Introduction
Colorectal cancer is a leading cause of cancer-related death worldwide [1]. In China, colorectal cancer ranked fifth among the national death rates for both genders, causing approximate 191,000 million deaths in 2015 [2]. Colorectal cancer is frequently diagnosed at advanced stages, and survival rates largely depend on cancer stages at diagnosis [3]. According to the staging system defined by the American Joint Committee on Cancer (AJCC) 6th edition system, the 5-year survival rate of colorectal cancer was 93.2% for stage I, 84.7% for stage IIa, 72.2% for stage IIb, 83.4% for stage IIIa, 64.1% for stage IIIb, 44.3% for stage IIIc and 8.1% for stage IV [4]. Because advanced patients often have poor prognosis, it is important to develop targeted prevention and early intervention strategies in control of colorectal cancer. From a clinical standpoint, surgery has long been established as the mainstay treatment for colorectal cancer [5]. The fact of matter, however, is that even after the surgery, colorectal cancer prognosis still is far from satisfying [6,7]. Consequently, current research interest has shifted to devise rational and effective strategies for improving the long-term prognosis of colorectal cancer.
Hypertension and diabetes mellitus are worldwide epidemics [8], and they are also regarded as common comorbidities of colorectal cancer [9-12]. A large cohort study by van Leersum et al. showed that hypertension and diabetes mellitus respectively affected about 22% and 11% of primary colorectal cancer patients [13]. Our recent findings revealed that pre-surgical hypertension and hyperglycemia were significant predictors for the poor prognosis of colorectal cancer after radical resection by prospectively inspecting the survival of 1,318 patients with median follow-up time of 58.6 months [14], which prompted us to speculate that long-term antihypertensive or antidiabetic medications can improve colorectal cancer survival. Indeed, some studies have suggested that the intake of antihypertensive and antidiabetic drugs was closely correlated with clinical outcomes of solid tumors at many sites, including colon and rectum [15,16]. For example, an analysis in 235 metastatic colorectal cancer patients indicated that the intake of beta-blockers in patients under chemotherapy played a potential prognostic role [17]. Moreover, a meta-analysis of seven cohorts demonstrated that metformin can prolong overall survival of diabetic patients with colorectal cancer, whereas the impact on colorectal cancer-specific survival remained non-significant [18]. However, the impact of antihypertensive and antidiabetic medications on post-surgical colorectal cancer survival is currently not fully understood.
In this prospective cohort study, we therefore aimed to investigate the impact of long-term antihypertensive and antidiabetic medications on colorectal cancer-specific survival and recurrence of post-surgical patients by analyzing data from the Fujian prospective investigation of cancer (FIESTA) study.
Results
Baseline characteristics
In total, 713 participants with complete data on hypertension and diabetes mellitus, as well as medications were analyzed in this study, and baseline characteristics are shown in Table 1 and Table 2. There were 384, 246 and 83 colorectal cancer patients without hypertension, with untreated hypertension and with treated hypertension, respectively, and the corresponding numbers for diabetes mellitus were 385, 285 and 43. Patients without hypertension or diabetes mellitus were relatively younger and leaner (indexed by BMI) than those under treatment (p <0.001). Also, recurrence rate was significantly lower in patients without hypertension or diabetes mellitus than those under treatment (p <0.001), whereas the percentage of patients with distant metastasis was significantly higher in patients with untreated hypertension or diabetes mellitus (p <0.001). No significance was detectable for other characteristics across three groups for both hypertension and diabetes mellitus.
Table 1. Baseline characteristics of colorectal cancer patients per hypertension and medications.
| Characteristics | Patients w/o HT | Patients w/t untreated HT | Patients w/t treated HT | p | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Number | 384 | 246 | 83 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age, years | 54.59 (12.74) | 62.36 (11.77) | 67.99 (8.85) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Male gender | 221 (57.55%) | 135 (54.88%) | 54 (65.06%) | 0.268 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking | 36 (9.38%) | 32 (13.01%) | 12 (14.46%) | 0.226 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Drinking | 5 (1.30%) | 8 (3.25%) | 1 (1.20%) | 0.198 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Family history | 27 (7.03%) | 13 (5.28%) | 3 (3.61%) | 0.407 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BMI, kg/m2 | 22.24 (3.03) | 23.21 (3.17) | 24.12 (2.99) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Antihypertensive medications | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| CCB | NA | NA | 30 (36.14%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ACEI or ARB or beta-blocker | NA | NA | 8 (9.64%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other treatment | NA | NA | 11 (13.25%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unclear | NA | NA | 34 (40.96%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Recurrence | 17 (4.43%) | 33 (13.41%) | 10 (12.05%) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Recurrence time (months) | 14.1 (12.5, 14.1) | 17.7 (12.3, 33.1) | 21.95 (18.6, 43.5) | 0.775 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TNM stage | 0.188 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I/II | 205 (53.39%) | 128 (52.03%) | 53 (63.86%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| III/IV | 179 (46.61%) | 118 (47.97%) | 30 (36.14%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Invasion depth | 0.236 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T1/T2 | 96 (25.00%) | 55 (22.36%) | 27 (32.53%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T3/T4 | 288 (75.00%) | 191 (77.64%) | 56 (67.47%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Regional LNM | 0.056 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N0 | 207 (53.91%) | 138 (56.10%) | 56 (67.47%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N1/N2 | 177 (46.09%) | 108 (43.90%) | 27 (32.53%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Distant metastasis | 14 (3.65%) | 30 (12.20%) | 8 (9.64%) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Differentiation | 0.679 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Low | 279 (72.66%) | 182 (73.98%) | 57 (68.67%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Moderate/High | 105 (27.34%) | 64 (26.02%) | 26 (31.33%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tumor embolus | 58 (15.10%) | 44 (17.89%) | 14 (16.87%) | 0.495 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tumor size (cm) | 5 (3.5, 6) | 4.5 (3.5, 4.5) | 4.5 (3.5, 6) | 0.456 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Number of LNM | 0 (0, 2) | 0 (0, 2) | 0 (0, 1) | 0.186 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: w/t, with; w/o, without; HT, hypertension; BMI, body mass index; NA, not available; CCB, calcium channel blocker; ACEI, angiotensin converting enzyme inhibitor; ARB, angiotensin receptor blocker; TNM, tumor node metastasis; LNM, lymph node metastasis. Data are expressed as mean (standard deviation) or median (interquartile range) or count (percentage). | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Table 2. Baseline characteristics of colorectal cancer patients per diabetes mellitus and medications.
