Introduction
Plant growth regulators (PGRs) are commonly applied in agriculture to increase the yield and improve the quality of fruits and vegetables. Gibberellin, one of the most common PGRs, can improve the fruit rate, enable the production of parthenocarpic fruits and increase the harvest index [1]. Recently, gibberellin has been widely used in the cultivation of off-season fruit. The global market for gibberellin is in the range of US $500 million [2], and in China, the annual output of gibberellin is estimated to be over 4000 tons. Although gibberellin is usually considered to be non-toxic, especially when applied in the correct dose for the proper amount of time, high doses or inappropriate applications may have adverse effects on humans and animals. A previous study revealed that gibberellin in the drinking water reduced the body weights and food consumption levels of mice [3]. Gibberellin exposure can cause oxidative stress, increase the levels of malondialdehyde and reduce the levels of anti-oxidant enzymes such as catalase (CAT), superoxide dismutase (SOD) and glutathione peroxidase (GPX) [4, 5]. Furthermore, gibberellin has been found to be hepatotoxic, nephrotoxic, genotoxic, neurotoxic and even carcinogenic and teratogenic [6–9].
Long-term exposure to PGRs may substantially impair male reproduction. The herbicide 2,4-dichlorophenoxyacetic acid, a PGR used globally in agriculture, was reported to reduce sperm motility dose-dependently in vitro. Specifically, 2,4-dichlorophenoxyacetic acid reduced the ability of human spermatozoa to penetrate a viscous medium and diminished their capacitation abilities and acrosome reaction rates [10]. Another PGR, 4-chlorophenoxy acetic acid, was found to induce apoptosis in rat gonads [11]. Ethephon, still another PGR, induced chromosomal aberrations (structural and numerical) in spermatocytes and stimulated the expression of γH2AX, an early and sensitive biomarker of genotoxic damage [12, 13]. Gibberellin reduced sperm production and increased the number of immature and abnormal sperm in different model animals [14]. Additionally, gibberellin caused subfertility and even infertility by inhibiting spermatogenesis and impairing testicular tissue [15].
Recently, oxidative stress has been recognized as a major cause of spontaneous male infertility [16]. Environmental pollution can promote reactive oxygen species (ROS) production in male germ cells, and the overproduction of seminal ROS is associated with teratozoospermia and oxidative DNA damage in sperm [17, 18]. The accumulation of active oxygen damages the cell membrane and reduces the activity of the Na+/K+- and Ca2+- adenosine triphosphatases (ATPases), which critically influence sperm motility by maintaining ion homeostasis. Lestari et al. suggested that Na+/K+-ATPase and Ca2+-ATPase activity could predict sperm motility disorder [19]. In addition, adenosine 5′-monophosphate (AMP)-activated protein kinase (AMPK), a protein that regulates energy balance and metabolism, is crucial for maintaining sperm cellular energy homeostasis. In our previous study and this study, we found that the AMPK signaling pathway was associated with sperm motility [20].
Although several studies have investigated the effects of gibberellin on sperm motility in model animals, the impacts of gibberellin on human sperm are not yet understood. Therefore, in the current investigation, we evaluated the effects of gibberellin on the motility of human sperm in vitro and explored the underlying mechanisms.
Results
Gibberellin exposure reduced human sperm motility in vitro
The impacts of gibberellin on sperm were determined at different concentrations and different exposure times (0, 1, 5, 9 and 19 h). Sperm motility was determined with an SCA sperm-class analyzer (Figure 1A and 1B; Supplementary Tables 2 and 3). The motility of sperm exposed to low doses of gibberellin (0.01, 0.04 or 0.08 mM) did not differ from that of the control group; however, high doses of gibberellin (0.1, 0.2, 0.4 and 0.8 mM) dose-dependently reduced sperm motility. Gibberellin exhibited highly acute toxicity at high doses (0.2, 0.4 and 0.8 mM), with a sharp decrease in spermatozoa motility in the first hour in vitro (Supplementary Figure 1A; Supplementary Table 3). Therefore, the experimental time was set to 1 h in further analyses.
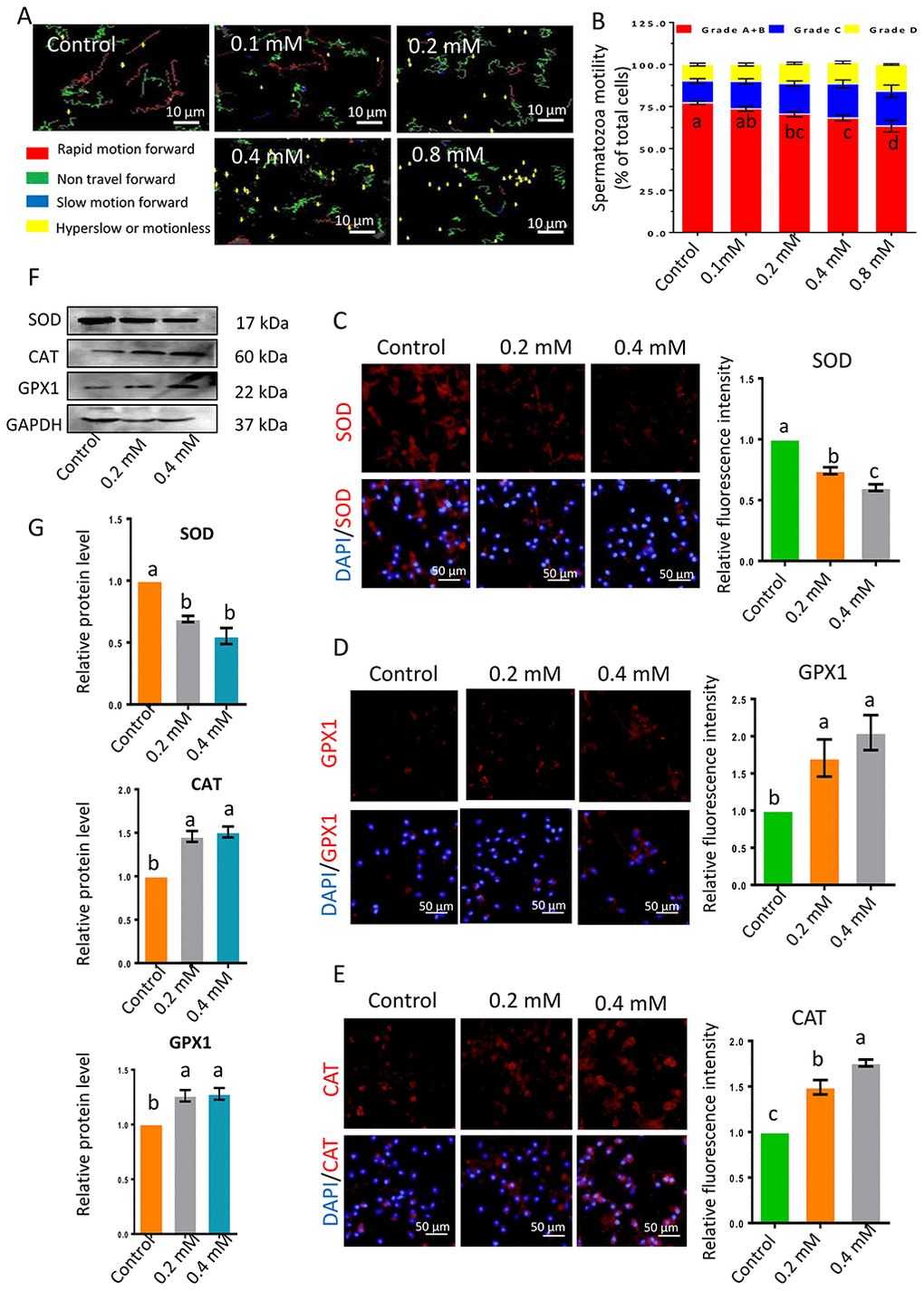
Figure 1. (A) Images depicting sperm motility after 1-h exposure to gibberellin. (B) Quantitative analysis of sperm motility after gibberellin exposure for 1 h. (C–E) The relative protein levels of SOD1, GPX1 and CAT in sperm were detected by immunocytochemistry, and data were quantified based on the relative fluorescence intensity. (F–G) The relative protein levels of SOD1, GPX1 and CAT in sperm were detected by Western blotting. The results are presented as the mean ± SEM.
