Introduction
Primary liver cancer is estimated to be the sixth most common cancer worldwide and the second main reason behind cancer mortality. With Hepatocellular Carcinoma (HCC) making up to 80% of its cases, the incidence of liver cancer varies significantly among continents and countries. 75% of all cases are recorded in Asia alone, with China standing for more than 50% of all patients diagnosed [1,2]. Gender, geographic location and ethnicity also affect the incidence rates (with male patients, and Asian populations followed by Hispanics having the highest incidence). Risk factors for HCC also differ among countries. For example, hepatitis B virus (HBV) and aflatoxin B1 (AFB1) are considered as major factors in Asian and African countries [1].
China stands for almost 22% of new cancer cases and 27% of cancer mortality worldwide. The growth and aging of the Chinese population have led to a continuous increase in cancer cases diagnosed [3]. The high incidence of liver cancer in China makes up to 55% of all cases in the world, with HCC ranking as the second major cause in cancer-mortality in males and the third in females. It is the most common type of cancer in Chinese patients under the age of 60 years, with a low incidence before the age of 35 that rises significantly after that age to reach a peak at 80-84 years with higher mortality rates in male patients [3–5].
Since 70% of HBV cases in high incidence areas are infected in perinatal stages or early childhood, the efforts to prevent HBV via vaccination programs in infants and children have shown a huge impact on the incidence rates of HBV (and HBV-related HCC) in targeted age groups. Although vaccinations may be ineffective in almost 5% of cases, still, a decrease of almost 70-85% of HBV-related HCC incidence has been recorded in Asia, and a 95% decrease in HCC related deaths has been recorded in Chinese patients younger than 19 years [1,3].
The success of HCC treatment is associated with early diagnosis. However, in many patients, the diagnosis is only established in advanced stages when a curative treatment is not an available option anymore. Therefore, advanced screening programs have been developed in many Asian countries such as Japan, South Korea, and China. Guidelines suggested that ultrasonography and measurements of blood levels of α-fetoprotein (AFP), AFP-L3, and des-γ-carboxyprothrombin (DCP) can be useful in screening for early stages’ HCC patients [4,6].
In the light of the continuous growing and aging of populations worldwide, and the significant developments of treatment options and disease control strategies, longer life expectancy has been achieved for many diseases, which translated into growing numbers of elderly patients. That created a continuous change in the cut-off of age groups in general and elderly patients in particular. Increased numbers of patients over 70 and 75 years have been recorded, with average life expectancy exceeding 80 years in some countries like South Korea. Therefore, the age cut-off of 80 years for elderly patients group has been accepted and proposed in many reports [2,7 ,8].
The process of aging can be defined as a continuous loss of physiological and biological functions of human organs that increases susceptibility for diseases and injuries. Many theories have been proposed to explain the aging effects on human cells, tissues, and organs. As for the liver, aging can affect both the function and structure of this organ. Changes in metabolic functions, synthesizing proteins abilities, sinusoids and kupffer cells, liver volume and blood flow have already been linked to aging in several reports [8–11].
One theory that has been proposed is that metabolic errors that stand behind the process of aging can actually be caused by both environmental and genetic factors, which may explain the differences among individuals and populations in the susceptibility and the speed of aging [10]. In general, genome alterations (such as DNA damage, mutations, and epi-mutations) are well-known reasons for diseases like cancer; however, they are usually monitored and fixed by the DNA repairing system that preserves the integrity of the genome [12]. Many studies tried to figure out the genetic connection between cancer and the process of aging, considering that the incidence of cancer increases significantly with age. Although cancer is associated with cellular “gain of function“ while aging is defined as a “loss of function“; however, the two have been considered as results of accumulation of cellular damages. Cancers are associated with accumulated mutations in the genome prior to any phenotype changes. Such accumulations have also been linked to aging, considering the time needed for those mutations to happen. However, such a connection has been challenged with findings which suggest that up to 50% of somatic mutations occur in early life of the human body, followed by decreased rate of mutations afterwards. Yet, the changing in tissue context with age can explain the high susceptibility and increased selection for oncogenic cells in advanced age. Another contradiction between aging and cancer is the chromosomal telomeres. The maintenance of telomeres is considered as a hallmark of cancer, while in aged cells short telomeres are a reason for ending the replication of cells. However, studies have found that cancers can indeed arise from short telomeres’ cells, but telomerase activation is still essential for the progression of cancers in the body [11,13–15].
HCC in children is a topic with limited data that requires greater attention and efforts to be explored. Liver cancer is considered to be the third common type of abdominal cancers in children, with HCC ranking second in common liver cancer types (making up to 23% of all cases). However, the high chemoresistance and low cure rates associated with HCC make it an important target for further investigation and research. The majority of pediatric HCC cases occur in patients older than 10 years but the rarity of this disease in childhood makes it very challenging to collect enough cases to carry a wide investigation. Therefore, most of the studies available are retrospective analyses with limited data [16–19].
The majority of pediatric HCC cases lack other background liver diseases such as cirrhosis. Yet, a stronger correlation with HBV infection has been found in childhood HCC, which translated into a significant decrease in incidence following the HBV vaccination programs in infants and children in Asia and the world. That created an urgent need to understand the role of other possible mechanisms of childhood HCC such as mutations and genetic alterations, which are still poorly investigated and addressed [16–22].
Recently, several genes and pathways have been investigated for a potential role in the development of HCC to clarify the genetic background of the disease, one of which is AKR1B10. Located at 7q33, AKR1B10 contains 10 exons and 9 introns, with a length of 13.8 kb and several putative oncogenic and tumor suppressor protein binding sites in its promoter [23]. The detoxification enzyme AKR1B10 is a well-known aldo-keto reductase family member that has been linked to hepatocellular carcinoma and proposed as a new possible biomarker and an independent risk factor [24–26]. In general, the NAD(P)H-dependent oxidoreductase catalyzes carbonyl compounds and reduces xenobiotic substrates by converting highly reactive aldehydic and ketonic groups into hydroxy groups, which is a form of cell protection against carbonyl toxicity. AKR1B10 is also linked to retinoid metabolism and fatty acid synthesis [23,24,27]. AKR1B10 helps in converting retinals to retinols instead of retinoic acids which are considered to be important anti-neoplastic signaling molecules [25,28,29]. Therefore, it is known for its role in intracellular detoxification, cellular proliferation and carcinogenesis [23,30]. Clinically, studies have found that higher levels of AKR1B10 expression are associated with HBV, HCV, and liver cirrhosis, which may suggest that the expression may be stimulated by the presence of such pre-HCC conditions, with a potential role in early hepatocarcinogenesis [24,26,27]. However, it was also found that high AKR1B10 expression may indicate lower risks of recurrence in HBV-related HCC patients after curative resection [29]. On another note, the relation between AKR1B10 expression and other clinical aspects like tumor size, lesions number, microvascular invasion, and metastasis is yet to be fully determined [24,29]. Yet, such a relation with tumor grade has been a subject for several studies and a correlation between AKR1B10 expression and lower tumor stage has been found which may make AKR1B10 a possible marker for HCC differentiation level [24,28,30]. Several studies have also concentrated on the links between AKR1B10 expression and drug metabolism, which is extremely important in some therapeutic approaches like chemotherapy. AKRs in general are considered as metabolizing enzymes for carbonyl-containing drugs; therefore, the family of enzymes has been investigated for a potential involvement in chemoresistance [24,28]. Studies showed that AKR1B10 in specific is linked to drug resistance [31] and its knockdown experiments showed better sensitivity to chemotherapy [28].
Although current studies and researches are devoting greater efforts to uncover more information regarding HCC both clinically and genetically, only few studies are addressing HCC in specific age groups like children and elderly patients, and even fewer studies are exploring this topic from a genetic point of view. Therefore, we, in this study, are shedding more lights on HCC patients by analyzing both their clinical and genetic characteristics in age-specific manner, in order to further investigate the differences among age groups of HCC patients and evaluate the need to address such differences in the diagnosis, treatment, and prevention strategies of HCC.
Results
Clinical analysis
Analyzing the clinical variables showed a domination of male patients in both children and elderly groups. However, a higher percentage was recorded in elderly patients. The children group had higher prevalence of HBV positive history than the elderly group, but the sample size of elderly group was much larger. Relevant clinical variables in both groups are summarized in (Table 1).
Table 1. A summary of relevant clinical variables in both children and elderly patients that shows the characteristics and differences between the two groups.
| Characteristics | Children Group (n=33) | Elderly Group (n=98) | P value | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N (%) | N (%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gender | Male | 26 (79) | 77 (92) | 0.979 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Female | 7 (21) | 21 (8) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age (y) | Range | 10~20 | 80~92 | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mean | 16.03±2.92 | 82.49±2.48 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HBV History * | (+) | 14 (42.42) | 38 (39.17) | 0.742 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| (-) | 19 (57.58) | 59 (60.82) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Over-all Survival (OS) (%)** | (1 Year) | 21 (67.74) (n=31) | 84 (92.31) (n=91) | 0.002 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mortality (%)** | (1 Year) | 10 (32.26) (n=31) | 7 (7.69) (n=91) | 0.002 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tumor Lesions | Single | 26 (79) | 82 (84) | 0.523 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Multiple | 7 (21) | 16 (16) | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tumor Size (cm) | Range | 1.2~18 | 1~15 | 0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Mean | 8.73±4.82 | 5.51±3.12 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| * Data for one case in elderly group were lost (n=97). | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ** Data for some cases were lost, the total numbers of cases available for each group are shown in the table. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The elderly group of patients had better early survival rates (1 year after the surgery) compared to children group. In children group, younger patients (≤15 years old) had decreased survival rates when compared to older children (≥15y) (survival rates were 50% in younger children compared to 76.19% in older children). In the elderly group, survival rates for patients ≤85y were 92.41%, compared to 91.67% in patients ≥86. Survival curves of both groups show that elderly patients have better early survival than children group (Figure 1).
