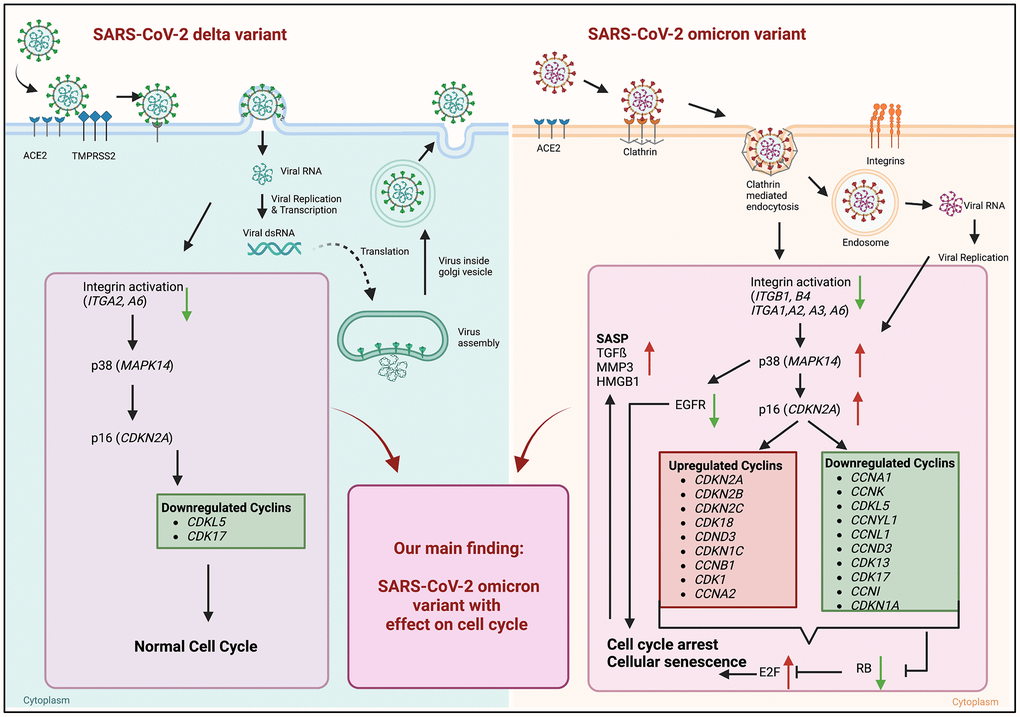
Figure 5.Graphical abstract of known differences of SARS-CoV-2 delta and omicron variant entry and findings of our study obtained from mRNA sequence data of 24 h post infection. Schematic overview of entry mechanisms of SARS-CoV-2 delta and omicron variants [5]. Delta variant (right panel) uses cell surface entry, by ACE2 and the protease TMPRSS2. Own data indicate a downregulation of the integrin activation without affecting p38 and p16 expression resulting in normal cell cycle. Omicron variant (right panel) prefers to use the clathrin-mediated endocytosis (CME) and cathepsin L as protease. Our results suggest the expression and activation of integrins (ITGB1, ITGB4, ITGA1, ITGA2, ITGA3, ITGA6) resulting an increase in p38 and p16. That increase in central kinases affects several cyclins, which in turn downregulates the retinoblastoma, increases the E2F transcription factors and results in cell cycle arrest and cellular senescence. Additionally, these changes lead to an increase in senescence-associated secretory phenotype (SASP). Thus, our findings indicate an influence of the altered cell entry mechanism of the omicron variant on the cell cycle. Created with https://www.BioRender.com.