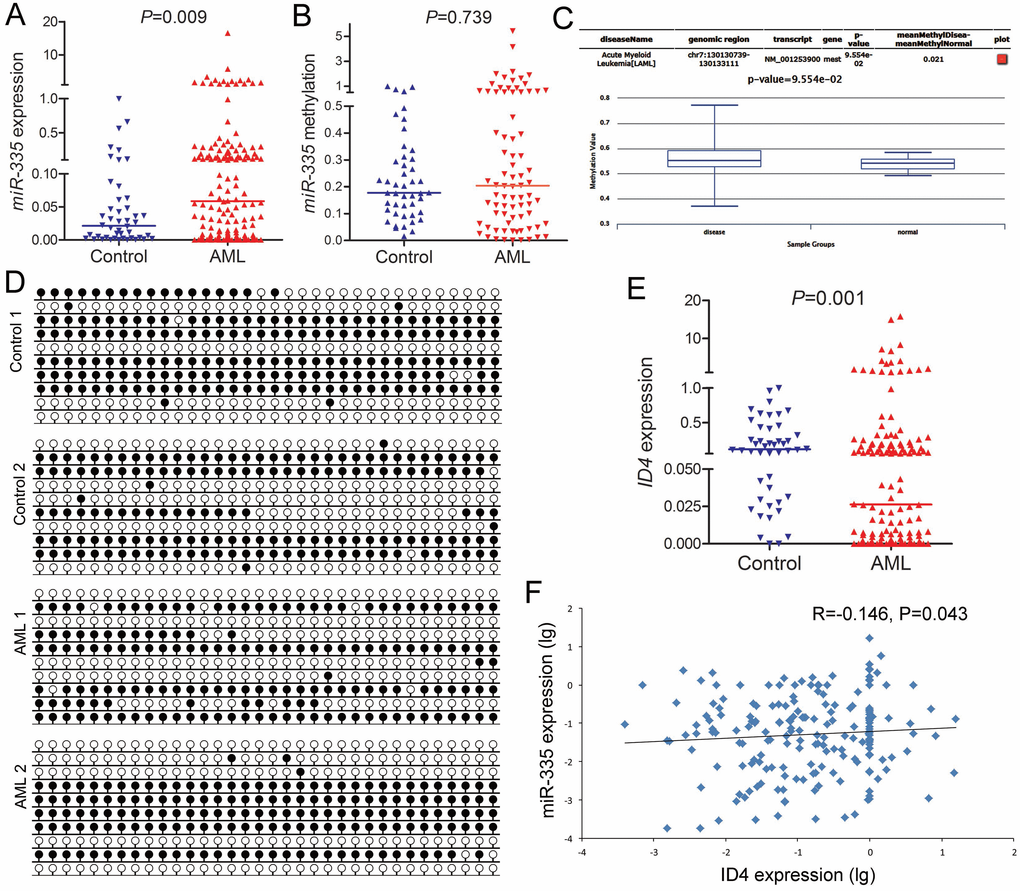
Introduction
Acute myeloid leukemia (AML), the most common adult leukemia, is an aggressive blood cancer with variable clinical outcome [1, 2]. The pathogenesis of AML is a complex process including diverse molecular events and signaling pathways [3]. Despite recent advances in the treatment of leukemia including targeted drugs, the overall prognosis for AML remains unsatisfactory [1, 2]. The karyotypes together with age and white blood cells of AML patients assessed at diagnosis are generally recognized as the three main prognostic factors [4, 5]. Recently, mounting evidences showed that molecular biomarkers such as mutations in NPM1, CEBPA, FLT3, and C-KIT as well as BAALC, MN1, EVI1, and ERG overexpression also provide powerful prognostic information [6]. Therefore, understanding molecular mechanism and finding effective prognostic biomarkers has been being one of the most urgent clinical needs and research hotspots.
MicroRNAs (miRNAs) are small non-coding RNAs that post-transcriptionally regulate gene expression, often targeting hundreds of different mRNAs with both temporal and spatial specificity [7, 8]. Increasing data show that many types of cancers were accompanied with the dysregulation of miRNAs, which contributing to tumorigenesis through various critical processes, including cell differentiation, apoptosis, proliferation and hematopoiesis [9, 10]. Notably, individual miRNAs may play distinct roles in cancers originating from different tissues or even from different lineages of the same cancer [11]. MircoRNA-335 (miR-335) has been reported as a significant cancer-associated miRNA, which was often epigenetically downregulated and acted as a tumor suppressor gene in diverse human cancers [12]. Interestingly, two recent studies showed that overexpression of miR-335 was identified in both adult and pediatric AML, and correlated with poor clinical outcome [13, 14]. However, the potential role of miR-335 in AML remains to be elucidated.
In the present study, we first validated that miR-335 expression was significantly increased and negatively correlated with decreased ID4 expression in AML. Moreover, aberrant miR-335/ID4 expression independently affected chemotherapy response and survival in patients with AML. Next, functional experiments in vitro showed the oncogenic role of miR-335 in AML, and could be rescued by ID4. Lastly, we identified that miR-335 contributed to leukemogenesis through PI3K/Akt signaling pathway.
Results
Aberrant miR-335/ID4 expression affected chemotherapy response in AML
In order to analyze the association of miR-335/ID4 with clinical characteristics of AML, total patients were firstly divided according to miR-335 expression (the cutoff value 0.048 was based on the ROC curve, the sensitivity and specificity were 55.5% and 73.9%, groups named miR-335high and miR-335low) and ID4 expression (the cutoff value 0.017 was based on the ROC curve, the sensitivity and specificity were 45.2% and 95.3%, groups named ID4high and ID4low). Finally, we further divided patients into three groups (miR-335lowID4high, miR-335lowID4low/miR-335highID4high, and miR-335highID4low) to determine the clinical significance. No significant differences were observed between miR-335/ID4 expressions with all the patient's parameters except for complete remission (CR) (Table 1). The rate of CR in patients with miR-335highID4low was the lowest (34.1%, 14/41), higher in patients with miR-335lowID4low/miR-335highID4high (40%, 26/65), and the highest in patients with miR-335lowID4high (62.5%, 25/40). Also, significant differences were also observed among both non-M3-AML and CN-AML (Table 1). Furthermore, Logistic regression analysis further confirmed that miR-335/ID4 expression independently affected CR in both non-M3-AML and CN-AML after receiving induction therapy (Table 2).
Table 1. Comparison of clinical/laboratory features of AML patients with aberrant miR-335/ID4 expression.
| Patient's parameters | miR-335lowID4high (n=40) | miR-335highID4high or miR-335lowID4low (n=65) | miR-335highID4low (n=41) | P value | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Sex, male/female | 20/20 | 39/26 | 26/15 | 0.443 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age, median (range), years | 58.5 (15-93) | 55 (17-81) | 61 (14-87) | 0.286 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| WBC, median (range), ×109/L | 6.7 (0.3-528.0) | 12.2 (0.8-201.0) | 27.8 (0.9-136.1) | 0.253 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Hemoglobin, median (range), g/L | 76 (42-133) | 75 (32-142) | 78 (40-126) | 0.377 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Platelets, median (range),×109/L | 41 (5-447) | 42 (3-399) | 33 (7-234) | 0.623 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BM blasts, median (range), % | 47.5 (1.0-97.5) | 42.5 (5.5-94.5) | 35.0 (3.0-99.0) | 0.893 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FAB subtypes | 0.399 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| M0 | 0 (0%) | 1 (2%) | 0 (0%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| M1 | 1 (3%) | 4 (6%) | 4 (10%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| M2 | 16 (40%) | 28 (43%) | 15 (36%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| M3 | 7 (17%) | 13 (20%) | 9 (22%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| M4 | 7 (17%) | 15 (23%) | 9 (22%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| M5 | 8 (20%) | 2 (3%) | 4(10%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| M6 | 1 (3%) | 2 (3%) | 0 (0%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Karyotypes | 0.493 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| normal | 19 (47%) | 30 (46%) | 20 (49%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| t(8;21) | 4 (10%) | 4 (6%) | 0 (0%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| t(15;17) | 7 (17%) | 13 (20%) | 9 (22%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| +8 | 1 (3%) | 3 (5%) | 1 (2%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| -5/5q- | 0 (0%) | 2 (3%) | 1 (2%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| -7/7q- | 0 (0%) | 1 (2%) | 0 (0%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| others | 4 (10%) | 3 (5%) | 6 (16%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| complex | 4 (10%) | 8 (11%) | 1 (2%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| No data | 1 (3%) | 1 (2%) | 3 (7%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gene mutations | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CEBPA (+/-) | 4/32 | 6/55 | 5/32 | 0.938 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NPM1 (+/-) | 4/32 | 8/53 | 3/34 | 0.735 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FLT3-ITD (+/-) | 6/30 | 6/55 | 5/32 | 0.570 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| c-KIT (+/-) | 2/34 | 2/59 | 0/37 | 0.286 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NRAS/KRAS (+/-) | 5/31 | 5/56 | 1/36 | 0.214 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IDH1/2 (+/-) | 4/32 | 3/58 | 2/35 | 0.511 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| DNMT3A (+/-) | 3/33 | 4/57 | 3/34 | 0.920 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| U2AF1 (+/-) | 1/35 | 3/58 | 2/35 | 1.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SRSF2 (+/-) | 2/34 | 3/58 | 1/36 | 0.880 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CR (+/-), whole-cohort | 25/15 | 26/39 | 14/27 | 0.024 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CR (+/-), non-M3-AML | 18/15 | 16/36 | 8/24 | 0.029 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CR (+/-), CN-AML | 13/6 | 11/19 | 5/15 | 0.018 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| WBC: white blood cells; BM: bone marrow; FAB: French-American-British classification; CR: complete remission; non-M3-AML: acute myeloid leukemia without FAB-M3; CN-AML: cytogenetically normal AML. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 2. Multivariate analyses of variables for overall survival in AML patients.