Gibberellin exposure increased the ROS levels in human sperm in vitro
As ROS significantly influence sperm motility, the ROS levels in sperm were analyzed by flow cytometry. ROS levels were significantly greater in sperm exposed to 0.2- or 0.4-mM gibberellin for 1 h than in the control group (P < 0.05; Figure 2A and Supplementary Figure 1B).
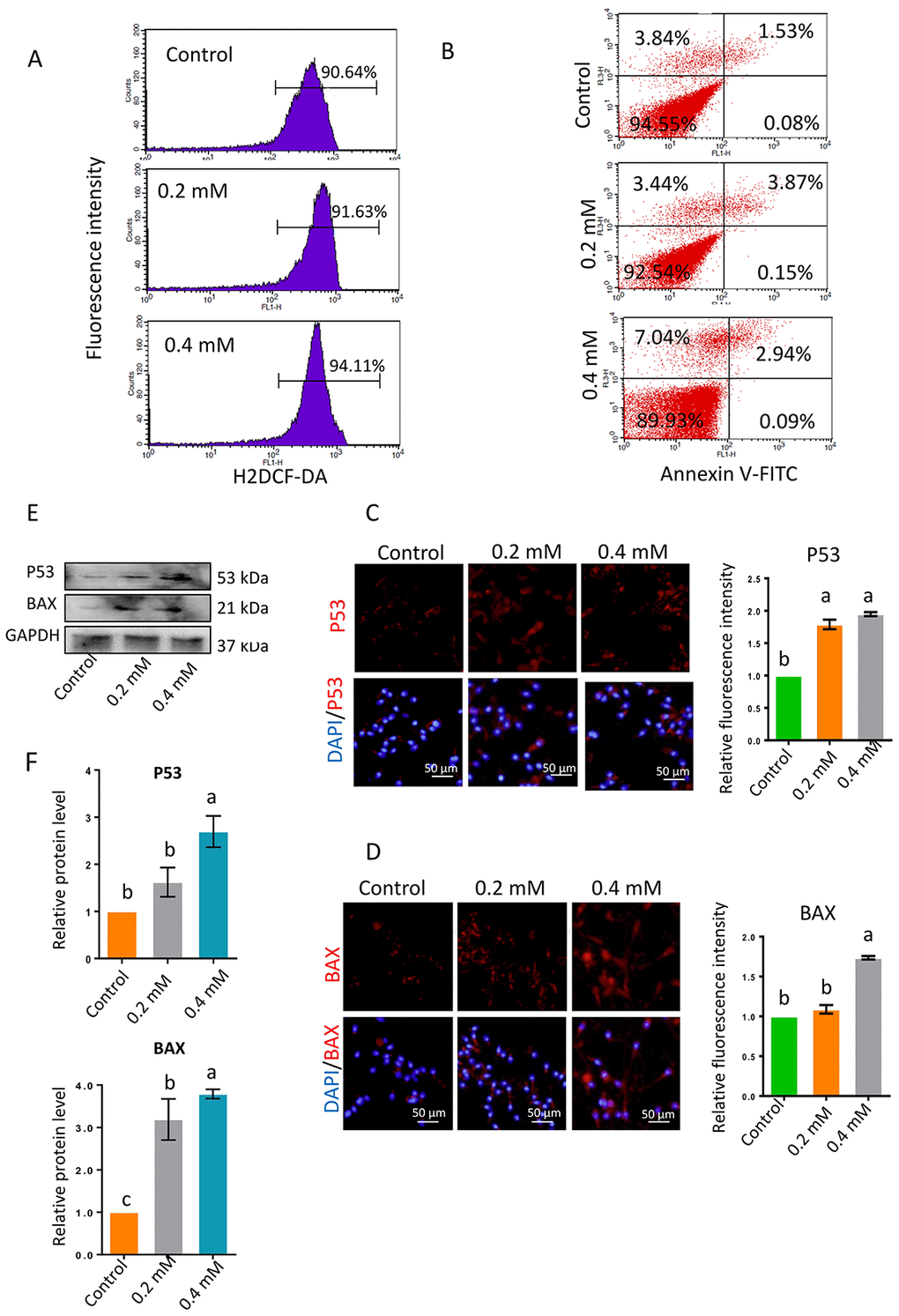
Figure 2. (A) ROS levels in sperm determined by flow cytometry with DCFH-DA after 1-h gibberellin treatment. (B) Spermatozoon plasma membrane phosphatidylserine externalization, determined by flow cytometry with an Annexin V-FITC apoptosis detection kit. (C–D) The relative protein levels of the apoptotic markers BAX and P53 in sperm were detected by immunocytochemistry and quantified based on the relative fluorescence intensity. (E–F) The relative protein levels of BAX and P53 in sperm were detected by Western blotting. The results are presented as the mean ± SEM.
Next, the levels of the anti-oxidant enzymes CAT, SOD and GPX1 were detected by immunocytochemistry and Western blotting. SOD levels were lower in the 0.2- and 0.4-mM gibberellin-exposed groups than in the control group (Figure 1C, 1F and 1G), while CAT and GPX1 levels were significantly increased in the gibberellin-exposed groups compared to the control group (Figure 1D–1G; P < 0.05).