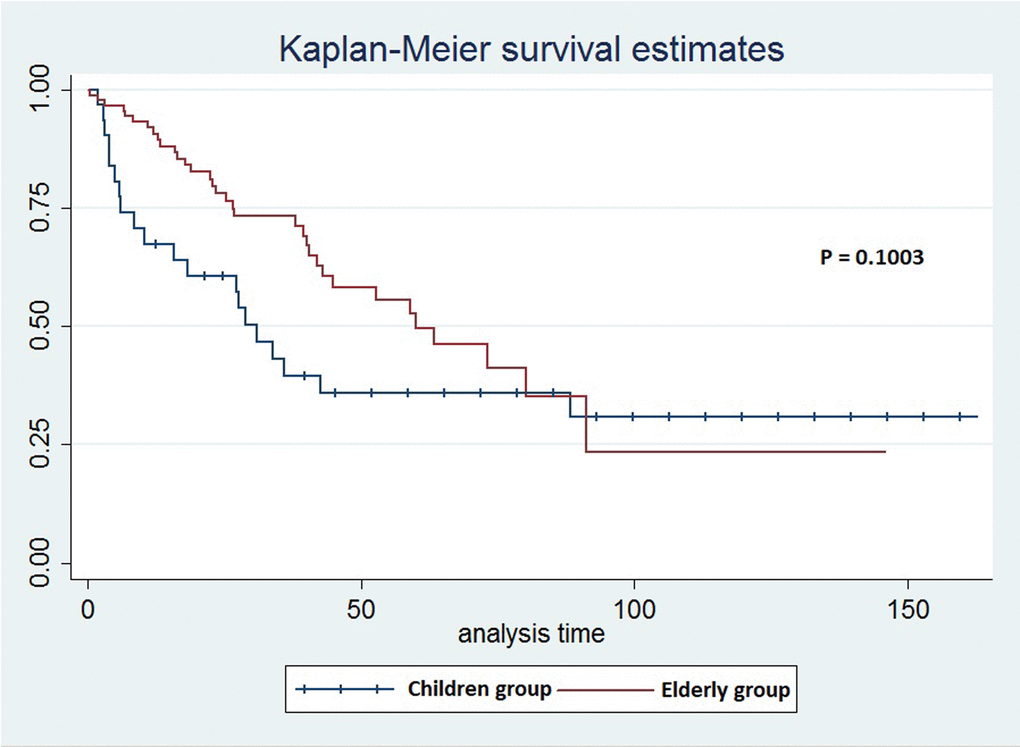
Figure 1. The survival curves for children and elderly groups of patients in the clinical analysis.
Single lesion tumors were more common than multiple lesions in both groups with a slightly higher rate in elderly patients. However, the average tumor size was bigger in children compared to elderly patients with a difference of more than 3 cm (P=0.001)
As expected, the previous results showed that factors like tumor size are associated with increased risks of mortality in both groups. Tumor size larger than 5 cm was associated with 39.13% mortality rate in children group compared to 12.5% in patients with tumor size ≤ 5 cm (P=0.172). As in elderly group, the difference was statistically significant (P=0.018) with tumors larger than 5 cm being associated with 15.39% mortality rate compared to only 1.93% in ≤ 5 cm tumors. The age influence on the survival of patients with tumors larger than 5 cm was also significant (P=0.036), with elderly patients having a 84.61% 1 year survival rate compared to 60.87% in children.
Genetic and mRNA alterations
365 patients from TCGA database [32,33] were included in the analysis of genetic alterations, all diagnosed with HCC liver cancer. Male patients were 247 (67.7%) and female patients were 118 (32.3%). All ethnicities were included in the analysis. The children group included 5 patients aged 16 to 20 years, with 2 Asian patients and 3 Caucasians. The elderly group included 14 patients aged 80 to 90 years, with a majority of Caucasian patients. Three aspects of genetic alterations were considered in our study: gene mutations, copy number variants (CNVs), and mRNA expression.
Genes mutations
Data of all patients in both children and elderly groups were checked for gene mutations. In children group, only three genes (BIRC6, NRXN1, and ZNF676) were found to be mutated in more than one patient (each gene was mutated in two cases). Due to the presence of mutation patterns in patients, the mutual exclusivity between genes was also investigated and only one pair of genes (NRXN1/ ZNF676) showed significant relation (P=0.008). Same method was carried in the elderly group and 14 genes were found to be mutated in at least three patients of this group. The genes are: ALB, FRAS1, TP53, TTN, CACNA1E, CCDC141, CTNNB1, DNAJC28, KMT2D, LRP2, MSLN, NBAS, RP1, and TEP1. Among those genes, three (TP53, CTNNB1, and KMT2D) were associated with driver mutations. Of which, driver mutations of TP53 and CTNNB1 were present simultaneously with mRNA expression alterations. Mutation rates of previous genes in all age groups are shown in (Table 2), while driver mutation rates are shown in (Table 3).
Table 2. All age groups mutation rates of the genes mutated in children and elderly groups.
| Gene | All Patients (%) (N=365) | Group ≤20 (%) (N=5) | Group 21~40 (%) (N=26) | Group 41~60 (%) (N=145) | Group 61~79 (%) (N=175) | Group 80≤ (%) (N=14) |
| TP53 | 30.96 | 0 | 30.77 | 36.55 | 27.43 | 28.57 |
| TTN | 27.39 | 0 | 23.07 | 22.06 | 33.14 | 28.57 |
| CTNNB1 | 26.57 | 0 | 26.92 | 22.06 | 31.42 | 21.42 |
| ALB | 11.23 | 0 | 7.69 | 6.89 | 13.71 | 35.71 |
| CACNA1E | 8.49 | 0 | 3.84 | 4.83 | 11.43 | 21.42 |
| FRAS1 | 6.85 | 0 | 7.69 | 3.45 | 8 | 28.57 |
| KMT2D | 5.75 | 20 | 7.69 | 4.14 | 5.14 | 21.42 |
| BIRC6 | 5.48 | 40 | 7.69 | 2.76 | 6.29 | 7.14 |
| LRP2 | 4.93 | 0 | 3.84 | 4.14 | 4.57 | 21.42 |
| RP1 | 3.56 | 0 | 0 | 1.38 | 4.57 | 21.42 |
| NBAS | 3.28 | 0 | 3.84 | 2.07 | 2.85 | 21.42 |
| TEP1 | 3.28 | 0 | 3.84 | 1.38 | 3.42 | 21.42 |
| CCDC141 | 2.46 | 0 | 0 | 2.76 | 1.14 | 21.42 |
| NRXN1 | 2.19 | 40 | 3.85 | 3.44 | 0 | 0 |
| ZNF676 | 1.91 | 40 | 0 | 0 | 2.86 | 0 |
| DNAJC28 | 1.09 | 0 | 3.84 | 0 | 0 | 21.42 |
| MSLN | 1.09 | 0 | 0 | 0.69 | 0 | 21.42 |
Table 3. All age groups driver mutation rates of the three genes found with driver mutations in children and elderly groups.
| Gene | All Patients (%) (N=365) | Group ≤20 (%) (N=5) | Group 21~40 (%) (N=26) | Group 41~60 (%) (N=145) | Group 61~79 (%) (N=175) | Group 80≤ (%) (N=14) |
| CTNNB1 | 24.65 | 0 | 23.07 | 20 | 29.71 | 21.43 |
| TP53 | 12.6 | 0 | 7.69 | 11.03 | 14.85 | 14.28 |
| KMT2D | 2.19 | 0 | 3.84 | 1.38 | 2.28 | 7.14 |
Copy Number Variants (CNVs)
All patients in children group were checked for CNVs and 6 genes were found altered in more than one patient. The genes are AHCTF1, CNST, KIF26B, SCCPDH, SMYD3, and TFB2M. Alterations included both deletions and amplifications. All six genes shared the same cyto-band (1q44) and were shared by the same two patients with a significant tendency towards co-occurrence among all genes (P<0.001). The alteration rates of the genes in all age groups were 40% for children group (≤20y), 11.54% for (21-40y) group, 7.58% for (41-60y) group, 9.71% for (41-60y) group, and 7.14% for elderly group (≥80y). In the elderly group, 299 genes were altered in three or more patients. Of those genes, 170 were presented in five patients (35.7%), all of which were deletions associated with cyto-bands: 8p21.2 , 8p21.3 , 8p22 , 8p23.1 , 8p23.2 , and 8p23.3. 62 genes were presented in four patients (28.57%), deletions, on cyto-bands: 8p12 , 8p21.1 , 8p21.2, 8p23.1, 8p23.2, and 8p23.3. Genes presented in three patients (21.43%) included 37 amplified genes (associated with cyto-bands: 6p21.1, 6p22.3, 6p24.1, 6p25.2, 6p25.3, and 17q25.3) and 30 deletions (cyto-bands: 6p12.3, 8p11.22, 8p11.23 , 8p12).
mRNA expression
25 genes that are commonly over-expressed in HCC patients [34] were selected to be analyzed. The genes are: GPC3, LCN2, SPP1, UBE2C, PTTG1, SFN, MDK, UBE2T, CCNB1, AKR1B10, NDUFA4L2, NT5DC2, PLVAP, G6PD, PDZK1IP1, CENPW, SPARCL1, SPINK1, UBD, THY1, PTP4A3, TK1, TACC3, GMNN, and STMN1. Expression patterns of all 25 genes in TCGA liver cancer patients along with tumor type and the diagnosis age of patients are shown in (Figure 2) [32,33]. The overall survival (OS) analysis showed that patients with alterations in selected genes expression levels had lower survival rates than those cases without alterations, the differences were statically significant (P=0.0011) (Figure 3) [32,33]. A further investigation of mRNA expression patterns in children group showed that 8 genes were found over-expressed in patients from this group: one patient had both GPC3 and UBD upregulated, UBE2C and UBE2T in one patient, PLVAP and THY1 in one patient, and SPARCL1 and PTP4A3 in one patient. Based on those results, the four pairs of genes were investigated for mutual exclusivity or correlation in expression patterns. Two pairs (UBE2C / UBE2T and PLVAP / THY1) were found to have significant tendency towards co-occurrence (P<0.001).