| Variables | Non-M3-AML | CN-AML | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CR | OS | CR | OS | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| OR (95% CI) | P | HR (95% CI) | P | OR (95% CI) | P | HR (95% CI) | P | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MiR-335/ID4 | 2.010 (1.057–3.823) | 0.033 | 1.894 (1.392–2.579) | 0.000 | 3.224 (1.335–7.784) | 0.009 | 2.376 (1.574–3.586) | 0.000 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age | 4.079 (1.594–10.435) | 0.003 | 1.481 (0.945–2.320) | 0.087 | 3.915 (1.206–12.703) | 0.023 | 1.770 (0.970–3.231) | 0.063 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| WBC | 1.469 (0.532–4.054) | 0.458 | 1.415 (0.917–2.183) | 0.117 | 2.056 (0.536–7.885) | 0.293 | 1.551 (0.853–2.820) | 0.150 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Karyotype | 4.214 (1.508–11.774) | 0.006 | 1.595 (1.143–2.227) | 0.006 | – | – | – | – | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CEBPA mutations | 0.529 (0.132–2.114) | 0.367 | 0.796 (0.405–1.565) | 0.509 | 2.293 (0.365–14.393) | 0.376 | 1.377 (0.559–3.394) | 0.487 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NPM1 mutations | 1.144 (0.227–5.765) | 0.871 | 0.972 (0.449–2.101) | 0.942 | 1.955 (0.289–13.204) | 0.492 | 1.383 (0.558–3.427) | 0.484 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| FLT3–ITD mutations | 2.428 (0.541–10.897) | 0.247 | 1.141 (0.567–2.294) | 0.712 | 3.598 (0.610–21.229) | 0.157 | 1.080 (0.380–3.068) | 0.885 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| C–KIT mutations | 3.130 (0.139–70.308) | 0.472 | 1.523 (0.340–6.824) | 0.583 | 0.785 (0.010–64.537) | 0.914 | 1.181 (0.146–9.894) | 0.876 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N/K–RAS mutations | 7.147 (1.137–44.916) | 0.036 | 1.902 (0.917–3.944) | 0.084 | 12.082 (1.001–145.767) | 0.050 | 2.904 (1.061–7.948) | 0.038 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IDH1/2 mutations | 3.746 (0.349–40.186) | 0.275 | 2.903 (1.289–6.537) | 0.010 | 6.979 (0.534–91.233) | 0.139 | 3.154 (1.248–7.969) | 0.015 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| DNMT3A mutations | 0.651 (0.104–4.076) | 0.646 | 0.914 (0.408–2.048) | 0.827 | 0.410 (0.050–3.346) | 0.405 | 0.652 (0.262–1.624) | 0.358 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| U2AF1 mutations | undetermined | 0.999 | 1.815 (0.751–4.386) | 0.186 | undetermined | 0.999 | 1.480 (0.423–5.179) | 0.539 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| SRSF2 mutations | undetermined | 0.999 | 1.906 (0.788–4.610) | 0.153 | undetermined | 0.999 | 1.141 (0.215–6.057) | 0.877 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CR, complete remission; OS, overall survival; OR, odd ratio; HR, hazard ratio; CI, confidence interval. Variables in multivariate analysis including miR-335/ID4 expression (Low vs. High), age (≤60 vs. >60 years), WBC (≥30×109 vs. <30×109 /L), karyotype (favorable vs. intermediate vs. poor), and gene mutations (mutant vs. wild-type). | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Dysregulated miR-335/ID4 expression predicted clinical outcome in AML
Due to independent disease entity, AML-M3/acute promyelocytic leukemia (APL) was excluded from the survival analysis. Firstly, we determined the prognostic value of miR-335 and ID4 expression in AML, respectively. Patients with miR-335high presented significantly shorter LFS and OS than those with miR-335low in both non-M3-AML and CN-AML (Figure 2A). Similarly, ID4low patients also showed significantly shorter LFS and OS than those with ID4high among both non-M3-AML and CN-AML (Figure 2B). Moreover, the prognostic impact of ID4 expression on OS among CN-AML patients was also confirmed by TCGA data and GEO data analyzed though online tool GenomicScape (Figure 2C and 2D). Secondly, we further evaluated the prognostic significance of miR-335/ID4 expression for LFS and OS based on the three groups (miR-335lowID4high, miR-335lowID4low/miR-335highID4high, and miR-335highID4low), and the statistic value was enhanced (Figure 2E). Moreover, Cox regression multivariate analysis further confirmed that miR-335/ID4 expression was an independent prognostic biomarker for OS among both non-M3-AML and CN-AML (Table 2).
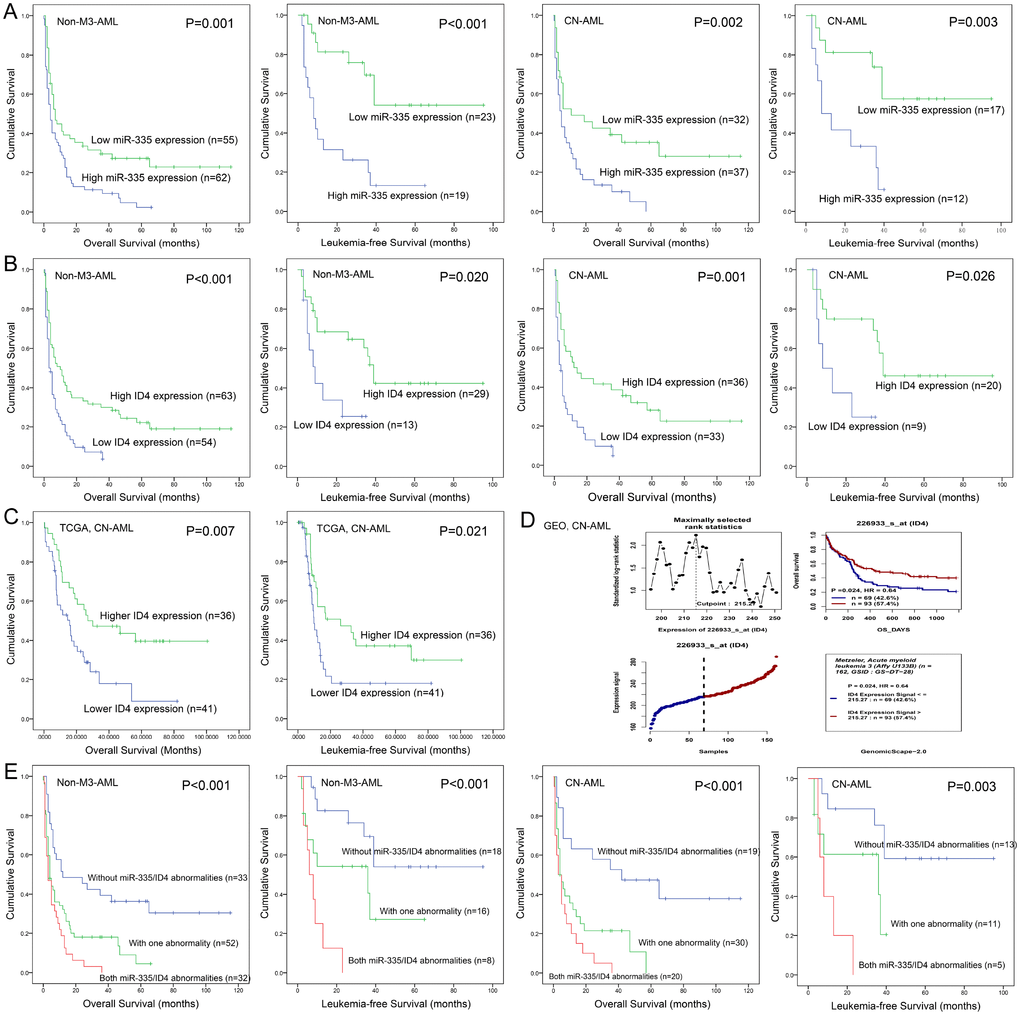
Figure 2. Aberrant miR-335/ID4 expression predicted clinical outcome in AML. (A) The prognostic value of miR-335 expression for OS and LFS in non-M3-AML and CN-AML patients. (B) The prognostic value of ID4 expression for OS and LFS in non-M3-AML and CN-AML patients. (C) The prognostic value of ID4 expression for OS and LFS among CN-AML patients based on TCGA databases. (D) The prognostic value of ID4 expression for OS among CN-AML patients obtained by bioinformatics analysis. The effect of ID4 expression on prognosis was determined by the Genomicscape (http://genomicscape.com/microarray/survival.php). (E) The prognostic value of combined miR-335/ID4 expression for OS and LFS in non-M3-AML and CN-AML patients.
