Introduction
Plasma cell dyscrasias are a rare heterogeneous group of disorders characterized by the expansion of monoclonal bone marrow plasma cells. Incidence increases considerably with age and the complex question of “how to best manage elderly patients with plasma cell dyscrasias” becomes even more relevant in the era of the aging population [1]. The introduction of novel agent combinations in recent years has improved overall survival (OS) and progression free survival (PFS) in patients with plasma cell dyscrasias. The older population has been however underrepresented in clinical trials and robust data to guide treatment decisions in the elderly diagnosed with plasma cell dyscrasias is lacking. In particular, the percentage of patients >75 years of age is particularly low as the majority will fail to meet inclusion criteria for clinical trial enrolment. In addition, disease-specific frailty assessment tools need to be developed and integrated in clinical practice to allow optimum management approaches that are not solely based on chronological age [2]. Frailty is a state characterized by decreased organ reserves due to “disease, lack of activity, inadequate nutritional intake, stress, and/or the physiologic changes of aging”. Chronological age, comorbidities and the patient’s performance status alone have limited quality in capturing the heterogeneity of the older patient group and measures of function are required [3,4]. Geriatric assessment (GA) has become increasingly important in oncology, their use increases treatment tolerance and completion rates and should therefore be included in the complex treatment decision making process [5]. GA tools assess the patients’ functional and global health status allowing fine tuning of management plans and avoiding over or under-treatment [6,7]. This review aims to discuss issues that are specific to the management of the heterogeneous group of older patients diagnosed with plasma cell dyscrasias. The discussion will focus on the three most common plasma cell dyscrasias, Multiple Myeloma (MM), Waldenstrom Macroglobulinemia (WM) and systemic AL Amyloidosis. What will become increasingly evident is the need for frailty assessment tools, the complexities of defining frailty within the context of each disease, the need of having clear treatment goals that can guide clinical practice (balance between treatment effectiveness and toxicity) and the lack of evidence from clinical trials specific to this patient population.
Multiple Myeloma
Median age at diagnosis of patients with MM is 70 years and about 34-40% of patients with MM are older than 75 years [8,9]. Outcomes have considerably improved over the last years for patients with newly diagnosed MM (NDMM) due to better understanding of disease biology, improvements in supportive care but mostly due to the addition of novel agents like proteasome inhibitors (PI), immunomodulatory drugs (IMiDs) and monoclonal antibodies (MoAbs) in the MM armamentarium [10,11]. The improvement in PFS and OS have been reflected in the older cohort of patients as well [12] but the added benefit has been less pronounced [11–14]. Survival is poorer for patients with NDMM over the age of 70 and the risk of early death is twice as high [15–18]. In a recent analysis of 827 consecutive NDMM patients, median survival in the 110 patients who were ≥80 years old was 22 months and early mortality within 2 months post diagnosis was 20% [18]. The use of less effective drug combinations, comorbidities, less favourable disease biology, increased toxicity, lower physiological reserves and early treatment discontinuation all possibly contribute to the worse outcomes associated with increased age [19].Patients >75 years’ old who receive treatment with novel agents often have similar PFS but lower OS than younger patients which can be partly explained by the effect of first line toxicities in the choice of second line treatment [20].
The challenge is to develop sensitive tools that are clinically validated to assess frailty in the heterogeneous elderly population and to move away from making clinical decisions based on chronological age and performance status [21]. Increasingly treatment decisions based on physiological age and geriatric assessments are being incorporated in clinical practice. Frail patients however continue to be underrepresented in clinical trials [22]. Frailty tools need to be used to set treatment goals and clinical trials tailored to the frailty status of the patient [23,24].
Initial evaluation of the MM patient
Disease evaluation in MM dose not change with age and diagnosis of symptomatic disease is based on the International Myeloma Working Group (IMWG) 2014 criteria [25]. Determining whether the cause of end-organ damage is secondary to the effects of the malignant plasma cell clone rather than inter-current illness, physiological decline of organ function or comorbidities may however confound the evaluation and can be a challenge [26]. The utility of the International Stating System (ISS) in risk stratifying older patients with MM has also been questioned by some authors as beta-2-microglobulin can be increased with impaired renal function and serum albumin which can be lower secondary to malnutrition [27,28]. The Revised-International Staging System however incorporates also high risk cytogenetics, was developed using 4000 patients, one-third was over 65 years old and its prognostic value remained independent of age [29].
Geriatric assessment tools (GCAs) in patients with Multiple Myeloma (Table 1)
Geriatric assessment (GA) is time and manpower consuming and a challenge to incorporate it in everyday practice. A more targeted and disease specific approach that uses a limited number of indicators to assess frailty is required. In the field of MM a number of frailty scoring systems have been developed [21].
IMWG- frailty index
The IMWG developed in 2015 an additive scoring system (0-5) that assesses age, comorbidities and functional conditions assessed by the Charlson Comorbidity Index (CCI), Katz Activities of Daily Living (ADL) and Lawton’s Instrumental Activities of Daily Living (IADL). It categorizes patients with MM at diagnosis as fit, intermediate and frail [30]. The score is available online (http://www.myeloma frailtyscorecalculator.net/). It should be noted that age >80 highly drives the score and that it has not been validated in “real world” myeloma patients [18].
R-MCI frailty score
A German cohort of 801 consecutive NDMM patients was used to develop the revised myeloma comorbidity index (R-MCI). Multivariate analysis determined impaired lung and renal function, the Karnofsky Performance status (KPS), frailty and age as highly significant for OS and these were combined to form three categories; fit (R-MCI 1-3), intermediate-fit (R-MCI 4-6) and frail (R-MCI 7-9). A web-based application is also available. (www.myeloma comorbidityindex.org)
Another group developed the Mayo frailty staging which uses the NTproBNP as an additive biomarker of frailty as it predicts survival independent of age and performance status [21,31]. Imaging techniques are also becoming of value in determining patient frailty. Recent data from a small study demonstrated a relationship between low subcutaneous adipose tissue index and poor overall survival [32]. Ongoing clinical trials are designed to tailor treatment to the frailty status of the patient.
How we treat elderly patients with MM
Newly diagnosed patients
Multiple trials have demonstrated the superior PFS and OS with Melphalan-Prednisone-Thalidomide (MPT) or bortezomib-melphalan-prednisone (VMP) over Melphalan and Prednisone (MP) alone. Survival benefit has been demonstrated across subgroups including patients ≥ 75 years old despite an increased incidence of grade 3 toxicities and treatment discontinuation, particularly in older patients [33–36]. Continuous Revlimid-dexamethasone (Rd) was shown to be superior to Rd for 18 cycles and MPT for 12 cycles in the FIRST trial. Carfilzomib (K), a second generation PI, was compared to bortezomib in the CLARION study (K-MP vs VMP) but PFS was comparable and AEs ≥ grade 3 (hypertension, acute renal and cardiac failure) and the number of deaths were higher in the KMP arm. Given its toxicity profile and the twice weekly treatment regimen carfilzomib can be a challenging treatment option for elderly patients [37]. The phase 3 SWOG-S0777 study demonstrated a significantly improved PFS and median OS for patients who receive bortezomib/revlimid/dexamethasone (VRd) over Rd, the benefit remained for the ≥ 65 years old cohort and toxicity profile of VRd was worse but was considered acceptable [38]. It should be noted that patients enrolled were not necessarily transplant ineligible and age was not a stratification factor. The monoclonal antibody (anti-CD38) daratumumab was assessed in non-transplant eligible > 65 years patients in the ALCYONE trial in combination with VMP (dara-VMP vs VMP) followed by daratumumab maintenance. PFS was significantly improved and the advantage was also evident for the ≥ 75 years’ old cohort [39]. The goal of maintenance post induction is to retain and further deepen the response achieved. Prolonged treatment is however associated with toxicity and can adversely affect quality of life particularly in the elderly population. So far clinical trials have shown that maintenance improves PFS but not OS in transplant-ineligible patients although this is even less clear for patients over 75 years of age [40–42].
The results of ongoing trials that aim to address treatment options specifically in frail patients and make use of GA tools are much awaited but more trials need to be designed. The ongoing TOURMALINE-MM4 trial (NCT02312258) compares oral Ixazomib versus placebo as a 2 year maintenance option in transplant ineligible patients. Another on-going phase III trial aims to address the role of ASCT in older patients and compares Rd plus/minus intensification by high-dose melphalan in patients aged 60-75 years who are fit. (NCT01090089) A dose reduced Rd schedule versus the standard Rd schedule in NDMM is being compared in a phase III trial in patients >65 years who are considered unfit and unsuitable based on the investigator’s opinion to receive approved first line treatments. Risk stratification includes the use of GA tools (NCT02215980).
Recommendations for treatment of the elderly at diagnosis
Randomized phase 3 trials have established that the addition of novel agents to the MP backbone improves outcomes for older MM patients but other independent trials have failed to demonstrated this advantage particularly in frail patients [43]. An optimal balance between treatment efficacy and toxicity must be achieved as higher rates of AEs might translate into higher discontinuation rates and inferior survival benefits [44,45]. IMWG-frailty index and the R-MCI are both recommended tools for the identification of fit, intermediate and frail patient [21]. Based on this categorization one can adapt treatment goals and select less intensive or dose-reduced treatment schedules as appropriate. According to the recent EMN guidelines for patients categorized as fit, treatment efficacy and deep remission (defined as complete response (CR) or minimal residual disease (MRD) negativity) should be the priority. They should receive full-dose therapy including VMP, Rd or VRD. In intermediate-fitness patients one should aim to achieve a balance between safety and efficacy by using doublets and or even low dose triplets. Finally, in frail patients doing no harm and preserving quality of life should be prioritized and doublet combinations at lower doses might be required. (Table 1) In terms of maintenance, trials have demonstrated a benefit in PFS but not an OS advantage in transplant-ineligible patients although time to next treatment is prolonged.
Table 1. Frailty status definition and treatment goals, treatment options and dose adjustments based on frailty status in NDMM elderly patients. Adapted from Larocca et al 2018.