Gibberellin exposure increased apoptosis in human sperm in vitro
To investigate whether apoptosis was involved in the reduction in sperm motility following gibberellin exposure, we used flow cytometry to evaluate the phosphatidylserine externalization of the sperm plasma membrane, a measure of apoptosis in human sperm. Gibberellin exposure induced plasma membrane scrambling, although the level of phosphatidylserine externalization in the sperm plasma membrane remained very low (Annexin V+/PI- ratio < 5% of total viable sperm). Exposure to 0.4-mM gibberellin triggered sperm apoptosis (Figure 2B and Supplementary Figure 1C; P < 0.01).
In addition, the protein levels of P53 and BAX were detected by immunocytochemistry and Western blotting. The expression of these proteins was significantly greater in sperm exposed to 0.4-mM gibberellin than in the control group (Figure 2C–2F; P < 0.01).
Gibberellin exposure in vitro reduced ATP synthase protein levels and activated the AMPK signaling pathway
Na+/K+-ATPase and Ca2+-ATPase activity are vital for sperm motility. In the current investigation, Na+/K+-ATPase and Ca2+-ATPase activity decreased significantly following gibberellin exposure (Figure 3A and Supplementary Table 4; P < 0.01). The protein levels of two major adenosine triphosphate (ATP) synthases, ATP-5A and ATP-5B, were also significantly reduced by gibberellin exposure (Figure 3B–3E; P < 0.01). Despite these changes, gibberellin exposure did not alter the population of sperm with a high mitochondrial membrane potential (ΔΨm, Supplementary Figure 1D).
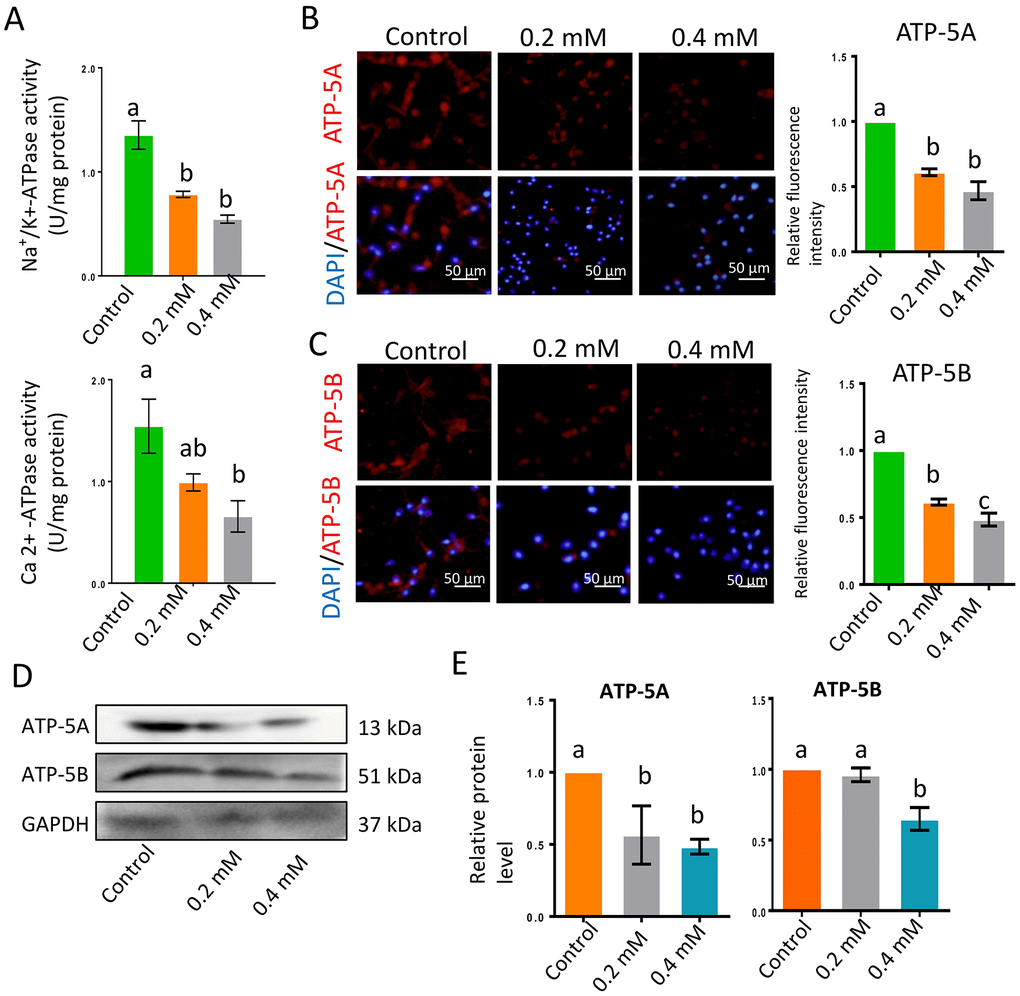
Figure 3. Gibberellin exposure reduced ATPase activity. (A) Na+/K+-ATPase and Ca2+-ATPase activity in sperm were detected with an ATP assay kit. (B–C) The protein levels of ATP-5A and ATP-5B in sperm were detected by immunocytochemistry and quantified based on the relative fluorescence intensity. (D–E) The relative protein levels of ATP-5A and ATP-5B in sperm were detected by Western blotting. The results are presented as the mean ± SEM.
The AMPK signaling pathway is very important for sperm motility and is highly sensitive to the AMP/ATP or ADP/ATP ratio. Inhibited ATP production or increased ATP consumption can reduce the ATP/AMP ratio, thus activating the AMPK pathway by stimulating the phosphorylation of AMPK (p-AMPK). Gibberellin exposure significantly increased the protein levels of AMPK and p-AMPK (Figure 4A–4E; P < 0.01).