MiR-335 exhibited pro-proliferative and anti-apoptotic effects in leukemic cell-lines
To investigate the biological role of miR-335 in AML, gain-of-function experiments were performed in leukemic cell-lines K562 and HL60 since they showed low miR-335 expression. We transfected miR-335 plasmid into K562 and HL60 (called K562/miR-335 and HL60/miR-335, controls as K562/miR-NC and HL60/miR-NC), and miR-335 overexpression was confirmed by RT-qPCR (Figure 3A and 3B). Overexpression of miR-335 significantly increased the proliferation ability in K562 and HL60 cell-lines (Figure 3C and 3D), together with increased expression of proliferation-related proteins PCNA and Cyclin-D1 (Figure 3G). Moreover, the ratio of apoptosis was significantly reduced in K562 and HL60 cell-lines when overexpressing miR-335 (Figure 3E and 3F), and confirmed by decreased expression of apoptosis-related Caspase-3 (Figure 3G). In addition, we did not observe the effect of miR-335 overexpression on cell differentiation based on the expression of CD11b (data not shown).
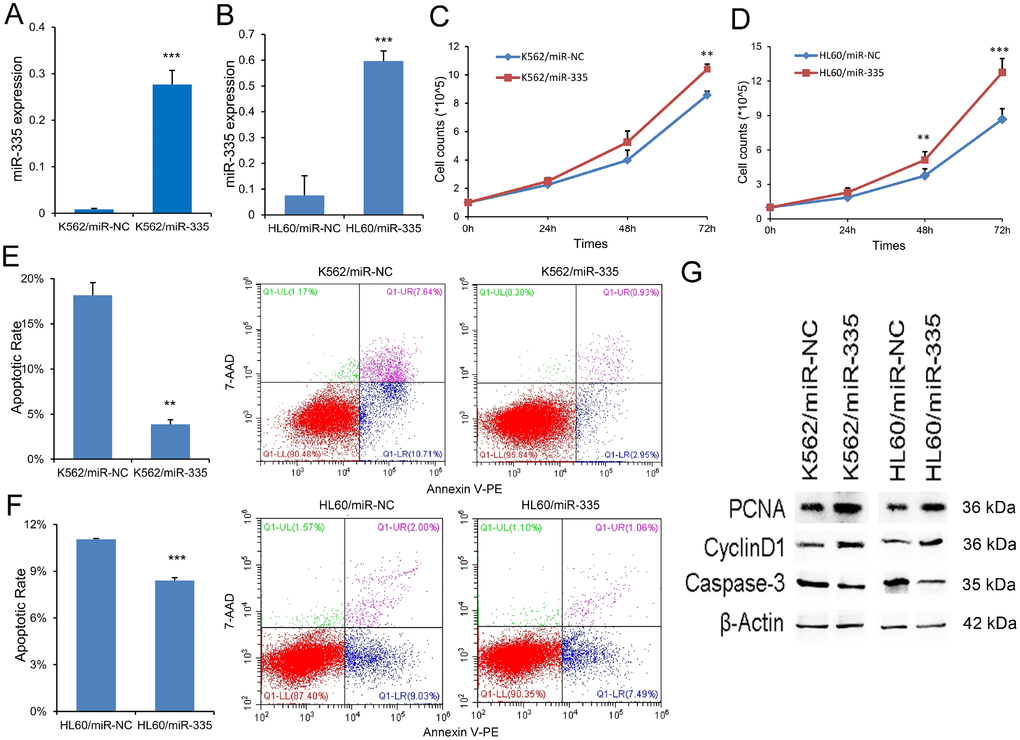
Figure 3. MiR-335 exhibited pro-proliferative and anti-apoptotic effects in leukemic cell-lines. (A–B) Confirmation of miR-335 expression after miR-335 transfection. MiR-335 expression was significantly upregulated after miR-335 transfection in both K562 and HL60 cell-lines. (C–D) The effect of miR-335 overexpression on cell proliferation. Overexpression of miR-335 significantly increased the proliferation ability in K562 and HL60 cell-lines. (E–F) The effect of miR-335 overexpression on cell apoptosis. Overexpression of miR-335 significantly decreased the apoptosis ratio in K562 and HL60 cell-lines. (G) The expression of proliferation-related proteins (PCNA and Cyclin D1) and apoptosis-related proteins (Caspase-3) affected by miR-335 overexpression. The expression of proliferation-related proteins (PCNA and Cyclin D1) was increased, whereas the apoptosis-related proteins (Caspase-3) expression was decreased after miR-335 overexpression in K562 and HL60 cell-lines. *, P<0.05; **, P<0.01; ***, P<0.001.
ID4 rescued the pro-leukemia effects of miR-335 in leukemic cell-lines
Emerging evidence showed that ID4 was direct target of miR-335, acting as a tumor suppressor in leukemia. Herein, we also observed that ID4 expression at mRNA and protein level was significantly reduced by miR-335 overexpression in both K562 and HL60 (Figure 4A, 4B, and 4E). In order to determine the role of ID4 in the process of leukemogenesis caused by miR-335 overexpression, we restored ID4 expression in K562/miR-335 and HL60/miR-335 cells. ID4 overexpression was confirmed by RT-qPCR and western blot (Figure 4C–4E). Restoration of ID4 expression significantly inhibited the proliferation ability in K562/miR-335 and HL60/miR-335 cells (Figure 4F and 4G), at the same time, with decreased expression of proliferation-related proteins PCNA and Cyclin-D1 (Figure 4J). Moreover, the apoptosis ratio was significantly upregulated in K562/miR-335 and HL60/miR-335 cells when overexpressing ID4 (Figure 4H and 4I), and confirmed by increased expression of apoptosis-related Caspase-3 (Figure 4G).