| Characteristics | Patients w/o DM | Patients w/t untreated DM | Patients w/t treated DM | p | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Number | 385 | 285 | 43 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Age, years | 54.62 (12.74) | 63.49 (11.58) | 65.49 (9.86) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Male | 222 (57.66%) | 165 (57.89%) | 23 (53.49%) | 0.858 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Smoking | 36 (9.35%) | 39 (13.68%) | 5 (11.63%) | 0.213 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Drinking | 5 (1.30%) | 8 (2.81%) | 1 (2.33%) | 0.374 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Family history | 27 (7.03%) | 14 (4.91%) | 2 (4.65%) | 0.484 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BMI, kg/m2 | 22.24 (3.03) | 23.33 (3.20) | 24.14 (2.71) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Antidiabetic medications | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Metformin | NA | NA | 5 (11.63%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Non-Metformin | NA | NA | 21 (48.84%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Other treatment | NA | NA | 9 (20.93%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Unclear | NA | NA | 8 (18.60%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Recurrence | 17 (4.42%) | 38 (13.33%) | 5 (11.63%) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Recurrence time (months) | 14.1 (12.5, 49.8) | 19.4 (12.4, 33.1) | 48.8 (8.3, 81.3) | 0.102 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TNM stage | 0.502 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I/II | 205 (53.52%) | 153 (53.68%) | 27 (62.79%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| III/IV | 178 (46.48%) | 132 (46.32%) | 16 (37.21%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Invasion depth | 0.665 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T1/T2 | 97 (25.19%) | 68 (23.86%) | 13 (30.23%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T3/T4 | 288 (74.81%) | 217 (76.14%) | 30 (69.77%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Regional LNM | 0.249 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N0 | 208 (54.03%) | 166 (58.25%) | 28 (65.12%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N1/N2 | 177 (45.97%) | 119 (41.75%) | 15 (34.88%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Distant metastasis | 14 (3.64%) | 36 (12.63%) | 2 (4.65%) | <0.001 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Differentiation | 0.088 | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Low | 279 (72.47%) | 199 (87.37%) | 40 (93.02%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Moderate/High | 106 (27.53%) | 36 (12.63%) | 3 (6.98%) | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tumor embolus | 58 (15.06%) | 54 (18.95%) | 4 (9.30%) | 0.038 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tumor size (cm) | 5 (3.5, 6) | 4.5 (3.5, 6) | 4 (3, 6) | 0.517 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Number of LNM | 0 (0, 2) | 0 (0, 2) | 0 (0, 1) | 0.337 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: w/t, with; w/o, without; DM, diabetes mellitus; BMI, body mass index; NA, not available; TNM, tumor-node-metastasis; LNM, lymph node metastasis. Data are expressed as mean (standard deviation) or median (interquartile range) or count (percentage) if appropriate. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
Overall survival comparison
Figure 1 shows the comparison of cumulative survival rates per hypertension and diabetes mellitus in colorectal cancer patients. As expected, colorectal cancer patients without hypertension had better survival than those with hypertension (median survival time (MST): 190.3 months versus 99.0 months, Log-rank test p <0.001 in Supplementary Table 1). Although antihypertensive medications seemed to be beneficial on colorectal cancer prognosis, the impact was not statistically significant (Log-rank test p: 0.205). By contrast, in colorectal cancer patients with diabetes mellitus, the impact of medications on survival was statistically significant, that is, patients receiving antidiabetic medications had longer survival time than untreated diabetic patients (MST: 135.8 months versus 80.2 months, Log-rank test p: 0.007), whereas the prognosis was greatly improved in colorectal cancer patients without diabetes mellitus (MST: 170.3 months, Log-rank test p <0.001 in Supplementary Table 1).
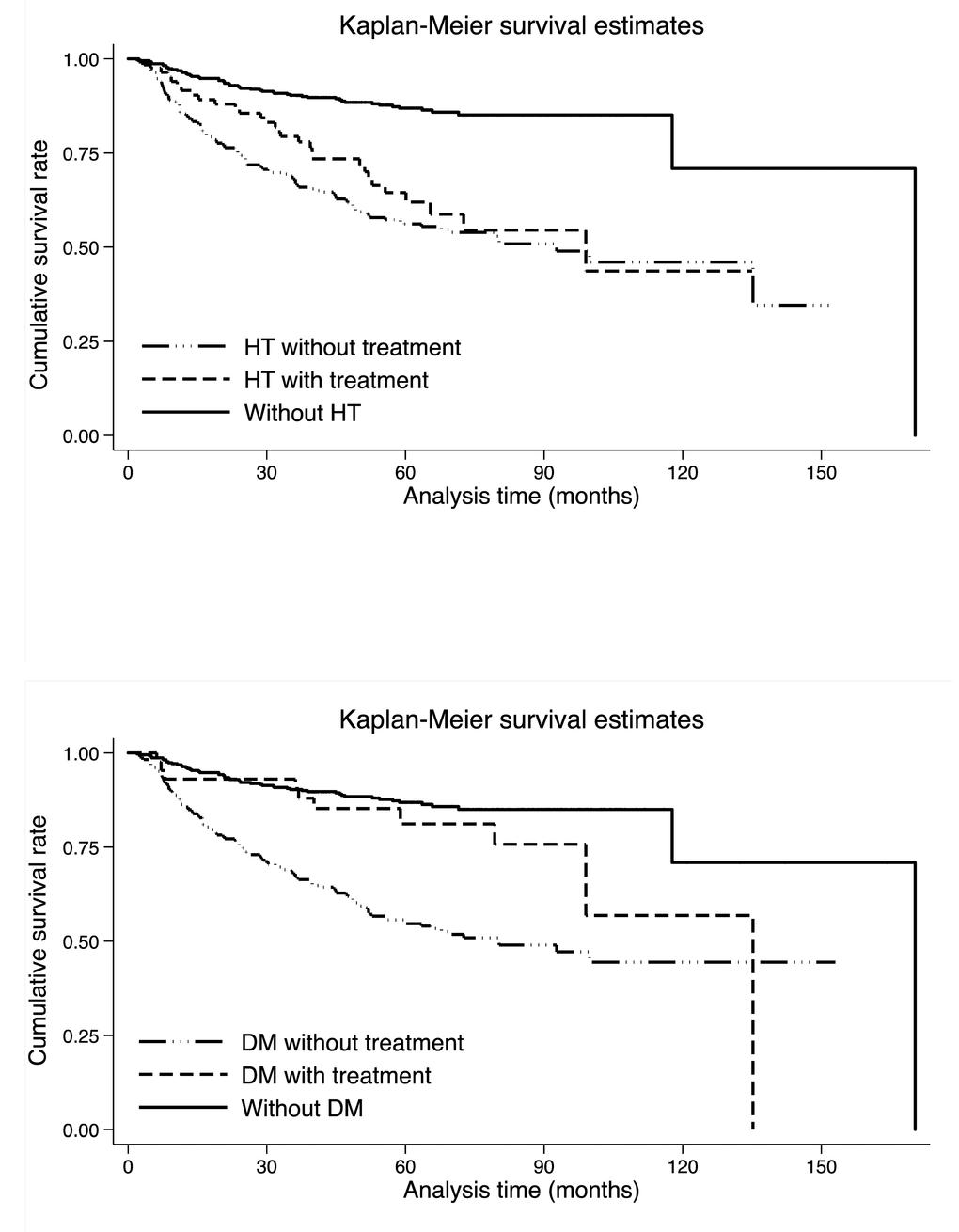
Figure 1. Kaplan-Meier survival curves per hypertension (the upper panel) and diabetes mellitus (the lower panel). Abbreviations: HT, hypertension; DM, diabetes mellitus. There were 384, 246 and 83 colorectal cancer patients without hypertension, with untreated hypertension and with treated hypertension, respectively. There were 385, 285 and 43 colorectal cancer patients without diabetes mellitus, with untreated diabetes mellitus and with treated diabetes mellitus, respectively. The Log-rank test was statistically significant for both hypertension and diabetes mellitus medications (p <0.001).
