Introduction
Periodontitis is a widespread infectious human chronic inflammatory disease which is characterized by progressive disruption of the connective tissue surrounding the teeth and also the inflammatory reaction mostly caused by an oral microbial biofilm [1]. Globally, serious periodontitis infects 9% to 11% of adult population in the world, especially in adult population over 50 years of age [2–4]. If untreated, periodontitis will induce the destruction of the periodontal structures, including alveolar bone, periodontal ligament, and root cementum, which will eventually lead to tooth-loss [5]. Therefore, investigating the pathogenesis of periodontitis is extremely important in the prevention and remedy of the disease.
Periodontal ligament stem cells (PDLSCs) are a group of mesenchymal stem cells (MSCs) derived from the periodontal ligament, which show multiple differentiation capability and a promising potential of being used to regenerate supporting tissues for the patients recently [6, 7]. Current researches have demonstrated that activating phosphatidylinositol 3-kinase (PI3K) / Akt signaling pathway could regulate the osteogenic differentiation of PDLSCs [8]. Though most of the studies focus on cancers, there are still some of the researches for periodontitis [9–11].
LncRNAs, a class of RNAs that are >200 bases in length, have recently drawn wide attention in describing the complex mechanisms underlying malignant processes, such as carcinogenesis and periodontitis. LncRNAs have been reported to engage in numerous biological processes, such as transcription, post-transcription, and translational regulation of gene expression [12]. For example, PTENP1, a pseudogene transcript of the tumor-inhibiting factor phosphatase and tensin homolog (PTEN), was targeted by endogenous miRNA, and quenched the endogenous miRNAs [13]. Though many current studies of lncRNAs focus on cancer, some researches have reported roles in lncRNAs in periodontitis [9–11]. LncRNAs were indicated by microarray analysis to exert partial or crucial roles in periodontitis pathogenesis [14]. For one, previous study had demonstrated that lncRNA ANCR played a vital role in regulating the proliferation and osteogenic differentiation of PDLSCs [15, 16]. In addition, H19, another important lncRNA, was reported to play important roles in osteogenic differentiation of PDLSCs via wnt/β-cantein signaling pathway [17]. In brief, studies mentioned above have tentatively shed light on the roles of lncRNAs in osteogenic differentiation of PDLSCs and our study will further investigate the effects of lncRNA MEG3 in osteogenic differentiation of PDLSCs via PI3K/Akt signaling pathway.
Furthermore, a new regulation in which lncRNAs can cross-talk with mRNAs through competition for shared miRNA-response elements has been certified recently. In this circumstance, lncRNAs function as ceRNAs which means miRNA sponges or antagomirs, to affect the expression levels and activities of miRNAs, thereby repressing miRNA targets and causing an additional level of posttranscriptional regulation [18]. The overall activity and functional balance of gene networks was maintained by the lncRNA/miRNA/mRNA regulatory interactions, and any destabilization of this system may lead to pathological changes [19], and the lncRNA POIR/miR-182 regulatory network was found to play significant roles in osteogenic differentiation of PDLSCs in periodontitis [20]. Hence, a fresh perspective would be produced to research the complex mechanism underlying lncRNA/miRNA/mRNA in PDLSCs.
In this study, microarray and KEGG analysis helped identify differentially expressed lncRNA MEG3 and mRNA IGF1 in PDLSCs, as well as a regulated PI3K/Akt pathway. As co-expression networks predicted correlation between LncRNA MEG3 and IGF1, and between IGF1 and PI3K/Akt related miRNAs were screened and miR-27a-3p stood out as a possible bridge between lncRNA MEG3 and IGF1. Besides, we determined the levels of lncRNA MEG3, miR-27a-3p and IGF1 in PDLSCs, and investigated the cross-talk on osteogenic differentiation of PDLSCs. In addition, mechanism analysis revealed that lncRNA MEG3 may serve as a ceRNA to regulate the expression of IGF1 by competing for miR-27a-3p, thus effecting osteogenic differentiation via PI3K/Akt signaling pathway. These findings shed new light on the diagnosis and therapy for periodontitis. Above all, we proposed that there was an association between osteogenic differentiation of PDLSCs and lncRNA MEG3.
Results
IGF1 was down-regulated and PI3K/Akt signaling pathway was dys-regulated in periodontitis periodontal tissues compared with healthy periodontal tissues
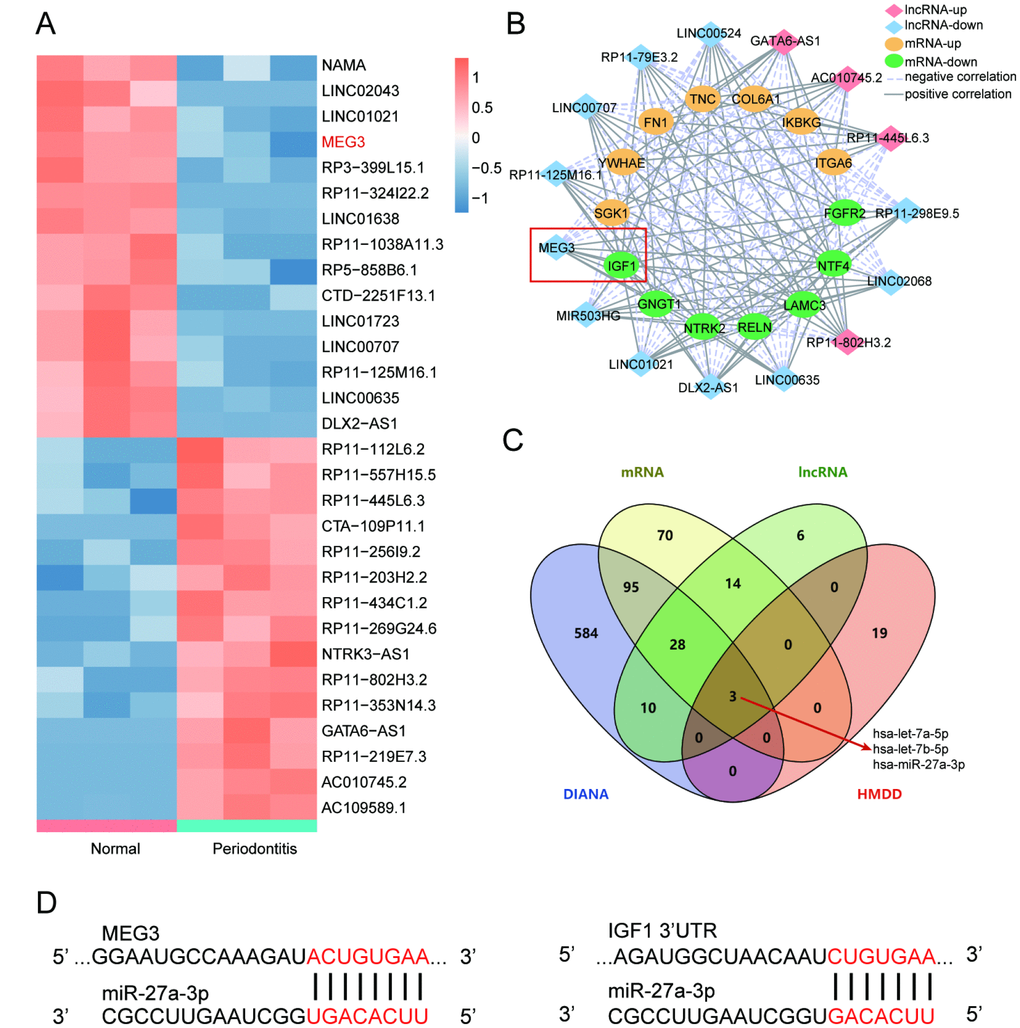
Bioinformatic differential analysis selected out significantly differentially expressed genes (DEGs) between periodontitis periodontal tissues and healthy periodontal tissues. Top 15 up-regulated and down-regulated mRNAs were visualized by heatmaps as shown in Figure 1A. Thereafter, KEGG pathway enrichment analysis using gene set enrichment analysis (GSEA) software suggested that PI3K/Akt signaling pathway was inactivated in periodontal tissues with periodontitis compared with healthy samples (Figure 1B, 1C), which was better visualized by joyplot and dotplot as shown in Figure 1D–1F. Taken together, IGF1 down-regulated in PI3K/Akt signaling pathway could be inactivated in periodontitis periodontal tissues compared with healthy periodontal tissues.