| FIT | INTERMEDIATE | FRAIL | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| IMWG-frailty index score | 0 | 1 | 2-5 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CCI 2 :1 IADL <5: 1 ADL <4: 1 Age 76-80: 1, >80:2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Revised myeloma comorbidity index (R-MCI) | 0-3 | 4-6 | 7-9 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age 60-69 KPS: 80-90%: 2, <70%: 3 Renal disease: eGFR <60:1 Lung disease: moderate/severe:1 Frailty: moderate or severe:1 ± cytogenetic unfavourable: 1 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MAYO FRAILTY INDEX | 0 Age 70: 1 ECOG PS 2 :1 NT-proBNP 300 mg/L | 1 (Stage I) 2 (Stage II) | 3 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Goal of Treatment | Efficacy: deep response | Balance efficacy and toxicity | Conservative approach, low toxicity | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Treatment Options | Full dose therapy ASCT Triplet regimens: VMP, VRD doublet regimens: Rd | Full or reduced dose therapy Doublet regimens Rd Vd Reduced-dose triplet | Reduced dose therapy Reduced dose doublet regimens: Rd, Vd Palliative + supportive care | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dosing Regimens – dose Levels | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| 0 | -1 | -2 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Dexamethasone | 40 mg d1,8,15,22 in 28 day cycle | 20 mg d1,8,15,22 q28 day | 10 mg d1,8,15,22 q28 day cycle | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Melphalan | 0.25mg/kg on days 1-4 on a 4-6 week schedule | 0.18mg/kg on days 1-4 on a 4-6 week schedule | 0.13mg/kg on days 1-4 on a 4-6 week schedule | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Prednisone | 2mg/kg days 1-4 q 4-6 weeks | 1mg/kg days 1-4 q4-6 weeks | 0.5mg/kg days 1-4 q 4-6 weeks | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thalidomide | 100 -200 mg/day | 50 -100 mg/day | 50 mg qother day –qday | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Lenalidomide | 25mg d1-21 q28 | 15mg d1-21 q28d | 10mg d1-21 d28d | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Pomalidomide | 4mg d1-21 q28 day | 3mg d1-21 q28 days | 2mg d1-21 q28 days | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Bortezomib | 1.3mg/m2 d 1,4,8,11 q 3 weeks | 1.3mg/m2 d 1,8,15, 22 q 5 weeks | 1.0mg/m2 d 1,8,15,22 q 5 weeks | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Carfilzomib | 20mg/m2 d1,2,8,9,15,16 in cycle 1 then 27mg/m2 cycle 2 every 4 weeks | 20mg/m2 d1,2 then 27mg/m2 d 1,8,15 q 4 weeks | 20mg/m2 d1,8,15 every 4 or 5 weeks | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Ixazomib | 4mg d 1,8,15 q28d | 3mg d 1,8,15 | 2.3mg d 1,8,15 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Daratumumab | 16mg/kg weeks 1-8, weeks 9-24 d1+15 and week 25 onwards q 4 weeks | 16mg/kg weeks 1-8, weeks 9-24 d1+15 and week 25 onwards q 4 weeks | 16mg/kg weeks 1-8, weeks 9-24 d1+15 and week 25 onwards q4weeks | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Elotuzumab | 10mg/kg d1,8,15,11 cycles 1+2, and cycle 3 d1+15 | 10mg/kg d1,8,15,11 cycles 1+2, and cycle 3 d1+15 | 10mg/kg d1,8,15,11 cycles 1+2, and cycle 3 d1+15 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| NDMM: newly diagnosed multiple myeloma, ADL: Activity of Daily Living, IADL: Instrumental Activity of Daily Living, | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CCI: Charlson Comorbidity Index, KPS: Karnofsky Performance Status, ECOG PS: ECOG Performance Status, ASCT: | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Autologous Stem Cell Transplantation, VMP: bortezomib-melphalan-prednisone, VRD: bortezomib lenalidomidedexamethasone, | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rd lenalidomide-dexamethasone, Vd: bortezomib-dexamethasone | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Treating elderly MM patients in the relapse and/or refractory setting
Treatment at relapse can often be more challenging due to prior line toxicities, comorbidities, advancing age and aggressive patterns of relapse [46]. The percentage of patients >75 years are enrolled in clinical trials at relapse is even lower given that most of them will fail to meet inclusion criteria at this point. Evidence supports a prolonged OS in elderly relapsed and/or refractory MM (RRMM) patients who achieve a CR so the treatment goal should be achieving a deep response in fit patients [47]. On the other hand preserving QoL and minimizing treatment toxicity should be the main goal in frail patients [30]. Unfortunately data from clinical trials and on GA assessments in the RRMM patients is even more scarce [26]. The benefit in PFS seen in the carfilzomib-dexamethasone (Kd) versus Vd arm in the ENDEAVOR study and in the carfilzomib-lenalidomide-dexamethasone (KRd) versus Rd arm in the ASPIRE trial was maintained for patients ≥75 years [48–50]. In the POLLUX trial RRMM patients were randomized to receive Rd versus Daratumumab-Rd and the PFS benefit was even more pronounced ≥ 75-year-old patients, with higher rate of AEs but comparable discontinuation rates [51]. The PFS advantage also persisted for the older subgroup in the CASTOR trial which compared Dara-Vd vs Vd [52]. Finally the impressive overall response rate of 60% seen in RRMM patients who received pomalidomide-daratumumab in the phase 1 EQUULEUS study was the same across all age groups [53]. Pomalidomide (Pd) has also significant activity in heavily pre-treated RRMM patients and advantages seem to be similar for patients older and younger than 65 years but limited data is available for patients aged ≥ 75 years. In the phase 3 ELOQUENT-2 trial which led to FDA approval of Elotuzumab in combination with Rd for the RRMM, 20% of patients were ≥ 75 years old [54].
Recommendations for treatment of elderly patients with RRMM
Due to the lack of clinical trials designed specifically for the elderly in the RRMM setting recommendations are mostly expert-opinion-based [21]. Patients who are non-PI refractory following lenalidomide can receive Kd or DaraVd. KRd is an option for patients’ sensitive to lenalidomide. Careful cardiovascular assessment prior to carfilzomib treatment initiation and close monitoring is required. Dara-Rd or Elo-Rd are recommended for patients who are bortezomib but not lenalidomide refractory. Fit patients should receive full-dose combinations. Elo-Rd or IRd are appropriate options for Intermediate-fit patients. KRd can be considered in intermediate-fit patients with no cardiac comorbidities. Dara-Rd or Dara-Vd triplets can improve effectiveness without increasing toxicity compared to their respective doublets even in frail patients. For IMiD and PI refractory fit patients Pd, Pd+cyclosphosphamide, single–agent daratumumab and inclusion in clinical trials are possible options. In double refractory frail patients low dose oral combinations of cyclophosphamide or melphalan +/- thalidomide can be tried.
The role of autologous stem cell transplantation (ASCT) in the elderly population
Single-centre retrospective transplant registry analyses have demonstrated that ASCT is feasibly in the elderly fit MM patients [55,56]. Candidates should however meet strict selection criteria as the risk of toxicities may counteract the potential benefits [30,57–59]. The results of DSMM XIII study which assesses continuous Rd treatment vs Rd induction, tandem melphalan 140mg/m2-ASCT consolidation and R-maintenance in 60-75 year-old patients are eagerly awaited [60].
Considerations in managing treatment toxicity and supportive care
Frailty determines to a considerable extent patient susceptibility to treatment related side-effects. Dose reductions or interruptions can jeopardize response rates, deter quality of life and affect OS. Prompt and effective toxicity management to achieve an optimal balance between toxicity and effectiveness is key to treatment success. Dosing alternations depend on the type of the AE and its grading [61,62] (Table 2).
Table 2. Management of drug related toxicities in the elderly patients with MM and recommended dose modifications.
| Adverse event | Suspected agent | Grading of AE | Management | Dose modification | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Neutropenia | Bortezomib Lenalidomide | Grade 4 or grade 2-3 with infection | G-CSF until recovery | 25-50% reduction | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Anemia | Bortezomib Lenalidomide | Grade 2-4 | Erythropoetin or Darbopoetin for Hb <10g/dl | 25-50% reduction | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Thrombo-cytopenia | Bortezomib Lenalidomide | Grade 3 and 4 | Drug interruptions Platelet transfusion for Grade 4 AE | 25-50% reduction | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Bone disease | None | Intravenous zolendronic acid, or pamindronate. Vertebroplasty if indicated, analgesia as appropriate | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Venous thrombo- embolism (VTE) – prophylaxis | IMiDs | ≤ 1 risk factor for VTE: aspirin 100mg >1 risk factors for VTE: low molecular weight heparin at prophylactic dose | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| VTE management | IMiDs | Therapeutic dose of LMWH or warfarin | Temporary drug interruption and full anticoagulation | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Neuropathy | Thalidomide Bortezomib | Grade 2 PN Grade 3 PN Grade 1 with pain or Gr 2 Grade 2 with pain or Gr3 Grade 4 | Neurological assessment during treatment, immediate dose reductions recommended + Gabapetin, pregabalin, Acetyl-L-carnitin and alpha lipoic acid, opiods, calcium channel blockers, sodium channel blockers, serotonin reuptake inhibitors | 50% dose reduction Treatment discontinuation until Gr 2 25-50% dose reduction Dose interruption until Gr 1 and 50% dose reduction Treatment discontinuation | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Skin toxicity | Lenalidomide thalidomide | Grade 2 Grade 3-4 | Antihistamines and steroids | 50% drug reduction Interruption | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Infection | Bortezomib Lenalidomide Thalidomide | Grade 2-4 | Prophylaxis: Trimethoprim-cotrimoxazole for Pneumocystis carinii prophylacis during high dose dexamethasone. Acyclovir or valacyclovir for HVZ prophylaxis during PI-containing therapy, seasonal influenza vaccination, pneumococcal vaccination , Haemophilus influenza vaccination | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Adapted from Cerrato et al. [146] and Palumbo et al. [62]. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Hematologic toxicities
Bortezomib causes cyclical neutropenia and thrombocytopenia [35,63]. Hematologic toxicities are amongst the most common AEs associated with thalidomide and lenalidomide use [64,65]. Granulocyte-colony stimulating factor therapy (G-CSF) should be used as primary or secondary prophylaxis for neutropenia and erythropoiesis-stimulating agents (ESA) can be used to manage anemia which does not respond to treatment.
Peripheral neuropathy (PN)
Bortezomib and thalidomide-based therapies are associated with considerable rates of PN. In frail patients PN increases risk of falls and impairs function [66]. Toxicity is cumulative and dose-dependent but reversible for bortezomib contrary to the permanent neuropathy often associated with thalidomide [67,68]. Careful monitoring and patient education are imperative. The initial dosing schedule should be guided by the patients’ frailty status and immediate dose reductions are recommended. Options for management exist have not been evaluated prospectively (Acetyl-L-carnitin, alpha lipoic acid, opioids, gabapentin, pregabalin) [61].
Thrombosis
The incidence of venous thromboembolism (VTE) in MM patients ranges approximately from 8 to 22/1000-person years. The risk is disease, patient and treatment-dependent [69]. IMiDs, multi-agent chemotherapy regimens and high dose dexamethasone increase VTE risk substantially [70]. VTE risk assessment and appropriate thromboprophylaxis is imperative. IMWG guidelines recommend low dose aspirin for patients on IMiDs with none or one risk factor and low molecular weight heparin or full dose warfarin when more than one VTE risk factors is present.
Infections
Disease- related immunoparesis and myelosuppression secondary to treatment regimens render MM patients at high risk of infection. The risk is higher at diagnosis and mostly associated with IMiDs [71,72]. Infection prophylaxis has been shown to decrease treatment associated morbidity in MM patients. Prophylactic antiviral medication to reduce PI associated herpes zoster infection [73,74], seasonal influenza, streptococcal pneumonia and Hemophilus influenza vaccination [61] and prophylactic trimethoprim-sulfamethoxazole are all recommended [75]. A recent abstract presentation from a phase 3 clinical trial demonstrated that prophylactic use of levofloxacin reduces febrile episodes and death in NDMM patients [76].
In the case of frail patients, decision-making capacity should also be assessed to determine whether there is need for a surrogate decision-maker [77]. Finally issues related to adherence to oral treatment and the presence of appropriate socioeconomic support also come into play [78,79].
Commentary
The complexities of managing the patient with MM are increasing in the era of the aging population and increasing treatment options. Older patients have been up to date underrepresented in clinical trials and evidence to guide management is therefore lacking. Frailty assessment tools are being developed and should be increasingly incorporated in clinical trials and clinical practice. Management of the older patient with MM should be in all contexts tailored to the patients’ frailty status. The results of current ongoing clinical trials that incorporate tools that assess frailty and are tailored to older MM patients are eagerly awaited.
Waldenstrom Macroglobulinemia
Waldenstrom’s Macroglobulinemia (WM) is a rare lymphoproliferative disorder characterized by the proliferation of lymphoplasmacytic elements and the presence of monoclonal immunoglobulin M (IgM) gammopathy. It is a disease of the elderly (median age at diagnosis 63-75 years) and comes with the age-related comorbidities which complicate patient management [80,82]. It is considered an indolent yet incurable disease with median disease-specific survival of 10-11 years. Despite an increase in therapeutic options there is no precise treatment algorithm due to a paucity of comparative high quality data. Older and medically non-fit patients are underrepresented in the existing clinical trials [83]. There are no frailty scores available specifically for patients with WM and up to date clinical trials have not included any scoring systems to categorize patients as fit or unfit. The ESMO consensus proposes three different categories of fitness in the context of treatment feasibility. For an “elderly fit” patient the treatment related and unrelated AEs would be comparable to those for a young “fit” patient. In contrast the “vulnerable patient” would have higher risk of treatment related and unrelated AEs. Finally, the “terminally ill” patient has a short life expectancy and will only benefit from best supportive care [84]. There is however no tool to categorize patients and therefore frailty assessment is left to clinical judgement. In the International prognostic scoring system for WM age has significant weighting. Patients aged ≥65 years are categorized as at least intermediate risk [85]. Patients with asymptomatic disease should be closely monitored and treatment initiation should be based on the IWWM-8 consensus criteria [86]. Care is required to exclude other possible diagnoses related to comorbidities of the older patient with WM.