Combined impact of hypertension and diabetes mellitus medications on colorectal cancer survival is presented in Supplementary Figure 1.
Overall recurrence comparison
The comparison of cumulative recurrence rates per hypertension and diabetes mellitus in colorectal cancer patients is provided in Figure 2, and no significance was observed across hypertension or diabetes mellitus, as well as across medications (Log-rank test p: 0.899 for hypertension and 0.169 for diabetes mellitus).
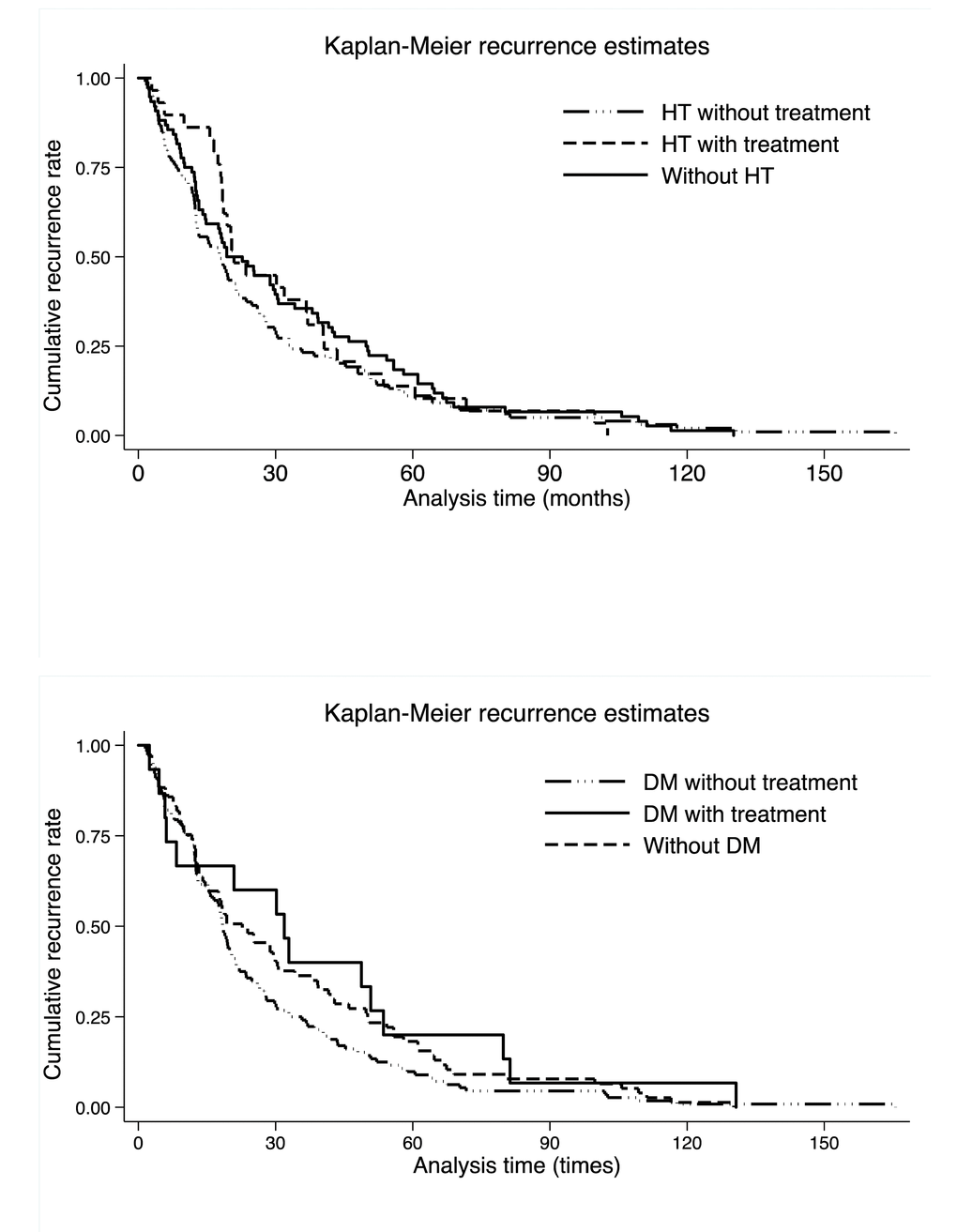
Figure 2. Kaplan-Meier recurrence curves per hypertension (the upper panel) and diabetes mellitus (the lower panel). Abbreviations: HT, hypertension; DM, diabetes mellitus. There were 33, 10 and 17 colorectal cancer patients without hypertension, with untreated hypertension and with treated hypertension, respectively. There were 17, 38 and 5 colorectal cancer patients without diabetes mellitus, with untreated diabetes mellitus and with treated diabetes mellitus, respectively. Log-rank test was not significant for both hypertension and diabetes mellitus medications (p: 0.899 for hypertension and 0.169 for diabetes mellitus).
Survival comparison per medications
Shown in Figure 3 is the comparison of cumulative survival rates per antihypertensive and antidiabetic medications in colorectal cancer patients. Upon stratification by antihypertensive medications, colorectal cancer patients with other medications seemed to have better survival within 60 months after the surgery than those receiving CCBs and ACEIs or ARBs or beta-blockers, whereas there was no detectable significance (Log-rank test p: 0.978 in Supplementary Table 1). By contrast, upon stratification by antidiabetic medications, colorectal cancer patients with metformin only seemed to have better survival within 40 months after the surgery than patients with non-metformin and the other medications, and similarly no significance was noted (Log-rank test p: 0.778 in Supplementary Table 1).