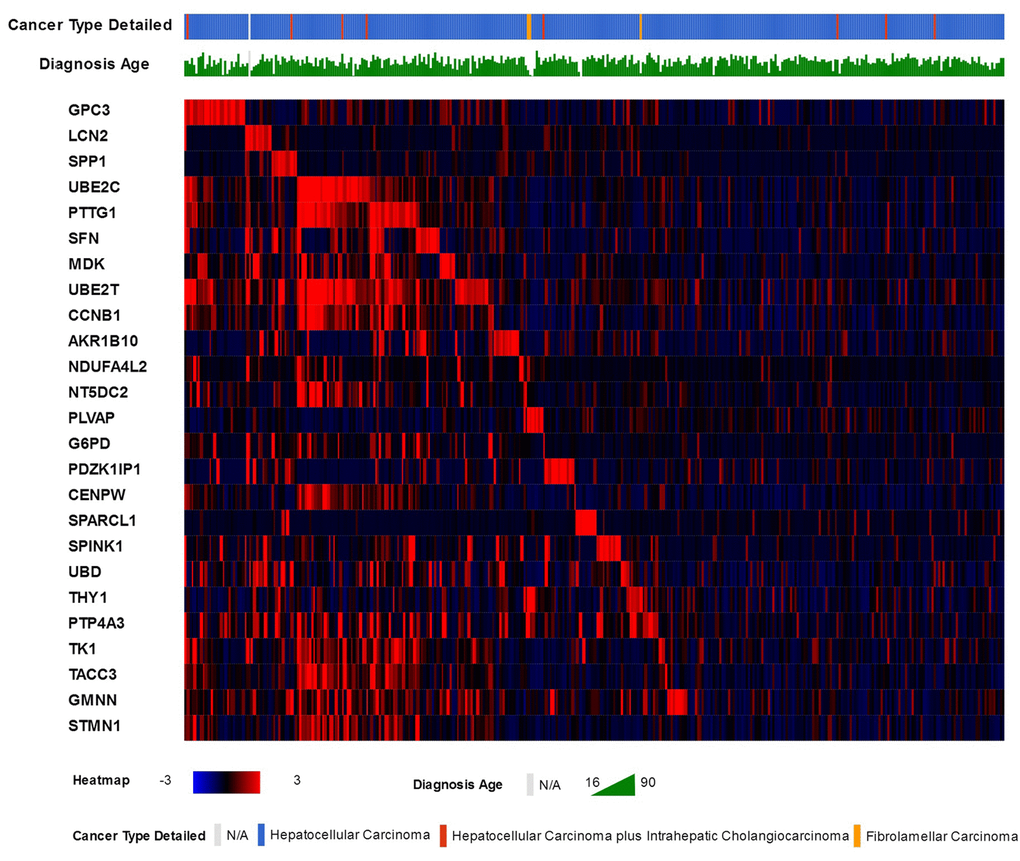
Figure 2. The 25 commonly over-expressed genes in HCC patients and the heat-map of their expression patterns in TCGA liver cancer patients, along with patients liver cancer type and age of diagnosis.
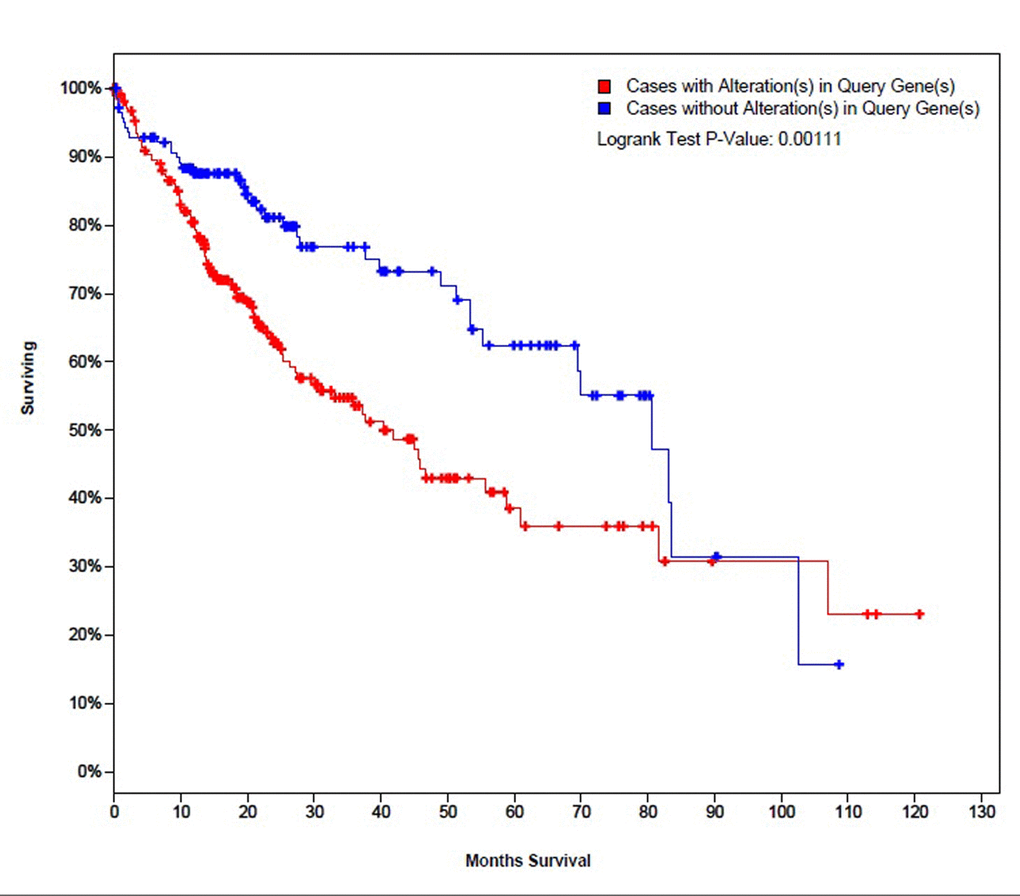
Figure 3. The survival curves of TCGA liver cancer patients with and without alterations in mRNA expression patterns of the selected 25 genes.
As for the elderly group, 12 genes were over-expressed in patients: GPC3, LCN2, SFN, MDK, AKR1B10, PLVAP, G6PD, PDZK1IP1, SPINK1, UBD, PTP4A3, and TACC3. SFN had the highest rate of alterations in this group (21.42%) followed by GPC3 and AKR1B10 (14.28% each). After analyzing the expression patterns in each patient of this group, 4 couples of genes were found to have significant tendency towards co-occurrence: MDK / TACC3, SFN / G6PD, SFN / AKR1B10 (P<0.001), and SFN / UBD (P=0.025). The alteration rates of expression of all 25 genes in all groups are shown in (Table 4). The results showed that AKR1B10 up-regulation rates increased significantly with the age of patients; therefore, we selected this gene as a subject for a further IHC analysis to investigate the relation between its expression levels and the age of patients.
Table 4. The alteration rates of mRNA expression of the selected 25 genes in the five age groups of the genetic analysis.
| Gene | All Patients (%) (N=365) | Group ≤20 (%) (N=5) | Group 21~40 (%) (N=26) | Group 41~60 (%) (N=145) | Group 61~79 (%) (N=175) | Group 80≤ (%) (N=14) |
| UBE2T | 16.99 | 20 | 23.08 | 17.93 | 16.57 | 0 |
| PTTG1 | 11.51 | 0 | 11.54 | 12.41 | 7.43 | 0 |
| PTP4A3 | 11.51 | 20 | 11.54 | 13.1 | 10.28 | 7.14 |
| UBE2C | 9.59 | 20 | 11.54 | 12.41 | 7.43 | 0 |
| UBD | 9.04 | 20 | 7.69 | 10.34 | 8.57 | 7.14 |
| GMNN | 8.77 | 0 | 15.38 | 12.41 | 5.71 | 0 |
| TK1 | 7.73 | 0 | 7.69 | 8.96 | 7.43 | 0 |
| CCNB1 | 7.39 | 0 | 3.85 | 7.59 | 8.57 | 0 |
| GPC3 | 7.12 | 20 | 15.38 | 7.59 | 4.57 | 14.28 |
| AKR1B10 | 7.12 | 0 | 3.85 | 5.52 | 8.57 | 14.28 |
| PDZK1IP1 | 7.12 | 0 | 0 | 7.59 | 8 | 7.14 |
| SPINK1 | 7.12 | 0 | 3.85 | 7.59 | 7.43 | 7.14 |
| SFN | 6.85 | 0 | 15.38 | 5.52 | 5.71 | 21.42 |
| MDK | 6.85 | 0 | 7.69 | 8.27 | 5.71 | 7.14 |
| NT5DC2 | 6.57 | 0 | 7.69 | 9.65 | 4.57 | 0 |
| TACC3 | 4.93 | 0 | 3.85 | 6.89 | 3.43 | 7.14 |
| STMN1 | 4.93 | 0 | 3.85 | 6.89 | 4 | 0 |
| G6PD | 4.11 | 0 | 3.85 | 4.83 | 3.43 | 7.14 |
| CENPW | 4.11 | 0 | 7.69 | 4.14 | 4 | 0 |
| NDUFA4L2 | 3.56 | 0 | 7.69 | 3.45 | 3.43 | 0 |
| LCN2 | 3.29 | 0 | 3.85 | 4.83 | 1.71 | 7.14 |
| SPP1 | 3.29 | 0 | 3.85 | 2.76 | 4 | 0 |
| SPARCL1 | 3.01 | 20 | 3.85 | 1.38 | 4 | 0 |
| THY1 | 3.01 | 20 | 0 | 3.45 | 2.86 | 0 |
| PLVAP | 1.64 | 20 | 3.85 | 0.69 | 1.14 | 7.14 |
AKR1B10 expression
According to TCGA database and the pathology atlas of the human cancer transcriptome, the overall survival (OS) and disease free survival (DFS) analyses showed that patients with normal AKR1B10 expression levels have better survival rates than those with over-expression (Figure 4) [32,33,35]. Therefore, and based on our previous results, we chose to carry a further IHC analysis of AKR1B10 to investigate a potential relationship between its expression levels and the age of patients. Representative images of AKR1B10 immunohistochemistry staining in tested HCC tissues are provided in (Figure 5). Samples from 280 patients (280 cancer tissue samples and 168 near-tumor samples) were collected and tested. All relevant clinical data are summarized in Table 5.