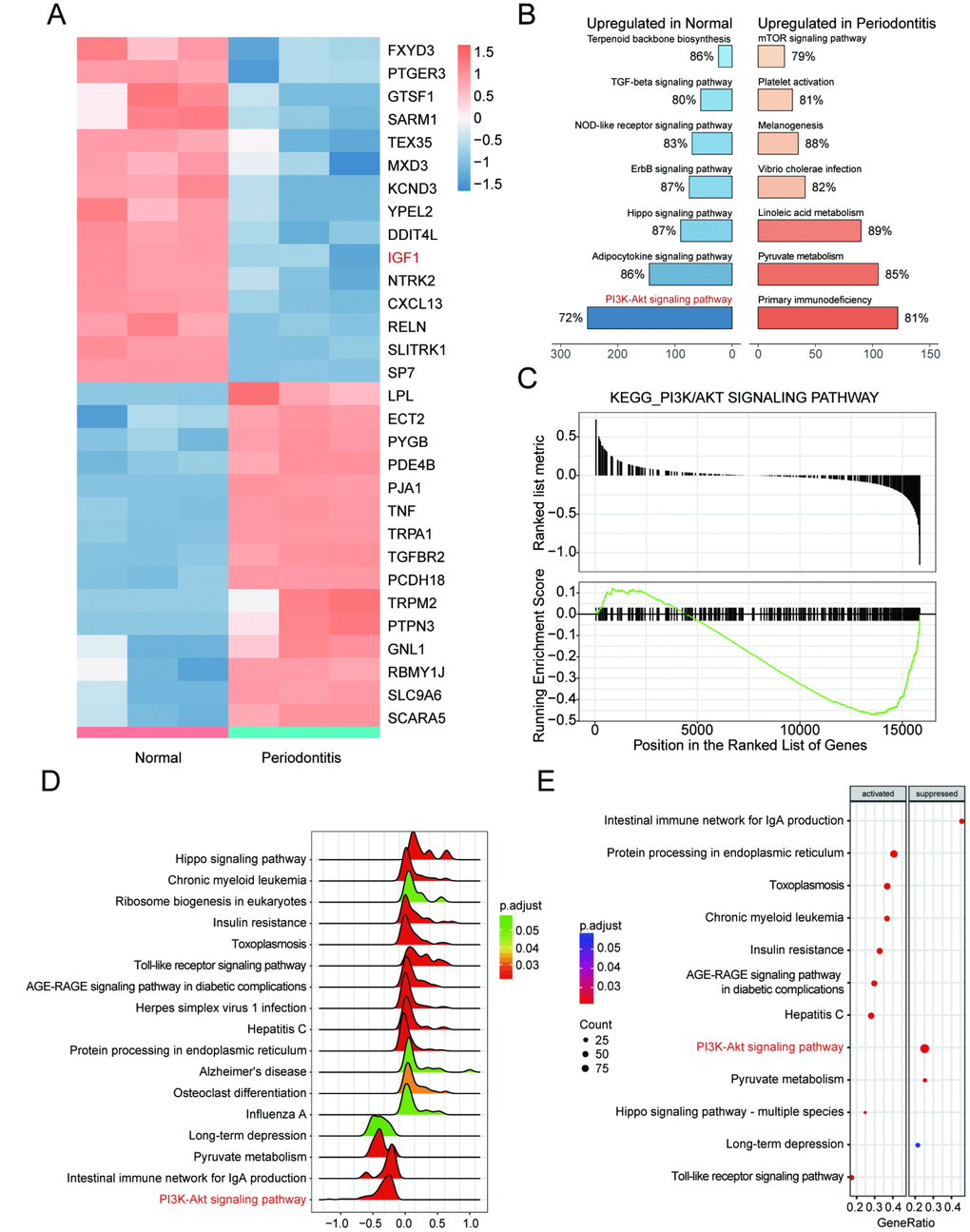
Figure 1. IGF1 was down-regulated and PI3K/Akt signaling pathway was dys-regulated in periodontitis periodontal tissues compared with healthy periodontal tissues. (A) Heatmap of top 15 up-regulated or down-regulated mRNAs between periodontitis periodontal tissues and healthy periodontal tissues. (B, C) KEGG pathway enrichment analysis using GSEA. PI3K/Akt signaling pathway was one of pathways that were inactivated in periodontal tissues with periodontitis compared with healthy samples. (D–E) Joyplot and dotplot of abnormally regulated signaling pathway in periodontitis tissues compared with the that in healthy samples.
LncRNA MEG3 regulated PDLSC osteogenic differentiation through miR-27a-3p
Opposite to the qRT-PCR results of lncRNA MEG3 expression, miR-27a-3p expression in PDLSCs was up-regulated in periodontitis periodontal tissues compared with healthy periodontal tissues, and down-regulated after 14 days of osteogenic induction (Figure 4A, 4B). To further illustrate the targeting relationship between lncRNA MEG3 and miR-27a-3p, dual luciferase reporter gene assay was employed. Co-transfection of lncRNA MEG3-3’UTR-wt and agomiR-27a-3p significantly reduced relative luciferase activity, which confirmed that lncRNA MEG3 could directly target miR-27a-3p (Figure 4C, 4D). In group of agomiR-27a-3p transfection, miR-27a-3p expression was promoted, which could be neutralized by overexpression of lncRNA MEG3; in group of antagomiR-27a-3p transfection, miR-27a-3p expression declined, which could be weaken by silencing of lncRNA MEG3. In addition, MEG3 expression showed no significant change under down-regulation or up-regulation of miR-27a-3p (Figure 4E). Expression of Runx2, Osterix, Osteocalcin and Colla1 were down-regulated by agomiR-27a-3p, while up-regulated by antagomiR-27a-3p, indicating the negative correlation between miR-27a-3p and PDLSC osteogenic differentiation (Figure 4F). Subsequently, as shown by ALP and Alizarin Red staining, osteogenic activities could be suppressed in the agomiR-27a-3p group, and the suppression was relieved by lncRNA MEG3 overexpression; osteogenic activities boosted in the antagomiR-27a-3p group, and were reversed by silencing of lncRNA MEG3(Figure 4G). In addition, Figure 4H showed that with promotion of miR-27a-3p, protein expressions of certain genes in PI3k/Akt signaling pathway, including IGF1, IRS1, p-Akt, declined, which went the opposite way with suppression of miR-27a-3p. Taken together, these results indicated that miR-27a-3p negatively correlated with PDLSC osteogenic differentiation, as a target of lncRNA MEG3.