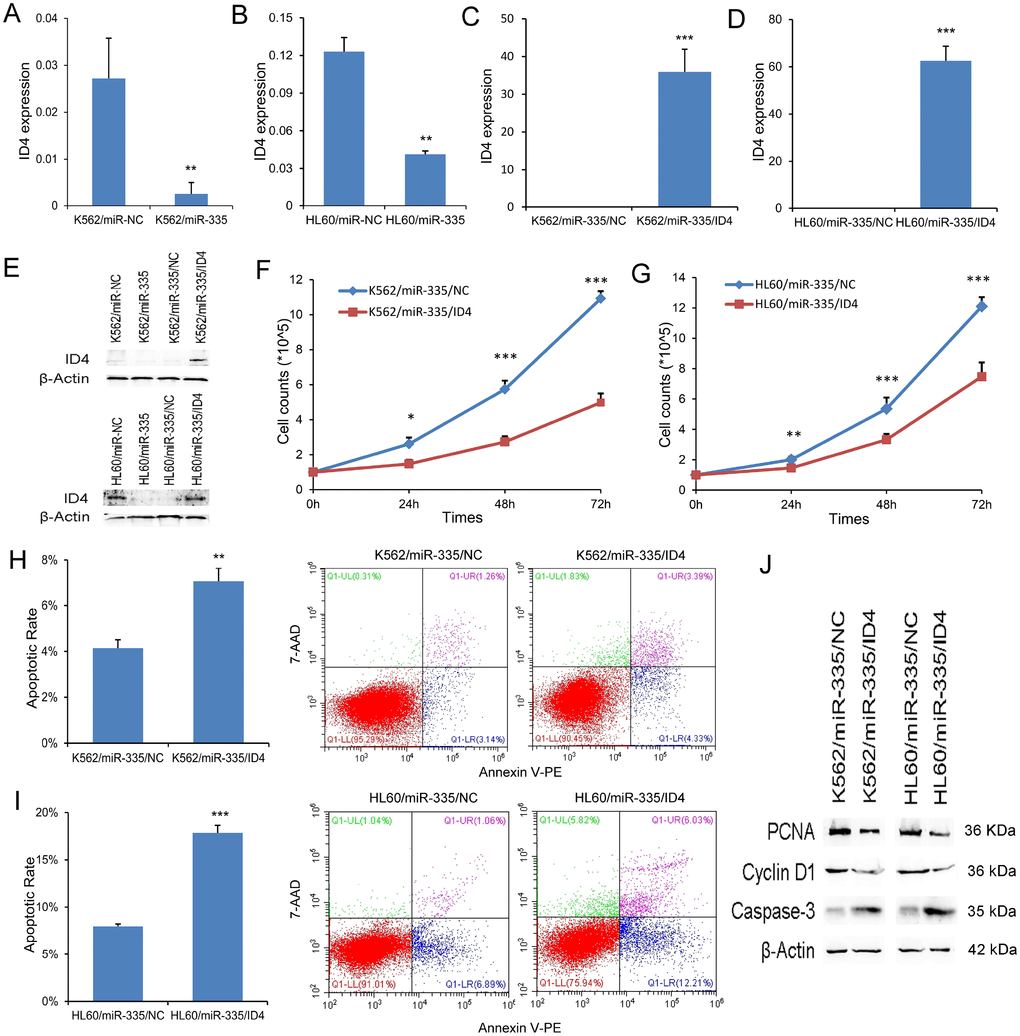
Figure 4. ID4 rescued the pro-leukemia effects of miR-335 in leukemic cell-lines. (A–B) ID4 mRNA expression affected by miR-335 transfection. ID4 mRNA expression was significantly reduced after miR-335 overexpression in both K562 and HL60 cell-lines. (C–D) Confirmation of ID4 mRNA expression after ID4 restoration. ID4 mRNA expression was significantly upregulated after ID4 transfection in both K562/miR-335 and HL60/miR-335 cells. (E) ID4 protein expression affected by miR-335 overexpression and ID4 restoration. ID4 protein expression was significantly reduced after miR-335 overexpression in both K562 and HL60 cell-lines, and was increased after ID4 restoration. (F–G) The effect of ID4 restoration on cell proliferation. Restoration of ID4 significantly reduced the proliferation ability in K562/miR-335 and HL60/miR-335 cells. (H–I) The effect of ID4 restoration on cell apoptosis. Restoration of ID4 significantly increased the apoptosis ratio in K562/miR-335 and HL60/miR-335 cells. (J) The expression of proliferation-related proteins (PCNA and Cyclin D1) and apoptosis-related proteins (Caspase-3) affected by ID4 restoration. The expression of proliferation-related proteins (PCNA and Cyclin D1) was decreased, whereas the apoptosis-related proteins (Caspase-3) expression was increased after ID4 restoration in K562/miR-335 and HL60/miR-335 cells. *, P<0.05; **, P<0.01; ***, P<0.001.
MiR-335/ID4 contributed to leukemogenesis through PI3K/Akt signaling pathway
To further identify the biological network and potential mechanism involved in the pro-leukemia effects of miR-335/ID4, we first analyzed the differentially expressed genes (DEGs) between ID4low and ID4high groups of AML patients among TCGA datasets. A total of 998 DEGs (892 positively correlated and 106 negatively correlated, FDR<0.05, P<0.05 and |log2 FC|>1.5) were identified (Figure 5A and 5B). To gain further insight into the function of identified DEGs, functional and pathway enrichment analysis was performed. We found that the DEGs mainly involved in PI3K/Akt signaling pathway (Figure 5C). Moreover, GSEA showed the significantly enriched in genes down-regulated in mouse prostate by transgenic expression of human AKT1 gene in ID4high AML (Figure 5D). In addition, we also observed that the negative association between ID4 and AKT1/AKT2 expression in AML patients among TCGA datasets (Figure 5E, Spearman test).
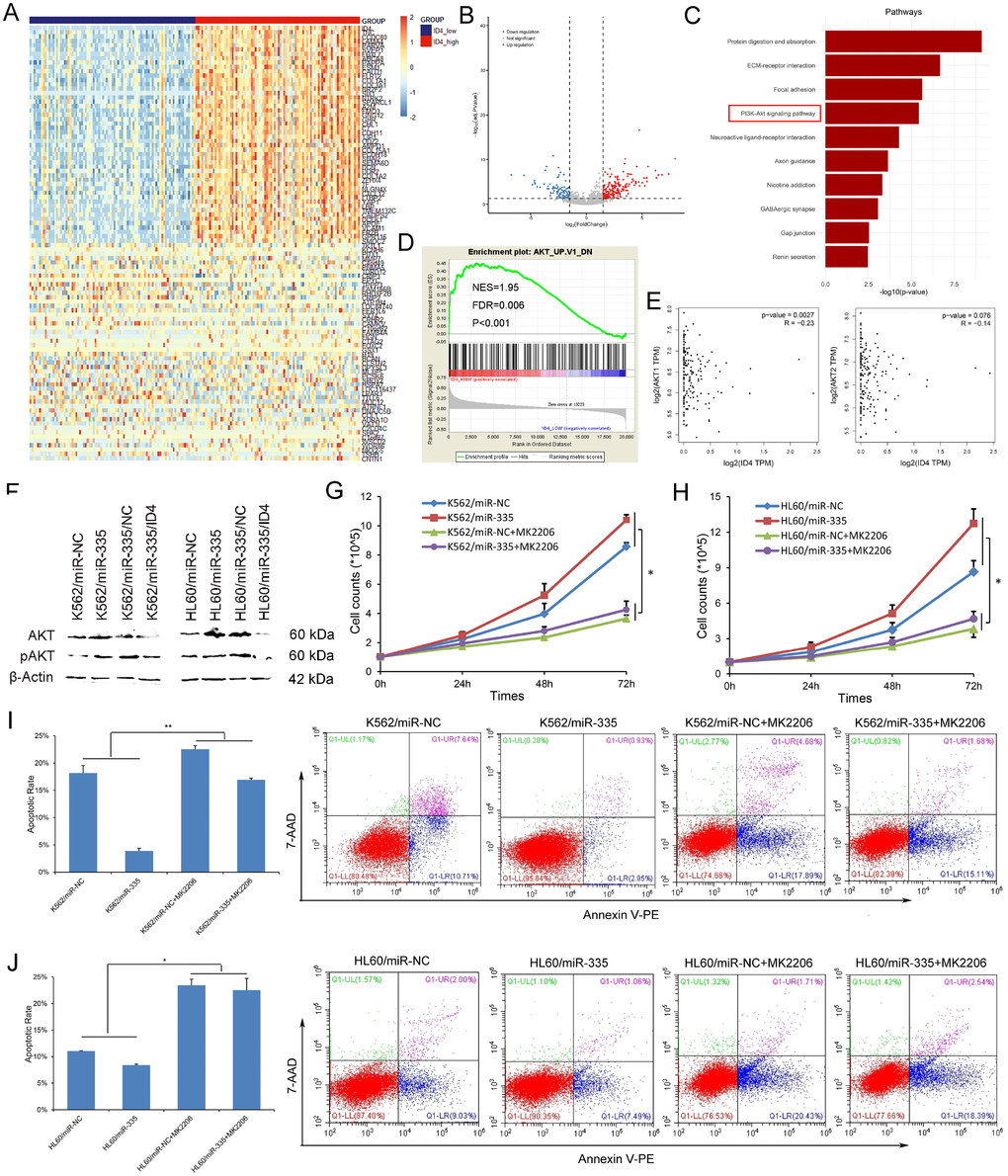
Figure 5. MiR-335 contributed to leukemogenesis through PI3K/Akt signaling pathway. (A) Expression heatmap of top 50 differentially expressed genes (DEGs) between ID4low and ID4high AML patients among TCGA datasets (FDR<0.05, P<0.05 and |log2 FC|>1.5). (B) Volcano plot of DEGs. (C) Significantly enriched pathway terms of DEGs in AML. DEGs functional and signaling pathway enrichment was conducted using online website of STRING (http://string-db.org). (D) GSEA showed the significantly enriched in genes down-regulated in mouse prostate by transgenic expression of human AKT1 gene in ID4high AML. (E) The association between ID4 expression and AKT1/AKT2 expression in AML among TCGA datasets. A negative correlation was observed between ID4 and AKT1/AKT2 expression. The correlation analysis conducted through online web GEPIA (http://gepia.cancer-pku.cn/detail.php?clicktag=correlation) using Spearman test. (F) The expression of Akt and pAkt affected by miR-335 overexpression and ID4 restoration. Akt and pAkt protein expression was significantly increased after miR-335 overexpression in both K562 and HL60 cell-lines, and was decreased after ID4 restoration. (G–H) The effect of miR-335 on proliferation of K562 and HL60 cell-lines with/without Akt inhibitor MK2206 2HCL. MK2206 2HCL remarkably reversed and impaired the pro-proliferative effect in K562 and HL60 cell-lines. (I–J) The effect of miR-335 on apoptosis of K562 and HL60 cell-lines with/without Akt inhibitor MK2206 2HCL. MK2206 2HCL remarkably reversed and impaired the pro-proliferative effect in K562 and HL60 cell-lines. *, P<0.05; **, P<0.01; ***, P<0.001.
