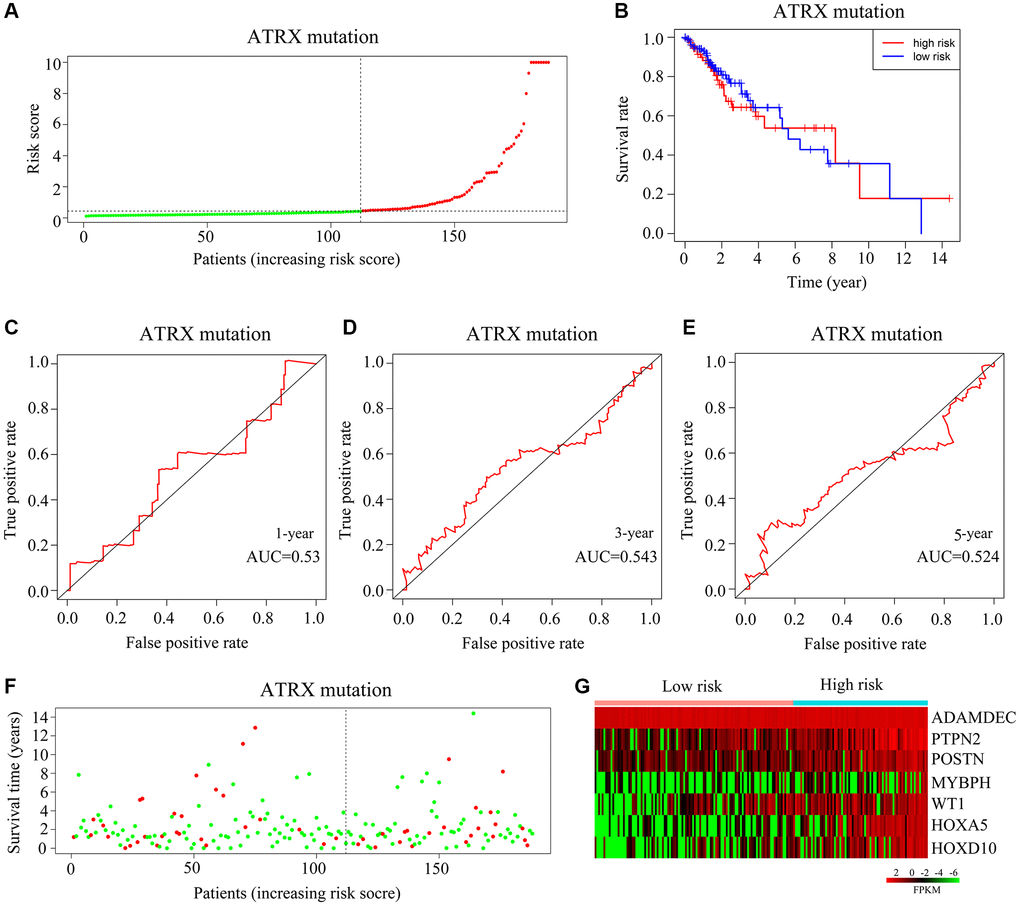
Figure 7.Validation of the applicability of the risk model in ATRX-mt glioma patients from TCGA. (A) ATRX-mt glioma patients from TCGA were divided into high- and low-risk-score groups based on the median risk score. (B) Survival differences between the high- and low-risk-score groups of ATRX-mt glioma patients from TCGA. (C–E) Prognostic value of the risk model for the one-, three- and five-year survival of ATRX-mt glioma patients from TCGA. (F) Deaths of ATRX-mt glioma patients from TCGA in the high- and low-risk-score groups (green dots represent living cases; red dots represent dead cases). (G) Expression of HOXA5, PTPN2, WT1, HOXD10, POSTN, ADAMDEC1 and MYBPH in ATRX-mt glioma tissues from the high- and low-risk-score groups.