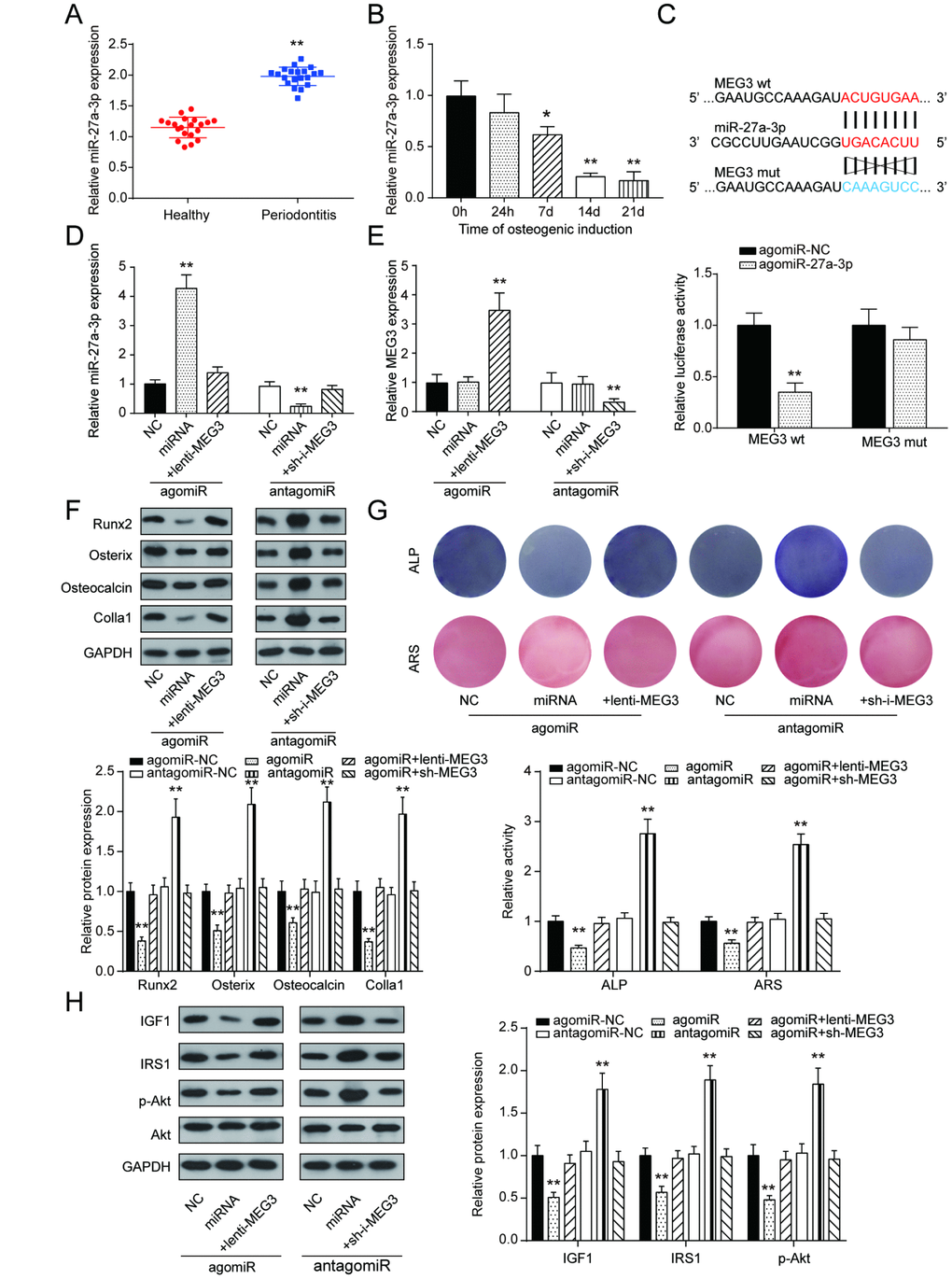
Figure 4. LncRNA MEG3 regulated PDLSC osteogenic differentiation through miR-27a-3p. (A) MiR-27a-3p expression in PDLSCs was up-regulated in periodontitis periodontal tissues compared with healthy periodontal tissues. (B) Expression of miR-27a-3p in 0h, 24h, 7d, 14d and 21d after osteogenic induction. (C) MiR-27a-3p directly targeted MEG3. Co-transfection of lncRNA MEG3-3’UTR-wt and agomiR-27a-3p significantly reduced relative luciferase activity. (D) Expression of miR-27a-3p in group of agomiR-27a-3p or antagomiR-27a-3p transfection (E) Expression of MEG3 in group of agomiR-27a-3p or antagomiR-27a-3p transfection. (F) Western blot of osteoblast makers including Runx2, Osterix, Osteocalcin and Colla1 in group of agomiR-27a-3p or antagomiR-27a-3p transfection. (G) ALP and Alizarin Red staining of osteogenic activities in the agomiR-27a-3p group and antagomiR-27a-3p group. (H) Protein expression of certain genes in PI3k/Akt signaling pathway, including IGF1, IRS1, p-Akt, and Akt. ** P < 0.01, compared with the NC group
MiR-27a-3p regulated PDLSC osteogenic differentiation through targeting IGF1
Similar to the expression of lncRNA MEG3, IGF1 expression was lower in periodontitis periodontal tissues, compared with healthy periodontal tissues (Figure 5A). In older donors, expression of IGF1 were lower compared with the young donors (Supplementary Figure 3B). After 7 days of osteogenic induction, IGF expression in PDSLCs was tuned up (Figure 5B). Dual luciferase reporter gene assay confirmed the targeting relationship between miR-27a-3p and IGF1 (Figure 5C). Deliberate suppression of IGF1 expression by sh-IGF1 were neutralized by co-transfection of antagomiR-27a-3p; promotion of IGF1 expression by lenti-IGF1 could be reversed by agomiR-27a-3p. However, little change in the expression of miR-27a-3p nor lncRNA MEG3 was shown under overexpression or suppression of IGF1 (Figure 5D–5F). Western blot analysis showed that the inhibitory effects of agomiR-27a-3p on the expression of Runx2, Osterix, Osteocalcin and Colla1 could reverse the promoting effect by up-regulation of IGF1, while antagomiR-27a-3p could reverse the suppression of expressions of the three by sh-IGF1 (Figure 6A). Furthermore, ALP and Alizarin Red staining provided similar results that IGF1 could counteract the inhibiting effect of miR-27a-3p on osteogenic activities (Figure 6B–6D). Besides, down-regulation of IGF1 greatly suppressed relative protein expression of IGF1, IRS1 and p-Akt (Figure 6E). In short, miR-27a-3p regulated PDLSC osteogenic differentiation through targeting IGF1.
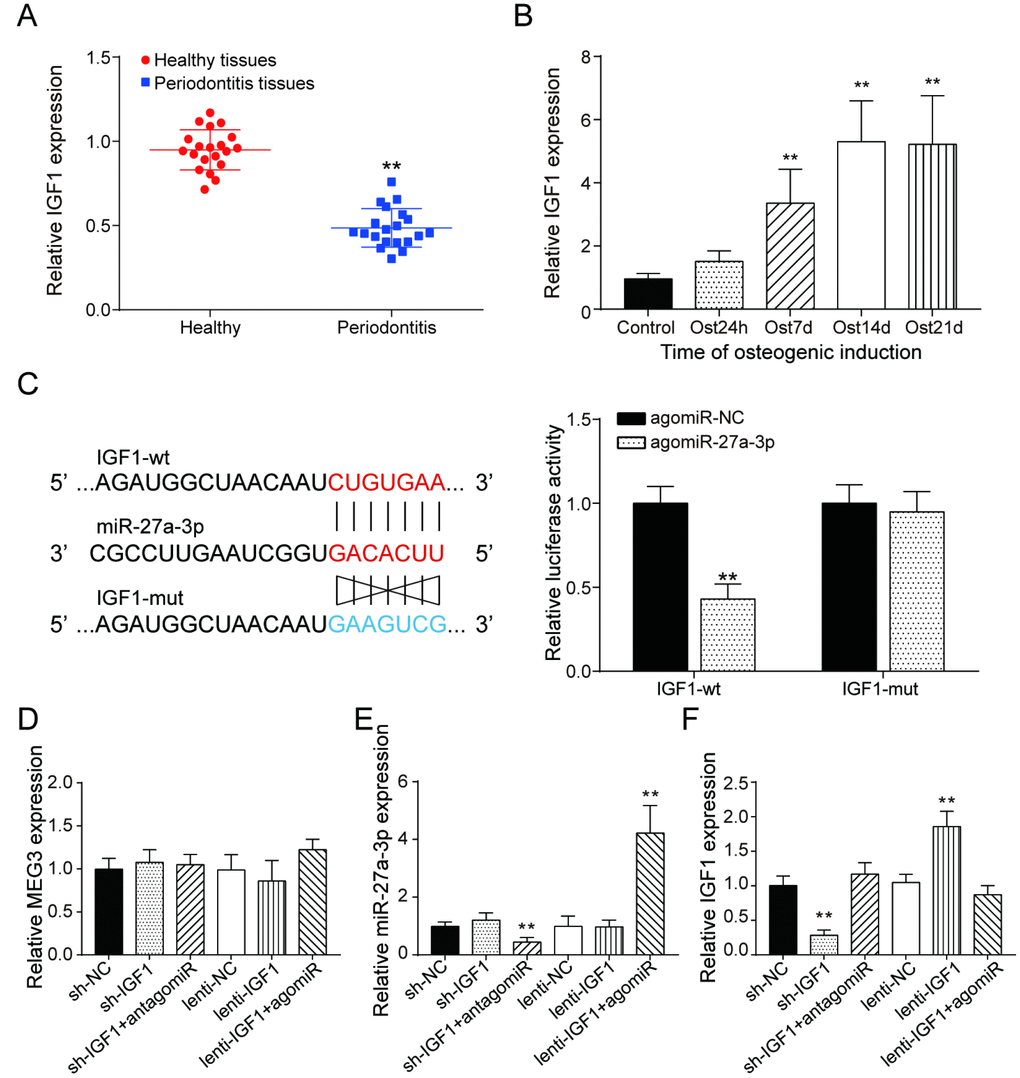
Figure 5. LncRNA MEG3 was correlated to IGF1 via MiR-27a-3p. (A) IGF1 expression in periodontitis periodontal tissues compared with healthy periodontal tissues. (B) Expression of IGF1 in 0h, 24h, 7d, 14d and 21d after osteogenic induction. (C) MiR-27a-3p directly targeted IGF1. (D–F) Expression of MEG3, miR-27a-3p and IGF1 in PDLSCs of each group. ** P < 0.01, compared with the NC group.