Next, we confirmed that miR-335 overexpression significantly upregulated the expression of both Akt and pAkt, which was reversed by ID4 overexpression in both K562 and HL60 leukemic cell-lines (Figure 5F). To further validate whether PI3K/Akt signaling was involved in the pro-leukemia effects of miR-335 overexpression on K562 and HL60 cell-lines, MK-2206 2HCL which is an inhibitor of Akt was further used to block PI3K/Akt signaling pathway. We found that MK-2206 2HCL remarkably reversed and impaired the pro-proliferative and anti-apoptotic effects in K562 and HL60 cell-lines (Figure 5G–5J). All the results suggested that miR-335/ID4 contributed to leukemogenesis through activating PI3K/Akt signaling pathway.
Discussion
MiR-335, which is transcribed from the chromosome 7q32.2, has been observed to aberrantly express in diverse cancers including hematological malignances and to play a crucial role in cancer initiation and progression [12]. MiR-335 was identified to be involved in tumor proliferation, apoptosis, migration, invasion and metastasis by targeting multiple genes and some and some cell signaling pathways in vitro or/and in vivo [12]. the present study, we found that miR-335 was overexpressed in AML patients, and increased expression of miR-335 expression was negatively associated with decreased expression of ID4, which acted as tumor suppressor in AML [19]. Further functional studies demonstrated that miR-335 exhibited pro-proliferative and anti-apoptotic effects by targeting ID4 in AML. Two recent studies also showed that overexpression of miR-335 was a frequent event in both adult and pediatric AML [13, 14]. Similarly, miR-335 was also a promoter of neurologic tumors and multiple myeloma [20–22]. In contrast, miR-335 was frequently downregulated and functioned mainly as a suppressor in a majority of the solid tumors, such as hepatocellular cancer, prostate cancer, clear cell renal cell carcinoma, colorectal cancer, breast cancer, cervical cancer, ovarian cancer, and lung cancer [12]. The conflicting results showed that miR-335 played either as tumor suppressor genes or oncogenes mainly depending on various cancer types.
With progress in genomics technology has tremendously increased our knowledge of the molecular heterogeneity of AML, and has these insights been translated into improved disease classification, clinical care, and novel therapeutic approaches [23, 24]. Herein, we showed that combined miR-335 and ID4 expression was associated chemotherapy response in AML. Several studies showed the direct role of miR-335 in regulating drug resistance. Martin et al. reported that miR-335 played an oncogenic role in promoting agonistic estrogen signaling in a cancerous setting, and resulted in the enhanced resistance of MCF-7 cells to the growth inhibitory effects of tamoxifen [25]. Moreover, miR-335 was identified to regulate the chemo-radioresistance of small cell lung cancer cells by targeting PARP-1 [26]. Kim et al. also revealed that miR-335 conferred sensitivity to anti-cancer drugs by increasing the expression of HDAC3 [27]. As a result, it was not surprising that miR-335/ID4 expression correlated with clinical outcome. Similarly, the effect of miR-335 overexpression on both CR and OS in AML was also confirmed by the two previously studies regarding miR-335 expression in AML [13, 14]. All the results suggested miR-335/ID4 expression were potential prognostic biomarkers in AML.
The PI3K/Akt signaling pathway is crucial to widely divergent physiological processes including cell proliferation, cell cycle progression, cell apoptosis, cell survival, differentiation, angiogenesis and drug resistance [28]. Approximately 60% of AML patients showed Akt phosphorylated on Thr308 and/or Ser473, indicating the activation of PI3K/Akt signaling is a frequent event in AML [29]. In addition, both LFS and OS were significantly shorter in AML cases where pathway up-regulation was documented [30]. Activation of PI3K/Akt signaling in AML may be resulted from several factors, including mutations of FLT3, c-Kit, and N/K-Ras mutations, PI3K p110β/δ overexpression [31]. Herein, although we did not observe the associations of aberrant miR-335/ID4 expression with these gene mutations in AML, evidence shwed that miR-335/ID4 expression was associated Akt/pAkt expression in leukemic cell-lines and AML patients, and inhibition of PI3K/Akt signaling reversed and impaired the pro-proliferative and anti-apoptotic effects mediated by miR-335 overexpression in K562 and HL60 cell-lines. All the results provided new insights into the underlying mechanism of PI3K/Akt activation in AML, and gave potentially novel targets for drug development to treat AML patients in the future.
Taken together, our findings revealed that aberrant miR-335/ID4 expression was a frequent event, and independently affected clinical outcome in AML. Moreover, miR-335/ID4 dysregulation facilitated leukemogenesis through the activation of PI3K/Akt signaling pathway.
Materials and Methods
Patients and treatment
The present study was approved by Institutional Ethics Committee of the Affiliated People’s Hospital of Jiangsu University. After written informed consents were obtained from all participants, a total of 146 bone marrow (BM) samples collected from AML patient and 46 BM collected from healthy donors were included in this study. The diagnosis and classification of AML patients were established according to the French-American-British (FAB) classification and the revised World Health Organization (WHO) criteria [32, 33]. The characteristics of AML patients were summarized in Table 1. Treatment regimens for AML patients were induction chemotherapy and subsequent consolidation chemotherapy as reported [34, 35].
Cytogenetic analysis and BMMNCs samples preparation
Karyotypes were analyzed at the newly diagnosis time by conventional R-banding method according to the previous literature [36]. BM mononuclear cells (BMMNCs) were separated by density-gradient centrifugation using Lymphocyte Separation Medium (Absin, Shanghai, China), and used for RNA and DNA extraction once BMMNCs were isolated.
Cell line and cell culture
Human leukemic cell lines K562 and HL60 were cultured in RPMI 1640 medium (BOSTER, Wuhan, China) containing 10% fetal calf serum (ExCell Bio, Shanghai, China) and grown at 37°C in 5% CO2 humidified atmosphere.
RNA isolation and reverse transcription
Total RNA isolation was conducted as reported previously [37]. Reverse transcription obtained cDNA used for miRNAs was performed using MiScript Reverse Transcription Kit (Qiagen, Duesseldorf, Germany), whereas cDNA used for mRNAs was performed using random primers as reported [19, 38]. cDNA was stored at -80°C until used.
