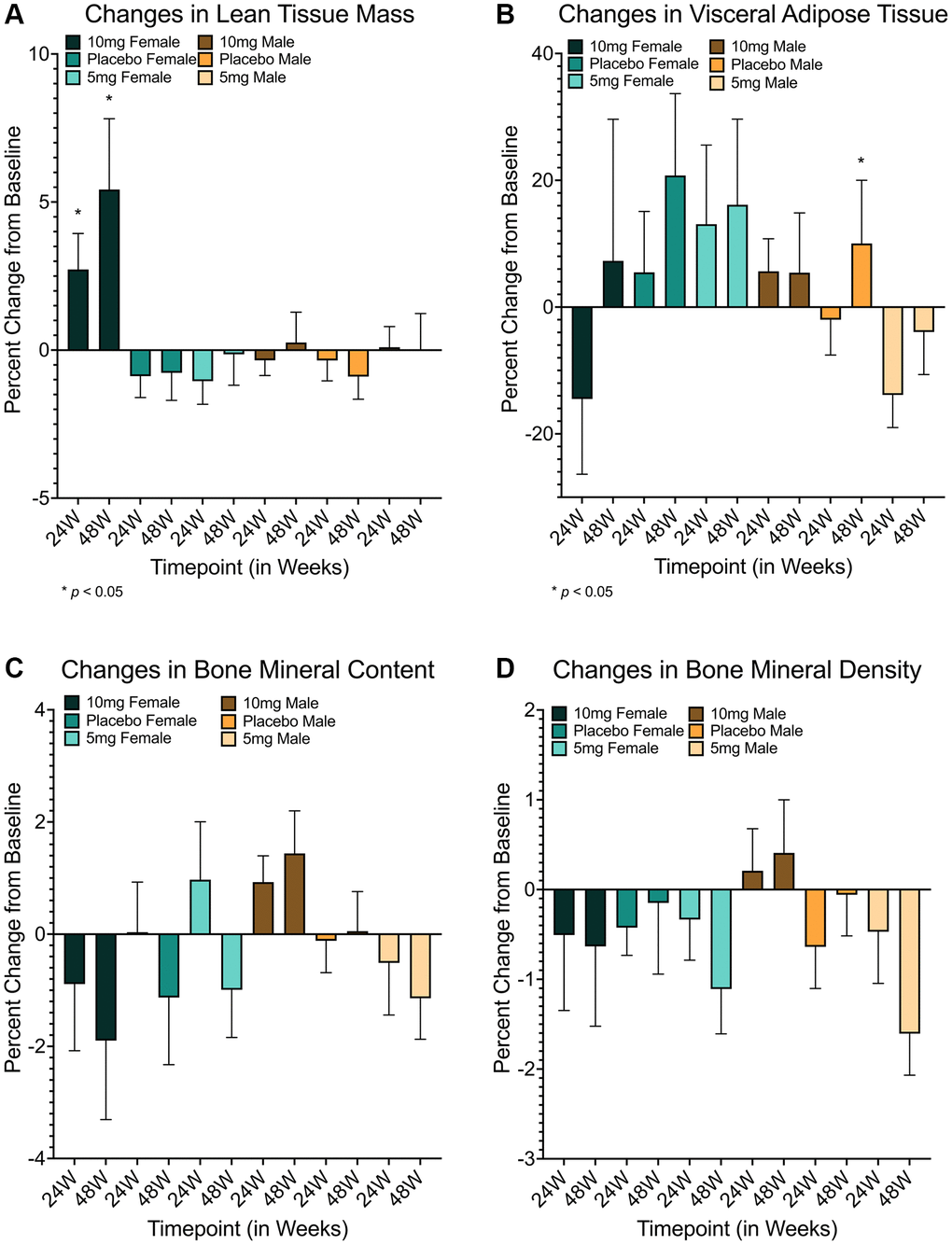
Figure 1.Changes in body composition measures in response to rapamycin use. Females using 10 mg of rapamycin had significant improvements in lean tissue mass at 24 and 48 weeks relative to both placebo (24 week: md = 3.60472 (95% CI = 0.0913–7.1182), p = 0.043; 48 week: md = 6.194 (95% CI = 0.8773–11.5105), p = 0.018) and 5mg groups (24 week: md = 3.774 (95% CI = 0.3271–7.2212), p = 0.028; 48 week: md = 5.565 (95% CI = 0.5311–10.5979), p = 0.026) (A). Improvements in visceral adiposity (measured by VAT) were clear for males in the 5 mg cohort relative to the 10 mg cohort (md = -19.520 (95% CI = −37.6513–−1.3893), p = 0.031) but not placebo (md = −11.866 (95% CI = −30.4152–6.6813), p = 0.362) at 24 weeks, but reverted to non-significance after 48 weeks (B). While no other measures showed significant differences (C, D), trending differences were observed in BMC for males at 48 weeks in 10 mg versus 5 mg groups (md = 2.580 (95% CI = −0.0600–5.2198), p = 0.057) but not placebo (md = 1.383 (95% CI = −1.1092–3.8757), p = 0.527) (C). md = mean difference, *=p ≤ 0.05. Error bars represent standard error of the mean.