Management of elderly patients with WM
Overview of treatment options
Patients with symptomatic hyperviscosity should undergo plasmapheresis followed rapidly by cytoreductive treatment. The mainstay of treatment and backbone to most treatment combinations are anti-CD20 monoclonal antibodies (Rituximab). Rituximab monotherapy has a very safe toxicity profile but is inferior to combinations [87]. To avoid IgM flare, rituximab monotherapy is not recommended for patients with high IgM levels but is indicated for patients with WM-related immunologic disorders [88,89]. The combination of Rituximab, dexamethasone and cyclophosphamide (DRC) achieves better responses, has a favourable short and long term toxicity profile and is an option for patients with comorbidities and low tumour burden [90]. Another well-tolerated effective combination is Bendamustine+Rituximab (BR) but dose adjustments might be necessary in older medically non-fit patients due to associated myelosuppression risk [91]. Bortezomib (V) is also very active in patients with WM (subcutaneously, weekly, at 1.6mg/m2 dose) [92–94]. Bortezomib containing combinations are first choice in patients with hyperviscosity, high IgM levels, renal impairment, cryoglobulinemia or cold agglutinemia [95]. Carfilzomib in the relapse setting is an alternative neurotoxicity-sparing option among PI inhibitors [96,97]. More intensive chemotherapy regimens can induce high response rates but are not favoured for use in first line due to the associated significant toxicity. These include R-CHOP and nucleoside analogues (fludarabine/rituximab (FR) or fludarabine/cyclophosphamide/ rituximab FCR) or chlorambucil [97,98]. Ibrutinib (bruton’s tyrosine kinase inhibitor) is a very effective oral option in WM patients [99,100]. In the USA, FDA has granted approval for both first line and relapse. In Europe the EMA has licensed the drug for use at relapse and as first line for patients who are non-eligible for “immunochemotherapy”. The INNOVATE study compared rituximab/ibrutinib to rituximab monotherapy in pre-treated rituximab sensitive patients and in untreated patients. At 30 months PFS was 82% in the ibrutinib/rituximab arm versus 28% in the rituximab arm [101]. The advantage was also seen in previously untreated patients. Maintenance treatment with rituximab is currently not recommended due to the lack of prospective data [102]. ASCT is also an option for salvage therapy in WM in young patients with refractory disease or multiple relapses [97].
Recommendations for treating elderly patients with WM
The recent ESMO and EMN recommendations base treatment recommendations on tumour burden assessment/clinical presentation and the patient’s fitness status [95,103]. PI-combinations like bortezomib alone, BDR and bortezomib-rituximab are recommended for patients with hyperviscosity or bulky disease in fit patients. Ibrutinib is another option but responses are slower. In patients with cytopenias DRC, PI-based therapy, BR or ibrutinib are all appropriate. If neuropathy bortezomib should be avoided and for AL amyloidosis related to WM, PI-based therapy or BR are recommended [95]. For the unfit patient oral fluradarabine for 6 cycles or chlorambucil for 12 cycles, DRC for 6 cycles and rituximab monotherapy are all acceptable options if disease burden is low. VR or ibrutinib are preferred for higher tumour burdens (Figure 1). For early relapse (<1year after R-based therapy) patients should be included in a clinical trial. Ibrutinib is appropriate for early or late relapses and should be given until disease progression as drug discontinuation leads to frequent relapses. The safety profile is safe and therefore it is appropriate for medically unfit patients [100,104]. Note that ibrutinib is not recommended for patients with MYD88WT disease. For relapses > 2 years post the previous R-based therapy one can repeat or alternate all the available treatment options at first line [90]. Lenalidomide alone or in combination with DRC has been used in clinical trials in heavily pre-treated populations and the combination achieved a 80% response rate and a median PFS of 24 months [105] (Figure 2).
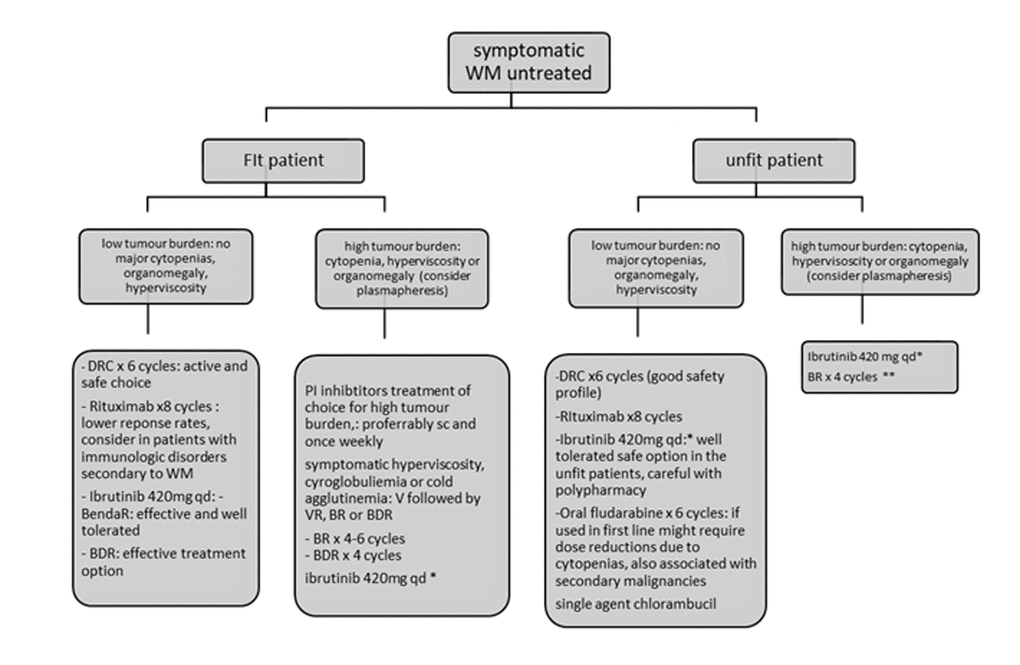
Figure 1. Recommendations for the treatment of newly diagnosed patients with Waldenstrom’s Macroglobulinemia. Figure adjusted from ESMO guidelines for WM 2018 and EMN recommendations for treatment of rare plasma cell dyscrasias. *Approved in USA by FDA for first line and only for patients unfit for immunochemotherapy in Europe by EMA. ** BR for unfit patients may require dose reductions for bendamustine and use of G-CSF and antibiotic prophylaxis. BDR: bortezomib, dexamethasone, rituximab, BR: bendamustine, rituximab; DRC: dexamethasone, rituximab, cyclophosphamide; AF: atrial fibrillation, V: bortezomib.
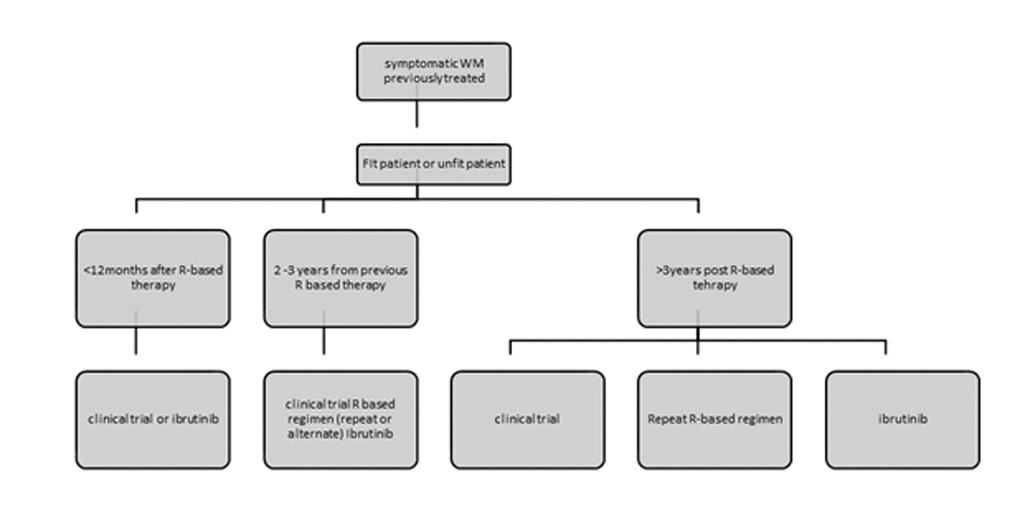
Figure 2. Recommendations for the treatment of previously treated patients with Waldenstrom’s Macroglobulinemia. Figure adjusted from ESMO guidelines for WM 2018 and EMN recommendations for treatment of rare plasma cell dyscrasias. R: rituximab.
Commentary
The indolent nature of the disease, the reasonably safe toxicity profile of the therapeutic regimens together with appropriate reasoning on the choice of the regimen and dose adjustments can allow for effectiveness and acceptable quality of life for both fit and unfit elderly patients with WM. Median survival for younger patients exceeds 10 years. It is shorter for elderly patients but a significant proportion will die due to reasons unrelated to disease [82]. Treatment induced myelodysplasia or WM transformation to more aggressive lymphoma are commonly seen in the older patient cohorts. The disease does however in the elderly commonly transform or can develop to myelodysplasia [106–108]. Secondary non-hematological malignancies develop in about 18% of patients secondary to treatment toxicity [109]. It is realistic to pursue the development of more effective and at the same time minimally toxic treatment strategies [83]. Current recommendations distinguish between fit and non-fit patients but no specific tool is available to guide stratification. The success of such a pursue requires the development of disease specific frailty assessment tools and more clinical trials that test regimens in patient subgroups including the elderly.
Primary AL Amyloidosis
Immunoglobulin light chain (AL) amyloidosis is the most common type of amyloidosis in Western countries. A plasma cell clone secretes a patient specific monoclonal light chain which is amyloidogenic and causes progressive decline of vital organ function [110]. Median age of diagnosis is 62 years and the disease presents most commonly in the seventh decade of life [111]. Incidence increases with age as proteostasis progressively declines but also due to aforementioned increased incidence of associated and co-existent plasma cell dyscrasias [112,113]. Frailty in amyloidosis is associated with the type and degree of organ involvement rather than chronological age of the patient per se more so than in other hematological malignancies. The systemic nature of the disease, the rapidly progressive decline in vital organ function and the often delayed diagnosis make a large proportion of patients frail at diagnosis. Elderly patients represent however an even frailer group [114]. Older patients with AL are also underrepresented in clinical trials and GA tools are lacking. An additional complexity lays in the diagnosis of AL amyloidosis as its presentation can mimic conditions that are more prevalent in the elderly population. High clinical suspicion and appropriate diagnostic investigations are necessary to set the correct diagnosis in a timely manner.
Clinical picture and diagnosis
The presentation of AL amyloidosis is heterogeneous and depends on organ involvement. Target organs include the heart, kidneys, soft tissues, liver, peripheral and autonomic nervous system [115]. Cardiac involvement is seen in 82% of patients and presents as restrictive cardiomyopathy. The revised Mayo staging system includes 4 stages of cardiac disease severity using cardiac biomarker assessment (NT-proBNP and cardiac troponins I or T or high sensitivity cTnT) and measurement of the free light chains (FLC) [116]. NT-proBNP can also increase in other more common cardiac conditions and due to renal impairment [117]. The value of the prognostic system has therefore been questioned in older patients [118]. Renal involvement is seen in approximately 68% of patients and presents as albuminuria and progressive decline in renal function [119]. Kidney biopsy is often required to determine the cause of albuminuria, particularly in patients with comorbidities such as chronic hypertension and diabetes. Caution is required as other types of amyloidosis are included in the differential diagnosis particularly in the elderly population. All elderly Caucasian males with cardiac involvement should have a Tc-99m 3,3-diphosphono-1,2-propanodicarboxylic acid (Tc-99m DPD) bone scan to exclude transthyretin-related cardiac amyloidosis (ATTR). About 39% of patients with ATTR will also have a plasma cell dyscrasia leading to increased chances of misdiagnosis [120,121]. Cases of hereditary amyloidosis coexisting with monoclonal gammopathy of undetermined significance and AA amyloidosis coexisting with IgM monoclonal gammopathy have also been reported [122–124]. Amyloid typing is mandatory in all the above cases using mass spectrometry, immnunoelectron microscopy or immunohistochemistry.