RT-qPCR
The expression of miR-335 was detected by real-time quantitative PCR (RT-qPCR) using miScript SYBR green PCR kit (Qiagen, Duesseldorf, Germany). U6 detected by the same reagent was used to calculate the abundance of miR-335 level. The primers for miR-335 and U6 were listed in Supplementary Table 1. ID4 expression was examined by RT-qPCR using AceQ qPCR SYBR Green Master Mix (Vazyme Biotech Co., Piscataway, NJ, USA). The housekeeping gene ABL1 detected by 2×SYBR Green PCR Mix (Multisciences, Hangzhou, China) was used to calculate the abundance of ID4 transcript. The primers for ID4 and ABL1 were shown in Supplementary Table 1. Relative miR-335 and ID4 expression was calculated using 2-ΔΔCT method.
DNA isolation, bisulfite modification, and RQ-MSP
Genomic DNA isolation and modification were performed as reported previously [19], and modified DNA were stored at -80°C until used. The level of miR-335 methylation was detected by real-time quantitative methylation-specific PCR (RQ-MSP) using AceQ qPCR SYBR Green Master Mix (Vazyme Biotech Co., Piscataway, NJ, USA) with primers showed in Supplementary Table 1. ALU detected by 2×SYBR Green PCR Mix (Multisciences, Hangzhou, China) with primers (Supplementary Table 1) was used to calculate the abundance of miR-335 methylation level. Relative miR-335 methylation level was calculated using 2-ΔΔCT method.
BSP
TaKaRa TaqTM Hot Start Version kit (Tokyo, Japan) was used for bisulfite sequencing PCR (BSP) reaction with primers showed in Supplementary Table 1. BSP products cloning sequencing was performed as described [19]. Eight independent clones from each specimen were sequenced (BGI Tech Solutions Co., Shanghai, China).
Gene mutation detection
Gene mutations including CEBPA, NPM1, FLT3-ITD, C-KIT, NRAS/KRAS, IDH1/2, DNMT3A, U2AF1, and SRSF2 were detected by high-resolution melting analysis (HRMA) and/or direct DNA sequencing as reported [39–44].
Plasmid construction and transfection
Human mature miR-335 sequences were cloned in pGCMV/EGFP/miR/Blasticidin expression vector, whereas human full-length ID4 CDS sequences were cloned in PEX-2 expression vector, and all of them were purchased from GenePharma (Shanghai, China). Cell transfection was peformed using HiPerFect Transfection Reagent (Qiagen, Duesseldorf, Germany). MiR-335/ID4 stably expressed cells were selected by Blasticidin and G418 (InvivoGen, San Diego, CA, USA), respectively, and flow sorting (BD FACSAriall, San Jose, CA, USA).
Treatment with Akt inhibitor MK-2206 2HCL
The Akt inhibitor MK-2206 2HCL (Selleck Chemicals, Houston, TX, USA) was used to block PI3K/Akt signaling in leukemic cell-lines at a final concentrations of 5 μM.
Western blot
Western blotting was performed as described previously [45]. The antibodies were mouse anti-β-actin (BOSTER, Wuhan, China), rabbit anti-ID4 (Abcam, Cambridge, MA, USA), rabbit anti-PCNA (Cell Signaling Technology, Danvers, MA, USA), rabbit anti-Cyclin D1 (Cell Signaling Technology, Danvers, MA, USA), rabbit anti-Caspase-3 (Cell Signaling Technology, Danvers, MA, USA), rabbit anti-Akt/pAkt (Beijing Solarbio Science & Technology Co., Ltd., Beijing, China), and anti-mouse/anti-rabbit secondary antibodies (Fcmacs, Nanjing, China).
Cell proliferation assays
Cells (1×105 cells/ml) were seeded onto a 6-well plate in RPMI 1640 medium containing 10% fetal calf serum. After culturing for 0, 1, 2, and 3 days, cells were counted in counting board for three times.
Cell apoptosis analysis
Cells (2×105 cells/ml) were seeded onto a 6-well plate in RPMI 1640 medium containing 1% fetal calf serum for 2 days. Annexin V-PE/7-AAD apoptosis detection kits (BD Pharmingen, San Diego, CA, USA) were used to analyze the apoptosis rate according to the manufacturer’s protocols, and then analyzed via flow cytometry (Beckman Coulter, Miami, FL, USA). Each experiment was repeated three times.
Immunophenotypic analysis by flow cytometry
Flow cytometric analysis was performed to determine surface marker expression. Cells at logarithmic growth phase (5×105 cells/ml) were stained with antibodies in phosphatebuffered saline (PBS) at 4°C for 30 min, and then washed and resuspended in 300 μL FACS buffer. The antibody was anti-human/mouse CD11b PE (Biogems, Rocky Hill, NJ, USA).
TCGA and GEO datasets
A cohort of 200 AML patients (NEJM 2013) from The Cancer Genome Atlas (TCGA) [23] was downloaded via cBioPortal (http://www.cbioportal.org) [46, 47]. A cohort of 162 cytogenetically normal AML (CN-AML) patients (GSE12417) from Gene Expression Omnibus (GEO) data was also used (https://www.ncbi.nlm.nih.gov/geo/).
Bioinformatics analyses
The human disease methylation database DiseaseMeth version 2.0 (http://bio-bigdata.hrbmu.edu.cn/diseasemeth/) was used for differential methylation analysis for miR-335/MEST (miR-335 host gene). The prognostic impact of ID4 expression in CN-AML patients for GSE12417 analyzed through the online web Genomicscape (http://genomicscape.com/microarray/survival.php) [48, 49]. The correlation between ID4 and AKT1/AKT2 expression was analyzed through online web GEPIA (http://gepia.cancer-pku.cn/detail.php?clicktag=correlation) [50]. Gene Set Enrichment Analysis (GSEA) was performed using GSEA v3.0 software (http://www.broad.mit.edu/gsea). Functional and signaling pathway enrichment was conducted using online website of STRING (http://string-db.org).
Statistical analyses
SPSS 20.0 software package (IBM, Armonk, NY, USA) was applied to statistical analyses. Student's T test/Mann-Whitney’s U test was performed to compare the differences of continuous variables. The difference of categorical variables was analyzed using Pearson Chi-square analysis/Fisher exact test. Spearman correlation test was conducted to evaluate the correlation between continuous variables. The receiver operating characteristic (ROC) curve and area under the ROC curve (AUC) were carried out to assess the discriminative capacity of miR-335/ID4 expression between patients and controls. Kaplan-Meier and Cox regression analysis (univariate and multivariate analysis) were used to analyze the impact of miR-335/ID4 expression on survival. Statistical significance was set at P<0.05 and all tests were two sided.
Supplementary Materials
Conflicts of Interest
None of the authors has any conflict of interest to disclose.
Funding
This work was supported by National Natural Science foundation of China (81270630), Medical Innovation Team of Jiangsu Province (CXTDB2017002), Six Talent Peaks Project in Jiangsu Province (2015-WSN-115), Zhenjiang Clinical Research Center of Hematology (SS2018009), Postgraduate Research & Practice Innovation Program of Jiangsu Province (KYCX17_1821, KYCX18_2281), Social Development Foundation of Zhenjiang (SH2016045, SH2017040, SH2018044), Clinical Medical Science Development Foundation of Jiangsu University (JLY20160011).