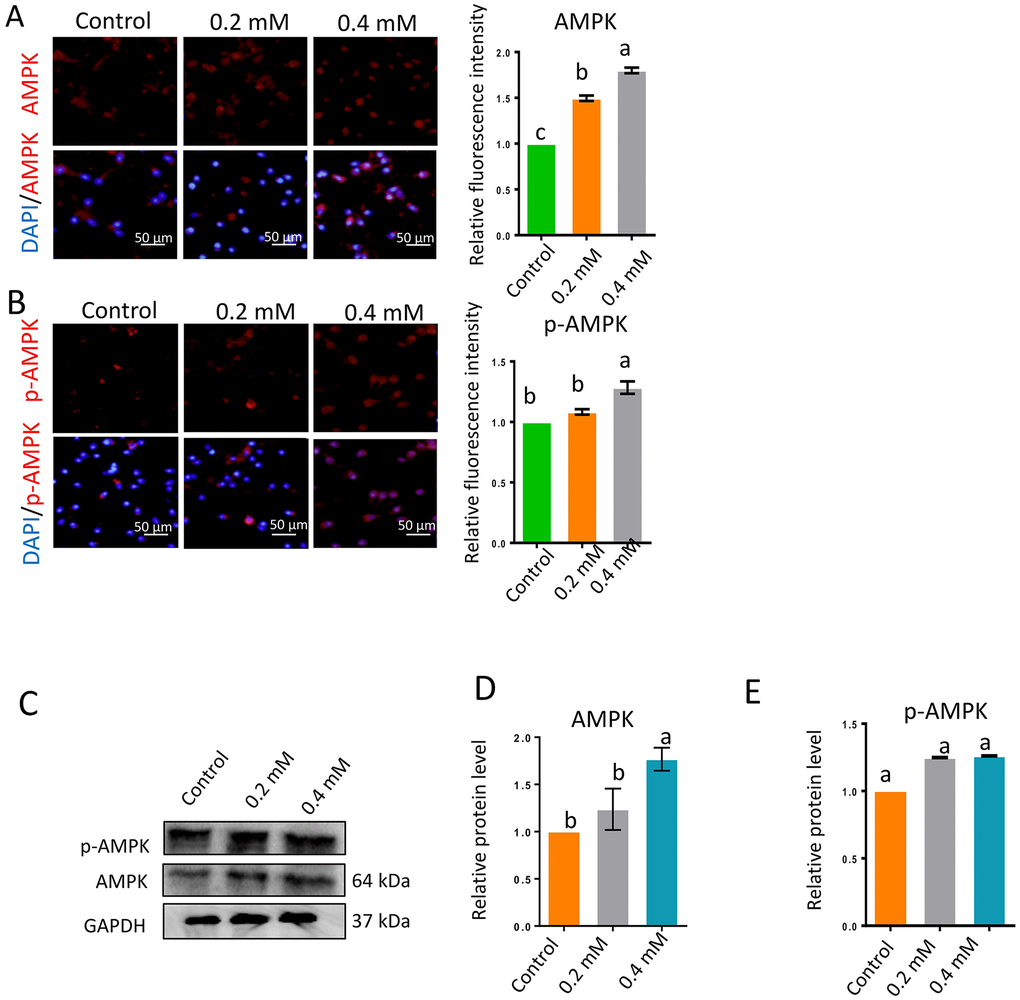
Figure 4. Gibberellin exposure increased the protein levels of p-AMPK and AMPK. (A–B) The relative protein levels of p-AMPK and AMPK in sperm were detected by immunocytochemistry and quantified based on the relative fluorescence intensity. (C–E) The relative protein levels of p-AMPK and AMPK in sperm were detected by Western blotting. The results are presented as the mean ± SEM.
Discussion
Recently, many reports have suggested that semen quality is declining all over the world [21–24]. Wang et al. collected sperm samples from 5210 men who were qualified to be sperm donors in China, and found that the sperm volume, sperm motility, semen concentration and total sperm count had all declined between 2008 and 2014 [25]. The reasons for these declines are not fully understood, although studies have indicated that reduced sperm quality might be associated with human papilloma virus and chlamydia trachomatis infections, environmental pollution, drug abuse, smoking, drinking and staying up late [26–31]. The toxicity of gibberellin to the male reproductive system has been a subject of growing concern, and the results of this study confirmed that gibberellin can reduce human sperm motility in vitro.
Human spermatozoa are very susceptible to oxidative stress [32], and several studies have demonstrated that the lipid peroxide content of human sperm correlates with severe motility loss [33–36]. Appropriate ROS levels are needed for sperm capacitation, the acrosome reaction and other molecular events in human fertility [37]. However, high ROS levels can break the fluidity and integrity of the sperm membrane, leading to sperm DNA damage and mitochondrial dysfunction [38, 39].
In the current investigation, gibberellin exposure significantly inhibited human sperm motility, increased ROS levels and reduced SOD expression in vitro, indicating that gibberellin induced oxidative stress in human sperm. Although many studies have shown that the increase of ROS will lead to the decreased of CAT and GPXI expression [40–42], some studies have proved that high stress also response to try and increase antioxidant to scavenge ROS [43, 44]. SOD and CAT, anti-oxidant enzymes, help to maintain the stability of the sperm membrane by eliminating oxygen free-radicals. Our study demonstrated that gibberellin disrupted the balance of the oxidation/anti-oxidation system. In other studies, gibberellin exposure significantly reduced anti-oxidant enzyme activity in the kidney, ovary, liver, pancreas and brain [45, 46]. Gibberellin was found to accelerate lipid peroxidation up to 65-fold, leading to oxidative damage [47].
The proper concentration of ions in spermatozoa facilitates sperm flagellar movement. Na+/K+-ATPase and Ca2+-ATPase are important for maintaining the membrane gradient and sperm motility [48, 49]. Under normal physiologic conditions, the CatSper, a ligand-gated Ca2+ channel, can be activated by lipophilic compounds. This induces Ca2+ inflow and helps to maintain normal sperm motility and function [50]. Lestari et al. demonstrated that Na+/K+-ATPase activity was lower in men with asthenozoospermia than in those with normozoospermia [51]. Furthermore, Na+/K+-ATPase and Ca2+-ATPase activity are thought to predict sperm motility disorder [52]. The accumulation of active oxygen species can damage the structure of the sperm cell membrane and reduce Na+/K+-ATPase and Ca2+-ATPase activity. Likewise, Ca2+ overload can damage and reduce the motility of human sperm.
In this investigation, gibberellin exposure in vitro increased the levels of ROS and the expression of the apoptosis-related proteins P53 and BAX. This indicated that gibberellin induced apoptosis in sperm cells. Sperm cell apoptosis is known to be associated with mitochondrial dysfunction [53]. In sperm, mitochondria are likely the major sources of ROS [54]. Oxidative stress promotes the opening of the mitochondrial permeability transition pore, an inner mitochondrial membrane channel, which results in matrix swelling and outer membrane rupture. Consequently, mitochondrial cytochrome C is released, which causes ATP depletion and apoptosis [55, 56].
AMPK is an essential regulatory kinase determining human sperm motility [57, 58], and its signaling pathway is crucial for the maintenance of human sperm cellular energy homeostasis. Reduced ATP levels and increased AMP/ATP or ADP/ATP ratios activate the AMPK pathway. In the current study, the protein levels of two major ATP synthases, ATP-5A and ATP-5B, were significantly reduced by gibberellin exposure in vitro. Gibberellin may prevent the formation of ATP, thus increasing AMPK protein expression. Further studies are needed to delineate the toxic effects of gibberellin on sperm mitochondria.
In conclusion, our study demonstrated that gibberellin exposure reduced human sperm motility and Na+/K+-ATPase and Ca2+-ATPase activity while increasing human sperm apoptosis in vitro. The AMPK signaling pathway seems to be an important contributor to the toxicity of gibberellin. Investigations are ongoing to determine the effects of gibberellin on other sperm parameters, such as DNA fragmentation and mitochondrial structure and function. Furthermore, it is essential to study gibberellin exposure in vivo to determine the concentration of this molecule in the male and female reproductive tracts.
