bock C, Rossiter SJ, Faulkes CG, Ozanne SE, Walter L, Odom DT, Mellersh C, Rakyan VK. Ageing-associated DNA methylation dynamics are a molecular readout of lifespan variation among mammalian species. Genome Biol. 2018; 19:22. https://doi.org/10.1186/s13059-018-1397-1 [PubMed]
- 18. Polanowski AM, Robbins J, Chandler D, Jarman SN. Epigenetic estimation of age in humpback whales. Mol Ecol Resour. 2014; 14:976–87. https://doi.org/10.1111/1755-0998.12247 [PubMed]
- 19. Horvath S. DNA methylation age of human tissues and cell types. Genome Biol. 2013; 14:R115. https://doi.org/10.1186/gb-2013-14-10-r115 [PubMed]
- 20. Bennett NC, Jarvis JU, Wallace DB. The relative age structure and body masses of complete wild-captured colonies of two social mole-rats, the common mole-rat, Cryptomys hottentotus hottentotus and the Damaraland mole-rat, Cryptomys damarensis. J Zool (Lond). 1990; 220:469–85. https://doi.org/10.1111/j.1469-7998.1990.tb04319.x
- 21. Christiansen L, Lenart A, Tan Q, Vaupel JW, Aviv A, McGue M, Christensen K. DNA methylation age is associated with mortality in a longitudinal Danish twin study. Aging Cell. 2016; 15:149–54. https://doi.org/10.1111/acel.12421 [PubMed]
- 22. Day K, Waite LL, Thalacker-Mercer A, West A, Bamman MM, Brooks JD, Myers RM, Absher D. Differential DNA methylation with age displays both common and dynamic features across human tissues that are influenced by CpG landscape. Genome Biol. 2013; 14:R102. https://doi.org/10.1186/gb-2013-14-9-r102 [PubMed]
- 23. Maegawa S, Hinkal G, Kim HS, Shen L, Zhang L, Zhang J, Zhang N, Liang S, Donehower LA, Issa JP. Widespread and tissue specific age-related DNA methylation changes in mice. Genome Res. 2010; 20:332–40. https://doi.org/10.1101/gr.096826.109 [PubMed]
- 24. Thompson RF, Atzmon G, Gheorghe C, Liang HQ, Lowes C, Greally JM, Barzilai N. Tissue-specific dysregulation of DNA methylation in aging. Aging Cell. 2010; 9:506–18. https://doi.org/10.1111/j.1474-9726.2010.00577.x [PubMed]
- 25. Zhu T, Zheng SC, Paul DS, Horvath S, Teschendorff AE. Cell and tissue type independent age-associated DNA methylation changes are not rare but common. Aging (Albany NY). 2018; 10:3541–57. https://doi.org/10.18632/aging.101666 [PubMed]
- 26. Rodriguez KA, Edrey YH, Osmulski P, Gaczynska M, Buffenstein R. Altered composition of liver proteasome assemblies contributes to enhanced proteasome activity in the exceptionally long-lived naked mole-rat. PLoS One. 2012; 7:e35890. https://doi.org/10.1371/journal.pone.0035890 [PubMed]
- 27. Andziak B, Buffenstein R. Disparate patterns of age-related changes in lipid peroxidation in long-lived naked mole-rats and shorter-lived mice. Aging Cell. 2006; 5:525–32. https://doi.org/10.1111/j.1474-9726.2006.00246.x [PubMed]
- 28. Salmon AB, Sadighi Akha AA, Buffenstein R, Miller RA. Fibroblasts from naked mole-rats are resistant to multiple forms of cell injury, but sensitive to peroxide, ultraviolet light, and endoplasmic reticulum stress. J Gerontol A Biol Sci Med Sci. 2008; 63:232–41. https://doi.org/10.1093/gerona/63.3.232 [PubMed]
- 29. Lewis KN, Mele J, Hornsby PJ, Buffenstein R. Stress resistance in the naked mole-rat: the bare essentials - a mini-review. Gerontology. 2012; 58:453–62. https://doi.org/10.1159/000335966 [PubMed]
- 30. MacRae SL, Croken MM, Calder RB, Aliper A, Milholland B, White RR, Zhavoronkov A, Gladyshev VN, Seluanov A, Gorbunova V, Zhang ZD, Vijg J. DNA repair in species with extreme lifespan differences. Aging (Albany NY). 2015; 7:1171–84. https://doi.org/10.18632/aging.100866 [PubMed]
- 31. Field AE, Robertson NA, Wang T, Havas A, Ideker T, Adams PD, Adams PD. DNA methylation clocks in aging: categories, causes, and consequences. Mol Cell. 2018; 71:882–95. https://doi.org/10.1016/j.molcel.2018.08.008 [PubMed]
- 32. Maierhofer A, Flunkert J, Oshima J, Martin GM, Haaf T, Horvath S. Accelerated epigenetic aging in Werner syndrome. Aging (Albany NY). 2017; 9:1143–52. https://doi.org/10.18632/aging.101217 [PubMed]
- 33. Quach A, Levine ME, Tanaka T, Lu AT, Chen BH, Ferrucci L, Ritz B, Bandinelli S, Neuhouser ML, Beasley JM, Snetselaar L, Wallace RB, Tsao PS, et al. Epigenetic clock analysis of diet, exercise, education, and lifestyle factors. Aging (Albany NY). 2017; 9:419–46. https://doi.org/10.18632/aging.101168 [PubMed]
- 34. Wagner W. The Link Between Epigenetic Clocks for Aging and Senescence. Front Genet. 2019; 10:303. https://doi.org/10.3389/fgene.2019.00303 [PubMed]
- 35. Krueger F, Andrews SR. Bismark: a flexible aligner and methylation caller for Bisulfite-Seq applications. Bioinformatics. 2011; 27:1571–72. https://doi.org/10.1093/bioinformatics/btr167 [PubMed]
- 36. Langmead B, Salzberg SL. Fast gapped-read alignment with Bowtie 2. Nat Methods. 2012; 9:357–59. https://doi.org/10.1038/nmeth.1923 [PubMed]
- 37. R Core Team. R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. 2013; URL http://www.R-project.org/.