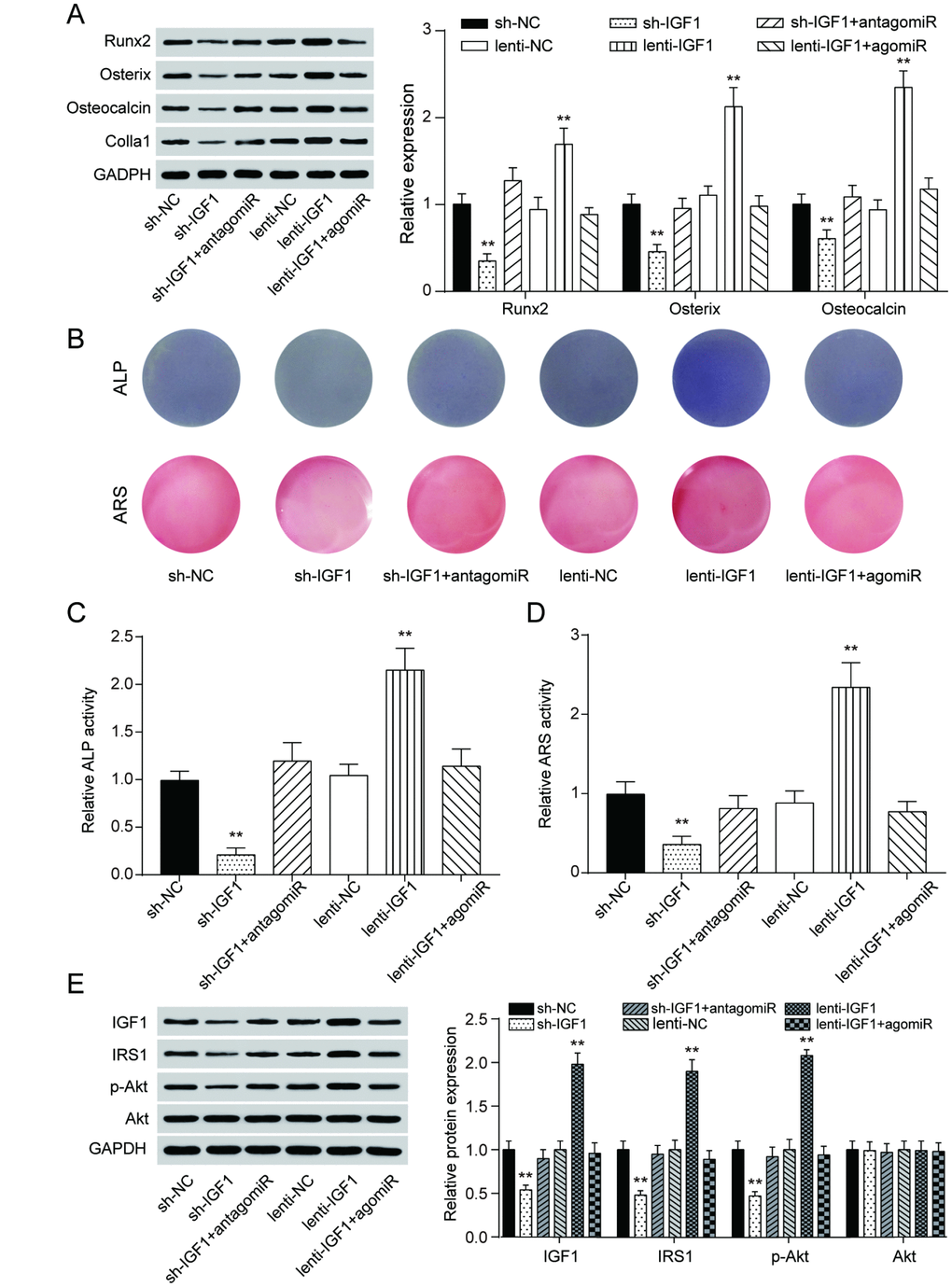
Figure 6. MiR-27a-3p regulated PDLSC osteogenic differentiation through targeting IGF1. (A) Western blot of Runx2, Osterix, Osteocalcin and Colla1 expression in PDLSCs with up-regulation or down-regulation of IGF1. (B–D) ALP and Alizarin Red staining of PDLSCs with different regulation of IGF1. (E) Protein expression of IGF1, IRS1, p-Akt and Akt in PDLSCs with different IGF1 regulation treatment. ** P < 0.01, compared with the NC group.
LncRNA MEG3/miR-27a-3p/IGF1 axis regulated PI3K/Akt signaling pathway to affect PDLSC osteogenic differentiation
PI3K inhibitor LY294002 (10 nM) was used to explore the effects of lncRNA MEG3/miR-27a-3p/IGF1 axis on the PI3K/Akt signaling pathway. The results of western blot analysis indicated that overexpression of either lncRNA MEG3 or IGF1 promoted expression of PI3K/Akt-related proteins IRS1 and p-Akt, while agomiR-27a-3p and LY294002 did the opposite (Figure 7A, 7B), indicating that lncRNA MEG3 might sponge miR-27a-3p for up-regulation of IGF1, leading to the activation of PI3K/Akt signaling pathway. Alizarin Red and ALP staining results validated this conclusion on a cellular level – osteogenic activities increased with lncRNA MEG3 or IGF1 upregulated, and declined under effect of agomiR-27a-3p or LY294002 (Figure 7C, 7D). To conclude, lncRNA MEG3/miR-27a-3p/IGF1 axis regulated PI3K/Akt signaling pathway to affect PDLSC osteogenic differentiation.
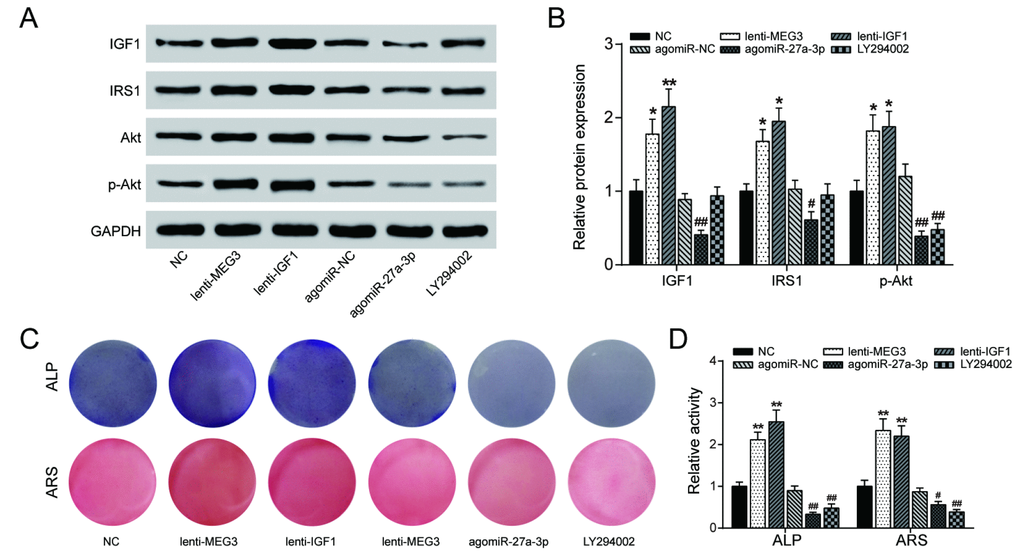
Figure 7. LncRNA MEG3/miR-27a-3p/IGF1 axis regulated PI3K/Akt signaling pathway to affect PDLSC osteogenic differentiation. (A, B) The results of western blot analysis indicated that overexpression of either lncRNA MEG3 or IGF1 promoted expression of PI3K/Akt-related proteins IRS1 and phosphorylated Akt (p-Akt), while agomiR-27a-3p and LY294002 blocked their expression. (C, D) Alizarin Red and ALP staining were also performed to validate this conclusion on cellular level. Increased calcium nodule formation and ALP activity were detected in lncRNA MEG3/IGF1 overexpression groups, while agomiR-27a-3p/LY294002 groups showed reverse results. ** P < 0.01, compared with the NC group. ## P<0.01, compared with agomiR-NC group.