Materials and Methods
Collection of human sperm samples
This study was carried out at the Center for Reproductive Medicine, Qingdao Women’s and Children’s Hospital, Qingdao, China. 78 ejaculated human sperm samples were collected in sterile containers by masturbation after three to seven days of sexual abstinence from healthy normozoospermic subjects (average age: 32 years old, range: 27-47 years old) living in Qingdao, China. Semen samples were collected in accordance with the World Health Organization guidelines [59]. After complete liquefaction of the semen samples at 37 °C for 30 min, microscopic analysis was performed to determine the following parameters: total sperm motility ≥ 32%, sperm concentration ≥ 1.5×107 cells/mL, sperm viability ≥ 58%, normal sperm forms ≥ 4%, leukocyte concentration < 1×106/mL and lack of sexually transmitted disease (HIV, syphilis and hepatitis B or genital tract infection diseases). In order to minimize individual variation, semen samples obtained from different men were blended for the following test. Each semen donor provided written informed consent for his inclusion in the study. The consent forms and employed methodologies involving human samples were approved by the Ethics Committee of Qingdao Women’s and Children’s Hospital.
Sperm sample processing, and motility evaluation of sperm exposed to gibberellin
Sperm samples were transferred from sterile containers to 15-mL centrifuge tubes, and fractions of highly motile sperm were separated via discontinuous Percoll gradient (45–90%) centrifugation. These fractions were resuspended at an approximate concentration of 10×106 sperm/mL in G-IVF PLUS medium (Vitrolife, Göteborg, Sweden), which is normally used for the preparation and handling of gametes for in vitro fertilization. The resuspended spermatozoa were divided into 2-mL tubes with 1 mL/tube, and three tubes were allocated to each treatment group. The gibberellin (Pribolab Inc., IAC-040-3, Singapore) stock solutions were made by pre-dissolving 100 mg powders in 20 μL 95% ethanol and then diluting them to 0.1mM, 0.2mM, 0.4mM and 0.8mM by deionized water, respectively.
After gibberellin exposure, sperm motility parameters were assessed by computer-aided sperm analysis in accordance with the World Health Organization guidelines (2010). The computer-aided sperm analysis system was operated by an SCA sperm-class analyzer (Microptic SL, Spain). Sperm motility was classified into four types: grade A, linear velocity > 22 μm/s; grade B, linear velocity < 22 μm/s and curvilinear velocity > 5 μm/s; grade C, curvilinear velocity < 5 μm/s; and grade D, immotile. In the clinic, the (A+B)% was used as an index of sperm motility. Sperm samples were stored at 17 °C in a portable incubator (Fuyilian, FYL-19MC-B4, Beijing, China) before motility detection [60]. Prior to the analysis of the gibberellin-exposed samples, the sperm motility of the control group was tested to ensure that the percentage of sperm in grade A + B + C was greater than or equal to 70%. The data were expressed as the mean ± standard error of the mean (SEM).
Immunocytochemistry
After gibberellin exposure, the samples were washed three times with phosphate-buffered saline (PBS) and fixed in 4% paraformaldehyde for at least 1 h. The sperm cells were then spread on microscope slides and air-dried at room temperature. After three washes with PBS (5 min each), the slides were dipped in 2% Triton X-100 (Solarbio, T8200, Beijing, China) in PBS for 30 min at room temperature. After three washes with PBS, the slides were blocked with PBS containing 10% bovine serum albumin (BSA, Solarbio, A8020) for 30 min at room temperature. Then, the samples were incubated with primary antibodies (1:100; Supplementary Table 1) diluted in blocking solution for 2 h at 37 °C. After three washes with 1% BSA, the slides were incubated with Cy3-labeled goat anti-rabbit IgG (Beyotime, A0516, Nantong, China) (1:150) for 1 h in the dark at 37 °C. The negative controls were incubated with the secondary antibody without the primary antibody.
After three washes with PBS, the slides were incubated with 1 μg/mL Hoechst33342 (Beyotime, C1022) for 5 min at 37 °C. Then, the slides were covered with an anti-fading mounting medium (Vector, H-1000, Burlingame, USA). After the staining, the fluorescent images were viewed through an Olympus microscope BX51 (Olympus, Japan) or a Leica Laser Scanning Confocal Microscope (Leica TCS SP5 II, Germany).
Western blotting
Total proteins from sperm samples were isolated in radioimmunoprecipitation assay lysis buffer (Beyotime, P0013C) with a protease inhibitor cocktail (Sangon, Shanghai, China) for 30 min on ice. Then, an ultrasonic technique was used to break up the sperm cells, and the total protein concentration was determined with a bicinchoninic acid kit (Thermo Fisher, 23209, USA). Each protein extract was then mixed with sodium dodecyl sulfate-polyacrylamide at a ratio of 1:4 and boiled for 5 min. Then, the lysates were collected by centrifugation (14,000 rpm for 5 min). The total proteins were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis with a 3% stacking gel and a 10% separating gel for 40 min at 100 V, followed by 1.5 h at 120 V.
The proteins were transferred to polyvinylidene fluoride membranes at 300 mA for 2 h. Then, the membranes were blocked with 5% non-fat milk (Solarbio, D8340) for 4 h at room temperature, and washed three times with Tris-buffered saline containing 0.1% Tween-20 (TBST). The membranes were incubated with primary antibodies (1:1000 dilution) in TBST containing 1% BSA overnight at 4 °C. The next morning, after three times washes with TBST, the membranes were incubated for 1 h at room temperature with horseradish peroxidase-conjugated goat anti-mouse IgG (Beyotime, A0216) or goat anti-rabbit IgG (Beyotime, A0208) diluted 1:2000 in TBST. After the membranes were washed three times with TBST, a BeyoECL Plus kit (Beyotime, P0018) was used for signal detection. Glyceraldehyde 3-phosphate dehydrogenase (GAPDH) was used as a loading control, and the results were analyzed with AlphaView SA software.
Flow cytometry analysis
For flow cytometry, a FACSCaliburTM flow cytometer (BD Bioscience, Mississauga, USA) equipped with a 488-nm laser, a forward-scatter diode detector and a photomultiplier tube SSC detector was used. The data were collected with CellQuest software and further analyzed with ModiFit LT (ModiFit LT for Maclntel). About 20,000 sperm were analyzed in each group.
Annexin V-FITC apoptosis detection kit
Sperm plasma membrane phosphatidylserine externalization (the translocation of phosphatidylserine from the inner to the outer leaflet of the sperm plasma membrane) was detected with an Annexin-V-FITC apoptosis detection kit (TransGen, FA101, Beijing, China) as previously described [22]. After gibberellin exposure, sperm were collected, washed with cold PBS, centrifuged at 1,500 rpm for 3 min and resuspended in 1× Annexin V binding buffer (100 μL). Then, Annexin V (5 μL) and propidium iodide (PI) (5 μL) were added, and the samples were incubated for 15 min in the dark at room temperature. Lastly, 400 μL of binding buffer was added. Samples were analyzed on a FACSCalibur flow cytometer (BD Bioscience) within 30 min. Apoptotic cells were counted and reported as a percentage of the total cell count. The data were expressed as the mean ± SEM.