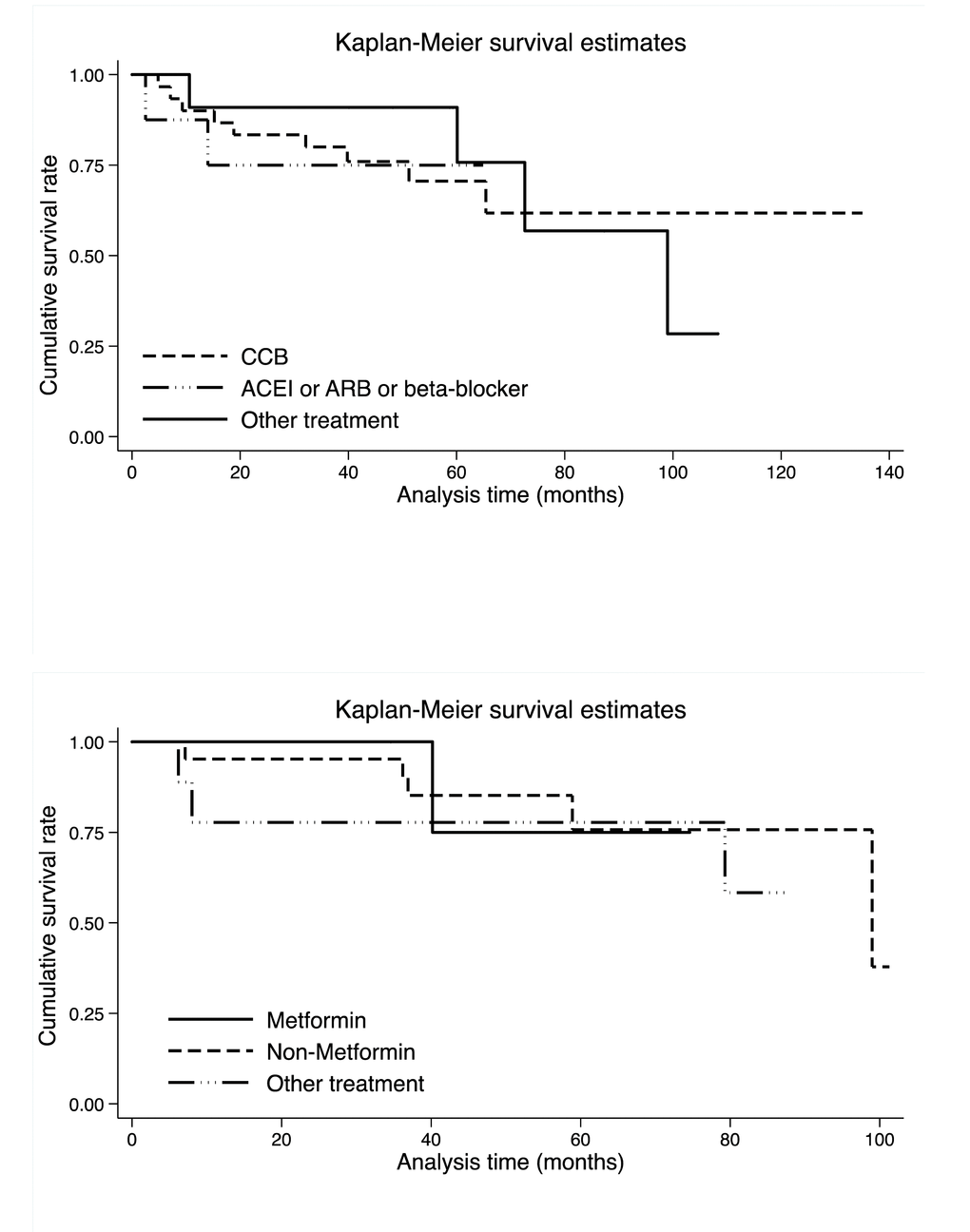
Figure 3. Kaplan-Meier survival curves per antihypertensive (the upper panel) and antidiabetic (the lower panel) medications. Abbreviations: CCB, calcium channel blocker; ACEI, angiotensin converting enzyme inhibitor; ARB, angiotensin receptor blocker. There were 30, 8 and 11 colorectal cancer patients who took ACEI or ARB or beta-blocker, CCB and other antihypertensive drugs, respectively. There were 5, 21 and 9 colorectal cancer patients who took metformin, non-metformin and other antidiabetic drugs, respectively. Log-rank test was not significant for both antihypertensive (p: 0.978) and antidiabetic (p: 0.778) medications.
Mortality risk estimation
To see whether antihypertensive or antidiabetic medications can improve the prognosis of post-surgical colorectal cancer, we first tested whether proportional-hazards assumption was satisfied. As shown in Supplementary Figure 2, this assumption was violated for the comparison of colorectal cancer patients with and without antihypertensive or antidiabetic medications because the two curves were not parallel. As the ln(-ln(S(t))) is a linear function of ln(t) (here, t is the time variable and S(t) is the survival function), we therefore adopted the multivariate Weibull proportional hazards regression model to derive survival estimates.
Table 3 presents the overall and stratified estimates for colorectal cancer mortality risk per hypertension and diabetes mellitus, respectively. All risk estimates were adjusted for age, gender, BMI, smoking, drinking and family history of cancer. Overall, medical treatment for hypertension and diabetes mellitus was associated with 28% (HR: 0.72; 95% CI: 0.47-1.10; p: 0.131) and 57% (HR: 0.43; 95% CI: 0.22-0.82; p: 0.010) reduced risk of dying from colorectal cancer relative to those without medications, respectively, especially for diabetes mellitus. In case of diabetes mellitus, the power to detect this significant association was over 99.0%.
Table 3. Overall and stratified analyses of antihypertensive and antidiabetic medications in predicting the mortality risk of post-surgical colorectal cancer.