How we treat elderly patients with AL amyloidosis
The primary aim of therapy is to achieve a hematologic response by reducing the amyloidogenic light chain production via plasma cell clone eradication. A deep haematological response is a prerequisite for the secondary aim which is the gradual restoration of the target organ function [125]. Treatment is guided on the basis of cardiac staging and assessment of patient fitness and clinical trials for the older AL patients are lacking [126]. There are strict eligibility criteria for ASCT in patients with AL amyloidosis [114]. A cut-off at 70 years is used by most centres and patients > 65 years receive reduced conditioning [127]. Non-eligible patients will receive risk-adapted therapy. Conventional treatment is based on alkylating agents (melphalan and cyclophosphamide), PIs and IMiDs [128,129]. Retrospective studies have demonstrated up to 90% hematologic response with the triplet combination of Bortezomib-Cyclophosphamide-Dexamethasone (VCD) in first line treatment [130,131]. Interim analysis of a phase 3 clinical trial comparing melphalan-dexamethasone (MDex) to bortezomib-MDex showed higher responses with the latter. Ixazomib compared to physicians’ best choice is being assessed currently [132] and IMiDs are used mainly in the relapsed/refractory setting [133,134]. Addition of alkylating agents to IMiDs can achieve even higher responses but myelotoxicity is a concern [135]. Daratumumab monotherapy also yielded haematological responses in heavily pre-treated AL patients [136]. Contraindications to agent use include neuropathy for bortezomib, renal failure for lenalidomide. Agents that target the amyloid deposits (passive immunotherapy) with the aim of accelerating removal and restoring organ function are currently being investigated in clinical trials [137–139]. Current EMN guidelines suggest a risk-adapted stratification treatment approach which includes age (Figure 3). For low risk-transplant eligible patients VCD induction followed by MEL 200 mg/m2 plus bortezomib post ASCT if complete response has not been achieved. Intermediate risk patients should receive MDex or BMDex. ΜDex alone for patients with neuropathy and t(11;14) translocation and for patients with 1q21 gain or renal failure VCD [140]. Close monitoring of dFLC is required to initiate treatment at relapse without waiting for organ progression but no consensus exists on timepoint of treatment re-initiation. Lenalidomide, pomalidomide ixazomib or bendamustine are all indicated at relapse [141]. Frontline treatment can be repeated or switched to another option for refractory patients. The complex nature of the disease makes supportive management imperative to treatment-related toxicity minimization and increased tolerability. The poor physiological reserves associated with target organ dysfunction require a multidisciplinary approach that requires heart failure specialists, nephrologists, haematologists and nutrition specialists [142–145].
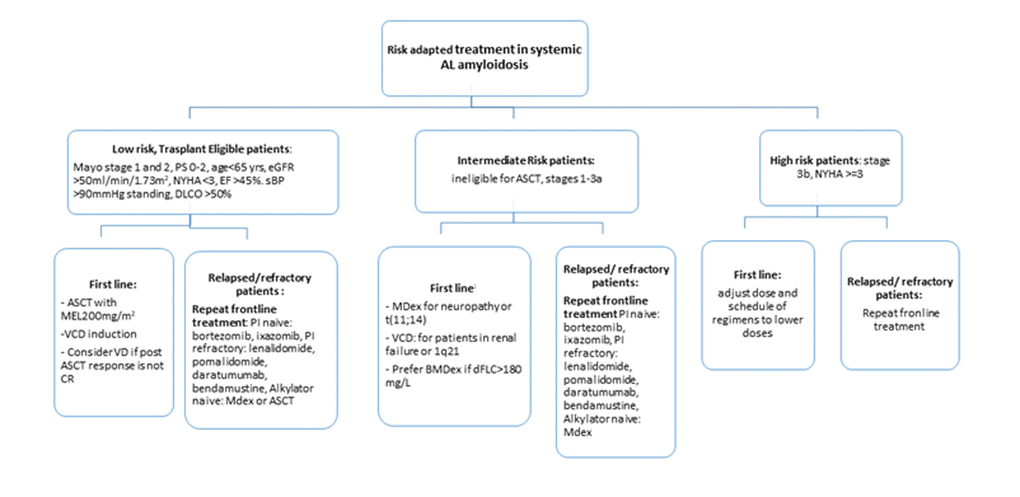
Figure 3. Risk adapted treatment recommendations in systemic AL amyloidosis. Adapted from Palladini et al 2016(115) and Gavriatopoulou et al 2018. Data mainly comes from uncontrolled trials. ASCT: autologous stem cell transplant, DLCO: lung diffusion of CO, EF: ejection fraction, MEL: melphalan, NYHA: New York Heart Association, OS: performance status by ECOG, sBP: systolic blood pressure, Stage is Mayo Clinic cardiac stage, VCD: velcade+cyclophosphamide+dexamethasone, CD: velcade+ dexamethasone, MDex: melphalan+dexamethasone, CR: complete response, PI: proteasome inhibitor, BMDex: bortezomib+ melphalan+dexamethasone.
Commentary
Frailty is a more complex construct in AL amyloidosis compared to other plasma cell dyscrasias owing to the nature of the disease. Frailty assessment tools are lacking and older patients are underrepresented in clinical trials. Development of GA tools will be a more difficult endeavour for this heterogeneous disease as multiple physiological parameters will need to be accurately assessed to appropriately categorize patients and set treatment goals. Clinical trials designed specifically for older or more frail patients are needed to guide evidence based medicine in these patients with poor overall outcomes. Increased awareness of the entity of AL amyloidosis and consideration in the differential diagnosis of physicians is imperative for timely diagnosis. Finally, pre-emptive supportive measures is key to maximize treatment tolerance and improve treatment outcomes.
Conflicts of Interest
M.G has received honoraria from Takeda, Amgen and Research grant from Novartis, MAD has received honoraria from Celgene, BMS, Janssen, Takeda, Amgen, E.K has received honoraria from Genesis Pharma, Janssen, Amgen, Takeda, Prothena and E.T has received honoraria form Celgene, Genesis Pharma, Janssen, Takeda. DF and INS have nothing to disclose.
References
- 1. United Nations. 2018. http://www.un.org/en/sections/issues-depth/ageing/.
- 2. Abel GA, Klepin HD. Frailty and the management of hematologic malignancies. Blood. 2018; 131:515–24. https://doi.org/10.1182/blood-2017-09-746420 [PubMed]
- 3. Hamaker ME, Prins MC, Stauder R. The relevance of a geriatric assessment for elderly patients with a haematological malignancy--a systematic review. Leuk Res. 2014; 38:275–83. https://doi.org/10.1016/j.leukres.2013.12.018 [PubMed]
- 4. Fried LP, Ferrucci L, Darer J, Williamson JD, Anderson G. Untangling the concepts of disability, frailty, and comorbidity: implications for improved targeting and care. J Gerontol A Biol Sci Med Sci. 2004; 59:255–63. https://doi.org/10.1093/gerona/59.3.M255 [PubMed]
- 5. Hamaker ME, Te Molder M, Thielen N, van Munster BC, Schiphorst AH, van Huis LH. The effect of a geriatric evaluation on treatment decisions and outcome for older cancer patients - A systematic review. J Geriatr Oncol. 2018; 9:430–40. https://doi.org/10.1016/j.jgo.2018.03.014 [PubMed]
- 6. Extermann M, Hurria A. Comprehensive geriatric assessment for older patients with cancer. J Clin Oncol. 2007; 25:1824–31. https://doi.org/10.1200/JCO.2007.10.6559 [PubMed]
- 7. Pallis AG, Wedding U, Lacombe D, Soubeyran P, Wildiers H. Questionnaires and instruments for a multidimensional assessment of the older cancer patient: what clinicians need to know? Eur J Cancer. 2010; 46:1019–25. https://doi.org/10.1016/j.ejca.2010.01.006 [PubMed]
- 8. Palumbo A, Bringhen S, Ludwig H, Dimopoulos MA, Bladé J, Mateos MV, Rosiñol L, Boccadoro M, Cavo M, Lokhorst H, Zweegman S, Terpos E, Davies F, et al. Personalized therapy in multiple myeloma according to patient age and vulnerability: a report of the European Myeloma Network (EMN). Blood. 2011; 118:4519–29. https://doi.org/10.1182/blood-2011-06-358812 [PubMed]
- 9. Rosenberg PS, Barker KA, Anderson WF. Future distribution of multiple myeloma in the United States by sex, age, and race/ethnicity. Blood. 2015; 125:410–12. https://doi.org/10.1182/blood-2014-10-609461 [PubMed]
- 10. van de Donk NW, Lokhorst HM. New developments in the management and treatment of newly diagnosed and relapsed/refractory multiple myeloma patients. Expert Opin Pharmacother. 2013; 14:1569–73. https://doi.org/10.1517/14656566.2013.805746 [PubMed]
- 11. Kumar SK, Dispenzieri A, Lacy MQ, Gertz MA, Buadi FK, Pandey S, Kapoor P, Dingli D, Hayman SR, Leung N, Lust J, McCurdy A, Russell SJ, et al. Continued improvement in survival in multiple myeloma: changes in early mortality and outcomes in older patients. Leukemia. 2014; 28:1122–28. https://doi.org/10.1038/leu.2013.313 [PubMed]
- 12. Pozzi S, Marcheselli L, Bari A, Liardo EV, Marcheselli R, Luminari S, Quaresima M, Cirilli C, Ferri P, Federico M, Sacchi S. Survival of multiple myeloma patients in the era of novel therapies confirms the improvement in patients younger than 75 years: a population-based analysis. Br J Haematol. 2013; 163:40–46. https://doi.org/10.1111/bjh.12465 [PubMed]
- 13. Mateos MV, Oriol A, Martínez-López J, Gutiérrez N, Teruel AI, de Paz R, García-Laraña J, Bengoechea E, Martín A, Mediavilla JD, Palomera L, de Arriba F, González Y, et al. Bortezomib, melphalan, and prednisone versus bortezomib, thalidomide, and prednisone as induction therapy followed by maintenance treatment with bortezomib and thalidomide versus bortezomib and prednisone in elderly patients with untreated multiple myeloma: a randomised trial. Lancet Oncol. 2010; 11:934–41. https://doi.org/10.1016/S1470-2045(10)70187-X [PubMed]
- 14. Verlest S, Karim-Kos H,Blommenstein H, et al. Are we making progress? Survival in plasma cell malignancies in the era of novel treatments. A population based study of 17790 patients in the Netherlands [abstract]. . Haematologica. 2012; 97:242. Abstract 0592 .
- 15. Anagnostopoulos A, Gika D, Symeonidis A, Zervas K, Pouli A, Repoussis P, Grigoraki V, Anagnostopoulos N, Economopoulos T, Maniatis A, Dimopoulos MA. Multiple myeloma in elderly patients: prognostic factors and outcome. Eur J Haematol. 2005; 75:370–75. https://doi.org/10.1111/j.1600-0609.2005.00532.x [PubMed]
- 16. Siegel DS, Desikan KR, Mehta J, Singhal S, Fassas A, Munshi N, Anaissie E, Naucke S, Ayers D, Spoon D, Vesole D, Tricot G, Barlogie B. Age is not a prognostic variable with autotransplants for multiple myeloma. Blood. 1999; 93:51–54. [PubMed]
- 17. Chng WJ, Dispenzieri A, Chim CS, Fonseca R, Goldschmidt H, Lentzsch S, Munshi N, Palumbo A, Miguel JS, Sonneveld P, Cavo M, Usmani S, Durie BG, Avet-Loiseau H, and International Myeloma Working Group. IMWG consensus on risk stratification in multiple myeloma. Leukemia. 2014; 28:269–77. https://doi.org/10.1038/leu.2013.247 [PubMed]
- 18. Gavriatopoulou M, Fotiou D, Roussou M, Migkou M, Ntanasis-Stathopoulos I, Kanellias N, Ziogas DC, et al. Vulnerability variables among Octagenarians Myeloma patients: A Single -Center Analysis in 110 Patients. Leuk Lymphoma. 2017; in press.