Reactive oxygen species assay
ROS were detected with 2,7-dichlorodihydrofluorescein-diacetate (DCFH-DA, Jiancheng, E004, Nanjing, China). After gibberellin exposure, the sperm were stained with DCFH-DA (10 μM) in the dark at 37 °C for 30 mihen, fluorescence was assessed with a FACSCalibur flow cytometer with a 530-nm LP filter. The data were presented as the mean fluorescence intensity ± SEM.
Mitochondrial membrane potential assay
The mitochondrial membrane potential (△Ψm) was measured with the specific probe JC-1 (Thermo Fisher, M34152). After 1-h gibberellin treatment, the sperm cells were collected and resuspended in 1 mL of PBS. Then, 10 μL of a 200-μM JC-1 solution was added, and the samples were incubated in the dark at 37 °C for 30 min. The fluorescence was assessed in a FACSCalibur flow cytometer with a 525-nm filter, and the percentage of stained cells was recorded as the population of sperm with a high △Ψm. The data were presented as the average percentage of high-△Ψm sperm ± SEM.
Measurement of Na+/K+-ATPase and Ca2+-ATPase activity
The activity of the Na+/K+-ATPase and the Ca2+-ATPase were determined with a kit from Nanjing Jiancheng Biochemistry Co. in accordance with the manufacturer’s instructions. After 1 h of gibberellin treatment, sperm were collected and lysed in 0.9% NaCl. Then, the enzyme activity in the lysate was determined by the kit, and the optical density was measured with an ultraviolet spectrophotometer (Thermo Fisher, MA, USA) at a wavelength of 660 nm. The protein concentration was determined with a bicinchoninic acid kit (Thermo Fisher, 23209, USA). The data were expressed as the mean ± SEM.
Statistical analysis
All the experiments were repeated at least three times, and the data were expressed as the mean ± SEM. The data were statistically analyzed with SPSS statistical software (IBM Co., NY). First, the data were evaluated for a normal distribution, and then analysis of variance (ANOVA) was performed. If the ANOVA test was significant, group-to-group comparisons were made with the Least Significant Difference test. Differences were considered significant if P < 0.05.
Acknowledgments
We thank the other members of Dr. Shen’s laboratory for their discussion and help.
Conflicts of Interest
The authors have no conflicts of interest to declare.
Funding
This work was supported by the National Key Research and Development Program of China (2016YFD0501207).
References
- 1. Rokaya PR, Baral DR, Gautam DM, Shrestha AK, Paudyal KP. Effect of pre-harvest application of gibberellic acid on fruit quality and shelf life of mandarin (citrus reticulatablanco). Am J Plant Sci. 2016; 7:1033–39. https://doi.org/10.4236/ajps.2016.77098
- 2. Rademacher W. Chemical Regulators of Gibberellin Status and Their Application in Plant Production. Annual Plant Reviews. John Wiley & Sons, Ltd, 2017.
- 3. El-Okazy AM. The effects of combination of gibberellic acid-3 (ga3) and ethephon (2-chloroethyl phosphonic acid) (plant growth regulators) on some physiological parameters in mice. J Egypt Public Health Assoc. 2008; 83:67–86. [PubMed]
- 4. Erin N, Afacan B, Ersoy Y, Ercan F, Balci MK. Gibberellic acid, a plant growth regulator, increases mast cell recruitment and alters Substance P levels. Toxicology. 2008; 254:75–81. https://doi.org/10.1016/j.tox.2008.09.020 [PubMed]
- 5. Celik I, Turker M, Tuluce Y. Abcisic acid and gibberellic acid cause increased lipid peroxidation and fluctuated antioxidant defense systems of various tissues in rats. J Hazard Mater. 2007; 148:623–29. https://doi.org/10.1016/j.jhazmat.2007.03.018 [PubMed]
- 6. Isik I, Celik I. Investigation of neurotoxic and immunotoxic effects of some plant growth regulators at subacute and subchronic applications on rats. Toxicol Ind Health. 2015; 31:1095–105. https://doi.org/10.1177/0748233713487247 [PubMed]
- 7. Hussein WF, Farahat FY, Abass MA, Shehata AS. Hepatotoxic potential of gibberellic acid (ga(3)) in adult male albino rats. Life Sci J. 2011; 8:373–83.
- 8. Troudi A, Amara IB, Samet AM, Fetoui H, Soudani N, Guermazi F, Boudawara T, Zeghal N. Oxidative stress and thyroid impairment after gibberellic acid treatment in pregnant and lactating rats and their offspring. Biofactors. 2011; 37:429–38. https://doi.org/10.1002/biof.178 [PubMed]
- 9. El-Mofty MM, Sakr SA, Rizk AM, Moussa EA. Carcinogenic effect of gibberellin A3 in Swiss albino mice. Nutr Cancer. 1994; 21:183–90. https://doi.org/10.1080/01635589409514316 [PubMed]
- 10. Tan Z, Zhou J, Chen H, Zou Q, Weng S, Luo T, Tang Y. Toxic effects of 2,4-dichlorophenoxyacetic acid on human sperm function in vitro. J Toxicol Sci. 2016; 41:543–49. https://doi.org/10.2131/jts.41.543 [PubMed]
- 11. Yeşilkaya E, Bideci A, Ozer C, Elmas C, Camurdan O, Giray SG, Boyraz M, Vurucu S, Cinaz P. Plant growth regulator (4-chlorophenoxy acetic acid) increases apoptosis in gonads of rats without changing hormonal levels. Horm Res. 2009; 72:225–35. https://doi.org/10.1159/000236084 [PubMed]
- 12. Nada AT, Saleha YM. Genotoxic effect of an organophosphorus pesticide “ethephon” on somatic and germ cells of male mice. Biosci Biotechnol Res Asia. 2016; 5:1–8.
- 13. Hodjat M, Baeeri M, Rezvanfar MA, Rahimifard M, Gholami M, Abdollahi M. On the mechanism of genotoxicity of ethephon on embryonic fibroblast cells. Toxicol Mech Methods. 2017; 27:173–80. https://doi.org/10.1080/15376516.2016.1273425 [PubMed]
- 14. Hosseinchi M, Soltanalinejad F, Najafi G, Roshangar L. Effect of gibberellic acid on the quality of sperm and in vitro fertilization outcome in adult male rats. Vet Res Forum. 2013; 4:259–64. [PubMed]
- 15. Soltanalinejad F, Ghr N, Hosseinchi MR, Hoshyari A, Najafi A, Babaei M. Evaluation of effects of gibberellic acid on testicular tissue of adult rats. Int J Fertil Steril. 2013; 47:1022–30.