| Groups | Antihypertensive medications | Antidiabetic medications | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Deaths/Patients | HR | 95% CI | p | Deaths/Patients | HR | 95% CI | p | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Overall | 140/327 | 0.72 | 0.47-1.10 | 0.131 | 140/327 | 0.43 | 0.22-0.82 | 0.010 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Gender | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Males | 85/187 | 0.72 | 0.42-1.21 | 0.213 | 85/187 | 0.54 | 0.25-1.18 | 0.125 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Females | 55/140 | 0.71 | 0.34-1.47 | 0.356 | 55/140 | 0.30 | 0.01-0.97 | 0.045 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| TNM stage | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| I/II | 45/180 | 0.81 | 0.41-1.60 | 0.544 | 45/180 | 0.68 | 0.26-1.74 | 0.415 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| III/IV | 95/147 | 0.86 | 0.49-1.51 | 0.606 | 95/147 | 0.30 | 0.12-0.73 | 0.009 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Invasion depth | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T1/T2 | 21/81 | 1.39 | 0.54-3.58 | 0.495 | 21/81 | 0.38 | 0.09-1.65 | 0.196 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| T3/T4 | 119/246 | 0.65 | 0.40-1.07 | 0.090 | 119/246 | 0.46 | 0.22-0.95 | 0.037 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Regional LNM | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N0 | 58/194 | 0.79 | 0.43-1.45 | 0.455 | 58/194 | 0.64 | 0.27-1.51 | 0.313 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| N1/N2 | 39/71 | 1.14 | 0.50-2.57 | 0.757 | 39/71 | 0.40 | 0.14-1.12 | 0.081 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Distant metastasis | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Negative | 104/209 | 0.74 | 0.46-1.20 | 0.222 | 104/290 | 0.44 | 0.21-0.91 | 0.027 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Positive | 36/37 | 0.59 | 0.21-1.68 | 0.325 | 36/37 | 1.86 | 0.34-10.15 | 0.476 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tumor differentiation | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Low | 93/238 | 0.52 | 0.29-0.93 | 0.028 | 93/238 | 0.39 | 0.19-0.80 | 0.011 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Moderate/High | 22/36 | 0.44 | 0.14-1.35 | 0.153 | 22/36 | 6.40 | 0.72-56.63 | 0.095 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tumor embolus | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Positive | 38/57 | 0.32 | 0.11-0.95 | 0.040 | 38/57 | 0.16 | 0.02-1.18 | 0.072 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Negative | 55/171 | 0.79 | 0.43-1.48 | 0.468 | 55/171 | 0.60 | 0.28-1.27 | 0.182 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Tumor size | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| ≤ 4.5 cm | 64/170 | 0.92 | 0.51-1.66 | 0.782 | 64/170 | 0.59 | 0.26-1.31 | 0.193 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| > 4.5 cm | 75/155 | 0.52 | 0.28-0.98 | 0.043 | 75/155 | 0.30 | 0.09-0.96 | 0.042 | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Abbreviations: HR, hazard ratio; 95% CI, 95% confidence interval; TNM, tumor-node-metastasis; LNM, lymph node metastasis. Besides stratification upon gender, p value was adjusted for age, gender, body mass index, smoking, drinking and family history of cancer. | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
By gender, TNM stage and invasion depth, the reduced risk was only significant when analysis was restricted to antidiabetic medications (HR: 0.30, 0.30 and 0.46; 95% CI: 0.01-0.97, 0.12-0.73 and 0.22-0.95; p: 0.045, 0.009 and 0.037, respectively) (Table 3). By distant metastasis, antidiabetic medications had a significantly reduced risk only in patients without distant metastasis (HR: 0.44; 95% CI: 0.21-0.91; p: 0.027). By tumor differentiation, both antihypertensive and antidiabetic medications can significantly reduce the risk of colorectal cancer-specific mortality (HR: 0.52 and 0.39; 95% CI: 0.29-0.93 and 0.19-0.80; p: 0.028 and 0.011, respectively) in patients with low differentiation. By tumor embolus, risk reduction was only seen in positive embolus for the comparison of patients with antihypertensive medications than those without (HR: 0.32; 95% CI: 0.11-0.95; p: 0.040). By tumor size, both antihypertensive and antidiabetic medications can reduce the mortality risk of colorectal cancer significantly in patients with tumor size greater than 4.5 cm (HR: 0.52 and 0.30; 95% CI: 0.28-0.98 and 0.09-0.96; p: 0.043 and 0.042, respectively).
Discussion
As an extension of our previous FIESTA study [14,19-26], we investigated the impact of long-term antihypertensive and antidiabetic medications on colorectal cancer-specific survival and recurrence in post-surgical patients. Noteworthily, our data indicate that long-term antidiabetic medications can significantly prolong the survival and improve the prognosis of post-surgical colorectal cancer, whereas the anticancer impact of long-term antihypertensive medications was not obvious. Our findings highlight the importance of enhanced screening and targeted management of diabetes mellitus in colorectal cancer patients, especially after radical resection.
The association of diabetes mellitus with the risk and prognosis of colorectal cancer has been widely evaluated in the literature [27-29]. Mills et al. conducted a meta-analysis of 26 studies and found that colorectal cancer patients complicated with diabetes mellitus were at 17% and 12% significantly increased risk of all-cause and cancer-specific mortality, respectively [30]. In this present study, we found that diabetic patients had worse cancer-specific prognosis of post-surgical colorectal cancer than patients without diabetes mellitus, yet failed to detect any significance in association with recurrence, possibly due to small sample size involved. Our findings further revealed that long-term antidiabetic medications can prolong the survival and reduce colorectal cancer-specific mortality risk by about 50%, whereas there was no distinction in prognosis between metformin, a widely used oral antidiabetic agent, and non-metformin medications. In support of our findings, a recent meta-analysis of 7 cohort studies conducted by Meng et al. indicated that relative to non-metformin, metformin had no benefits for cancer-specific survival, whereas metformin can remarkably prolong the overall survival of colorectal cancer patients with comorbid diabetes mellitus [18]. There is mounting evidence underscoring the important anticancer impact of metformin on carcinogenesis, likely through the upregulation of AMP-activated protein kinase (AMPK) activity and the downstream suppression of signaling through the mTOR [31,32]. In addition, animal studies provided evidence that metformin can inhibit both colon carcinoma and intestinal polyp growth [33,34]. Although epidemiologic evidence failed to support the beneficial superiority of metformin over non-metformin, we cannot completely negate its existence, as diabetic patients may take more than one medication to lower blood glucose at the same time. In fact, during the follow-up of this study, we found that patients receiving either metformin or non-metformin usually took other types of antiglycemic drugs to better control glucose. Even in clinical settings, it is impractical and unethical to assign study participants to mono-medical treatment. It is also evidenced that metformin was associated with an increased risk of colorectal cancer in males, but not in females. In view of small sample sizes in the present study, we cannot interrogate the gender-specific difference between metformin and non-metformin in colorectal cancer survival [35]. We agree that future large prospective studies are needed to see whether gender plays a role in predicting the risk and prognosis of colorectal cancer.
By contrast, our results did not explicitly support a favorable benefit profile of antihypertensive medications for the prognosis of post-surgical colorectal cancer, and distinction in prognosis between antihypertensive drugs was nonsignificant. Epidemiological evidence suggests a potential prognostic role of beta-blockers in colorectal cancer patients under chemotherapy. Moreover, two meta-analyses consistently demonstrated that bevacizumab-induced hypertension may represent a prognostic factor in patients with metastatic colorectal cancer [36,37]. However, no report has thus far addressed the relationship between antihypertensive medications and post-surgical colorectal cancer prognosis. So, whether improvement in colorectal cancer survival with antihypertensive medications is because of only blood pressure lowering or additional anticancer mechanisms is still an open question, and further validation of our findings will be helpful.
Finally, several limitations should be acknowledged. Firstly, this study was carried out in a mono-center, and our findings could better be generalized pending consistently validated in other cohorts. However, mono-center design may represent an important indicator of daily clinical practice. Secondly, all colorectal cancer patients were enrolled between January 2000 and December 2008, and during the 9-year period remarkable advances in surgical techniques might introduce a possible bias, which may underestimate the impact of antihypertensive or antidiabetic medications during follow-up on colorectal cancer mortality. Thirdly, all participants were followed up at the Outpatient Department of Fujian Cancer Hospital or through calling or letters to obtain information on antihypertensive and antidiabetic medications, whereas their blood pressure and glucose were not measured. In case of poorly controlled blood pressure or glucose, participants were recommended to comprehensive or specialized hospitals. Fourthly, the findings presented in this study are based on only colorectal cancer patients who are operable for radical resection and therefore cannot be directly extrapolated to the general population. For this reason, validation in other large cohorts is required.