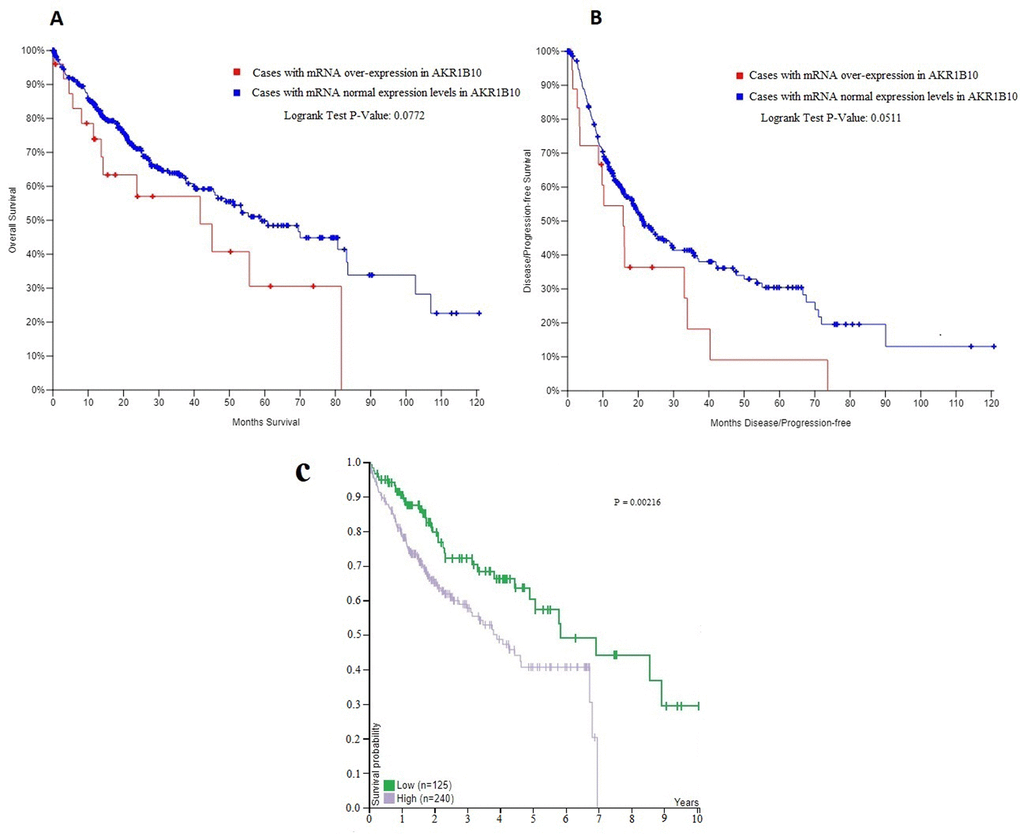
Figure 4. (A, B) The overall survival (OS) and disease free survival (DFS) curves in patients with and without mRNA over-expression in AKR1B10. (C) The correlation between AKR1B10 mRNA expression level in liver cancer and patient survival from the pathology atlas of the human cancer transcriptome .Corresponding expression cutoff= 26.8 FPKM. 5-year survival for patients with high expression= 41% , 5-year survival for patients with low expression= 57%, and log-rank P value = 0.00216. URL: https://www.proteinatlas.org/ENSG00000198074-AKR1B10/pathology/tissue/liver+cancer#ihc.
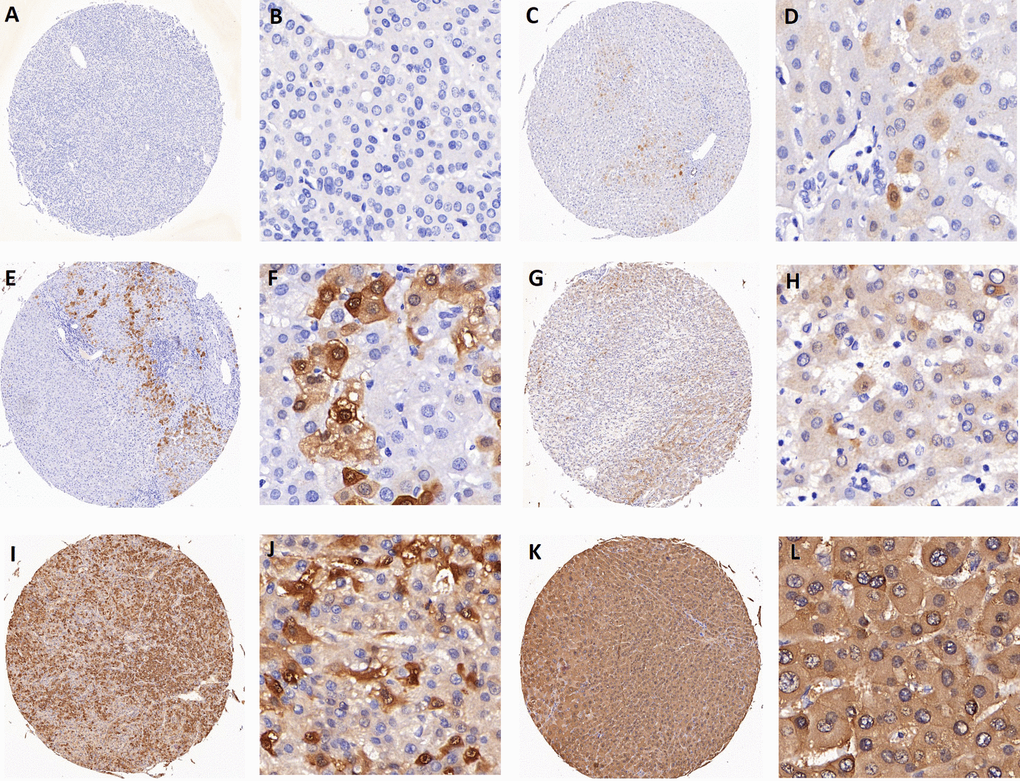
Figure 5. Representative images of AKR1B10 immunohistochemistry staining in tissues. (A-B) represent negative staining (score: 0). (C-D) (score: 1), (E-F) (score: 2), (G-H) (score: 4) all represent weak staining. (I-J) (score: 6) and (K-L) (score: 8) represent strong staining. Scores 0 to 4 are categorized as low expression of AKR1B10, while scores 6 to 8 are considered as high expression.
Table 5. A summary of relevant clinical data of 280 patients in AKR1B10 IHC expression analysis.
| N (%) (280) | Low expression 141 (50.36) | High expression 139 (49.64) | P | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GENDER (M/F) | 120/21 | 123/16 | 0.403 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| HBsAg (+/-) | 117/24 | 116/23 | 0.915 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Cirrhosis (+/-) | 85/56 | 85/54 | 0.882 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| MVI * (181) (+/-) | 41/50 | 36/54 | 0.492 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BCLC (0+A/B+C) | 53/88 | 60/79 | 0.342 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tumor Differentiation * (278) (I+II/III+IV) | 89/50 | 97/42 | 0.308 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AFP (Normal/ Elevated) | 47/94 | 58/81 | 0.147 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CEA * (181) (Normal/ Elevated) | 84/7 | 82/8 | 0.770 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CA19-9 * (181) (Normal/ Elevated) | 68/23 | 71/19 | 0.507 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Number of lesions (single/multiple) | 117/24 | 108/31 | 0.266 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tumor Size (<=5/>5) | 74/67 | 80/59 | 0.394 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Encapsulation (yes/no) | 78/63 | 78/61 | 0.893 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tumor thrombus (yes/no) | 26/115 | 32/107 | 0.344 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ALT (Normal/ Elevated) | 90/51 | 79/60 | 0.232 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| AST * (181) (Normal/ Elevated) | 62/29 | 57/33 | 0.496 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ALP * (181) (Normal/ Elevated) | 62/29 | 64/26 | 0.663 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| GGT (Normal/ Elevated) | 61/80 | 45/94 | 0.06 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TB * (273) (Normal/ Elevated) | 123/16 | 119/15 | 0.934 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| DB * (174) (Normal/ Elevated) | 82/7 | 77/8 | 0.716 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ALB * (271) (Normal/ Elevated) | 8/130 | 9/124 | 0.742 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| * represents the total number of patients data are available for. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
The results showed statistically significant differences between expression levels in HCC tissues and near-tumor tissues (P<0.001), with a high expression rate of 49.64% in HCC samples compared to only 6.58% in near-tumor samples (Figure 6A). We also found that expression levels differed significantly (P=0.043) at the age of 40 (Figure 6B), with a rate of 30.77% of high expression in patients younger than 40 compared to 51.57% in older patients.