- 16. Aitken RJ, Baker MA. Oxidative stress, sperm survival and fertility control. Mol Cell Endocrinol. 2006; 250:66–69. https://doi.org/10.1016/j.mce.2005.12.026 [PubMed]
- 17. Agarwal A, Makker K, Sharma R. Clinical relevance of oxidative stress in male factor infertility: an update. Am J Reprod Immunol. 2008; 59:2–11. https://doi.org/10.1111/j.1600-0897.2007.00559.x [PubMed]
- 18. Gavriliouk D, Aitken RJ. Damage to sperm DNA mediated by reactive oxygen species: its impact on human reproduction and the health trajectory of offspring. Adv Exp Med Biol. 2015; 868:23–47. https://doi.org/10.1007/978-3-319-18881-2_2 [PubMed]
- 19. Lestari SW, Miati DN, Seoharso P, Sugiyanto R, Pujianto DA. Sperm Na+, K+-ATPase α4 and plasma membrane Ca2+-ATPase (PMCA) 4 regulation in asthenozoospermia. Syst Biol Reprod Med. 2017; 63:294–302. https://doi.org/10.1080/19396368.2017.1348565 [PubMed]
- 20. Zhang TY, Wu RY, Zhao Y, Xu CS, Zhang WD, Ge W, Liu J, Sun ZY, Zou SH, Shen W. Ochratoxin A exposure decreased sperm motility via the AMPK and PTEN signaling pathways. Toxicol Appl Pharmacol. 2018; 340:49–57. https://doi.org/10.1016/j.taap.2017.12.011 [PubMed]
- 21. Huang C, Li B, Xu K, Liu D, Hu J, Yang Y, Nie H, Fan L, Zhu W. Decline in semen quality among 30,636 young Chinese men from 2001 to 2015. Fertil Steril. 2017; 107:83–88.e2. https://doi.org/10.1016/j.fertnstert.2016.09.035 [PubMed]
- 22. Centola GM, Blanchard A, Demick J, Li S, Eisenberg ML. Decline in sperm count and motility in young adult men from 2003 to 2013: observations from a U.S. sperm bank. Andrology. 2016; 4:270–76. https://doi.org/10.1111/andr.12149 [PubMed]
- 23. Tortolero MI, Arata BG, Osuna JA. Semen quality of venezuelan men. Adv Reprod. 2016; 5:1–2.
- 24. Romero-Otero J, Medina-Polo J, García-Gómez B, Lora-Pablos D, Duarte-Ojeda JM, García-González L, García-Cruz E, Rodríguez-Antolín A. Semen quality assessment in fertile men in madrid during the last 3 decades. Urology. 2015; 85:1333–38. https://doi.org/10.1016/j.urology.2015.02.001 [PubMed]
- 25. Wang L, Zhang L, Song XH, Zhang HB, Xu CY, Chen ZJ. Decline of semen quality among Chinese sperm bank donors within 7 years (2008-2014). Asian J Androl. 2017; 19:521–25. https://doi.org/10.4103/1008-682X.179533 [PubMed]
- 26. Pang J, Zhou Q, Sun X, Li L, Zhou B, Zeng F, Zhao Y, Shen W, Sun Z. Effect of low-dose zearalenone exposure on reproductive capacity of male mice. Toxicol Appl Pharmacol. 2017; 333:60–67. https://doi.org/10.1016/j.taap.2017.08.011 [PubMed]
- 27. Asare-Anane H, Bannison SB, Ofori EK, Ateko RO, Bawah AT, Amanquah SD, Oppong SY, Gandau BB, Ziem JB. Tobacco smoking is associated with decreased semen quality. Reprod Health. 2016; 13:90. https://doi.org/10.1186/s12978-016-0207-z [PubMed]
- 28. Chiu YH, Gaskins AJ, Williams PL, Mendiola J, Jørgensen N, Levine H, Hauser R, Swan SH, Chavarro JE. Intake of fruits and vegetables with low-to-moderate pesticide residues is positively associated with semen-quality parameters among young healthy men. J Nutr. 2016; 146:1084–92. https://doi.org/10.3945/jn.115.226563 [PubMed]
- 29. Colín-Ferreyra MD, Domínguez-García MV, Flores-Merino MV. Decrease in Sperm Quality due to Infection of Human Papilloma Virus and Chlamydia trachomatis. Genita Infect Infertil. 2016; 6:69–81. https://doi.org/10.5772/63717
- 30. Fathi Najafi T, Latifnejad Roudsari R, Namvar F, Ghavami Ghanbarabadi V, Hadizadeh Talasaz Z, Esmaeli M. Air pollution and quality of sperm: a meta-analysis. Iran Red Crescent Med J. 2015; 17:e26930. https://doi.org/10.5812/ircmj.17(4)2015.26930 [PubMed]
- 31. Gaskins AJ, Mendiola J, Afeiche M, Jørgensen N, Swan SH, Chavarro JE. Physical activity and television watching in relation to semen quality in young men. Br J Sports Med. 2015; 49:265–70. https://doi.org/10.1136/bjsports-2012-091644 [PubMed]
- 32. Awda BJ, Mackenzie-Bell M, Buhr MM. Reactive oxygen species and boar sperm function. Biol Reprod. 2009; 81:553–61. https://doi.org/10.1095/biolreprod.109.076471 [PubMed]
- 33. Jahanian E, Nanaei HA, Kor NM. Influence of oxidative stress on sperm quality in animal. Int J Biosci. 2014; 4:10–16.