Taken together, our data indicate that long-term antidiabetic medications can significantly prolong the survival and improve the prognosis of post-surgical colorectal cancer, whereas the anticancer impact of long-term antihypertensive medications was not obvious. This study underscores the critical need for intensified screening programs and pharmacological management for diabetes mellitus in colorectal cancer patients to prolong the survival and improve the prognosis in China.
Materials and Methods
The FIESTA study
The FIESTA study is an ongoing prospective evaluation on the survival of post-surgical patients with common digestive tract cancer (such as esophageal cancer, gastric cancer and colorectal cancer) initiated in January 2000 and still recruiting [14,19-26]. Approval of this study was obtained from the Ethics Committees of Fujian Cancer Hospital. All participants gave written informed consent before recruitment.
Study participants
All participants were consecutively recruited from the Department of Thoracic Surgery at Fujian Cancer Hospital during the period between January 2000 and December 2008, and they were followed up until July 2017. Only participants of Han Chinese descent and without consanguinity were eligible for inclusion, and they for the first time underwent radical resection for colorectal cancer that was confirmed by pre-surgical biopsy or post-surgical pathologic tests. In addition, no participants had received chemotherapy and radiotherapy. Only participants with complete data on hypertension or diabetes mellitus and their medications were analyzed. Moreover, they must be followed up for one month or over.
Tissue collection
From each participant, both cancerous tissue and normal colonic or rectal tissue were resected during the surgery. Tissue samples were formalin-fixed and paraffin-embedded, and their pathological characteristics were analyzed at the Department of Pathology, Fujian Cancer Hospital.
Follow-up evaluation
After discharge from hospital, annual follow-up was carried out for qualified participants at the Out-Patient Department of Fujian Cancer Hospital or through calling or sending printed letters in case of no-show on scheduled time. In case of death events, the date was recorded from relatives or medical reports. The primary outcome was death from colorectal cancer. Survival time was calculated from the date of receiving radical resection to the date of death or last follow-up, whichever occurred first. Median follow-up time was also calculated, and it referred to the length of time from the start of radical resection for colorectal cancer to the time when half of the patients were still alive.
Baseline characteristics
A self-designed questionnaire was completed by each participant at the time of admission on age, gender, smoking, drinking, family cancer history, antihypertensive medications and antidiabetic medications. Meanwhile, body weight and height were measured with participants wearing light clothing and no shoes, and body mass index (BMI) was calculated as weight divided by height in meters-squared (kg/m2). Arterial blood pressure was measured with participants in the seated position using a mercury sphygmomanometer, and three readings were taken, each at least 5 minutes apart. Age was recorded at the date of receiving radical resection for colorectal cancer. Cigarette smoking was defined by indicator variables for never, former and current smoking. Alcohol drinking was defined by indicator variables for never, former and current drinking. Family cancer history was recorded for study participants with one or more affected relatives within three generations who suffered malignancies except non-melanoma skin cancer.
Hypertension and diabetes mellitus, as well as information on medications were recorded during the annual follow-up period. Hypertension was diagnosed if averaged blood pressure was at least 140 mm Hg systolic or 90 mm Hg diastolic, or when the participants were on antihypertensive medications. In view of sample sizes involved, the type of antihypertensive medications was classified into the calcium channel blocker (CCB) group, the angiotensin converting enzyme inhibitor (ACEI) or angiotensin receptor blocker (ARB) or beta-blocker group and the other treatment group (including irregular treatment and use of traditional Chinese medicine). Fasting venous blood samples were taken the morning of surgery day and used to measure blood glucose using an automated glucose oxidase method at Clinical Laboratory, Fujian Cancer Hospital. Diabetes mellitus was diagnosed if the fasting blood glucose ≥7.0 mmol/L or when the participants were on antidiabetic medications. In view of sample sizes involved, the type of antidiabetic medications was classified into the metformin group, the non-metformin group and the other treatment group (including irregular treatment and use of traditional Chinese medicine).
Clinicopathologic characteristics including tumor-node-metastasis (TNM) stage (I, II, III and IV) [38], depth of invasion (T1, T2, T3 and T4), regional lymph node metastasis (N0, N1, N2 and N3), distant metastasis (M0 and M1), tumor differentiation, tumor embolus and tumor size (in centimeters) were obtained from pathological reports.
Statistical analysis
Baseline characteristics were expressed as mean (standard deviation) or median (interquartile range) or count (percentage) if appropriate. Kaplan-Meier curves and Log-rank tests were used to present and test the differences in cumulative survival or recurrence rates. Tests of proportional-hazards assumption were performed to facilitate the selection of proper model. The risk prediction of pre-surgical hypertension or diabetes mellitus and their medications for colorectal cancer mortality was calculated under the Cox or Weibull, where appropriate, proportional hazards regression model, and the magnitude of risk prediction was denoted by hazard ratio (HR) and 95% confidence interval (95% CI).
All statistical analyses were done by the STATA/SE Release 14.0 (StataCorp, College Station, TX, USA). Study power was estimated by the PS Power and Sample Size Calculations software Release 3.0 [39].
Supplementary Materials
Author Contributions
XZ, JL, WN planned and designed the study, and directed its implementation; FP, DH, XZ drafted the protocol; DH, XL, BL, YC, HZ, YX obtained statutory and ethics approvals; DH, XL, BL, HZ, YX contributed to data acquisition; FP, DH, JL, XZ, WN had access to all raw data; FP, DH, WN did the data preparation, quality control and data analysis; FP, WN wrote the manuscript.
Acknowledgements
We thank Dr. Helen Colley for help with organotypic models and Brenka McCabe and Jason Heath for technical assistance.
Conflicts of Interest
The authors have no conflicts of interest to declare.
Funding
This study was financially supported by the Joint Funds for the Innovation of Science and Technology of Fujian Province (Grant No. 2017Y9090), the Natural Science Foundation of China (Grant No. 81770309) and the Natural Science Foundation of Fujian Province (Grant No. 2018Y0024). The funder of this study had no role in study design, data collection, data analysis, data interpretation, or writing of the report. The corresponding authors had full access to all the data in the study and had final responsibility for the decision to submit for publication.