References
- 1. Döhner H, Weisdorf DJ, Bloomfield CD. Acute Myeloid Leukemia. N Engl J Med. 2015; 373:1136–52. https://doi.org/10.1056/NEJMra1406184 [PubMed]
- 2. Döhner H, Estey E, Grimwade D, Amadori S, Appelbaum FR, Büchner T, Dombret H, Ebert BL, Fenaux P, Larson RA, Levine RL, Lo-Coco F, Naoe T, et al. Diagnosis and management of AML in adults: 2017 ELN recommendations from an international expert panel. Blood. 2017; 129:424–47. https://doi.org/10.1182/blood-2016-08-733196 [PubMed]
- 3. Bullinger L, Döhner K, Döhner H. Genomics of Acute Myeloid Leukemia Diagnosis and Pathways. J Clin Oncol. 2017; 35:934–46. https://doi.org/10.1200/JCO.2016.71.2208 [PubMed]
- 4. Avivi I, Rowe JM. Prognostic factors in acute myeloid leukemia. Curr Opin Hematol. 2005; 12:62–67. https://doi.org/10.1097/01.moh.0000148760.15412.df [PubMed]
- 5. Grimwade D. The clinical significance of cytogenetic abnormalities in acute myeloid leukaemia. Best Pract Res Clin Haematol. 2001; 14:497–529. https://doi.org/10.1053/beha.2001.0152 [PubMed]
- 6. Marcucci G, Haferlach T, Döhner H. Molecular genetics of adult acute myeloid leukemia: prognostic and therapeutic implications. J Clin Oncol. 2011; 29:475–86. https://doi.org/10.1200/JCO.2010.30.2554 [PubMed]
- 7. Farh KK, Grimson A, Jan C, Lewis BP, Johnston WK, Lim LP, Burge CB, Bartel DP. The widespread impact of mammalian MicroRNAs on mRNA repression and evolution. Science. 2005; 310:1817–21. https://doi.org/10.1126/science.1121158 [PubMed]
- 8. Lim LP, Lau NC, Garrett-Engele P, Grimson A, Schelter JM, Castle J, Bartel DP, Linsley PS, Johnson JM. Microarray analysis shows that some microRNAs downregulate large numbers of target mRNAs. Nature. 2005; 433:769–73. https://doi.org/10.1038/nature03315 [PubMed]
- 9. Lu J, Getz G, Miska EA, Alvarez-Saavedra E, Lamb J, Peck D, Sweet-Cordero A, Ebert BL, Mak RH, Ferrando AA, Downing JR, Jacks T, Horvitz HR, Golub TR. MicroRNA expression profiles classify human cancers. Nature. 2005; 435:834–38. https://doi.org/10.1038/nature03702 [PubMed]
- 10. Yeh CH, Moles R, Nicot C. Clinical significance of microRNAs in chronic and acute human leukemia. Mol Cancer. 2016; 15:37. https://doi.org/10.1186/s12943-016-0518-2 [PubMed]
- 11. Chen J, Odenike O, Rowley JD. Leukaemogenesis: more than mutant genes. Nat Rev Cancer. 2010; 10:23–36. https://doi.org/10.1038/nrc2765 [PubMed]
- 12. Luo LJ, Wang DD, Wang J, Yang F, Tang JH. Diverse roles of miR-335 in development and progression of cancers. Tumour Biol. 2016; 37:15399–410. Epub ahead of print. https://doi.org/10.1007/s13277-016-5385-3 [PubMed]
- 13. Yingchun L, Rong Z, Kun Y, Ying Y, Zhuogang L. Bone Marrow MicroRNA-335 Level Predicts the Chemotherapy Response and Prognosis of Adult Acute Myeloid Leukemia. Medicine (Baltimore). 2015; 94:e0986. https://doi.org/10.1097/MD.0000000000000986 [PubMed]
- 14. Lin X, Wang Z, Zhang R, Feng W. High serum microRNA-335 level predicts aggressive tumor progression and unfavorable prognosis in pediatric acute myeloid leukemia. Clin Transl Oncol. 2015; 17:358–64. https://doi.org/10.1007/s12094-014-1237-z [PubMed]
- 15. Sandoval-Bórquez A, Polakovicova I, Carrasco-Véliz N, Lobos-González L, Riquelme I, Carrasco-Avino G, Bizama C, Norero E, Owen GI, Roa JC, Corvalán AH. MicroRNA-335-5p is a potential suppressor of metastasis and invasion in gastric cancer. Clin Epigenetics. 2017; 9:114. https://doi.org/10.1186/s13148-017-0413-8 [PubMed]
- 16. Dohi O, Yasui K, Gen Y, Takada H, Endo M, Tsuji K, Konishi C, Yamada N, Mitsuyoshi H, Yagi N, Naito Y, Tanaka S, Arii S, Yoshikawa T. Epigenetic silencing of miR-335 and its host gene MEST in hepatocellular carcinoma. Int J Oncol. 2013; 42:411–18. https://doi.org/10.3892/ijo.2012.1724 [PubMed]
- 17. Png KJ, Yoshida M, Zhang XH, Shu W, Lee H, Rimner A, Chan TA, Comen E, Andrade VP, Kim SW, King TA, Hudis CA, Norton L, et al. MicroRNA-335 inhibits tumor reinitiation and is silenced through genetic and epigenetic mechanisms in human breast cancer. Genes Dev. 2011; 25:226–31. https://doi.org/10.1101/gad.1974211 [PubMed]
- 18. Heyn H, Engelmann M, Schreek S, Ahrens P, Lehmann U, Kreipe H, Schlegelberger B, Beger C. MicroRNA miR-335 is crucial for the BRCA1 regulatory cascade in breast cancer development. Int J Cancer. 2011; 129:2797–806. https://doi.org/10.1002/ijc.25962 [PubMed]
- 19. Zhou JD, Zhang TJ, Li XX, Ma JC, Guo H, Wen XM, Zhang W, Yang L, Yan Y, Lin J, Qian J. Epigenetic dysregulation of ID4 predicts disease progression and treatment outcome in myeloid malignancies. J Cell Mol Med. 2017; 21:1468–81. https://doi.org/10.1111/jcmm.13073 [PubMed]
- 20. Ronchetti D, Lionetti M, Mosca L, Agnelli L, Andronache A, Fabris S, Deliliers GL, Neri A. An integrative genomic approach reveals coordinated expression of intronic miR-335, miR-342, and miR-561 with deregulated host genes in multiple myeloma. BMC Med Genomics. 2008; 1:37. https://doi.org/10.1186/1755-8794-1-37 [PubMed]
- 21. Shu M, Zheng X, Wu S, Lu H, Leng T, Zhu W, Zhou Y, Ou Y, Lin X, Lin Y, Xu D, Zhou Y, Yan G. Targeting oncogenic miR-335 inhibits growth and invasion of malignant astrocytoma cells. Mol Cancer. 2011; 10:59. https://doi.org/10.1186/1476-4598-10-59 [PubMed]
- 22. Shi L, Jiang D, Sun G, Wan Y, Zhang S, Zeng Y, Pan T, Wang Z. miR-335 promotes cell proliferation by directly targeting Rb1 in meningiomas. J Neurooncol. 2012; 110:155–62. https://doi.org/10.1007/s11060-012-0951-z [PubMed]
- 23. Ley TJ, Miller C, Ding L, Raphael BJ, Mungall AJ, Robertson A, Hoadley K, Triche TJ
Jr , Laird PW, Baty JD, Fulton LL, Fulton R, Heath SE, et al, Cancer Genome Atlas Research Network. Genomic and epigenomic landscapes of adult de novo acute myeloid leukemia. N Engl J Med. 2013; 368:2059–74. https://doi.org/10.1056/nejmoa1301689 [PubMed] - 24. Papaemmanuil E, Gerstung M, Bullinger L, Gaidzik VI, Paschka P, Roberts ND, Potter NE, Heuser M, Thol F, Bolli N, Gundem G, Van Loo P, Martincorena I, et al. Genomic Classification and Prognosis in Acute Myeloid Leukemia. N Engl J Med. 2016; 374:2209–21. https://doi.org/10.1056/NEJMoa1516192 [PubMed]
- 25. Martin EC, Conger AK, Yan TJ, Hoang VT, Miller DF, Buechlein A, Rusch DB, Nephew KP, Collins-Burow BM, Burow ME. MicroRNA-335-5p and -3p synergize to inhibit estrogen receptor alpha expression and promote tamoxifen resistance. FEBS Lett. 2017; 591:382–92. https://doi.org/10.1002/1873-3468.12538 [PubMed]
- 26. Luo Y, Tong L, Meng H, Zhu W, Guo L, Wei T, Zhang J. MiR-335 regulates the chemo-radioresistance of small cell lung cancer cells by targeting PARP-1. Gene. 2017; 600:9–15. https://doi.org/10.1016/j.gene.2016.11.031 [PubMed]
- 27. Kim Y, Kim H, Park D, Jeoung D. miR-335 Targets SIAH2 and Confers Sensitivity to Anti-Cancer Drugs by Increasing the Expression of HDAC3. Mol Cells. 2015; 38:562–72. https://doi.org/10.14348/molcells.2015.0051 [PubMed]