- 19. Brenner H, Gondos A, Pulte D. Recent major improvement in long-term survival of younger patients with multiple myeloma. Blood. 2008; 111:2521–26. https://doi.org/10.1182/blood-2007-08-104984 [PubMed]
- 20. Panitsas F, Kothari J, Vallance G, Djebbari F, Ferguson L, Sultanova M, Ramasamy K. Treat or palliate: outcomes of very elderly myeloma patients. Haematologica. 2018; 103:e32–34. https://doi.org/10.3324/haematol.2017.173617 [PubMed]
- 21. Larocca A, Dold SM, Zweegman S, Terpos E, Wäsch R, D’Agostino M, Scheubeck S, Goldschmidt H, Gay F, Cavo M, Ludwig H, Straka C, Bringhen S, et al. Patient-centered practice in elderly myeloma patients: an overview and consensus from the European Myeloma Network (EMN). Leukemia. 2018; 32:1697–712. https://doi.org/10.1038/s41375-018-0142-9 [PubMed]
- 22. Hutchins LF, Unger JM, Crowley JJ, Coltman CA
Jr , Albain KS. Underrepresentation of patients 65 years of age or older in cancer-treatment trials. N Engl J Med. 1999; 341:2061–67. https://doi.org/10.1056/NEJM199912303412706 [PubMed] - 23. Rose JH, O’Toole EE, Dawson NV, Lawrence R, Gurley D, Thomas C, Hamel MB, Cohen HJ. Perspectives, preferences, care practices, and outcomes among older and middle-aged patients with late-stage cancer. J Clin Oncol. 2004; 22:4907–17. https://doi.org/10.1200/JCO.2004.06.050 [PubMed]
- 24. Fried TR, Bradley EH, Towle VR, Allore H. Understanding the treatment preferences of seriously ill patients. N Engl J Med. 2002; 346:1061–66. https://doi.org/10.1056/NEJMsa012528 [PubMed]
- 25. Rajkumar SV, Dimopoulos MA, Palumbo A, Blade J, Merlini G, Mateos MV, Kumar S, Hillengass J, Kastritis E, Richardson P, Landgren O, Paiva B, Dispenzieri A, et al. International Myeloma Working Group updated criteria for the diagnosis of multiple myeloma. Lancet Oncol. 2014; 15:e538–48. https://doi.org/10.1016/S1470-2045(14)70442-5 [PubMed]
- 26. Wildes TM, Campagnaro E. Management of multiple myeloma in older adults: gaining ground with geriatric assessment. J Geriatr Oncol. 2017; 8:1–7. https://doi.org/10.1016/j.jgo.2016.04.001 [PubMed]
- 27. Greipp PR, San Miguel J, Durie BG, Crowley JJ, Barlogie B, Bladé J, Boccadoro M, Child JA, Avet-Loiseau H, Kyle RA, Lahuerta JJ, Ludwig H, Morgan G, et al. International staging system for multiple myeloma. J Clin Oncol. 2005; 23:3412–20. https://doi.org/10.1200/JCO.2005.04.242 [PubMed]
- 28. Bataille R, Annweiler C, Beauchet O. Multiple myeloma international staging system: “staging” or simply “aging” system? Clin Lymphoma Myeloma Leuk. 2013; 13:635–37. https://doi.org/10.1016/j.clml.2013.07.003 [PubMed]
- 29. Palumbo A, Avet-Loiseau H, Oliva S, Lokhorst HM, Goldschmidt H, Rosinol L, Richardson P, Caltagirone S, Lahuerta JJ, Facon T, Bringhen S, Gay F, Attal M, et al. Revised International Staging System for Multiple Myeloma: A Report From International Myeloma Working Group. J Clin Oncol. 2015; 33:2863–69. https://doi.org/10.1200/JCO.2015.61.2267 [PubMed]
- 30. Palumbo A, Bringhen S, Mateos MV, Larocca A, Facon T, Kumar SK, Offidani M, McCarthy P, Evangelista A, Lonial S, Zweegman S, Musto P, Terpos E, et al. Geriatric assessment predicts survival and toxicities in elderly myeloma patients: an International Myeloma Working Group report. Blood. 2015; 125:2068–74. https://doi.org/10.1182/blood-2014-12-615187 [PubMed]
- 31. Milani P, Vincent Rajkumar S, Merlini G, Kumar S, Gertz MA, Palladini G, Lacy MQ, Buadi FK, Hayman SR, Leung N, Dingli D, Lust JA, Lin Y, et al. N-terminal fragment of the type-B natriuretic peptide (NT-proBNP) contributes to a simple new frailty score in patients with newly diagnosed multiple myeloma. Am J Hematol. 2016; 91:1129–34. https://doi.org/10.1002/ajh.24532 [PubMed]
- 32. Takeoka Y, Sakatoku K, Miura A, Yamamura R, Araki T, Seura H, Okamura T, Koh H, Nakamae H, Hino M, Ohta K. Prognostic Effect of Low Subcutaneous Adipose Tissue on Survival Outcome in Patients With Multiple Myeloma. Clin Lymphoma Myeloma Leuk. 2016; 16:434–41. https://doi.org/10.1016/j.clml.2016.04.010 [PubMed]
- 33. Palumbo A, Waage A, Hulin C, Beksac M, Zweegman S, Gay F, Gimsing P, Leleu X, Wijermans P, Sucak G, Pezzatti S, Juliusson G, Pégourié B, et al. Safety of thalidomide in newly diagnosed elderly myeloma patients: a meta-analysis of data from individual patients in six randomized trials. Haematologica. 2013; 98:87–94. https://doi.org/10.3324/haematol.2012.067058 [PubMed]
- 34. Fayers PM, Palumbo A, Hulin C, Waage A, Wijermans P, Beksaç M, Bringhen S, Mary JY, Gimsing P, Termorshuizen F, Haznedar R, Caravita T, Moreau P, et al, and Nordic Myeloma Study Group, and Italian Multiple Myeloma Network, and Turkish Myeloma Study Group, and Hemato-Oncologie voor Volwassenen Nederland, and Intergroupe Francophone du Myélome, and European Myeloma Network. Thalidomide for previously untreated elderly patients with multiple myeloma: meta-analysis of 1685 individual patient data from 6 randomized clinical trials. Blood. 2011; 118:1239–47. https://doi.org/10.1182/blood-2011-03-341669 [PubMed]
- 35. San Miguel JF, Schlag R, Khuageva NK, Dimopoulos MA, Shpilberg O, Kropff M, Spicka I, Petrucci MT, Palumbo A, Samoilova OS, Dmoszynska A, Abdulkadyrov KM, Schots R, et al, and VISTA Trial Investigators. Bortezomib plus melphalan and prednisone for initial treatment of multiple myeloma. N Engl J Med. 2008; 359:906–17. https://doi.org/10.1056/NEJMoa0801479 [PubMed]
- 36. San Miguel JF, Schlag R, Khuageva NK, Dimopoulos MA, Shpilberg O, Kropff M, Spicka I, Petrucci MT, Palumbo A, Samoilova OS, Dmoszynska A, Abdulkadyrov KM, Delforge M, et al. Persistent overall survival benefit and no increased risk of second malignancies with bortezomib-melphalan-prednisone versus melphalan-prednisone in patients with previously untreated multiple myeloma. J Clin Oncol. 2013; 31:448–55. https://doi.org/10.1200/JCO.2012.41.6180 [PubMed]
- 37. Facon T, Lee JH, Moreau P, Niesvizky R, Dimopoulos MA, Jahek R, et al. Phase 3 study (CLARION) of carfilzomib, melphalan, prednisone (KMP) v bortezomib, melphalan, prednisone (VMP) in newly diagnosed multiple myeloma (NDMM). International Myeloma Workshop2017.
- 38. Durie BG, Hoering A, Abidi MH, Rajkumar SV, Epstein J, Kahanic SP, Thakuri M, Reu F, Reynolds CM, Sexton R, Orlowski RZ, Barlogie B, Dispenzieri A. Bortezomib with lenalidomide and dexamethasone versus lenalidomide and dexamethasone alone in patients with newly diagnosed myeloma without intent for immediate autologous stem-cell transplant (SWOG S0777): a randomised, open-label, phase 3 trial. Lancet. 2017; 389:519–27. https://doi.org/10.1016/S0140-6736(16)31594-X [PubMed]
- 39. Mateos MV, Dimopoulos MA, Cavo M, Suzuki K, Jakubowiak A, Knop S, Doyen C, Lucio P, Nagy Z, Kaplan P, Pour L, Cook M, Grosicki S, et al, and ALCYONE Trial Investigators. Daratumumab plus Bortezomib, Melphalan, and Prednisone for Untreated Myeloma. N Engl J Med. 2018; 378:518–28. https://doi.org/10.1056/NEJMoa1714678 [PubMed]
- 40. Palumbo A, Hajek R, Delforge M, Kropff M, Petrucci MT, Catalano J, Gisslinger H, Wiktor-Jędrzejczak W, Zodelava M, Weisel K, Cascavilla N, Iosava G, Cavo M, et al, and MM-015 Investigators. Continuous lenalidomide treatment for newly diagnosed multiple myeloma. N Engl J Med. 2012; 366:1759–69. https://doi.org/10.1056/NEJMoa1112704 [PubMed]
- 41. Facon T, Dimopoulos MA, Dispenzieri A, Catalano JV, Belch A, Cavo M, Pinto A, Weisel K, Ludwig H, Bahlis NJ, Banos A, Tiab M, Delforge M, et al. Final analysis of survival outcomes in the phase 3 FIRST trial of up-front treatment for multiple myeloma. Blood. 2018; 131:301–10. https://doi.org/10.1182/blood-2017-07-795047 [PubMed]
- 42. Jackson G, Davies FE, Pawlyn C, Caims D, Striha A, Hockaday A, et al. Lenalidomide maintenance significantly improves outcomes compared to observation irrespective of cytogenetic risk. Results of the myeloma XI Trial. ASH 59th Annual Meeting2017.
- 43. Magarotto V, Bringhen S, Offidani M, Benevolo G, Patriarca F, Mina R, Falcone AP, De Paoli L, Pietrantuono G, Gentili S, Musolino C, Giuliani N, Bernardini A, et al. Triplet vs doublet lenalidomide-containing regimens for the treatment of elderly patients with newly diagnosed multiple myeloma. Blood. 2016; 127:1102–08. https://doi.org/10.1182/blood-2015-08-662627 [PubMed]
- 44. Bringhen S, Mateos MV, Zweegman S, Larocca A, Falcone AP, Oriol A, Rossi D, Cavalli M, Wijermans P, Ria R, Offidani M, Lahuerta JJ, Liberati AM, et al. Age and organ damage correlate with poor survival in myeloma patients: meta-analysis of 1435 individual patient data from 4 randomized trials. Haematologica. 2013; 98:980–87. https://doi.org/10.3324/haematol.2012.075051 [PubMed]
- 45. Ludwig H, Delforge M, Facon T, Einsele H, Gay F, Moreau P, Avet-Loiseau H, Boccadoro M, Hajek R, Mohty M, Cavo M, Dimopoulos MA, San-Miguel JF, et al. Prevention and management of adverse events of Novel agents in multiple myeloma: A consensus of the european myeloma network. Leukemia. 2017. https://doi.org/10.1038/leu.2017.353 [PubMed]
- 46. Cid Ruzafa J, Merinopoulou E, Baggaley RF, Leighton P, Werther W, Felici D, Cox A. Patient population with multiple myeloma and transitions across different lines of therapy in the USA: an epidemiologic model. Pharmacoepidemiol Drug Saf. 2016; 25:871–79. https://doi.org/10.1002/pds.3927 [PubMed]
- 47. Lopez A, Mateos MV, Oriol A, Valero M, Martínez J, Lorenzo JI, Perez M, Martinez R, de Paz R, Granell M, De Arriba F, Blanchard MJ, Peñalver FJ, et al. Patterns of relapse and outcome of elderly multiple myeloma patients treated as front-line therapy with novel agents combinations. Leuk Res Rep. 2015; 4:64–69. https://doi.org/10.1016/j.lrr.2015.09.002 [PubMed]