References
- 1. Douaiher J, Ravipati A, Grams B, Chowdhury S, Alatise O, Are C. Colorectal cancer-global burden, trends, and geographical variations. J Surg Oncol. 2017; 115:619–30. https://doi.org/10.1002/jso.24578 [PubMed]
- 2. Chen W, Zheng R, Baade PD, Zhang S, Zeng H, Bray F, Jemal A, Yu XQ, He J. Cancer statistics in China, 2015. CA Cancer J Clin. 2016; 66:115–32. https://doi.org/10.3322/caac.21338 [PubMed]
- 3. Liu CY, Chen WT, Kung PT, Chiu CF, Wang YH, Shieh SH, Tsai WC. Characteristics, survival, and related factors of newly diagnosed colorectal cancer patients refusing cancer treatments under a universal health insurance program. BMC Cancer. 2014; 14:446. https://doi.org/10.1186/1471-2407-14-446 [PubMed]
- 4. O’Connell JB, Maggard MA, Ko CY. Colon cancer survival rates with the new American Joint Committee on Cancer sixth edition staging. J Natl Cancer Inst. 2004; 96:1420–25. https://doi.org/10.1093/jnci/djh275 [PubMed]
- 5. Rentsch M, Schiergens T, Khandoga A, Werner J. Surgery for Colorectal Cancer - Trends, Developments, and Future Perspectives. Visc Med. 2016; 32:184–91. https://doi.org/10.1159/000446490 [PubMed]
- 6. Modest DP, Denecke T, Pratschke J, Ricard I, Lang H, Bemelmans M, Becker T, Rentsch M, Seehofer D, Bruns CJ, Gebauer B, Modest HI, Held S, et al. Surgical treatment options following chemotherapy plus cetuximab or bevacizumab in metastatic colorectal cancer-central evaluation of FIRE-3. Eur J Cancer. 2018; 88:77–86. https://doi.org/10.1016/j.ejca.2017.10.028 [PubMed]
- 7. Glockzin G, Rochon J, Arnold D, Lang SA, Klebl F, Zeman F, Koller M, Schlitt HJ, Piso P. A prospective multicenter phase II study evaluating multimodality treatment of patients with peritoneal carcinomatosis arising from appendiceal and colorectal cancer: the COMBATAC trial. BMC Cancer. 2013; 13:67. https://doi.org/10.1186/1471-2407-13-67 [PubMed]
- 8. Navar AM, Gallup DS, Lokhnygina Y, Green JB, McGuire DK, Armstrong PW, Buse JB, Engel SS, Lachin JM, Standl E, Van de Werf F, Holman RR, Peterson ED, and TECOS Study Group. Hypertension Control in Adults With Diabetes Mellitus and Recurrent Cardiovascular Events: Global Results From the Trial Evaluating Cardiovascular Outcomes With Sitagliptin. Hypertension. 2017; 70:907–14. https://doi.org/10.1161/HYPERTENSIONAHA.117.09482 [PubMed]
- 9. Ahmadi A, Mobasheri M, Hashemi-Nazari SS, Baradaran A, Choobini ZM. Prevalence of hypertension and type 2 diabetes mellitus in patients with colorectal cancer and their median survival time: A cohort study. J Res Med Sci. 2014; 19:850–54. [PubMed]
- 10. Wang JL, Chang CH, Lin JW, Wu LC, Chuang LM, Lai MS. Infection, antibiotic therapy and risk of colorectal cancer: a nationwide nested case-control study in patients with Type 2 diabetes mellitus. Int J Cancer. 2014; 135:956–67. https://doi.org/10.1002/ijc.28738 [PubMed]
- 11. Nakaya A, Kurata T, Yokoi T, Iwamoto S, Torii Y, Katashiba Y, Ogata M, Hamada M, Kon M, Nomura S. Retrospective analysis of bevacizumab-induced hypertension and clinical outcome in patients with colorectal cancer and lung cancer. Cancer Med. 2016; 5:1381–87. https://doi.org/10.1002/cam4.701 [PubMed]
- 12. Datta S, Chowdhury S, Roy HK. Metabolism, microbiome and colorectal cancer. Aging (Albany NY). 2017; 9:1086–87. https://doi.org/10.18632/aging.101234 [PubMed]
- 13. van Leersum NJ, Janssen-Heijnen ML, Wouters MW, Rutten HJ, Coebergh JW, Tollenaar RA, Lemmens VE. Increasing prevalence of comorbidity in patients with colorectal cancer in the South of the Netherlands 1995-2010. Int J Cancer. 2013; 132:2157–63. https://doi.org/10.1002/ijc.27871 [PubMed]
- 14. Peng F, Hu D, Lin X, Chen G, Liang B, Zhang H, Ji K, Huang J, Lin J, Zheng X, Niu W. Preoperative metabolic syndrome and prognosis after radical resection for colorectal cancer: the Fujian prospective investigation of cancer (FIESTA) study. Int J Cancer. 2016; 139:2705–13. https://doi.org/10.1002/ijc.30404 [PubMed]
- 15. Uzunlulu M, Telci Caklili O, Oguz A. Association between Metabolic Syndrome and Cancer. Ann Nutr Metab. 2016; 68:173–79. https://doi.org/10.1159/000443743 [PubMed]
- 16. Rosato V, Tavani A, Gracia-Lavedan E, Guinó E, Castaño-Vinyals G, Villanueva CM, Kogevinas M, Polesel J, Serraino D, Pisa FE, Barbone F, Moreno V, La Vecchia C, Bosetti C. Type 2 Diabetes, Antidiabetic Medications, and Colorectal Cancer Risk: Two Case-Control Studies from Italy and Spain. Front Oncol. 2016; 6:210. https://doi.org/10.3389/fonc.2016.00210 [PubMed]
- 17. Giampieri R, Scartozzi M, Del Prete M, Faloppi L, Bianconi M, Ridolfi F, Cascinu S. Prognostic Value for Incidental Antihypertensive Therapy With β-Blockers in Metastatic Colorectal Cancer. Medicine (Baltimore). 2015; 94:e719. https://doi.org/10.1097/MD.0000000000000719 [PubMed]
- 18. Meng F, Song L, Wang W. Metformin Improves Overall Survival of Colorectal Cancer Patients with Diabetes: A Meta-Analysis. J Diabetes Res. 2017; 2017:5063239. https://doi.org/10.1155/2017/5063239 [PubMed]
- 19. Hu D, Lin X, Chen Y, Chang Q, Chen G, Li C, Zhang H, Cui Z, Liang B, Jiang W, Ji K, Huang J, Peng F, et al. Preoperative blood-routine markers and prognosis of esophageal squamous cell carcinoma: the Fujian prospective investigation of cancer (FIESTA) study. Oncotarget. 2017; 8:23841–50. https://doi.org/10.18632/oncotarget.13318 [PubMed]