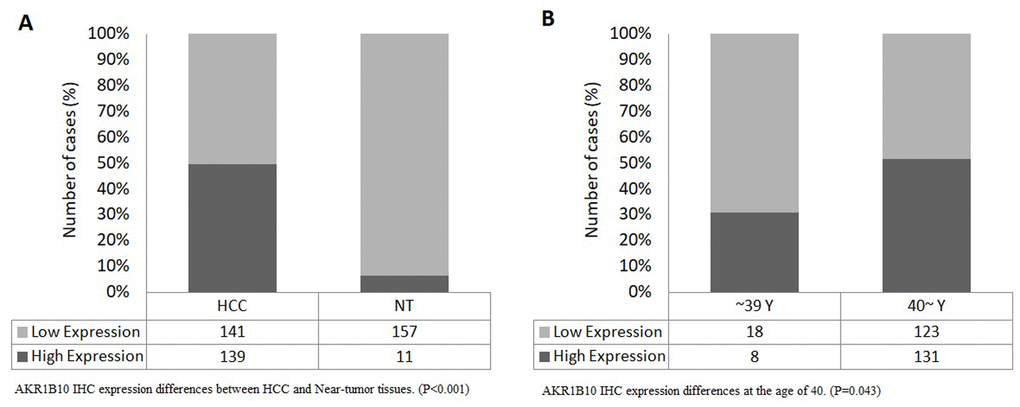
Figure 6. (A) AKR1B10 IHC expression differences between HCC and Near-tumor tissues. (P<0.001), (B) AKR1B10 IHC expression differences at the age of 40. (P=0.043).
Discussion
Our knowledge regarding HCC has been continuously expanding. Currently, more studies have been addressing new aspects such as HCC genetic background and novel treatments. Many genes have been explored and linked to a certain role in the development or treatments of HCC. However, only few efforts are dedicated to understand the differences and characteristics of HCC in certain groups like elderly patients or children, and even fewer studies are exploring the genetic features specific to them. Therefore, we carried, in this study, a clinical and genetic (gene mutations, CNVs, mRNA expression) analysis of elderly and young HCC patients, in order to contribute to the limited knowledge regarding this topic and help provide a solid base for further investigations.
Considering the growth and aging of populations all over the world (especially in China) and the rarity of HCC in certain age groups like children, we chose the age of 20 and 80 years as cut-off points for the young and elderly groups of patients.
In the clinical analysis, the sample size of the elderly group was larger than the children group due to the rarity of HCC in young patients after the national program of HBV vaccination in infants and children [17]. The majority of patients in both groups were males with a higher percentage in elderly group (92%) compared to (79%) in young patients. A possible explanation could be that several risk factors for HCC (like alcohol consuming and chronic HBV history) are more common in older males [9].
In general, the process of aging and the changes it brings can explain a lot of differences among elderly patients and other age groups. The accumulation of molecular abnormalities with time, the age-related alteration in immune functions and other changes have already been pointed out for their association with cancer morbidity and mortality [36]. However, when compared to children group, the elderly group in our study showed higher rates of early survival. The better liver functions in elderly patients and the heavier burden of the tumor on young livers can lead to such differences [8]. Besides, the tumor is unlikely to grow too large without leading to organ failure or death in earlier ages [9]. The differences between age subgroups within each group also indicated the impact of age on survival of patients, with older children and younger elderly having better survival rates than other patients in their groups. However, the effect of aging itself on survival rates and the role of other causes of death in elderly group prevent us from drawing a solid conclusion, and a further investigation with better controlled data is advised.
As expected, our results showed that tumor size and lesions number can affect the survival in both groups. The majority in both groups was presented with single lesions. The average tumor size, however, was bigger in children which translated into increased risk of death; an increase that can be expected considering the smaller size of liver in younger patients.
The number of studies addressing the genetic aspect of HCC is rapidly growing, expanding our knowledge of related genes and their role in diagnosis and treatment. However, the majority of studies are age-standardized rather than age-specific, leaving doctors and care givers with an only choice of applying the finding of those studies in special age groups like children and ignoring the potential differences related to the age of patients. In children, the family history can be taken into consideration; however, for De novo alterations such a history is not useful. Therefore, doctors tend to check for common alterations, considering the lack of age-specific data. As for elderly patients, aging has been investigated for its role in cancer development in general (such as the accumulation of alterations and the compromising of DNA repair system) [12,13]. However, the current knowledge in this field is still falling behind when compared to age-standardized data, and further efforts are required to optimize the diagnosis and treatment plans. Therefore, we carried an age-specific genetic analysis that covers three main aspects: gene mutations, copy number variants (CNVs), and mRNA expression patterns.
The analysis of gene mutation patterns in children group showed that only three genes were mutated in more than one patient (BIRC6, NRXN1, and ZNF676), with NRXN1 and ZNF676 showing a significant tendency towards co-occurrence. Although such findings could indicate a role of certain genes in young HCC patients; however, the limited number of patients in this group makes it hard to draw a conclusion, and further research is recommended. The protein encoded by BIRC6 is known for its role in inhibiting cell’s apoptosis, while proteins encoded by NRXN1 acts as adhesion molecules in cells of the nervous system [37–39]. For the elderly group, 14 genes were mutated in more than three cases (21.42%). ALB had the highest rate with (35.71%). The majority of genes were presented with passenger mutations except for TP53, CTNNB1, and KMT2D having driver mutations. Of which, TP53 and CTNNB1 mutations were associated with mRNA expression irregularities, which emphasize their role in cancer development. Mutations in these three genes have already been observed in different types of cancers [37–39]. As for TP53, P53 pathway is known for its association with aging and diseases development in advanced ages since P53 functions include suppressing tumors, preserving genome integrity, and restricting the proliferation of damages cells [40]. Genes like CCDC141, DNAJC28, and MSLN showed higher prevalence in elderly patients than other age groups, while the incidence of mutations in genes like CACNA1E, LRP2, and RP1 increased with the age of patients. Those mutation patterns show that the process of aging plays a different role in different age groups, with those genes being a potential key to uncover such a role. Therefore, searching for age-specific alterations is vital to optimize the care we give to patients of specific ages, like elderly patients.
Copy number variants (CNVs) of DNA sections can overlap with certain genes and, therefore, alter their functions, leading to diseases like cancer [12]. Six genes were found to be altered in more than one patient in children group. The significant tendency of co-occurrence among all six genes (P<0.001) led us to investigate the location of those genes on human chromosomes. All six genes shared the same cyto-band: 1q44; therefore, we suggest this location to be further investigated for a relation with HCC in all patients in general (since 9.31% of all patients had an alteration in this cyto-band) and children in specific. 1q44 has been linked in other studies to abnormalities like microcephaly, seizures, and other diseases [41]. More genes were found in the elderly group (299 genes). The majority of them were located in chromosome 6 and 8 in cyto-bands like: 6p21-25, 8p11-12, and 8p21-23. Abnormalities in some of those locations like 8p21-23 have already been associated with several cancers like lung, breast, colon, brain, prostate, and even liver cancer [42], and the deletion of 8p has been proposed as a prognostic predictor in HCC patients [43]. The importance of such findings is that it gets us closer to be able to achieve a diagnosis based not only on phenotypic or genetic changes but also karyotypical changes, which may save both efforts and resources.
Out of 25 genes that were considered for mRNA expression analysis, 8 were up-regulated in children group. Among those genes, three (PLVAP, SPARCL1, and THY1) were associated with low rates of up-regulation in other age groups. Therefore, a primary consideration of those genes in age-specific diagnosis is highly advised for further investigation. THY1 gene encodes a protein that is associated with cell adhesion and communication. The gene is also involved in tumor suppressing, metastasis, and prognosis of many cancer types like gallbladder and nasopharyngeal carcinoma [37–39,44]. PLVAP has also been investigated as a therapeutic target in HCC [45]. mRNA analysis also showed a tendency towards co-occurrence in two pairs of genes: (UBE2C / UBE2T and PLVAP / THY1). A further investigation of the mechanisms and the roles of the given genes could help uncover age-specific patterns of alterations. For example, both UBE2C and UBE2T encode ubiquitin-conjugating enzymes that are needed in ubiquitination and cell cycle progression [37–39]. The genes have already been linked to several types of malignancies such as bladder cancer, malignant glioma, and Fanconi anemia [46–49].
12 genes were up-regulated in elderly patients, with SFN, GPC3, AKR1B10 having the highest rates (21.42%, 14.28%, 14.28%, respectively). Comparing this group’s rates to other age groups’, we found that up-regulated patterns for genes like PLVAP and AKR1B10 are more common in elderly patients, which may indicate a certain relation with advanced age. The same could be considered for genes like PDZK1IP1 that was found to be up-regulated only in patients older than 40 years. Out of the 12 genes selected in elderly group, AKR1B10 stood out as its up-regulation rates increased significantly with the age of patients. As mentioned earlier, this gene has already been linked to cell proliferation and differentiation and addressed in HCC and other types of tumors like lung and breast cancer [49]. Among the four pairs of genes found to be significantly correlated, (SFN / G6PD) and (SFN / AKR1B10) could be of a certain use in future age-specific approaches, considering their high prevalence in elderly patients and stronger relation to aging.
Based on the previous findings, we selected (AKR1B10) as a subject for a further immunohistochemistry (IHC) testing in order to analyze the influence of its expression levels in HCC. Our results showed statistically significant differences between AKR1B10 expression levels in HCC tissues and near-tumor tissues (P<0.001), with a high expression rate of 49.64% in HCC samples compared to only 6.58% in near-tumor samples. Those findings are in line with other studies in which high expression levels of AKR1B10 have been linked to HCC while expression levels in near-tumor tissues, normal tissues, and even benign liver lesions showed minimal to no expression levels of the gene, which makes AKR1B10 a potential biomarker for the diagnosis of HCC [27,28,50].
Another finding of our study is that AKR1B10 expression levels showed a significant difference at the age of 40 (P=0.043), with a high expression rate of 30.77% in patients younger than 40 compared to 51.57% in older patients. These results validate and emphasize the proposed relationship between AKR1B10 expression and the age of patients. Due to the rarity of HCC in patients of certain ages (especially younger than 20 years old), an independent comparison of patients younger than 20y and older than 80y was hard to be carried.