Discussion
To sum up, the expression levels of lncRNAMEG3 and IGF1 were both down-regulated, while miR-27a-3p was up-regulated in periodontitis periodontal tissues compared with healthy periodontal tissues. Furthermore, both lncRNA MEG3 and IGF1 could be directly targeted by miR-27a-3p. In addition, lncRNA MEG3 sponged miR-27a-3p for up-regulation of IGF1 to result in activation of PI3K/Akt signaling pathway. Taken together, lncRNA MEG3/miR-27a-3p/IGF1 axis played a role in PDLSC osteogenesis differentiation via activating PI3K/Akt signaling pathway.
Bioinformatic analyses have greatly improved the ability of researchers in processing and analyzing biological data. The expression of the targets such as DNA [21], RNA [22] and signaling pathway [23] could be directly observed through microarray and gene network analysis to assess candidate mechanisms. Herein, gene set enrichment analysis (GSEA) was performed and a co-expression network of lncRNA/miRNA/mRNA was conducted for further annotation, visualization, and integrated discovery. The results that lncRNA influenced miRNA and miRNA influences mRNA subsequently, which were consistent with most of previous studies [24, 25].
Many recent researches have investigated human diseases through lncRNAs, leading to a new way to find the internal mechanism for better prognoses and treatments. However, few researchers aimed at studying lncRNAs in periodontitis. Most of the studies were related to the intrinsic mechanism of lncRNA from the perspective of periodontitis, such as chronic periodontitis (CP) [26] and aggressive periodontitis (AgP) [27], and seldom of them focused on PDLSCs. Besides, most of researches focusing on lncRNA MEG3 showed that lncRNA MEG3 involved in multifarious cancers was down-regulated in most cancers and affected cell proliferation, progression, and prognosis [28, 29] which indicated there was still a large research space on the influence mechanism of lncRNA MEG3 in periodontitis PDLSCs and our study was of great practical significance.
Furthermore, lncRNA/miRNA/mRNA regulatory network that lncRNA functioned as ceRNA to impact miRNAs at posttranscriptional processing steps has been widely identified, thus leading to inhibition of miRNA target genes recently [25]. For example, lncRNA POIR was found to form a regulatory network with miRNA-182 and FoxO1, which taken a notable up-regulated effect on periodontitis PDLSCs osteogenic differentiation [20] and miR-147 was down-regulated by up-regulation of lncRNA MEG3 to result in increased expression of Sox2, which activated NF-kB pathway and Wnt/β-catenin pathway in PC12 cells after hypoxia [30]. Besides, Galani V et al. discovered that lncRNA MEG3 genes and IGF signaling family genes included IGF1 are both associated with meningioma progression [31] which led to a guess that endogenous cross-talk maybe exist between lncRNA MEG3 and IGF1 in PDLSCs. Moreover, Yu Y et al. found that IGF1 can facilitate the osteogenic differentiation and osteogenesis of human PDLSCs via ERK and JNK MAPK pathway, suggesting that IGF1 was a potent endogenous agent for PDLSCs regeneration [32]. Based on these findings, we speculated that lncRNA MEG3 may participate in this system of periodontitis PDLSCs and act as a ceRNA, and searched for potential interactions between lncRNA MEG3 and IGF1. As expected, we found that lncRNA MEG3 could sponge miR-27a-3p, resulting in IGF1 up-regulation, and PI3K/Akt signaling pathway was activated, all which promoted PDLSC osteogenic differentiation ultimately.
Altogether, the findings presented in this study highlights that lncRNA MEG3 was determined as a biomarker as periodontitis inhibitor because of the new-found mechanism underlying lncRNA MEG3/miR-27a-3p/IGF1 axis via PI3K/Akt signaling pathway by promoting osteogenic differentiation in PDLSCs. Moreover, our result showed that lncRNA MEG3/miR-27a-3p/IGF1 axis definitely played a significant role in understanding the pathogenesis of periodontitis for better prognosis. However, because of the limitation of the samples we researched in this study, further confirmation should be needed in prospective researches of the pathological and physiological mechanism according to lncRNA MEG3/miR-27a-3p/IGF1 axis in periodontitis PDLSCs. Moreover, effects of MEG3/miR-27a-3p/IGF1 on osteoclast should also be investigated in further studies.
To conclude, lncRNA MEG3 sponged miR-27a-3p to up-regulate IGF1 to activate PI3k/Akt signaling pathway to promote PDLSC osteogenic differentiation in periodontitis.
Materials and Methods
Bioinformatic analysis
Microarray data of both lncRNA and mRNA expression profiles of 3 periodontitis periodontal tissues and 3 healthy periodontal tissues were downloaded from Gene Expression Omnibus database (GEO) (https://www.ncbi.nlm.nih.gov/geo/). The series accession number of microarray data in this study was GSE78074 and matched platform was GPL16976. Differentially expressed lncRNAs and mRNAs between periodontitis periodontal tissues and healthy periodontal tissues were selected out and visualized using R software (version 3.4.1) “limma” package. In brief, data was processed for selecting out differentially expressed genes (DEGs) with screening criteria adj P value <0.05 and |FC (Fold Change)|>2. In addition, Gene Set Enrichment Analysis (GSEA) software (http://software.broadinstitute.org/gsea) and kyoto Encyclopedia of Genes and Genomes (KEGG) database (https://www.genome.jp/kegg/) were adopted to unveil significantly enriched signaling pathways. Top enriched signaling pathways (adjusted P<0.01) from GSEA results were visualized by dotplot and joyplot using R software “ggplot2” and “easygplot2” packages. Cytoscape (version 3.6.0) was used to conduct co-expression network analysis based on differentially expressed lncRNAs and mRNAs. Node and edge files that revealed the coefficient factor threshold>0.7 and adjusted P<0.05 were produced using R project. Signaling pathway-related miRNAs or mRNAs were predicted using DIANA Tools (http://diana.imis.athena-innovation.gr/DianaTools) or string (https://string-db.org/) respectively.