- 34. Morielli T, O’Flaherty C. Oxidative stress promotes protein tyrosine nitration and s-glutathionylation impairing motility and capacitation in human spermatozoa. Free Radic Biol Med. 2012; 53:S137–137. https://doi.org/10.1016/j.freeradbiomed.2012.10.374
- 35. Kao SH, Chao HT, Chen HW, Hwang TI, Liao TL, Wei YH. Increase of oxidative stress in human sperm with lower motility. Fertil Steril. 2008; 89:1183–90. https://doi.org/10.1016/j.fertnstert.2007.05.029 [PubMed]
- 36. Aitken RJ, Clarkson JS. Cellular basis of defective sperm function and its association with the genesis of reactive oxygen species by human spermatozoa. J Reprod Fertil. 1987; 81:459–69. https://doi.org/10.1530/jrf.0.0810459 [PubMed]
- 37. Agarwal A, Tvrda E, Sharma R. Relationship amongst teratozoospermia, seminal oxidative stress and male infertility. Reprod Biol Endocrinol. 2014; 12:45. https://doi.org/10.1186/1477-7827-12-45 [PubMed]
- 38. Li GQ, He Y, Urology DO. [Sperm mitochondrial DNA and male infertility: an update]. Zhonghua Nan Ke Xue. 2017; 23:848–51. [PubMed]
- 39. Kim S, Agca C, Agca Y. Effects of various physical stress factors on mitochondrial function and reactive oxygen species in rat spermatozoa. Reprod Fertil Dev. 2013; 25:1051–64. https://doi.org/10.1071/RD12212 [PubMed]
- 40. Mookerjee A, Basu JM, Majumder S, Chatterjee S, Panda GS, Dutta P, Pal S, Mukherjee P, Efferth T, Roy S, Choudhuri SK. A novel copper complex induces ROS generation in doxorubicin resistant Ehrlich ascitis carcinoma cells and increases activity of antioxidant enzymes in vital organs in vivo. BMC Cancer. 2006; 6:267. https://doi.org/10.1186/1471-2407-6-267 [PubMed]
- 41. Kalmodia S, Vandhana S, Tejaswini Rama BR, Jayashree B, Sreenivasan Seethalakshmi T, Umashankar V, Yang W, Barrow CJ, Krishnakumar S, Elchuri SV. Bio-conjugation of antioxidant peptide on surface-modified gold nanoparticles: a novel approach to enhance the radical scavenging property in cancer cell. Cancer Nanotechnol. 2016; 7:1. https://doi.org/10.1186/s12645-016-0013-x [PubMed]
- 42. McFadden JW, Aja S, Li Q, Bandaru VV, Kim EK, Haughey NJ, Kuhajda FP, Ronnett GV. Increasing fatty acid oxidation remodels the hypothalamic neurometabolome to mitigate stress and inflammation. PLoS One. 2014; 9:e115642. https://doi.org/10.1371/journal.pone.0115642 [PubMed]
- 43. Zhang M, Liu L, Cheng L, Zhang P. [Express of plasma ROS, SOD and GSH-PX in patients with nasopharyngeal carcinoma]. Lin Chuang Er Bi Yan Hou Ke Za Zhi. 2003; 17:650–51. [PubMed]
- 44. Ma Y, Wang D, Xu X, Yang X, Wang X, Zhu Z, Zhao Y, Chen M, Xu F, Fu L, Zhu J, Gao F, An Y. [Dynamic changes of ROS, MDA and SOD during arsenic-induced neoplastic transformation in human keratinocytes]. Wei Sheng Yan Jiu. 2015; 44:456–461. [PubMed]
- 45. Troudi A, Ben Amara I, Soudani N, Samet AM, Zeghal N. Oxidative stress induced by gibberellic acid on kidney tissue of female rats and their progeny: biochemical and histopathological studies. J Physiol Biochem. 2011; 67:307–16. https://doi.org/10.1007/s13105-011-0076-4 [PubMed]
- 46. Tuluce Y, Celik I. Influence of subacute and subchronic treatment of abcisic acid and gibberellic acid on serum marker enzymes and erythrocyte and tissue antioxidant defense systems and lipid peroxidation in rats. Pestic Biochem Physiol. 2006; 86:85–92. https://doi.org/10.1016/j.pestbp.2006.01.009
- 47. Orrenius S, Zhivotovsky B, Nicotera P. Regulation of cell death: the calcium-apoptosis link. Nat Rev Mol Cell Biol. 2003; 4:552–65. https://doi.org/10.1038/nrm1150 [PubMed]
- 48. Hassan HA, Isa AM, El-Kholy WM, Nour SE. Testicular disorders induced by plant growth regulators: cellular protection with proanthocyanidins grape seeds extract. Cytotechnology. 2013; 65:851–62. https://doi.org/10.1007/s10616-012-9525-3 [PubMed]
- 49. Yamazaki D, Funato Y, Miyata H, Ikawa M, Miki H. Complementary role of CNNM2 in sperm motility and Ca(2+) influx during capacitation. Biochem Biophys Res Commun. 2016; 474:441–46. https://doi.org/10.1016/j.bbrc.2016.05.001 [PubMed]
- 50. Strünker T, Goodwin N, Brenker C, Kashikar ND, Weyand I, Seifert R, Kaupp UB. The CatSper channel mediates progesterone-induced Ca2+ influx in human sperm. Nature. 2011; 471:382–86. https://doi.org/10.1038/nature09769 [PubMed]
- 51. Lestari SW, Miati DN, Seoharso P, Sugiyanto R, Pujianto DA. Sperm Na+, K+-ATPase α4 and plasma membrane Ca2+-ATPase (PMCA) 4 regulation in asthenozoospermia. Syst Biol Reprod Med. 2017; 63:294–302. https://doi.org/10.1080/19396368.2017.1348565 [PubMed]
- 52. Lestari SW, Larasati MD, Mansur IG, Margiana R. Sperm Na+ K+-ATPase and Ca2+-ATPase Activities: A Potential Predictive Parameter of Sperm Motility Disorder in Infertile Men. Biomed Pharmacol J. 2018; 11. https://doi.org/10.13005/bpj/1388
- 53. Barroso G, Taylor S, Morshedi M, Manzur F, Gaviño F, Oehninger S. Mitochondrial membrane potential integrity and plasma membrane translocation of phosphatidylserine as early apoptotic markers: a comparison of two different sperm subpopulations. Fertil Steril. 2006; 85:149–54. https://doi.org/10.1016/j.fertnstert.2005.06.046 [PubMed]
- 54. Cassina A, Silveira P, Cantu L, Montes JM, Radi R, Sapiro R. Defective human sperm cells areassociated with mitochondrial dysfunction and oxidant production1. Biol Reprod. 2015; 93:119–28. https://doi.org/10.1095/biolreprod.115.130989 [PubMed]
- 55. Zhao Y, Ye L, Liu H, Xia Q, Zhang Y, Yang X, Wang K. Vanadium compounds induced mitochondria permeability transition pore (PTP) opening related to oxidative stress. J Inorg Biochem. 2010; 104:371–78. https://doi.org/10.1016/j.jinorgbio.2009.11.007 [PubMed]
- 56. Sahach VF, Kakhanovs’kyŭ EF, Horbovets’ VS. [The mitochondrial permeability transition pore opening under oxidative stress in ischemia/reperfusion in the tissue of the lower extremities]. Fiziol Zh. 2008; 54:47–51. [PubMed]
- 57. Calle-Guisado V, Hurtado de Llera A, González-Fernández L, Bragado MJ, Garcia-Marin LJ. Human sperm motility is downregulated by the AMPK activator A769662. Andrology. 2017; 5:1131–40. https://doi.org/10.1111/andr.12423 [PubMed]
- 58. Nguyen TM, Froment P, Combarnous Y, Blesbois É. AMPK, regulator of sperm energy and functions. Med Sci (Paris). 2016; 32:491–96. https://doi.org/10.1051/medsci/20163205016 [PubMed]
- 59. World Health Organization. WHO Laboratory Manual for the Examination and Processing of Human Semen. ( Ed.) 2010.
- 60. Zhao Y, Zhang WD, Liu XQ, Zhang PF, Hao YN, Li L, Chen L, Shen W, et al. Hydrogen Sulfide and/or Ammonia Reduces Spermatozoa Motilitythrough AMPK/AKT Related Pathways. Sci Rep. 2016; 6:378–84. https://doi.org/10.1038/srep37884 [PubMed]