- 48. Dimopoulos MA, Moreau P, Palumbo A, Joshua D, Pour L, Hájek R, Facon T, Ludwig H, Oriol A, Goldschmidt H, Rosiñol L, Straub J, Suvorov A, et al, and ENDEAVOR Investigators. Carfilzomib and dexamethasone versus bortezomib and dexamethasone for patients with relapsed or refractory multiple myeloma (ENDEAVOR): a randomised, phase 3, open-label, multicentre study. Lancet Oncol. 2016; 17:27–38. https://doi.org/10.1016/S1470-2045(15)00464-7 [PubMed]
- 49. Stewart AK, Rajkumar SV, Dimopoulos MA, Masszi T, Špička I, Oriol A, Hájek R, Rosiñol L, Siegel DS, Mihaylov GG, Goranova-Marinova V, Rajnics P, Suvorov A, et al, and ASPIRE Investigators. Carfilzomib, lenalidomide, and dexamethasone for relapsed multiple myeloma. N Engl J Med. 2015; 372:142–52. https://doi.org/10.1056/NEJMoa1411321 [PubMed]
- 50. Dimopoulos MA, Stewart AK, Masszi T, Špička I, Oriol A, Hájek R, Rosiñol L, Siegel D, Mihaylov GG, Goranova-Marinova V, Rajnics P, Suvorov A, Niesvizky R, et al. Carfilzomib, lenalidomide, and dexamethasone in patients with relapsed multiple myeloma categorised by age: secondary analysis from the phase 3 ASPIRE study. Br J Haematol. 2017; 177:404–13. https://doi.org/10.1111/bjh.14549 [PubMed]
- 51. Dimopoulos MA, Oriol A, Nahi H, San-Miguel J, Bahlis NJ, Usmani SZ, Rabin N, Orlowski RZ, Komarnicki M, Suzuki K, Plesner T, Yoon SS, Ben Yehuda D, et al, and POLLUX Investigators. Daratumumab, Lenalidomide, and Dexamethasone for Multiple Myeloma. N Engl J Med. 2016; 375:1319–31. https://doi.org/10.1056/NEJMoa1607751 [PubMed]
- 52. Palumbo A, Chanan-Khan A, Weisel K, Nooka AK, Masszi T, Beksac M, Spicka I, Hungria V, Munder M, Mateos MV, Mark TM, Qi M, Schecter J, et al, and CASTOR Investigators. Daratumumab, Bortezomib, and Dexamethasone for Multiple Myeloma. N Engl J Med. 2016; 375:754–66. https://doi.org/10.1056/NEJMoa1606038 [PubMed]
- 53. Miguel JS, Weisel K, Moreau P, Lacy M, Song K, Delforge M, Karlin L, Goldschmidt H, Banos A, Oriol A, Alegre A, Chen C, Cavo M, et al. Pomalidomide plus low-dose dexamethasone versus high-dose dexamethasone alone for patients with relapsed and refractory multiple myeloma (MM-003): a randomised, open-label, phase 3 trial. Lancet Oncol. 2013; 14:1055–66. https://doi.org/10.1016/S1470-2045(13)70380-2 [PubMed]
- 54. Lonial S, Dimopoulos M, Palumbo A, White D, Grosicki S, Spicka I, Walter-Croneck A, Moreau P, Mateos MV, Magen H, Belch A, Reece D, Beksac M, et al, and ELOQUENT-2 Investigators. Elotuzumab Therapy for Relapsed or Refractory Multiple Myeloma. N Engl J Med. 2015; 373:621–31. https://doi.org/10.1056/NEJMoa1505654 [PubMed]
- 55. Merz M, Jansen L, Castro FA, Hillengass J, Salwender H, Weisel K, Scheid C, Luttmann S, Emrich K, Holleczek B, Katalinic A, Nennecke A, Straka C, et al, and GEKID Cancer Survival Working Group and the DRST. Survival of elderly patients with multiple myeloma-Effect of upfront autologous stem cell transplantation. Eur J Cancer. 2016; 62:1–8. https://doi.org/10.1016/j.ejca.2016.04.004 [PubMed]
- 56. Auner HW, Iacobelli S, Sbianchi G, Knol-Bout C, Blaise D, Russell NH, Apperley JF, Pohlreich D, Browne PV, Kobbe G, Isaksson C, Lenhoff S, Scheid C, et al. Melphalan 140 mg/m(2) or 200 mg/m(2) for autologous transplantation in myeloma: results from the Collaboration to Collect Autologous Transplant Outcomes in Lymphoma and Myeloma (CALM) study. A report by the EBMT Chronic Malignancies Working Party. Haematologica. 2018; 103:514–21. https://doi.org/10.3324/haematol.2017.181339 [PubMed]
- 57. Straka C, Liebisch P, Salwender H, Hennemann B, Metzner B, Knop S, Adler-Reichel S, Gerecke C, Wandt H, Bentz M, Bruemmendorf TH, Hentrich M, Pfreundschuh M, et al. Autotransplant with and without induction chemotherapy in older multiple myeloma patients: long-term outcome of a randomized trial. Haematologica. 2016; 101:1398–406. https://doi.org/10.3324/haematol.2016.151860 [PubMed]
- 58. Kleber M, Ihorst G, Gross B, Koch B, Reinhardt H, Wäsch R, Engelhardt M. Validation of the Freiburg Comorbidity Index in 466 multiple myeloma patients and combination with the international staging system are highly predictive for outcome. Clin Lymphoma Myeloma Leuk. 2013; 13:541–51. https://doi.org/10.1016/j.clml.2013.03.013 [PubMed]
- 59. Engelhardt M, Domm AS, Dold SM, Ihorst G, Reinhardt H, Zober A, Hieke S, Baayen C, Müller SJ, Einsele H, Sonneveld P, Landgren O, Schumacher M, Wäsch R. A concise revised Myeloma Comorbidity Index as a valid prognostic instrument in a large cohort of 801 multiple myeloma patients. Haematologica. 2017; 102:910–21. https://doi.org/10.3324/haematol.2016.162693 [PubMed]
- 60. Straka C, Schafer-Eckart K, Bassermann F, HertesteinB, Engelhardt M, Salwender H, et al. Prospective randomized trial of Len.Dex induction followed by tandem MEL140 with autologous blood stem cell transplantation and len maintenance versus continued therapy with Len/Dex in myeloma patients age 60-75 years: protocol defined safety analysis [abstract].Blood. 2012; 120:2012.
- 61. Terpos E, Kleber M, Engelhardt M, Zweegman S, Gay F, Kastritis E, van de Donk NW, Bruno B, Sezer O, Broijl A, Bringhen S, Beksac M, Larocca A, et al, and European Myeloma Network. European Myeloma Network guidelines for the management of multiple myeloma-related complications. Haematologica. 2015; 100:1254–66. https://doi.org/10.3324/haematol.2014.117176 [PubMed]
- 62. Palumbo A, Mateos MV, Bringhen S, San Miguel JF. Practical management of adverse events in multiple myeloma: can therapy be attenuated in older patients? Blood Rev. 2011; 25:181–91. https://doi.org/10.1016/j.blre.2011.03.005 [PubMed]
- 63. Richardson PG, Sonneveld P, Schuster MW, Irwin D, Stadtmauer EA, Facon T, Harousseau JL, Ben-Yehuda D, Lonial S, Goldschmidt H, Reece D, San-Miguel JF, Bladé J, et al, and Assessment of Proteasome Inhibition for Extending Remissions (APEX) Investigators. Bortezomib or high-dose dexamethasone for relapsed multiple myeloma. N Engl J Med. 2005; 352:2487–98. https://doi.org/10.1056/NEJMoa043445 [PubMed]
- 64. Dimopoulos M, Spencer A, Attal M, Prince HM, Harousseau JL, Dmoszynska A, San Miguel J, Hellmann A, Facon T, Foà R, Corso A, Masliak Z, Olesnyckyj M, et al, and Multiple Myeloma (010) Study Investigators. Lenalidomide plus dexamethasone for relapsed or refractory multiple myeloma. N Engl J Med. 2007; 357:2123–32. https://doi.org/10.1056/NEJMoa070594 [PubMed]
- 65. Weber DM, Chen C, Niesvizky R, Wang M, Belch A, Stadtmauer EA, Siegel D, Borrello I, Rajkumar SV, Chanan-Khan AA, Lonial S, Yu Z, Patin J, et al, and Multiple Myeloma (009) Study Investigators. Lenalidomide plus dexamethasone for relapsed multiple myeloma in North America. N Engl J Med. 2007; 357:2133–42. https://doi.org/10.1056/NEJMoa070596 [PubMed]
- 66. Gewandter JS, Fan L, Magnuson A, Mustian K, Peppone L, Heckler C, Hopkins J, Tejani M, Morrow GR, Mohile SG. Falls and functional impairments in cancer survivors with chemotherapy-induced peripheral neuropathy (CIPN): a University of Rochester CCOP study. Support Care Cancer. 2013; 21:2059–66. https://doi.org/10.1007/s00520-013-1766-y [PubMed]
- 67. Richardson PG, Delforge M, Beksac M, Wen P, Jongen JL, Sezer O, Terpos E, Munshi N, Palumbo A, Rajkumar SV, Harousseau JL, Moreau P, Avet-Loiseau H, et al. Management of treatment-emergent peripheral neuropathy in multiple myeloma. Leukemia. 2012; 26:595–608. https://doi.org/10.1038/leu.2011.346 [PubMed]
- 68. Delforge M, Bladé J, Dimopoulos MA, Facon T, Kropff M, Ludwig H, Palumbo A, Van Damme P, San-Miguel JF, Sonneveld P. Treatment-related peripheral neuropathy in multiple myeloma: the challenge continues. Lancet Oncol. 2010; 11:1086–95. https://doi.org/10.1016/S1470-2045(10)70068-1 [PubMed]
- 69. De Stefano V, Za T, Rossi E. Venous thromboembolism in multiple myeloma. Semin Thromb Hemost. 2014; 40:338–47. https://doi.org/10.1055/s-0034-1370793 [PubMed]
- 70. Palumbo A, Rajkumar SV, Dimopoulos MA, Richardson PG, San Miguel J, Barlogie B, Harousseau J, Zonder JA, Cavo M, Zangari M, Attal M, Belch A, Knop S, et al, and International Myeloma Working Group. Prevention of thalidomide- and lenalidomide-associated thrombosis in myeloma. Leukemia. 2008; 22:414–23. https://doi.org/10.1038/sj.leu.2405062 [PubMed]
- 71. Dimopoulos MA, Palumbo A, Attal M, Beksaç M, Davies FE, Delforge M, Einsele H, Hajek R, Harousseau JL, da Costa FL, Ludwig H, Mellqvist UH, Morgan GJ, et al, and European Myeloma Network. Optimizing the use of lenalidomide in relapsed or refractory multiple myeloma: consensus statement. Leukemia. 2011; 25:749–60. https://doi.org/10.1038/leu.2011.3 [PubMed]
- 72. Dimopoulos MA, Leleu X, Palumbo A, Moreau P, Delforge M, Cavo M, Ludwig H, Morgan GJ, Davies FE, Sonneveld P, Schey SA, Zweegman S, Hansson M, et al. Expert panel consensus statement on the optimal use of pomalidomide in relapsed and refractory multiple myeloma. Leukemia. 2014; 28:1573–85. https://doi.org/10.1038/leu.2014.60 [PubMed]
- 73. Aoki T, Nishiyama T, Imahashi N, Kitamura K. Efficacy of continuous, daily, oral, ultra-low-dose 200 mg acyclovir to prevent herpes zoster events among bortezomib-treated patients: a report from retrospective study. Jpn J Clin Oncol. 2011; 41:876–81. https://doi.org/10.1093/jjco/hyr063 [PubMed]
- 74. Pour L, Adam Z, Buresova L, Krejci M, Krivanova A, Sandecka V, Zahradova L, Buchler T, Vorlicek J, Hajek R. Varicella-zoster virus prophylaxis with low-dose acyclovir in patients with multiple myeloma treated with bortezomib. Clin Lymphoma Myeloma. 2009; 9:151–53. https://doi.org/10.3816/CLM.2009.n.036 [PubMed]
- 75. Nucci M, Anaissie E. Infections in patients with multiple myeloma. Semin Hematol. 2009; 46:277–88. https://doi.org/10.1053/j.seminhematol.2009.03.006 [PubMed]
- 76. Bowcock SP, Iqbal G, Pratt G, Yong K, et al. Levofloxacin prophylaxis in newly diagnosed myeloma reduces febrile episodes and death without increasing healthcare associated infections: results from the TEAMM trial. EHA 2018.