Clinical tissues
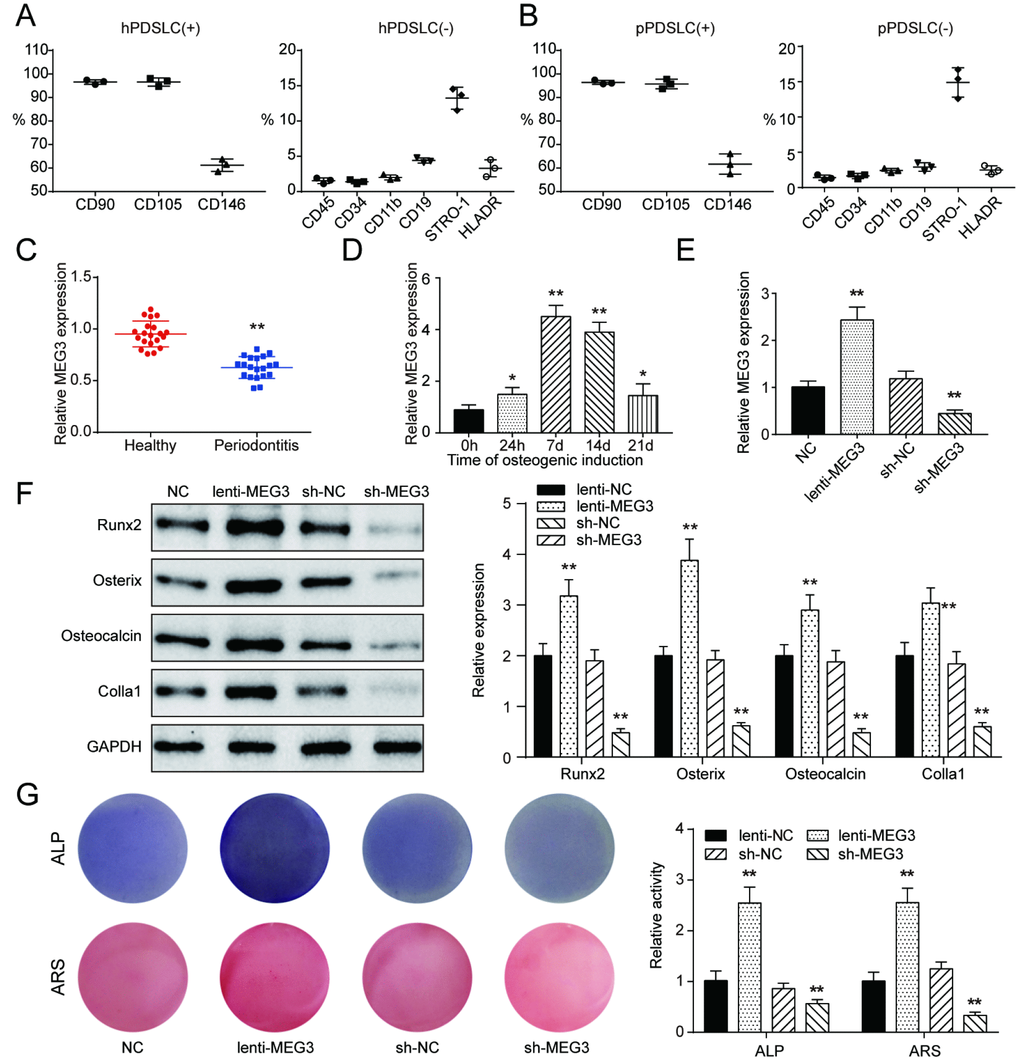
Human periodontitis PDLSCs (pPDLSCs) were obtained from periodontal ligament tissues of 20 periodontitis patients (10 males, 10 females, age 24-38), who received premolar extraction at stomatological hospital of Shandong University. Human healthy PDLSCs (hPDLSCs) were obtained from periodontal ligament tissues of premolars extracted for orthodontic treatment from 20 healthy individuals (10 males, 10 females, age 18-25). All procedures in the study were scrutinized and ratified by Shandong University & Department of Orthodontics Committee of stomatological hospital of Shandong University and tissue donors in the study signed informed consent allowing the use of their tissues for scientific researches.
Cell culture
Premolars were firstly disinfected with 75% ethanol and washed by PBS. Normal or periodontitis periodontal ligament tissues were then separated from middle 1/3 of teeth roots. Separated tissues were shred and digested using collagenase type I (1 mg/mL; Sigma-Aldrich, MO, USA) for 15 min at 37°C. Cell suspensions were then filtered and cultured in low-glucose DMEM (Gibco, Carlsbad, CA, USA) with 10% fetal bovine serum (FBS, Gibco, Carlsbad, CA, USA) at 37°C in a humidified atmosphere of 5% CO2. After 2-week incubation, cells were digested and single cell-derived PDLSC colony cultures were obtained by limiting dilution. PDLSCs were collected and utilized for following experiments after 5 times of passages. Human embryo kidney cell line HEK-293 used for dual-luciferase assay was purchased from BeNa Culture Collection (Beijing, China) and cultured in high-glucose DMEM (Gibco, Carlsbad, CA, USA) with 10 % FBS.
Flow cytometry analysis
A total of 1×106 PDLSCs was incubated with the antibodies against CD90, CD105, CD146, CD45, CD34, CD11b, CD19, STRO-1 and HLADR according to MSC characterization guidelines [33]. The isotype IgG1 antibodies were applied as controls (ab154450, 0.1 μg, Abcam). Thereafter, flow cytometry was operated on FACSCantoTM II Flow Cytometer (BD Biosciences, CA, USA). μμμμμ
Cell transfection
AgomiR-27a-3p, antagomiR-27a-3p, lentiviral-pEF-1a/Puro-MEG3 (lenti-MEG3), lentiviral-pGLVU6/Puro-sh-MEG3 (sh-MEG3), lentiviral-pEF-1a/Puro-IGF1 (lenti-IGF1), lentiviral-pGLVU6/Puro-sh-IGF1 (sh-IGF1) and their corresponding negative controls (NC) in this study were all bought from GenePharma (Shanghai, China). Before transfection, 2 × 105 cells in logarithmic growth phase were plated into six-well plates, with 2 mL complete medium for 16-18 h, until they were 30-40% confluent. AgomiR-27a-3p and antagomiR-27a-3p were transfected into pPDLSCs using Lipofectamine 3000 reagent (Invitrogen, Carlsbad, CA, USA), with all procedures following the manufacturer’s protocol. The transfection concentration was 60 nM. For lentivirus transfection of the PDLSCs, the supernatant cell culture medium was replaced with fresh cell culture medium containing 6 μg/mL polybrene and lentivirus particles (MOI=20). Then the cells were incubated for 24 h, before the medium was replaced with fresh medium. Stable transfected cells were filtered with puromycin. For LY294002 treatment, the pPDLSCs were cultured with 10 nM LY294002 (Beyotime) according to a previous study [34].
Osteoblastic induction
Osteoblastic induction was performed using StemProTM Osteogenesis Differentiation Kit (Gibco, Carlsbad, CA, USA). In brief, hPDLSCs were washed with PBS and seeded onto 12-well plates, then cultured with MSC Growth Medium at 37°C in a humidified atmosphere of 5% CO2 for 5 days. Afterwards, culture medium was replaced by Complete Osteogenesis Differentiation Medium (Invitrogen, Carlsbad, CA, USA) and PDLSCs would differentiate to osteoblasts.
Quantitative real-time PCR (qRT-PCR)
Total RNA of PDLSCs was extracted with TRIzol reagent (Invitrogen). Thereafter, about 2 μg of total RNA was reversely transcripted to cDNA using SuperScriptTM III Reverse Transcriptase Kit (Invitrogen) for lncRNA/mRNA reverse transcription and miRCURY LNA RT Kit (Qiagen, Duesseldorf, Germany) for miRNA reverse transcription. Subsequently, qRT-PCR was then performed using SYBR Green qPCR Master Mix (Takara, Tokyo, Japan) on LightCycler 480 PCR System (Roche, Rotkreuz, Switzerland). Normalization of mRNA and lncRNA expression values was performed against GAPDH and miRNA expression value was normalized against U6. Relative RNA expression was calculated with 2-ΔΔCt method. PCR primers were synthesized by Sangon Biotech (Shanghai, China) and primer sequences were shown in Table 1. Working concentration of primers used for qRT-PCR in this study was 0.5 μmol/L.