- 28. Vanhaesebroeck B, Guillermet-Guibert J, Graupera M, Bilanges B. The emerging mechanisms of isoform-specific PI3K signalling. Nat Rev Mol Cell Biol. 2010; 11:329–41. https://doi.org/10.1038/nrm2882 [PubMed]
- 29. Xu Q, Simpson SE, Scialla TJ, Bagg A, Carroll M. Survival of acute myeloid leukemia cells requires PI3 kinase activation. Blood. 2003; 102:972–80. https://doi.org/10.1182/blood-2002-11-3429 [PubMed]
- 30. Tamburini J, Elie C, Bardet V, Chapuis N, Park S, Broët P, Cornillet-Lefebvre P, Lioure B, Ugo V, Blanchet O, Ifrah N, Witz F, Dreyfus F, et al. Constitutive phosphoinositide 3-kinase/Akt activation represents a favorable prognostic factor in de novo acute myelogenous leukemia patients. Blood. 2007; 110:1025–28. https://doi.org/10.1182/blood-2006-12-061283 [PubMed]
- 31. Martelli AM, Evangelisti C, Chiarini F, Grimaldi C, Manzoli L, McCubrey JA. Targeting the PI3K/AKT/mTOR signaling network in acute myelogenous leukemia. Expert Opin Investig Drugs. 2009; 18:1333–49. https://doi.org/10.1517/14728220903136775 [PubMed]
- 32. Arber DA, Orazi A, Hasserjian R, Thiele J, Borowitz MJ, Le Beau MM, Bloomfield CD, Cazzola M, Vardiman JW. The 2016 revision to the World Health Organization classification of myeloid neoplasms and acute leukemia. Blood. 2016; 127:2391–405. https://doi.org/10.1182/blood-2016-03-643544 [PubMed]
- 33. Bennett JM, Catovsky D, Daniel MT, Flandrin G, Galton DA, Gralnick HR, Sultan C. Proposed revised criteria for the classification of acute myeloid leukemia. A report of the French-American-British Cooperative Group. Ann Intern Med. 1985; 103:620–25. https://doi.org/10.7326/0003-4819-103-4-620 [PubMed]
- 34. Zhang TJ, Zhou JD, Zhang W, Lin J, Ma JC, Wen XM, Yuan Q, Li XX, Xu ZJ, Qian J. H19 overexpression promotes leukemogenesis and predicts unfavorable prognosis in acute myeloid leukemia. Clin Epigenetics. 2018; 10:47. https://doi.org/10.1186/s13148-018-0486-z [PubMed]
- 35. Zhou JD, Wang YX, Zhang TJ, Li XX, Gu Y, Zhang W, Ma JC, Lin J, Qian J. Identification and validation of SRY-box containing gene family member SOX30 methylation as a prognostic and predictive biomarker in myeloid malignancies. Clin Epigenetics. 2018; 10:92. https://doi.org/10.1186/s13148-018-0523-y [PubMed]
- 36. Grimwade D, Hills RK, Moorman AV, Walker H, Chatters S, Goldstone AH, Wheatley K, Harrison CJ, Burnett AK, and National Cancer Research Institute Adult Leukaemia Working Group. Refinement of cytogenetic classification in acute myeloid leukemia: determination of prognostic significance of rare recurring chromosomal abnormalities among 5876 younger adult patients treated in the United Kingdom Medical Research Council trials. Blood. 2010; 116:354–65. https://doi.org/10.1182/blood-2009-11-254441 [PubMed]
- 37. Zhou JD, Yang L, Zhang YY, Yang J, Wen XM, Guo H, Yao DM, Ma JC, Chen Q, Lin J, Qian J. Overexpression of BAALC: clinical significance in Chinese de novo acute myeloid leukemia. Med Oncol. 2015; 32:386. https://doi.org/10.1007/s12032-014-0386-9 [PubMed]
- 38. Zhou JD, Zhang LC, Zhang TJ, Gu Y, Wu DH, Zhang W, Ma JC, Wen XM, Guo H, Lin J, Qian J. Dysregulation of miR-200s clusters as potential prognostic biomarkers in acute myeloid leukemia. J Transl Med. 2018; 16:135. https://doi.org/10.1186/s12967-018-1494-7 [PubMed]
- 39. Lin J, Yao DM, Qian J, Chen Q, Qian W, Li Y, Yang J, Wang CZ, Chai HY, Qian Z, Xiao GF, Xu WR. Recurrent DNMT3A R882 mutations in Chinese patients with acute myeloid leukemia and myelodysplastic syndrome. PLoS One. 2011; 6:e26906. https://doi.org/10.1371/journal.pone.0026906 [PubMed]
- 40. Lin J, Yao DM, Qian J, Chen Q, Qian W, Li Y, Yang J, Wang CZ, Chai HY, Qian Z, Xiao GF, Xu WR. IDH1 and IDH2 mutation analysis in Chinese patients with acute myeloid leukemia and myelodysplastic syndrome. Ann Hematol. 2012; 91:519–25. https://doi.org/10.1007/s00277-011-1352-7 [PubMed]
- 41. Yang X, Qian J, Sun A, Lin J, Xiao G, Yin J, Chen S, Wu D. RAS mutation analysis in a large cohort of Chinese patients with acute myeloid leukemia. Clin Biochem. 2013; 46:579–83. https://doi.org/10.1016/j.clinbiochem.2012.12.022 [PubMed]
- 42. Qian J, Yao DM, Lin J, Qian W, Wang CZ, Chai HY, Yang J, Li Y, Deng ZQ, Ma JC, Chen XX. U2AF1 mutations in Chinese patients with acute myeloid leukemia and myelodysplastic syndrome. PLoS One. 2012; 7:e45760. https://doi.org/10.1371/journal.pone.0045760 [PubMed]
- 43. Wen XM, Lin J, Yang J, Yao DM, Deng ZQ, Tang CY, Xiao GF, Yang L, Ma JC, Hu JB, Qian W, Qian J. Double CEBPA mutations are prognostically favorable in non-M3 acute myeloid leukemia patients with wild-type NPM1 and FLT3-ITD. Int J Clin Exp Pathol. 2014; 7:6832–40. [PubMed]
- 44. Yang J, Yao DM, Ma JC, Yang L, Guo H, Wen XM, Xiao GF, Qian Z, Lin J, Qian J. The prognostic implication of SRSF2 mutations in Chinese patients with acute myeloid leukemia. Tumour Biol. 2016; 37:10107–14. https://doi.org/10.1007/s13277-015-4716-0 [PubMed]
- 45. Lin J, Ma JC, Yang J, Yin JY, Chen XX, Guo H, Wen XM, Zhang TJ, Qian W, Qian J, Deng ZQ. Arresting of miR-186 and releasing of H19 by DDX43 facilitate tumorigenesis and CML progression. Oncogene. 2018; 37:2432–43. https://doi.org/10.1038/s41388-018-0146-y [PubMed]
- 46. Cerami E, Gao J, Dogrusoz U, Gross BE, Sumer SO, Aksoy BA, Jacobsen A, Byrne CJ, Heuer ML, Larsson E, Antipin Y, Reva B, Goldberg AP, et al. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov. 2012; 2:401–04. https://doi.org/10.1158/2159-8290.CD-12-0095 [PubMed]
- 47. Gao J, Aksoy BA, Dogrusoz U, Dresdner G, Gross B, Sumer SO, Sun Y, Jacobsen A, Sinha R, Larsson E, Cerami E, Sander C, Schultz N. Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci Signal. 2013; 6:pl1. https://doi.org/10.1126/scisignal.2004088 [PubMed]
- 48. Metzeler KH, Hummel M, Bloomfield CD, Spiekermann K, Braess J, Sauerland MC, Heinecke A, Radmacher M, Marcucci G, Whitman SP, Maharry K, Paschka P, Larson RA, et al, and Cancer and Leukemia Group B, and German AML Cooperative Group. An 86-probe-set gene-expression signature predicts survival in cytogenetically normal acute myeloid leukemia. Blood. 2008; 112:4193–201. https://doi.org/10.1182/blood-2008-02-134411 [PubMed]
- 49. Kassambara A, Rème T, Jourdan M, Fest T, Hose D, Tarte K, Klein B. GenomicScape: an easy-to-use web tool for gene expression data analysis. Application to investigate the molecular events in the differentiation of B cells into plasma cells. PLos Comput Biol. 2015; 11:e1004077. https://doi.org/10.1371/journal.pcbi.1004077 [PubMed]
- 50. Tang Z, Li C, Kang B, Gao G, Li C, Zhang Z. GEPIA: a web server for cancer and normal gene expression profiling and interactive analyses. Nucleic Acids Res. 2017; 45:W98–102. https://doi.org/10.1093/nar/gkx247 [PubMed]