- 77. Mohile SG, Velarde C, Hurria A, Magnuson A, Lowenstein L, Pandya C, O’Donovan A, Gorawara-Bhat R, Dale W. Geriatric Assessment-Guided Care Processes for Older Adults: A Delphi Consensus of Geriatric Oncology Experts. J Natl Compr Canc Netw. 2015; 13:1120–30. https://doi.org/10.6004/jnccn.2015.0137 [PubMed]
- 78. Teitelbaum A, Ba-Mancini A, Huang H, Henk HJ. Health care costs and resource utilization, including patient burden, associated with novel-agent-based treatment versus other therapies for multiple myeloma: findings using real-world claims data. Oncologist. 2013; 18:37–45. https://doi.org/10.1634/theoncologist.2012-0113 [PubMed]
- 79. Goren A, Gilloteau I, Lees M, DaCosta Dibonaventura M. Quantifying the burden of informal caregiving for patients with cancer in Europe. Support Care Cancer. 2014; 22:1637–46. https://doi.org/10.1007/s00520-014-2122-6 [PubMed]
- 80. Treon SP. How I treat Waldenström macroglobulinemia. Blood. 2015; 126:721–32. https://doi.org/10.1182/blood-2015-01-553974 [PubMed]
- 81. Teras LR, DeSantis CE, Cerhan JR, Morton LM, Jemal A, Flowers CR. 2016 US lymphoid malignancy statistics by World Health Organization subtypes. CA Cancer J Clin. 2016; Epub ahead of print. https://doi.org/10.3322/caac.21357 [PubMed]
- 82. Kastritis E, Kyrtsonis MC, Morel P, Gavriatopoulou M, Hatjiharissi E, Symeonidis AS, Vassou A, Repousis P, Delimpasi S, Sioni A, Michalis E, Michael M, Vervessou E, et al. Competing risk survival analysis in patients with symptomatic Waldenström macroglobulinemia: the impact of disease unrelated mortality and of rituximab-based primary therapy. Haematologica. 2015; 100:e446–49. https://doi.org/10.3324/haematol.2015.124149 [PubMed]
- 83. Buske C, Leblond V. How to manage Waldenstrom’s macroglobulinemia. Leukemia. 2013; 27:762–72. https://doi.org/10.1038/leu.2013.36 [PubMed]
- 84. Buske C, Hutchings M, Ladetto M, Goede V, Mey U, Soubeyran P, Spina M, Stauder R, Trnený M, Wedding U, Fields P, Buske C, Dreyling M, et al, and ESMO Lymphoma Consensus Conference Panel Members. ESMO Consensus Conference on malignant lymphoma: general perspectives and recommendations for the clinical management of the elderly patient with malignant lymphoma. Ann Oncol. 2018; 29:544–62. https://doi.org/10.1093/annonc/mdx413 [PubMed]
- 85. Morel P, Duhamel A, Gobbi P, Dimopoulos MA, Dhodapkar MV, McCoy J, Crowley J, Ocio EM, Garcia-Sanz R, Treon SP, Leblond V, Kyle RA, Barlogie B, Merlini G. International prognostic scoring system for Waldenstrom macroglobulinemia. Blood. 2009; 113:4163–70. https://doi.org/10.1182/blood-2008-08-174961 [PubMed]
- 86. Kyle RA, Treon SP, Alexanian R, Barlogie B, Björkholm M, Dhodapkar M, Lister TA, Merlini G, Morel P, Stone M, Branagan AR, Leblond V. Prognostic markers and criteria to initiate therapy in Waldenstrom’s macroglobulinemia: consensus panel recommendations from the Second International Workshop on Waldenstrom’s Macroglobulinemia. Semin Oncol. 2003; 30:116–20. https://doi.org/10.1053/sonc.2003.50038 [PubMed]
- 87. Santos-Lozano A, Morales-Gonzalez A, Sanchis-Gomar F, Cristi-Montero C, Fiuza-Luces C, Pareja-Galeano H, Martínez-López J, Garatachea N, Lucia A. Response rate to the treatment of Waldenström macroglobulinemia: A meta-analysis of the results of clinical trials. Crit Rev Oncol Hematol. 2016; 105:118–26. https://doi.org/10.1016/j.critrevonc.2016.06.004 [PubMed]
- 88. Dimopoulos MA, Kastritis E, Owen RG, Kyle RA, Landgren O, Morra E, Leleu X, García-Sanz R, Munshi N, Anderson KC, Terpos E, Ghobrial IM, Morel P, et al. Treatment recommendations for patients with Waldenström macroglobulinemia (WM) and related disorders: IWWM-7 consensus. Blood. 2014; 124:1404–11. https://doi.org/10.1182/blood-2014-03-565135 [PubMed]
- 89. Buske C, Leblond V, Dimopoulos M, Kimby E, Jäger U, Dreyling M, and ESMO Guidelines Working Group. Waldenstrom’s macroglobulinaemia: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2013 (Suppl 6); 24:vi155–59. https://doi.org/10.1093/annonc/mdt298 [PubMed]
- 90. Kastritis E, Gavriatopoulou M, Kyrtsonis MC, Roussou M, Hadjiharissi E, Symeonidis A, Repoussis P, Michalis E, Delimpasi S, Tsatalas K, Tsirigotis P, Vassou A, Vervessou E, et al. Dexamethasone, rituximab, and cyclophosphamide as primary treatment of Waldenström macroglobulinemia: final analysis of a phase 2 study. Blood. 2015; 126:1392–94. https://doi.org/10.1182/blood-2015-05-647420 [PubMed]
- 91. Rummel MJ, Niederle N, Maschmeyer G, Banat GA, von Grünhagen U, Losem C, Kofahl-Krause D, Heil G, Welslau M, Balser C, Kaiser U, Weidmann E, Dürk H, et al, and Study group indolent Lymphomas (StiL). Bendamustine plus rituximab versus CHOP plus rituximab as first-line treatment for patients with indolent and mantle-cell lymphomas: an open-label, multicentre, randomised, phase 3 non-inferiority trial. Lancet. 2013; 381:1203–10. https://doi.org/10.1016/S0140-6736(12)61763-2 [PubMed]
- 92. Gavriatopoulou M, García-Sanz R, Kastritis E, Morel P, Kyrtsonis MC, Michalis E, Kartasis Z, Leleu X, Palladini G, Tedeschi A, Gika D, Merlini G, Sonneveld P, Dimopoulos MA. BDR in newly diagnosed patients with WM: final analysis of a phase 2 study after a minimum follow-up of 6 years. Blood. 2017; 129:456–59. https://doi.org/10.1182/blood-2016-09-742411 [PubMed]
- 93. Ghobrial IM, Xie W, Padmanabhan S, Badros A, Rourke M, Leduc R, Chuma S, Kunsman J, Warren D, Poon T, Harris B, Sam A, Anderson KC, et al. Phase II trial of weekly bortezomib in combination with rituximab in untreated patients with Waldenström Macroglobulinemia. Am J Hematol. 2010; 85:670–74. https://doi.org/10.1002/ajh.21788 [PubMed]
- 94. Treon SP, Ioakimidis L, Soumerai JD, Patterson CJ, Sheehy P, Nelson M, Willen M, Matous J, Mattern J
2nd , Diener JG, Keogh GP, Myers TJ, Boral A, et al. Primary therapy of Waldenström macroglobulinemia with bortezomib, dexamethasone, and rituximab: WMCTG clinical trial 05-180. J Clin Oncol. 2009; 27:3830–35. https://doi.org/10.1200/JCO.2008.20.4677 [PubMed] - 95. Kastritis E, Leblond V, Dimopoulos MA, Kimby E, Staber P, Kersten MJ, Tedeschi A, Buske C. Waldenstrom’s macroglobulinaemia: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Ann Oncol. 2018. https://doi.org/10.1093/annonc/mdy466
- 96. Treon SP, Tripsas CK, Meid K, Kanan S, Sheehy P, Chuma S, Xu L, Cao Y, Yang G, Liu X, Patterson CJ, Warren D, Hunter ZR, et al. Carfilzomib, rituximab, and dexamethasone (CaRD) treatment offers a neuropathy-sparing approach for treating Waldenström’s macroglobulinemia. Blood. 2014; 124:503–10. https://doi.org/10.1182/blood-2014-03-566273 [PubMed]
- 97. Leblond V, Kastritis E, Advani R, Ansell SM, Buske C, Castillo JJ, García-Sanz R, Gertz M, Kimby E, Kyriakou C, Merlini G, Minnema MC, Morel P, et al. Treatment recommendations from the Eighth International Workshop on Waldenström’s Macroglobulinemia. Blood. 2016; 128:1321–28. https://doi.org/10.1182/blood-2016-04-711234 [PubMed]
- 98. Kapoor P, Ansell SM, Fonseca R, Chanan-Khan A, Kyle RA, Kumar SK, Mikhael JR, Witzig TE, Mauermann M, Dispenzieri A, Ailawadhi S, Stewart AK, Lacy MQ, et al. Diagnosis and Management of Waldenström Macroglobulinemia: Mayo Stratification of Macroglobulinemia and Risk-Adapted Therapy (mSMART) Guidelines 2016. JAMA Oncol. 2017; 3:1257–65. https://doi.org/10.1001/jamaoncol.2016.5763 [PubMed]
- 99. Treon SP, Tripsas CK, Meid K, Warren D, Varma G, Green R, Argyropoulos KV, Yang G, Cao Y, Xu L, Patterson CJ, Rodig S, Zehnder JL, et al. Ibrutinib in previously treated Waldenström’s macroglobulinemia. N Engl J Med. 2015; 372:1430–40. https://doi.org/10.1056/NEJMoa1501548 [PubMed]
- 100. Dimopoulos MA, Trotman J, Tedeschi A, Matous JV, Macdonald D, Tam C, Tournilhac O, Ma S, Oriol A, Heffner LT, Shustik C, García-Sanz R, Cornell RF, et al, and iNNOVATE Study Group and the European Consortium for Waldenström’s Macroglobulinemia. Ibrutinib for patients with rituximab-refractory Waldenström’s macroglobulinaemia (iNNOVATE): an open-label substudy of an international, multicentre, phase 3 trial. Lancet Oncol. 2017; 18:241–50. https://doi.org/10.1016/S1470-2045(16)30632-5 [PubMed]
- 101. Dimopoulos MA, Tedeschi A, Trotman J, García-Sanz R, Macdonald D, Leblond V, Mahe B, Herbaux C, Tam C, Orsucci L, Palomba ML, Matous JV, Shustik C, et al, and iNNOVATE Study Group and the European Consortium for Waldenström’s Macroglobulinemia. Phase 3 Trial of Ibrutinib plus Rituximab in Waldenström’s Macroglobulinemia. N Engl J Med. 2018; 378:2399–410. https://doi.org/10.1056/NEJMoa1802917 [PubMed]
- 102. Treon SP, Hanzis C, Manning RJ, Ioakimidis L, Patterson CJ, Hunter ZR, Sheehy P, Turnbull B. Maintenance Rituximab is associated with improved clinical outcome in rituximab naïve patients with Waldenstrom Macroglobulinaemia who respond to a rituximab-containing regimen. Br J Haematol. 2011; 154:357–62. https://doi.org/10.1111/j.1365-2141.2011.08750.x [PubMed]
- 103. Gavriatopoulou M, Musto P, Caers J, Merlini G, Kastritis E, van de Donk N, Gay F, Hegenbart U, Hajek R, Zweegman S, Bruno B, Straka C, Dimopoulos MA, et al. European myeloma network recommendations on diagnosis and management of patients with rare plasma cell dyscrasias. Leukemia. 2018; 32:1883–98. https://doi.org/10.1038/s41375-018-0209-7 [PubMed]
- 104. Moulopoulos LA, Dimopoulos MA, Weber D, Fuller L, Libshitz HI, Alexanian R. Magnetic resonance imaging in the staging of solitary plasmacytoma of bone. J Clin Oncol. 1993; 11:1311–15. https://doi.org/10.1200/JCO.1993.11.7.1311 [PubMed]
- 105. Dimopoulos MA, Gertz MA, Kastritis E, Garcia-Sanz R, Kimby EK, Leblond V, Fermand JP, Merlini G, Morel P, Morra E, Ocio EM, Owen R, Ghobrial IM, et al. Update on treatment recommendations from the Fourth International Workshop on Waldenstrom’s Macroglobulinemia. J Clin Oncol. 2009; 27:120–26. https://doi.org/10.1200/JCO.2008.17.7865 [PubMed]
- 106. Leblond V, Johnson S, Chevret S, Copplestone A, Rule S, Tournilhac O, Seymour JF, Patmore RD, Wright D, Morel P, Dilhuydy MS, Willoughby S, Dartigeas C, et al. Results of a randomized trial of chlorambucil versus fludarabine for patients with untreated Waldenström macroglobulinemia, marginal zone lymphoma, or lymphoplasmacytic lymphoma. J Clin Oncol. 2013; 31:301–07. https://doi.org/10.1200/JCO.2012.44.7920 [PubMed]
- 107. Castillo JJ, Gustine J, Meid K, Dubeau T, Hunter ZR, Treon SP. Histological transformation to diffuse large B-cell lymphoma in patients with Waldenström macroglobulinemia. Am J Hematol. 2016; 91:1032–35. https://doi.org/10.1002/ajh.24477 [PubMed]
- 108. Leleu X, Soumerai J, Roccaro A, Hatjiharissi E, Hunter ZR, Manning R, Ciccarelli BT, Sacco A, Ioakimidis L, Adamia S, Moreau AS, Patterson CJ, Ghobrial IM, Treon SP. Increased incidence of transformation and myelodysplasia/acute leukemia in patients with Waldenström macroglobulinemia treated with nucleoside analogs. J Clin Oncol. 2009; 27:250–55. https://doi.org/10.1200/JCO.2007.15.1530 [PubMed]