Table 1. Primer sequences for qRT-PCR
| Gene | Forward primer 5’-3’ | Reverse primer 5’-3’ |
| MEG3 | TACACCTCACGAGGGCACTA | CAGGGCTTAATGCCCAATGC |
| miR-27a-3p | GTTCACAGTGGCTAAGTTCCGC | Involved in the kit |
| IGF1 | ATAGAGCCTGCGCAATGGAA | ACTGAAGAGCATCCACCAGC |
| GAPDH | CCGCATCTTCTTTTGCGTCG | GGACTCCACGACGTACTCAG |
Western blot analysis
Total protein from PDLSCs (5×105 cells) was extracted 24h after transfection using RIPA lysis buffer (Beyotime, Shanghai, China) and quantified using Enhanced BCA Protein Assay Kit (Beyotime). 20 μg of total protein was then separated by SDS-PAGE and transferred onto PVDF membranes (Beyotime). 5% bovine serum albumin (BSA, Sigma-Aldrich) was used as a block for 30 min at room temperature, causing incubation of the membranes with primary antibodies overnight at 4°C (GAPDH was used for internal reference). Secondary antibody was then added and incubated for another 1 h at room temperature. HRP-labeled proteins were developed using BeyoECL Star Kit (Beyotime) and filmed after washed by TBST for 3 times. Primary antibodies were: rabbit anti-IGF1 (ab9572, 1:2000, Abcam), rabbit anti-IRS1 (ab40777, 1:2000), rabbit anti-pan-Akt (ab8805, 1:500), rabbit anti-pan-Akt (phospho T308; ab38449, 1:500), rabbit anti-Runx2 (ab23981, 1:1000), rabbit anti-Osterix (ab22552, 1:2000), rabbit anti-Osteocalcin (ab93876, 1:500), rabbit anti-type-1 collagen (Colla1, ab34710, 1:2000) and rabbit anti-GAPDH (ab181602, 1:10000). Secondary antibody was HRP labeled goat anti-rabbit IgG (ab6721, 1:2000). Antibodies in this study were purchased from Abcam (MA, USA). Proteins were visualized by ECL-plus reagents (Millipore, Billerica, MA, USA) and Image J software (Version1.48u, Bethesda, USA) was employed for the measurement of band density.
Dual luciferase reporter gene assay
Binding sites between miRNAs and lncRNAs were predicted using miRcode (http://www.mircode.org/) and targeted relationships between miRNAs and mRNAs were predicted using TargetScan 7.1 (http://www.targetscan.org/vert_71/). PmirGLO vectors (Promega, Madison, WI, USA) containing wild-type (wt) / mutation-type (mut) sequences of MEG3 or IGF1 3’-untranslated region (3’-UTR) were constructed using XL Site-directed Mutagenesis Kit (Qiagen). PmirGLO luciferase reporter gene vector loaded with MEG3-3’UTR-wt or MEG3-3’UTR-mut was introduced into HEK-293 cells with co-transfection of AgomiR-203b-3p or AgomiR-NC using Lipofectamine 3000 reagent (Invitrogen, Carksbad, CA, USA). Similarly, luciferase reporter gene vector loaded with IGF1-3’UTR-wt or IGF1-3’UTR-mut was introduced into HEK-293 cells with co-transfection of AgomiR-203b-3p or AgomiR-NC by using Lipofectamine 3000 reagent. Relative luciferase activity was assessed with Dual-luciferase Reporter Assay Kit (Promega) following the manufacturer’s protocol 24 h after transfection.
Alkaline Phosphatase (ALP) staining
ALP staining was performed using BCIP/NBT Alkaline Phosphatase Color Development Kit (Beyotime) at 14th day after osteoblastic induction. Cells cultured in 12-well plates were rinsed and fixed with PBS for 3 times and 4% paraformaldehyde for 30 min successively. Subsequently, BCIP/NBT ALP staining buffer was added and incubated for 2 h at room temperature in a dark room. Then reaction was terminated and the ALP activity was quantified by spectrophotometric absorbance at 405 nm.
Alizarin Red staining (ARS)
Alizarin red staining was performed at 21st day after osteoblastic induction. Cells cultured in 12-well plates were rinsed with Phosphate Buffered Saline (PBS) buffer (pH 7.4) for 3 times and fixed with 4% paraformaldehyde for 30 min. Afterwards, cells were stained with 1% alizarin red S solution (pH=8.4) (Sigma-Aldrich) for 5 min. Thereafter, cells were washed with PBS for 3 times and observed under a light microscope. To quantify mineralization, bound dye was extracted in 10 mM sodium phosphate containing 10% cetylpyridinium chloride and quantified spectrophotometrically at 562 nm.
Statistical analysis
All the experiments were repeated test at least thrice. Data was analyzed with GraphPad Prism version 6.0 (GraphPad Software, CA, USA) and presented as mean value ± standard deviation (mean ± SD) in this study. Multiple comparisons between two groups were implemented by student’s t test, and differences between three or more groups were evaluated by one-way ANOVA. P value less than 0.05 was deemed statistically significant.
Ethical statement
Investigation has been conducted in accordance with the ethical standards and according to the Declaration of Helsinki and according to national and international guidelines and has been approved by Shandong University & Department of Orthodontics committee.
Supplementary Materials
Conflicts of Interest
The authors declare that they have no conflict of interests.
Funding
This work was supported by grants from the National Natural Science Foundation of China (NO. 81701008, 81670993, 81873716); Natural Science Foundation of Shandong Province (NO. BS2015YY027); China Postdoctoral Science Foundation (NO.2014M561941); Special funds for Postdoctoral Innovation Projects of Shandong Province (NO.201402030); the Fundamental Research Funds of Shandong University (NO. 2016GN009) ; Students Research Fund of Shandong University (NO.2019298) and the Construction Engineering Special Fund of “Taishan Scholars” (NO. tsqn201611068); Key research and development program of Shandong Province (NO. 2019GSF108187).
References
- 1. Van Dyke TE. The management of inflammation in periodontal disease. J Periodontol. 2008 (Suppl); 79:1601–08. https://doi.org/10.1902/jop.2008.080173 [PubMed]
- 2. Ebersole JL, Graves CL, Gonzalez OA, Dawson D
3rd , Morford LA, Huja PE, Hartsfield JKJr , Huja SS, Pandruvada S, Wallet SM. Aging, inflammation, immunity and periodontal disease. Periodontol 2000. 2016; 72:54–75. https://doi.org/10.1111/prd.12135 [PubMed] - 3. Kassebaum NJ, Bernabé E, Dahiya M, Bhandari B, Murray CJ, Marcenes W. Global burden of severe periodontitis in 1990-2010: a systematic review and meta-regression. J Dent Res. 2014; 93:1045–53. https://doi.org/10.1177/0022034514552491 [PubMed]
- 4. Eke PI, Dye BA, Wei L, Thornton-Evans GO, Genco RJ, and CDC Periodontal Disease Surveillance workgroup: James Beck (University of North Carolina, Chapel Hill, USA), Gordon Douglass (Past President, American Academy of Periodontology), Roy Page (University of Washin). Prevalence of periodontitis in adults in the United States: 2009 and 2010. J Dent Res. 2012; 91:914–20. https://doi.org/10.1177/0022034512457373 [PubMed]
- 5. Chen FM, Sun HH, Lu H, Yu Q. Stem cell-delivery therapeutics for periodontal tissue regeneration. Biomaterials. 2012; 33:6320–44. https://doi.org/10.1016/j.biomaterials.2012.05.048 [PubMed]
- 6. Liu Y, Liu W, Hu C, Xue Z, Wang G, Ding B, Luo H, Tang L, Kong X, Chen X, Liu N, Ding Y, Jin Y. MiR-17 modulates osteogenic differentiation through a coherent feed-forward loop in mesenchymal stem cells isolated from periodontal ligaments of patients with periodontitis. Stem Cells. 2011; 29:1804–16. https://doi.org/10.1002/stem.728 [PubMed]