As we mentioned earlier, the promoter region of AKR1B10 includes several oncogenic and tumor suppressor protein binding sites, one of which is TP53 tumor suppressor [23,24,29]. Based on the previous results, the percentage of TP53 driver mutations was much higher after the age of 40. In addition, AKR1B10 was found to be significantly correlated with SFN regarding mRNA expression levels with high prevalence in elderly patients. SFN is one of the genes involved in G2 cell cycle arrest pathway that is regulated by TP53. The genes involved in this pathway and their interactions are shown in (Figure 7) [51]. All of that suggest a potential involvement of this specific pathway and other similar ones in the role AKR1B10 plays in different age groups of HCC patients.
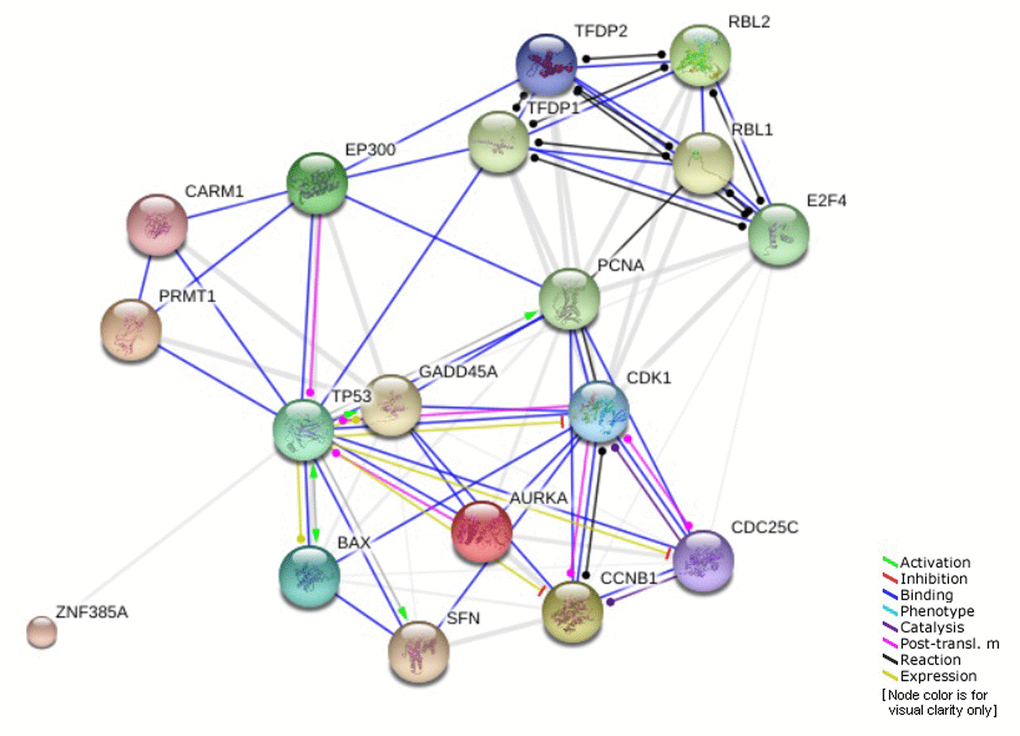
Figure 7. Genes involved in G2 cell cycle arrest pathway regulated by TP53.
The transcription factor nuclear factor erythroid 2-related factor (Nrf2) is known for its involvement in the regulation of AKR1B10 in addition to its role in the adoptive response to oxidative stress and damage [25,52], which is considered as one leading theory of aging [53]. Oxidative stress refers to the accumulation of reactive oxygen species (ROS) and reactive nitrogen species (RNS) in cells, exceeding the cellular antioxidant capacity, and therefore, damaging cellular DNA, lipids, and proteins [54,55]. Levels of accumulation increase with age, leading to a significant decline in cellular functions and repairing systems (including DNA repairing system) [53]. In general, hepatic cells have a high antioxidant ability when compared to other types of human cells; however, due to the functions of hepatic cells, accumulation of oxidative damage is quite easy to happen [56]. A study has shown that, with age, hepatic cells show a significant increase in protein carbonylation, lipid perioxidation, in addition to a decrease in antioxidant enzymes activities [57], which may easily explain how genes with a mechanism like AKR1B10 and their high expression can be involved in the increase of carcinogenesis with age.
The effect of genetic alterations and profile on the phenotype or clinical aspects of cancer patients is very important. However, genetic alterations or signatures in general can fall into three groups: Predictive, functional, or phenotypical alterations [58,59]. Therefore, passenger mutations can also be extremely useful for early screening and diagnosis of HCC which could enhance the success of treatments offered. We still encourage future studies and researches to explore possible relations between the genotype and phenotype of HCC patients in better controlled study designs.
Like all other researches in this field, we were faced with certain limitations in this study. The limited number of available data and patients (especially in children group) made it hard to draw definite conclusions based on the results. It also decreased our ability to control the conditions of the analysis to the level we wanted or find patients with certain level of similarity. The rarity of this disease in some age groups forced us to carry the analysis retrospectively. In addition, we decided to include patients from all ethnicities in the genetic analysis, considering the lack of relevant data. Such limitations are often encountered in studies like ours, which sometimes may translate into different results among different studies. However, we still believe that the contribution of this study to the limited data in this field and the results we found (especially the genetic findings) can indeed create a strong base for future researches and studies to uncover more age-specific abnormalities and carry deeper analyses in targeted age groups. Hopefully, similar future studies can optimize the diagnosis and treatment plans offered to young and elderly HCC patients in both Asian and global populations.
Materials and Methods
Patients
Different groups of patients were selected for each part of this study:
For the clinical analysis, data from Department of Liver Surgery, Zhongshan Hospital, Fudan University were reviewed for the selection of suitable patients. From 2000 to 2015, a total of 131 patients’ data were selected to be retrospectively analyzed. All patients were i) ≤20 or ≥80 years old (in order to include sufficient number of patients, considering the rarity of HCC in younger populations and the increased numbers of HCC patients older than 80y in Asia and China [2,7,8], ii) pathologically diagnosed with HCC liver cancer, and iii) underwent hepatectomy in Zhongshan Hospital, Fudan University. Patients were then divided into two groups according to their age at the time of surgery: children group (20 years old or younger, 33 patients) and elderly group (80 years old or older, 98 patients). Relevant clinical variables and information including age, gender, HBV status, tumor size, lesions number, and date of surgery were collected and analyzed. Survival periods of patients were calculated starting from the date of surgery according to follow-up results in 2016. The study was approved by the Ethics Committee of Zhongshan Hospital, Fudan University and carried in accordance with the Helsinki declaration.
For genetic characteristics, a total of 365 HCC patients (from all age groups and ethnicities) were selected from TCGA database to be genetically analyzed and compared, with the major attention directed to two groups in specific: childhood patients (≤20y, n=5) and elderly patients (≥80,n=14). Other age groups include: (21-40y,n=26), (41-60y,n=145), and (61-79y, n=175). The genetic analysis included three different aspects: genes mutations, copy number variants (CNV), and mRNA expression levels.
For the further investigation of AKR1B10, data and tissue samples from 280 patients (280 cancer tissue samples and 168 near-tumor samples) were collected from Department of Liver Surgery, Zhongshan Hospital, Fudan University. All patients were i) pathologically diagnosed with HCC liver cancer ii) underwent hepatectomy in our hospital between 2005 -2010. Immunohistochemistry was then performed and samples were divided into high expression and low expression groups.
Genetic and mRNA alterations
To address the limitations and lack of genetic data on HCC childhood and elderly patients worldwide, we based the genetic analysis in our study on TCGA global database for liver cancer. Platforms like cBioPortal (http://www.cbioportal.org) and UALCAN (http://ualcan.path.uab.edu/index.html) were used to collect and analyze relevant data. cBioPortal is a platform that helps download, analyze and visualize genetic data from large scale cancer databases like TCGA database [32,33]. UALCAN is another platform that provides gene expression information and survival analyses regarding different types of tumors [34].
Alterations from three different aspects were considered: genes mutations, copy number variants (CNV), and mRNA expression levels.
For genes mutations analysis, mutated genes in childhood and elderly groups were analyzed. Mutated genes shared by two or more patients in childhood patients group, or by three or more patients in elderly patients group were selected for further investigation. Mutation rates of selected genes were calculated in all age groups and the mutual exclusivity were determined. Driver mutations were then selected and analyzed to figure any possible connections to mRNA expression alterations.
For copy number variants (CNVs), CNVs shared among two or more patients in childhood patients group, or three or more patients in elderly patients group were selected. The CNVs were then assigned to their locations on human chromosomes and analyzed for insights on possible age-related variants.
As for the mRNA expression, 25 commonly over-expressed genes in HCC patients [34] were selected to be investigated. The expression patterns were then analyzed in all 365 patients and the effects of expression patterns on overall survivals (OS) were determined and visualized. Alteration rates in different age groups were then compared, to understand the age influence on expression patterns. Patients in childhood and elderly groups were individually analyzed and genes mutual over-expression exclusivity were determined to spot any age-relevant significant connections that can aid in identifying potential genes in the given age groups.
Immunohistochemistry (IHC)
Tissue samples were paraffin embedded; therefore, deparaffinization and then rehydration of samples in descending alcohol (ethanol) gradient were carried out first. Peroxidase activities were then blocked by using 3% hydrogen peroxide for 10 min. Samples were placed in 0.1 M citrate buffer and heated for 8 min in a microwave to induce antigen epitope retrieval. After that, 10% goat serum was used before the incubation of samples with primary antibody overnight at 4°C temperature, followed by secondary antibody for 30 min at room temperature. Finally, staining of samples was completed by using 3,3'-diaminobenzidineto visualize the results.
Staining results were assessed by two different doctors separately. The extent of staining was graded on a scale from 0 to 4 (0%, 1~5%, 6~25%, 26~75%, 76~100%) while a scale from 0 to 2 was used for intensity (0: negative, 1: weak, 2: strong). Final scores (0 to 8) were calculated (extent × intensity) and divided into two groups: low level of expression (0~4) and high level of expression (6~8).