- 109. Castillo JJ, Olszewski AJ, Hunter ZR, Kanan S, Meid K, Treon SP. Incidence of secondary malignancies among patients with Waldenström macroglobulinemia: an analysis of the SEER database. Cancer. 2015; 121:2230–36. https://doi.org/10.1002/cncr.29334 [PubMed]
- 110. Merlini G, Stone MJ. Dangerous small B-cell clones. Blood. 2006; 108:2520–30. https://doi.org/10.1182/blood-2006-03-001164 [PubMed]
- 111. Palladini G, Comenzo RL. The challenge of systemic immunoglobulin light-chain amyloidosis (Al). Subcell Biochem. 2012; 65:609–42. https://doi.org/10.1007/978-94-007-5416-4_22 [PubMed]
- 112. Kyle RA, Therneau TM, Rajkumar SV, Larson DR, Plevak MF, Offord JR, Dispenzieri A, Katzmann JA, Melton LJ
3rd . Prevalence of monoclonal gammopathy of undetermined significance. N Engl J Med. 2006; 354:1362–69. https://doi.org/10.1056/NEJMoa054494 [PubMed] - 113. Blumenthal HT. Amyloidosis: a universal disease of aging? J Gerontol A Biol Sci Med Sci. 2004; 59:361–69. https://doi.org/10.1093/gerona/59.4.M361 [PubMed]
- 114. Nuvolone M, Milani P, Palladini G, Merlini G. Management of the elderly patient with AL amyloidosis. Eur J Intern Med. 2018; 58:48–56. https://doi.org/10.1016/j.ejim.2018.05.004 [PubMed]
- 115. Palladini G, Merlini G. What is new in diagnosis and management of light chain amyloidosis? Blood. 2016; 128:159–68. https://doi.org/10.1182/blood-2016-01-629790 [PubMed]
- 116. Kumar S, Dispenzieri A, Lacy MQ, Hayman SR, Buadi FK, Colby C, Laumann K, Zeldenrust SR, Leung N, Dingli D, Greipp PR, Lust JA, Russell SJ, et al. Revised prognostic staging system for light chain amyloidosis incorporating cardiac biomarkers and serum free light chain measurements. J Clin Oncol. 2012; 30:989–95. https://doi.org/10.1200/JCO.2011.38.5724 [PubMed]
- 117. Palladini G, Foli A, Milani P, Russo P, Albertini R, Lavatelli F, Obici L, Perlini S, Moratti R, Merlini G. Best use of cardiac biomarkers in patients with AL amyloidosis and renal failure. Am J Hematol. 2012; 87:465–71. https://doi.org/10.1002/ajh.23141 [PubMed]
- 118. Sachchithanantham S, Offer M, Venner C, Mahmood SA, Foard D, Rannigan L, Lane T, Gillmore JD, Lachmann HJ, Hawkins PN, Wechalekar AD. Clinical profile and treatment outcome of older (>75 years) patients with systemic AL amyloidosis. Haematologica. 2015; 100:1469–76. https://doi.org/10.3324/haematol.2015.128025 [PubMed]
- 119. Palladini G, Hegenbart U, Milani P, Kimmich C, Foli A, Ho AD, Vidus Rosin M, Albertini R, Moratti R, Merlini G, Schönland S. A staging system for renal outcome and early markers of renal response to chemotherapy in AL amyloidosis. Blood. 2014; 124:2325–32. https://doi.org/10.1182/blood-2014-04-570010 [PubMed]
- 120. Geller HI, Singh A, Mirto TM, Padera R, Mitchell R, Laubach JP, Falk RH. Prevalence of Monoclonal Gammopathy in Wild-Type Transthyretin Amyloidosis. Mayo Clin Proc. 2017; 92:1800–05. https://doi.org/10.1016/j.mayocp.2017.09.016 [PubMed]
- 121. Grogan M, Scott CG, Kyle RA, Zeldenrust SR, Gertz MA, Lin G, Klarich KW, Miller WL, Maleszewski JJ, Dispenzieri A. Natural History of Wild-Type Transthyretin Cardiac Amyloidosis and Risk Stratification Using a Novel Staging System. J Am Coll Cardiol. 2016; 68:1014–20. https://doi.org/10.1016/j.jacc.2016.06.033 [PubMed]
- 122. Lachmann HJ, Booth DR, Booth SE, Bybee A, Gilbertson JA, Gillmore JD, Pepys MB, Hawkins PN. Misdiagnosis of hereditary amyloidosis as AL (primary) amyloidosis. N Engl J Med. 2002; 346:1786–91. https://doi.org/10.1056/NEJMoa013354 [PubMed]
- 123. Terrier B, Jaccard A, Harousseau JL, Delarue R, Tournilhac O, Hunault-Berger M, Hamidou M, Dantal J, Bernard M, Grosbois B, Morel P, Coiteux V, Gisserot O, et al. The clinical spectrum of IgM-related amyloidosis: a French nationwide retrospective study of 72 patients. Medicine (Baltimore). 2008; 87:99–109. https://doi.org/10.1097/MD.0b13e31816c43b6 [PubMed]
- 124. Palladini G, Merlini G. Diagnostic challenges of amyloidosis in Waldenström macroglobulinemia. Clin Lymphoma Myeloma Leuk. 2013; 13:244–46. https://doi.org/10.1016/j.clml.2013.02.001 [PubMed]
- 125. Palladini G, Dispenzieri A, Gertz MA, Kumar S, Wechalekar A, Hawkins PN, Schönland S, Hegenbart U, Comenzo R, Kastritis E, Dimopoulos MA, Jaccard A, Klersy C, Merlini G. New criteria for response to treatment in immunoglobulin light chain amyloidosis based on free light chain measurement and cardiac biomarkers: impact on survival outcomes. J Clin Oncol. 2012; 30:4541–49. https://doi.org/10.1200/JCO.2011.37.7614 [PubMed]
- 126. Kanapuru BS, Myers A, Beaver JA, Kwitkowski VE, Farrell AT, et al. Enrollment of older adults in clinical trials and evaluating paeints with hematologic malignancies - the Food and Drug Adminstration (FDA) experience [abstract]. Blood. 2017; 130:861.
- 127. Seldin DC, Anderson JJ, Skinner M, Malek K, Wright DG, Quillen K, Finn K, Oran B, Sanchorawala V. Successful treatment of AL amyloidosis with high-dose melphalan and autologous stem cell transplantation in patients over age 65. Blood. 2006; 108:3945–47. https://doi.org/10.1182/blood-2006-06-029728 [PubMed]
- 128. Sitia R, Palladini G, Merlini G. Bortezomib in the treatment of AL amyloidosis: targeted therapy? Haematologica. 2007; 92:1302–07. https://doi.org/10.3324/haematol.12136 [PubMed]
- 129. Palladini G, Milani P, Foli A, Obici L, Lavatelli F, Nuvolone M, Caccialanza R, Perlini S, Merlini G. Oral melphalan and dexamethasone grants extended survival with minimal toxicity in AL amyloidosis: long-term results of a risk-adapted approach. Haematologica. 2014; 99:743–50. https://doi.org/10.3324/haematol.2013.095463 [PubMed]
- 130. Venner CP, Lane T, Foard D, Rannigan L, Gibbs SD, Pinney JH, Whelan CJ, Lachmann HJ, Gillmore JD, Hawkins PN, Wechalekar AD. Cyclophosphamide, bortezomib, and dexamethasone therapy in AL amyloidosis is associated with high clonal response rates and prolonged progression-free survival. Blood. 2012; 119:4387–90. https://doi.org/10.1182/blood-2011-10-388462 [PubMed]
- 131. Mikhael JR, Schuster SR, Jimenez-Zepeda VH, Bello N, Spong J, Reeder CB, Stewart AK, Bergsagel PL, Fonseca R. Cyclophosphamide-bortezomib-dexamethasone (CyBorD) produces rapid and complete hematologic response in patients with AL amyloidosis. Blood. 2012; 119:4391–94. https://doi.org/10.1182/blood-2011-11-390930 [PubMed]
- 132. Merlini G, Sachorawala V, Jeffrey ZA., Kukreti V, Schoenland SO, Jaccard A, et al. Long-Term Outcome of a Phase 1 Study of the Investigational Oral Protea-some Inhibitor (PI) Ixazomib at the Recommended Phase 3 Dose (RP3D) in Patients (Pts) with Relapsed or Refractory Systemic Light-Chain (AL) Amyloidosis (RRAL) [abstract]. Blood. 2014; 124:3450.
- 133. Palladini G, Russo P, Foli A, Milani P, Lavatelli F, Obici L, Nuvolone M, Brugnatelli S, Invernizzi R, Merlini G. Salvage therapy with lenalidomide and dexamethasone in patients with advanced AL amyloidosis refractory to melphalan, bortezomib, and thalidomide. Ann Hematol. 2012; 91:89–92. https://doi.org/10.1007/s00277-011-1244-x [PubMed]
- 134. Dispenzieri A, Buadi F, Laumann K, LaPlant B, Hayman SR, Kumar SK, Dingli D, Zeldenrust SR, Mikhael JR, Hall R, Rajkumar SV, Reeder C, Fonseca R, et al. Activity of pomalidomide in patients with immunoglobulin light-chain amyloidosis. Blood. 2012; 119:5397–404. https://doi.org/10.1182/blood-2012-02-413161 [PubMed]
- 135. Kastritis E, Terpos E, Roussou M, Gavriatopoulou M, Pamboukas C, Boletis I, Marinaki S, Apostolou T, Nikitas N, Gkortzolidis G, Michalis E, Delimpasi S, Dimopoulos MA. A phase 1/2 study of lenalidomide with low-dose oral cyclophosphamide and low-dose dexamethasone (RdC) in AL amyloidosis. Blood. 2012; 119:5384–90. https://doi.org/10.1182/blood-2011-12-396903 [PubMed]
- 136. Kaufman GP, Schrier SL, Lafayette RA, Arai S, Witteles RM, Liedtke M. Daratumumab yields rapid and deep hematologic responses in patients with heavily pretreated AL amyloidosis. Blood. 2017; 130:900–02. https://doi.org/10.1182/blood-2017-01-763599 [PubMed]
- 137. Edwards CV, Langer AL, Mapara MY, Radhakrishman J. Maurer MS et al. Final analysis of the phase 1a/b study of chimeric fibril-reactive monoclonal antibody 11-1F4 in patients with relapsed or refractory AL amyloidosis. ASH 59th annual meeting and exposition; Atlanta: Blood 2017.
- 138. Richards DB, Cookson LM, Barton SV, Liefaard L, Lane T, Hutt DF, Ritter JM, Fontana M, Moon JC, Gillmore JD, Wechalekar A, Hawkins PN, Pepys MB. Repeat doses of antibody to serum amyloid P component clear amyloid deposits in patients with systemic amyloidosis. Sci Transl Med. 2018; 10:eaan3128. https://doi.org/10.1126/scitranslmed.aan3128 [PubMed]
- 139. Gertz MA, Landau H, Comenzo RL, Seldin D, Weiss B, Zonder J, Merlini G, Schönland S, Walling J, Kinney GG, Koller M, Schenk DB, Guthrie SD, Liedtke M. First-in-Human Phase I/II Study of NEOD001 in Patients With Light Chain Amyloidosis and Persistent Organ Dysfunction. J Clin Oncol. 2016; 34:1097–103. https://doi.org/10.1200/JCO.2015.63.6530 [PubMed]
- 140. Merlini G. AL amyloidosis: from molecular mechanisms to targeted therapies. Hematology (Am Soc Hematol Educ Program). 2017; 2017:1–12. [PubMed]
- 141. Palladini G, Milani P, Foli A, Basset M, Russo F, Perlini S, Merlini G. Presentation and outcome with second-line treatment in AL amyloidosis previously sensitive to nontransplant therapies. Blood. 2018; 131:525–32. https://doi.org/10.1182/blood-2017-04-780544 [PubMed]
- 142. Larocca A, Palumbo A. Optimizing Treatment for Elderly Patients With Newly Diagnosed Multiple Myeloma: A Personalized Approach. J Clin Oncol. 2016; 34:3600–04. https://doi.org/10.1200/JCO.2016.68.6113 [PubMed]
- 143. Rubinow A, Skinner M, Cohen AS. Digoxin sensitivity in amyloid cardiomyopathy. Circulation. 1981; 63:1285–88. https://doi.org/10.1161/01.CIR.63.6.1285 [PubMed]
- 144. Gertz MA, Falk RH, Skinner M, Cohen AS, Kyle RA. Worsening of congestive heart failure in amyloid heart disease treated by calcium channel-blocking agents. Am J Cardiol. 1985; 55:1645. https://doi.org/10.1016/0002-9149(85)90995-6 [PubMed]
- 145. Kristen AV, Dengler TJ, Hegenbart U, Schonland SO, Goldschmidt H, Sack FU, Voss F, Becker R, Katus HA, Bauer A. Prophylactic implantation of cardioverter-defibrillator in patients with severe cardiac amyloidosis and high risk for sudden cardiac death. Heart Rhythm. 2008; 5:235–40. https://doi.org/10.1016/j.hrthm.2007.10.016 [PubMed]
- 146. Cerrato C, Mina R, Palumbo A. Optimal management of elderly patients with myeloma. Expert Rev Anticancer Ther. 2014; 14:217–28. https://doi.org/10.1586/14737140.2014.856269 [PubMed]