- 7. Seo BM, Miura M, Gronthos S, Bartold PM, Batouli S, Brahim J, Young M, Robey PG, Wang CY, Shi S. Investigation of multipotent postnatal stem cells from human periodontal ligament. Lancet. 2004; 364:149–55. https://doi.org/10.1016/S0140-6736(04)16627-0 [PubMed]
- 8. Lee JS, Yi JK, An SY, Heo JS. Increased osteogenic differentiation of periodontal ligament stem cells on polydopamine film occurs via activation of integrin and PI3K signaling pathways. Cell Physiol Biochem. 2014; 34:1824–34. https://doi.org/10.1159/000366381 [PubMed]
- 9. Zhou S, Wang L, Yang Q, Liu H, Meng Q, Jiang L, Wang S, Jiang W. Systematical analysis of lncRNA-mRNA competing endogenous RNA network in breast cancer subtypes. Breast Cancer Res Treat. 2018; 169:267–75. https://doi.org/10.1007/s10549-018-4678-1 [PubMed]
- 10. Qu Q, Fang F, Wu B, Hu Y, Chen M, Deng Z, Ma D, Chen T, Hao Y, Ge Y. Potential Role of Long Non-Coding RNA in Osteogenic Differentiation of Human Periodontal Ligament Stem Cells. J Periodontol. 2016; 87:e127–37. https://doi.org/10.1902/jop.2016.150592 [PubMed]
- 11. Zhuang W, Ge X, Yang S, Huang M, Zhuang W, Chen P, Zhang X, Fu J, Qu J, Li B. Upregulation of lncRNA MEG3 Promotes Osteogenic Differentiation of Mesenchymal Stem Cells From Multiple Myeloma Patients By Targeting BMP4 Transcription. Stem Cells. 2015; 33:1985–97. https://doi.org/10.1002/stem.1989 [PubMed]
- 12. Quinn JJ, Chang HY. Unique features of long non-coding RNA biogenesis and function. Nat Rev Genet. 2016; 17:47–62. https://doi.org/10.1038/nrg.2015.10 [PubMed]
- 13. Poliseno L, Salmena L, Zhang J, Carver B, Haveman WJ, Pandolfi PP. A coding-independent function of gene and pseudogene mRNAs regulates tumour biology. Nature. 2010; 465:1033–38. https://doi.org/10.1038/nature09144 [PubMed]
- 14. Zou Y, Li C, Shu F, Tian Z, Xu W, Xu H, Tian H, Shi R, Mao X. lncRNA expression signatures in periodontitis revealed by microarray: the potential role of lncRNAs in periodontitis pathogenesis. J Cell Biochem. 2015; 116:640–47. https://doi.org/10.1002/jcb.25015 [PubMed]
- 15. Peng W, Deng W, Zhang J, Pei G, Rong Q, Zhu S. Long noncoding RNA ANCR suppresses bone formation of periodontal ligament stem cells via sponging miRNA-758. Biochem Biophys Res Commun. 2018; 503:815–21. https://doi.org/10.1016/j.bbrc.2018.06.081 [PubMed]
- 16. Jia Q, Chen X, Jiang W, Wang W, Guo B, Ni L. The Regulatory Effects of Long Noncoding RNA-ANCR on Dental Tissue-Derived Stem Cells. Stem Cells Int. 2016; 2016:3146805. https://doi.org/10.1155/2016/3146805 [PubMed]
- 17. Gong YY, Peng MY, Yin DQ, Yang YF. Long non-coding RNA H19 promotes the osteogenic differentiation of rat ectomesenchymal stem cells via Wnt/β-catenin signaling pathway. Eur Rev Med Pharmacol Sci. 2018; 22:8805–13. https://doi.org/10.26355/eurrev_201812_16648 [PubMed]
- 18. Ren K, Li Y, Lu H, Li Z, Li Z, Wu K, Li Z, Han X. Long Noncoding RNA HOTAIR Controls Cell Cycle by Functioning as a Competing Endogenous RNA in Esophageal Squamous Cell Carcinoma. Transl Oncol. 2016; 9:489–97. https://doi.org/10.1016/j.tranon.2016.09.005 [PubMed]
- 19. Ergun S, Oztuzcu S. Oncocers: ceRNA-mediated cross-talk by sponging miRNAs in oncogenic pathways. Tumour Biol. 2015; 36:3129–36. https://doi.org/10.1007/s13277-015-3346-x [PubMed]
- 20. Wang L, Wu F, Song Y, Li X, Wu Q, Duan Y, Jin Z. Long noncoding RNA related to periodontitis interacts with miR-182 to upregulate osteogenic differentiation in periodontal mesenchymal stem cells of periodontitis patients. Cell Death Dis. 2016; 7:e2327. https://doi.org/10.1038/cddis.2016.125 [PubMed]
- 21. Song BN, Kim SK, Chu IS. Bioinformatic identification of prognostic signature defined by copy number alteration and expression of CCNE1 in non-muscle invasive bladder cancer. Exp Mol Med. 2017; 49:e282. https://doi.org/10.1038/emm.2016.120 [PubMed]
- 22. Omori K, Mitsuhashi M, Todorov I, Rawson J, Shiang KD, Kandeel F, Mullen Y. Microassay for glucose-induced preproinsulin mRNA expression to assess islet functional potency for islet transplantation. Transplantation. 2010; 89:146–54. https://doi.org/10.1097/TP.0b013e3181c4218d [PubMed]
- 23. Kim WT, Seo SP, Byun YJ, Kang HW, Kim YJ, Lee SC, Jeong P, Seo Y, Choe SY, Kim DJ, Kim SK, Moon SK, Choi YH, et al. Garlic extract in bladder cancer prevention: evidence from T24 bladder cancer cell xenograft model, tissue microarray, and gene network analysis. Int J Oncol. 2017; 51:204–12. https://doi.org/10.3892/ijo.2017.3993 [PubMed]
- 24. Lee KT, Nam JW. Post-transcriptional and translational regulation of mRNA-like long non-coding RNAs by microRNAs in early developmental stages of zebrafish embryos. BMB Rep. 2017; 50:226–31. https://doi.org/10.5483/BMBRep.2017.50.4.025 [PubMed]
- 25. Xia T, Liao Q, Jiang X, Shao Y, Xiao B, Xi Y, Guo J. Long noncoding RNA associated-competing endogenous RNAs in gastric cancer. Sci Rep. 2014; 4:6088. https://doi.org/10.1038/srep06088 [PubMed]
- 26. Kornman KS, Polverini PJ. Clinical application of genetics to guide prevention and treatment of oral diseases. Clin Genet. 2014; 86:44–49. https://doi.org/10.1111/cge.12396 [PubMed]
- 27. Schaefer AS, Bochenek G, Manke T, Nothnagel M, Graetz C, Thien A, Jockel-Schneider Y, Harks I, Staufenbiel I, Wijmenga C, Eberhard J, Guzeldemir-Akcakanat E, Cine N, et al. Validation of reported genetic risk factors for periodontitis in a large-scale replication study. J Clin Periodontol. 2013; 40:563–72. https://doi.org/10.1111/jcpe.12092 [PubMed]
- 28. Chang L, Wang G, Jia T, Zhang L, Li Y, Han Y, Zhang K, Lin G, Zhang R, Li J, Wang L. Armored long non-coding RNA MEG3 targeting EGFR based on recombinant MS2 bacteriophage virus-like particles against hepatocellular carcinoma. Oncotarget. 2016; 7:23988–4004. https://doi.org/10.18632/oncotarget.8115 [PubMed]
- 29. Zhang J, Yao T, Wang Y, Yu J, Liu Y, Lin Z. Long noncoding RNA MEG3 is downregulated in cervical cancer and affects cell proliferation and apoptosis by regulating miR-21. Cancer Biol Ther. 2016; 17:104–13. https://doi.org/10.1080/15384047.2015.1108496 [PubMed]
- 30. Han L, Dong Z, Liu N, Xie F, Wang N. Maternally Expressed Gene 3 (MEG3) Enhances PC12 Cell Hypoxia Injury by Targeting MiR-147. Cell Physiol Biochem. 2017; 43:2457–69. https://doi.org/10.1159/000484452 [PubMed]
- 31. Galani V, Lampri E, Varouktsi A, Alexiou G, Mitselou A, Kyritsis AP. Genetic and epigenetic alterations in meningiomas. Clin Neurol Neurosurg. 2017; 158:119–25. https://doi.org/10.1016/j.clineuro.2017.05.002 [PubMed]
- 32. Yu Y, Mu J, Fan Z, Lei G, Yan M, Wang S, Tang C, Wang Z, Yu J, Zhang G. Insulin-like growth factor 1 enhances the proliferation and osteogenic differentiation of human periodontal ligament stem cells via ERK and JNK MAPK pathways. Histochem Cell Biol. 2012; 137:513–25. https://doi.org/10.1007/s00418-011-0908-x [PubMed]
- 33. Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, Deans R, Keating A, Prockop D, Horwitz E. Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy. 2006; 8:315–17. https://doi.org/10.1080/14653240600855905 [PubMed]
- 34. Chen X, Hu C, Wang G, Li L, Kong X, Ding Y, Jin Y. Nuclear factor-κB modulates osteogenesis of periodontal ligament stem cells through competition with β-catenin signaling in inflammatory microenvironments. Cell Death Dis. 2013; 4:e510. https://doi.org/10.1038/cddis.2013.14 [PubMed]