Statistical analysis
Statistical analysis was performed with STATA software (13.0, College Station, Texas 77845 USA) and SPSS software (21.0, IBM corp.). Variables were presented as means ±standard deviations (SD) (for continuous variables) or frequencies (%) (for categorical variables). Overall survival (OS) rates were analyzed in accordance with Kaplan-Meier’s method and the log-rank test. P values were considered statistically significant when <0.05.
Conflicts of Interest
All authors declare that they have no competing interests.
Funding
This work was supported by grants from the National Natural Science Foundation of China (81472672). The study was also funded by the National Key Reseach and Development Program of China (2017YFC0908402 and 2017YFC1308604).
References
- 1. McGlynn KA, Petrick JL, London WT. Global epidemiology of hepatocellular carcinoma: an emphasis on demographic and regional variability. Clin Liver Dis. 2015; 19:223–38. https://doi.org/10.1016/j.cld.2015.01.001 [PubMed]
- 2. Nozawa A, Kubo S, Takemura S, Sakata C, Urata Y, Nishioka T, Kinoshita M, Hamano G, Uenishi T, Suehiro S. Hepatic resection for hepatocellular carcinoma in super-elderly patients aged 80 years and older in the first decade of the 21st century. Surg Today. 2015; 45:851–57. https://doi.org/10.1007/s00595-014-0994-1 [PubMed]
- 3. Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F, Jemal A, Yu XQ, He J. Cancer statistics in China, 2015. CA Cancer J Clin. 2016; 66:115–32. https://doi.org/10.3322/caac.21338 [PubMed]
- 4. Song P, Feng X, Zhang K, Song T, Ma K, Kokudo N, Dong J, Yao L, Tang W. Screening for and surveillance of high-risk patients with HBV-related chronic liver disease: promoting the early detection of hepatocellular carcinoma in China. Biosci Trends. 2013; 7:1–6. [PubMed]
- 5. Fang JY, Wu KS, Zeng Y, Tang WR, Du PL, Xu ZX, Xu XL, Luo JY, Lin K. Liver cancer mortality characteristics and trends in China from 1991 to 2012. Asian Pac J Cancer Prev. 2015; 16:1959–64. https://doi.org/10.7314/APJCP.2015.16.5.1959 [PubMed]
- 6. Chen JG, Zhang SW. Liver cancer epidemic in China: past, present and future. Semin Cancer Biol. 2011; 21:59–69. https://doi.org/10.1016/j.semcancer.2010.11.002 [PubMed]
- 7. Kim YJ, Jang BK, Kim ES, Chung WJ, Park KS, Cho KB, Hwang JS. Hepatocellular carcinoma in the elderly: clinical characteristics, treatment, survival analysis in Korean patients older than 70 years. J Korean Med Sci. 2012; 27:1147–54. https://doi.org/10.3346/jkms.2012.27.10.1147 [PubMed]
- 8. Oishi K, Itamoto T, Kohashi T, Matsugu Y, Nakahara H, Kitamoto M. Safety of hepatectomy for elderly patients with hepatocellular carcinoma. World J Gastroenterol. 2014; 20:15028–36. https://doi.org/10.3748/wjg.v20.i41.15028 [PubMed]
- 9. Fan C, Jin G, Sun C, Ma D, Chen C, Qiao P, Wu D. Clinical characteristics, treatment patterns and survival outcome of hepatocellular carcinoma patients aged 70 years or older: a single-center retrospective study from China. Aging Clin Exp Res. 2014; 26:123–30. https://doi.org/10.1007/s40520-013-0142-6 [PubMed]
- 10. Moskalev AA, Aliper AM, Smit-McBride Z, Buzdin A, Zhavoronkov A. Genetics and epigenetics of aging and longevity. Cell Cycle. 2014; 13:1063–77. https://doi.org/10.4161/cc.28433 [PubMed]
- 11. López-Otín C, Blasco MA, Partridge L, Serrano M, Kroemer G. The hallmarks of aging. Cell. 2013; 153:1194–217. https://doi.org/10.1016/j.cell.2013.05.039 [PubMed]
- 12. Vijg J, Suh Y. Genome instability and aging. Annu Rev Physiol. 2013; 75:645–68. https://doi.org/10.1146/annurev-physiol-030212-183715 [PubMed]
- 13. de Magalhães JP. How ageing processes influence cancer. Nat Rev Cancer. 2013; 13:357–65. https://doi.org/10.1038/nrc3497 [PubMed]
- 14. Bernardes de Jesus B, Blasco MA. Telomerase at the intersection of cancer and aging. Trends Genet. 2013; 29:513–20. https://doi.org/10.1016/j.tig.2013.06.007 [PubMed]
- 15. Rozhok AI, DeGregori J. The evolution of lifespan and age-dependent cancer risk. Trends Cancer. 2016; 2:552–60. https://doi.org/10.1016/j.trecan.2016.09.004 [PubMed]
- 16. Allan BJ, Wang B, Davis JS, Parikh PP, Perez EA, Neville HL, Sola JE. A review of 218 pediatric cases of hepatocellular carcinoma. J Pediatr Surg. 2014; 49:166–71. https://doi.org/10.1016/j.jpedsurg.2013.09.050 [PubMed]
- 17. Zhang XF, Liu XM, Wei T, Liu C, Li MX, Long ZD, Lv Y. Clinical characteristics and outcome of hepatocellular carcinoma in children and adolescents. Pediatr Surg Int. 2013; 29:763–70. https://doi.org/10.1007/s00383-013-3334-4 [PubMed]
- 18. Kelly D, Sharif K, Brown RM, Morland B. Hepatocellular carcinoma in children. Clin Liver Dis. 2015; 19:433–47. https://doi.org/10.1016/j.cld.2015.01.010 [PubMed]
- 19. Tanaka Y, Inoue T, Horie H. International pediatric liver cancer pathological classification: current trend. Int J Clin Oncol. 2013; 18:946–54. https://doi.org/10.1007/s10147-013-0624-8 [PubMed]
- 20. Hung GY, Horng JL, Yen HJ, Lee CY, Lin LY. Changing incidence patterns of hepatocellular carcinoma among age groups in Taiwan. J Hepatol. 2015; 63:1390–96. https://doi.org/10.1016/j.jhep.2015.07.032 [PubMed]
- 21. Spector LG, Pankratz N, Marcotte EL. Genetic and nongenetic risk factors for childhood cancer. Pediatr Clin North Am. 2015; 62:11–25. https://doi.org/10.1016/j.pcl.2014.09.013 [PubMed]
- 22. Plon SE, Wheeler DA, Strong LC, Tomlinson GE, Pirics M, Meng Q, Cheung HC, Begin PR, Muzny DM, Lewis L, Biegel JA, Gibbs RA. Identification of genetic susceptibility to childhood cancer through analysis of genes in parallel. Cancer Genet. 2011; 204:19–25. https://doi.org/10.1016/j.cancergencyto.2010.11.001 [PubMed]
- 23. Liu Z, Zhong L, Krishack PA, Robbins S, Cao JX, Zhao Y, Chung S, Cao D. Structure and promoter characterization of aldo-keto reductase family 1 B10 gene. Gene. 2009; 437:39–44. https://doi.org/10.1016/j.gene.2009.02.007 [PubMed]
- 24. Schmitz KJ, Sotiropoulos GC, Baba HA, Schmid KW, Müller D, Paul A, Auer T, Gamerith G, Loeffler-Ragg J. AKR1B10 expression is associated with less aggressive hepatocellular carcinoma: a clinicopathological study of 168 cases. Liver Int. 2011; 31:810–16. https://doi.org/10.1111/j.1478-3231.2011.02511.x [PubMed]
- 25. Sato S, Genda T, Ichida T, Murata A, Tsuzura H, Narita Y, Kanemitsu Y, Ishikawa S, Kikuchi T, Mori M, Hirano K, Iijima K, Wada R, et al. Impact of aldo-keto reductase family 1 member B10 on the risk of hepatitis C virus-related hepatocellular carcinoma. J Gastroenterol Hepatol. 2016; 31:1315–22. https://doi.org/10.1111/jgh.13295 [PubMed]
- 26. Sato S, Genda T, Hirano K, Tsuzura H, Narita Y, Kanemitsu Y, Kikuchi T, Iijima K, Wada R, Ichida T. Up-regulated aldo-keto reductase family 1 member B10 in chronic hepatitis C: association with serum alpha-fetoprotein and hepatocellular carcinoma. Liver Int. 2012; 32:1382–90. https://doi.org/10.1111/j.1478-3231.2012.02827.x [PubMed]
- 27. Tsuzura H, Genda T, Sato S, Murata A, Kanemitsu Y, Narita Y, Ishikawa S, Kikuchi T, Mori M, Hirano K, Iijima K, Wada R, Ichida T. Expression of aldo-keto reductase family 1 member b10 in the early stages of human hepatocarcinogenesis. Int J Mol Sci. 2014; 15:6556–68. https://doi.org/10.3390/ijms15046556 [PubMed]
- 28. Matkowskyj KA, Bai H, Liao J, Zhang W, Li H, Rao S, Omary R, Yang GY. Aldoketoreductase family 1B10 (AKR1B10) as a biomarker to distinguish hepatocellular carcinoma from benign liver lesions. Hum Pathol. 2014; 45:834–43. https://doi.org/10.1016/j.humpath.2013.12.002 [PubMed]
- 29. Wang YY, Qi LN, Zhong JH, Qin HG, Ye JZ, Lu SD, Ma L, Xiang BD, Li LQ, You XM. High expression of AKR1B10 predicts low risk of early tumor recurrence in patients with hepatitis B virus-related hepatocellular carcinoma. Sci Rep. 2017; 7:42199. https://doi.org/10.1038/srep42199 [PubMed]