- 20. Hu D, Peng F, Lin X, Chen G, Liang B, Li C, Zhang H, Liao X, Lin J, Zheng X, Niu W. The elevated preoperative fasting blood glucose predicts a poor prognosis in patients with esophageal squamous cell carcinoma: the Fujian prospective investigation of cancer (FIESTA) study. Oncotarget. 2016; 7:65247–56. https://doi.org/10.18632/oncotarget.11247 [PubMed]
- 21. Hu D, Peng F, Lin X, Chen G, Zhang H, Liang B, Ji K, Lin J, Chen LF, Zheng X, Niu W. Preoperative Metabolic Syndrome Is Predictive of Significant Gastric Cancer Mortality after Gastrectomy: The Fujian Prospective Investigation of Cancer (FIESTA) Study. EBioMedicine. 2017; 15:73–80. https://doi.org/10.1016/j.ebiom.2016.12.004 [PubMed]
- 22. Hu D, Zhang H, Lin X, Chen G, Li C, Liang B, Chen Y, Cui Z, Peng F, Zheng X, Niu W. Elevated preoperative neutrophil-to-lymphocyte ratio can predict poor survival in early stage gastric cancer patients receiving radical gastrectomy: the Fujian prospective investigation of cancer (FIESTA) study. J Cancer. 2017; 8:1214–22. https://doi.org/10.7150/jca.18707 [PubMed]
- 23. Peng F, Hu D, Lin X, Chen G, Liang B, Li C, Chen Y, Cui Z, Zhang H, Lin J, Zheng X, Niu W. The monocyte to red blood cell count ratio is a strong predictor of postoperative survival in colorectal cancer patients: the Fujian prospective investigation of cancer (FIESTA) study. J Cancer. 2017; 8:967–75. https://doi.org/10.7150/jca.18000 [PubMed]
- 24. Peng F, Hu D, Lin X, Chen G, Liang B, Zhang H, Dong X, Lin J, Zheng X, Niu W. Analysis of Preoperative Metabolic Risk Factors Affecting the Prognosis of Patients with Esophageal Squamous Cell Carcinoma: The Fujian Prospective Investigation of Cancer (FIESTA) Study. EBioMedicine. 2017; 16:115–23. https://doi.org/10.1016/j.ebiom.2017.01.035 [PubMed]
- 25. Peng F, Hu D, Lin X, Chen G, Liang B, Chen Y, Li C, Zhang H, Xia Y, Lin J, Zheng X, Niu W. An in-depth prognostic analysis of baseline blood lipids in predicting postoperative colorectal cancer mortality: the FIESTA study. Cancer Epidemiol. 2018; 52:148–57. https://doi.org/10.1016/j.canep.2018.01.001 [PubMed]
- 26. Sha H, Hu D, Wu S, Peng F, Xu G, Fan G, Lin X, Chen G, Liang B, Chen Y, Li C, Zhang H, Xia Y, et al. Baseline Metabolic Risk Score and Postsurgical Esophageal Cancer-Specific Mortality: The Fujian Prospective Investigation of Cancer (FIESTA) Study. J Cancer. 2018; 9:1173–81. https://doi.org/10.7150/jca.23631 [PubMed]
- 27. Luo J, Lin HC, He K, Hendryx M. Diabetes and prognosis in older persons with colorectal cancer. Br J Cancer. 2014; 110:1847–54. https://doi.org/10.1038/bjc.2014.68 [PubMed]
- 28. Sehdev A, Shih YC, Vekhter B, Bissonnette MB, Olopade OI, Polite BN. Metformin for primary colorectal cancer prevention in patients with diabetes: a case-control study in a US population. Cancer. 2015; 121:1071–78. https://doi.org/10.1002/cncr.29165 [PubMed]
- 29. Fransgaard T, Thygesen LC, Gögenur I. Metformin Increases Overall Survival in Patients with Diabetes Undergoing Surgery for Colorectal Cancer. Ann Surg Oncol. 2016; 23:1569–75. https://doi.org/10.1245/s10434-015-5028-8 [PubMed]
- 30. Mills KT, Bellows CF, Hoffman AE, Kelly TN, Gagliardi G. Diabetes mellitus and colorectal cancer prognosis: a meta-analysis. Dis Colon Rectum. 2013; 56:1304–19. https://doi.org/10.1097/DCR.0b013e3182a479f9 [PubMed]
- 31. Zhou G, Myers R, Li Y, Chen Y, Shen X, Fenyk-Melody J, Wu M, Ventre J, Doebber T, Fujii N, Musi N, Hirshman MF, Goodyear LJ, Moller DE. Role of AMP-activated protein kinase in mechanism of metformin action. J Clin Invest. 2001; 108:1167–74. https://doi.org/10.1172/JCI13505 [PubMed]
- 32. Zhang HH, Zhang Y, Cheng YN, Gong FL, Cao ZQ, Yu LG, Guo XL. Metformin incombination with curcumin inhibits the growth, metastasis, and angiogenesis of hepatocellular carcinoma in vitro and in vivo. Mol Carcinog. 2018; 57:44–56. https://doi.org/10.1002/mc.22718 [PubMed]
- 33. Tomimoto A, Endo H, Sugiyama M, Fujisawa T, Hosono K, Takahashi H, Nakajima N, Nagashima Y, Wada K, Nakagama H, Nakajima A. Metformin suppresses intestinal polyp growth in ApcMin/+ mice. Cancer Sci. 2008; 99:2136–41. https://doi.org/10.1111/j.1349-7006.2008.00933.x [PubMed]
- 34. Harada K, Ferdous T, Harada T, Ueyama Y. Metformin in combination with 5-fluorouracil suppresses tumor growth by inhibiting the Warburg effect in human oral squamous cell carcinoma. Int J Oncol. 2016; 49:276–84. https://doi.org/10.3892/ijo.2016.3523 [PubMed]
- 35. Bodmer M, Becker C, Meier C, Jick SS, Meier CR. Use of metformin is not associated with a decreased risk of colorectal cancer: a case-control analysis. Cancer Epidemiol Biomarkers Prev. 2012; 21:280–86. https://doi.org/10.1158/1055-9965.EPI-11-0992-T [PubMed]
- 36. Chen C, Sun P, Ye S, Weng HW, Dai QS. Hypertension as a predictive biomarker for efficacy of bevacizumab treatment in metastatic colorectal cancer: a meta-analysis. J BUON. 2014; 19:917–24. [PubMed]
- 37. Cai J, Ma H, Huang F, Zhu D, Bi J, Ke Y, Zhang T. Correlation of bevacizumab-induced hypertension and outcomes of metastatic colorectal cancer patients treated with bevacizumab: a systematic review and meta-analysis. World J Surg Oncol. 2013; 11:306. https://doi.org/10.1186/1477-7819-11-306 [PubMed]
- 38. Edge SB, Compton CC. The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol. 2010; 17:1471–674. https://doi.org/10.1245/s10434-010-0985-4 [PubMed]
- 39. Dupont WD, Plummer WD
Jr . Power and sample size calculations for studies involving linear regression. Control Clin Trials. 1998; 19:589–601. https://doi.org/10.1016/S0197-2456(98)00037-3 [PubMed]