- 30. Heringlake S, Hofdmann M, Fiebeler A, Manns MP, Schmiegel W, Tannapfel A. Identification and expression analysis of the aldo-ketoreductase1-B10 gene in primary malignant liver tumours. J Hepatol. 2010; 52:220–27. https://doi.org/10.1016/j.jhep.2009.11.005 [PubMed]
- 31. Laffin B, Petrash JM. Expression of the Aldo-Ketoreductases AKR1B1 and AKR1B10 in Human Cancers. Front Pharmacol. 2012; 3:104. https://doi.org/10.3389/fphar.2012.00104 [PubMed]
- 32. Gao J, Aksoy BA, Dogrusoz U, Dresdner G, Gross B, Sumer SO, Sun Y, Jacobsen A, Sinha R, Larsson E, Cerami E, Sander C, Schultz N. Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci Signal. 2013; 6:pl1. https://doi.org/10.1126/scisignal.2004088 [PubMed]
- 33. Cerami E, Gao J, Dogrusoz U, Gross BE, Sumer SO, Aksoy BA, Jacobsen A, Byrne CJ, Heuer ML, Larsson E, Antipin Y, Reva B, Goldberg AP, et al. The cBio cancer genomics portal: an open platform for exploring multidimensional cancer genomics data. Cancer Discov. 2012; 2:401–04. https://doi.org/10.1158/2159-8290.CD-12-0095 [PubMed]
- 34. Chandrashekar DS, Bashel B, Balasubramanya SA, Creighton CJ, Ponce-Rodriguez I, Chakravarthi BV, Varambally S. UALCAN: A portal for facilitating tumor subgroup gene expression and survival analyses. Neoplasia. 2017; 19:649–58. https://doi.org/10.1016/j.neo.2017.05.002 [PubMed]
- 35. Uhlen M, Zhang C, Lee S, Sjöstedt E, Fagerberg L, Bidkhori G, Benfeitas R, Arif M, Liu Z, Edfors F, Sanli K, von Feilitzen K, Oksvold P, et al. A pathology atlas of the human cancer transcriptome. Science. 2017; 357:eaan2507. https://doi.org/10.1126/science.aan2507 [PubMed]
- 36. Pallis AG, Hatse S, Brouwers B, Pawelec G, Falandry C, Wedding U, Lago LD, Repetto L, Ring A, Wildiers H. Evaluating the physiological reserves of older patients with cancer: the value of potential biomarkers of aging? J Geriatr Oncol. 2014; 5:204–18. https://doi.org/10.1016/j.jgo.2013.09.001 [PubMed]
- 37. O’Leary NA, Wright MW, Brister JR, Ciufo S, Haddad D, McVeigh R, Rajput B, Robbertse B, Smith-White B, Ako-Adjei D, Astashyn A, Badretdin A, Bao Y, et al. Reference sequence (RefSeq) database at NCBI: current status, taxonomic expansion, and functional annotation. Nucleic Acids Res. 2016; 44:D733–45. https://doi.org/10.1093/nar/gkv1189 [PubMed]
- 38. Tatusova T, DiCuccio M, Badretdin A, Chetvernin V, Nawrocki EP, Zaslavsky L, Lomsadze A, Pruitt KD, Borodovsky M, Ostell J. NCBI prokaryotic genome annotation pipeline. Nucleic Acids Res. 2016; 44:6614–24. https://doi.org/10.1093/nar/gkw569 [PubMed]
- 39. Brister JR, Ako-Adjei D, Bao Y, Blinkova O. NCBI viral genomes resource. Nucleic Acids Res. 2015; 43:D571–77. https://doi.org/10.1093/nar/gku1207 [PubMed]
- 40. Wu D, Prives C. Relevance of the p53-MDM2 axis to aging. Cell Death Differ. 2018; 25:169–79. https://doi.org/10.1038/cdd.2017.187 [PubMed]
- 41. Raun N, Mailo J, Spinelli E, He X, McAvena S, Brand L, O’Sullivan J, Andersen J, Richer L, Tang-Wai R, Bolduc FV. Quantitative phenotypic and network analysis of 1q44 microdeletion for microcephaly. Am J Med Genet A. 2017; 173:972–77. https://doi.org/10.1002/ajmg.a.38139 [PubMed]
- 42. Wistuba II, Behrens C, Virmani AK, Milchgrub S, Syed S, Lam S, Mackay B, Minna JD, Gazdar AF. Allelic losses at chromosome 8p21-23 are early and frequent events in the pathogenesis of lung cancer. Cancer Res. 1999; 59:1973–79. [PubMed]
- 43. Pang JZ, Qin LX, Ren N, Hei ZY, Ye QH, Jia WD, Sun BS, Lin GL, Liu DY, Liu YK, Tang ZY. Loss of heterozygosity at D8S298 is a predictor for long-term survival of patients with tumor-node-metastasis stage I of hepatocellular carcinoma. Clin Cancer Res. 2007; 13:7363–69. https://doi.org/10.1158/1078-0432.CCR-07-0593 [PubMed]
- 44. Zhang DH, Yang ZL, Zhou EX, Miao XY, Zou Q, Li JH, Liang LF, Zeng GX, Chen SL. Overexpression of Thy1 and ITGA6 is associated with invasion, metastasis and poor prognosis in human gallbladder carcinoma. Oncol Lett. 2016; 12:5136–44. https://doi.org/10.3892/ol.2016.5341 [PubMed]
- 45. Wang YH, Cheng TY, Chen TY, Chang KM, Chuang VP, Kao KJ. Plasmalemmal Vesicle Associated Protein (PLVAP) as a therapeutic target for treatment of hepatocellular carcinoma. BMC Cancer. 2014; 14:815. https://doi.org/10.1186/1471-2407-14-815 [PubMed]
- 46. Ma R, Kang X, Zhang G, Fang F, Du Y, Lv H. High expression of UBE2C is associated with the aggressive progression and poor outcome of malignant glioma. Oncol Lett. 2016; 11:2300–04. https://doi.org/10.3892/ol.2016.4171 [PubMed]
- 47. Gong YQ, Peng D, Ning XH, Yang XY, Li XS, Zhou LQ, Guo YL. UBE2T silencing suppresses proliferation and induces cell cycle arrest and apoptosis in bladder cancer cells. Oncol Lett. 2016; 12:4485–92. https://doi.org/10.3892/ol.2016.5237 [PubMed]
- 48. Hira A, Yoshida K, Sato K, Okuno Y, Shiraishi Y, Chiba K, Tanaka H, Miyano S, Shimamoto A, Tahara H, Ito E, Kojima S, Kurumizaka H, et al. Mutations in the gene encoding the E2 conjugating enzyme UBE2T cause Fanconi anemia. Am J Hum Genet. 2015; 96:1001–07. https://doi.org/10.1016/j.ajhg.2015.04.022 [PubMed]
- 49. Cao Z, Zhou B, Chen X, Huang D, Zhang X, Wang Z, Huang H, Wang Y, Cao D. Statil suppresses cancer cell growth and proliferation by the inhibition of tumor marker AKR1B10. Anticancer Drugs. 2014; 25:930–37. https://doi.org/10.1097/CAD.0000000000000121 [PubMed]
- 50. Sonohara F, Inokawa Y, Hishida M, Kanda M, Nishikawa Y, Yamada S, Fujii T, Sugimoto H, Kodera Y, Nomoto S. Prognostic significance of AKR1B10 gene expression in hepatocellular carcinoma and surrounding non-tumorous liver tissue. Oncol Lett. 2016; 12:4821–28. https://doi.org/10.3892/ol.2016.5240 [PubMed]
- 51. Belinky F, Nativ N, Stelzer G, Zimmerman S, Iny Stein T, Safran M, Lancet D. PathCards: multi-source consolidation of human biological pathways. Database (Oxford). 2015; 2015:bav006. https://doi.org/10.1093/database/bav006 [PubMed]
- 52. Murata A, Genda T, Ichida T, Amano N, Sato S, Tsuzura H, Sato S, Narita Y, Kanemitsu Y, Shimada Y, Hirano K, Iijima K, Wada R, et al. Pretreatment AKR1B10 expression predicts the risk of hepatocellular carcinoma development after hepatitis C virus eradication. World J Gastroenterol. 2016; 22:7569–78. https://doi.org/10.3748/wjg.v22.i33.7569 [PubMed]
- 53. Shen Y, Liao DF, Cao D. AKR1B10 in gastrointestinal diseases. Aging (Albany NY). 2015; 7:221–22. https://doi.org/10.18632/aging.100737 [PubMed]
- 54. da Cruz AC, Petronilho F, Heluany CC, Vuolo F, Miguel SP, Quevedo J, Romano-Silva MA, Dal-Pizzol F. Oxidative stress and aging: correlation with clinical parameters. Aging Clin Exp Res. 2014; 26:7–12. https://doi.org/10.1007/s40520-013-0176-9 [PubMed]
- 55. Yan LJ. Positive oxidative stress in aging and aging-related disease tolerance. Redox Biol. 2014; 2:165–69. https://doi.org/10.1016/j.redox.2014.01.002 [PubMed]
- 56. Tanikawa K, Torimura T. Studies on oxidative stress in liver diseases: important future trends in liver research. Med Mol Morphol. 2006; 39:22–27. https://doi.org/10.1007/s00795-006-0313-z [PubMed]
- 57. Castro MR, Suarez E, Kraiselburd E, Isidro A, Paz J, Ferder L, Ayala-Torres S. Aging increases mitochondrial DNA damage and oxidative stress in liver of rhesus monkeys. Exp Gerontol. 2012; 47:29–37. https://doi.org/10.1016/j.exger.2011.10.002 [PubMed]
- 58. Hamed O, Kimchi ET, Sehmbey M, Gusani NJ, Kaifi JT, Staveley-O’Carroll K. Impact of genetic targets on cancer therapy: hepatocellular cancer. Adv Exp Med Biol. 2013; 779:67–90. https://doi.org/10.1007/978-1-4614-6176-0_4 [PubMed]
- 59. Woo HG, Park ES, Thorgeirsson SS, Kim YJ. Exploring genomic profiles of hepatocellular carcinoma. Mol Carcinog. 2011; 50:235–43. https://doi.org/10.1002/mc.20691 [PubMed]



