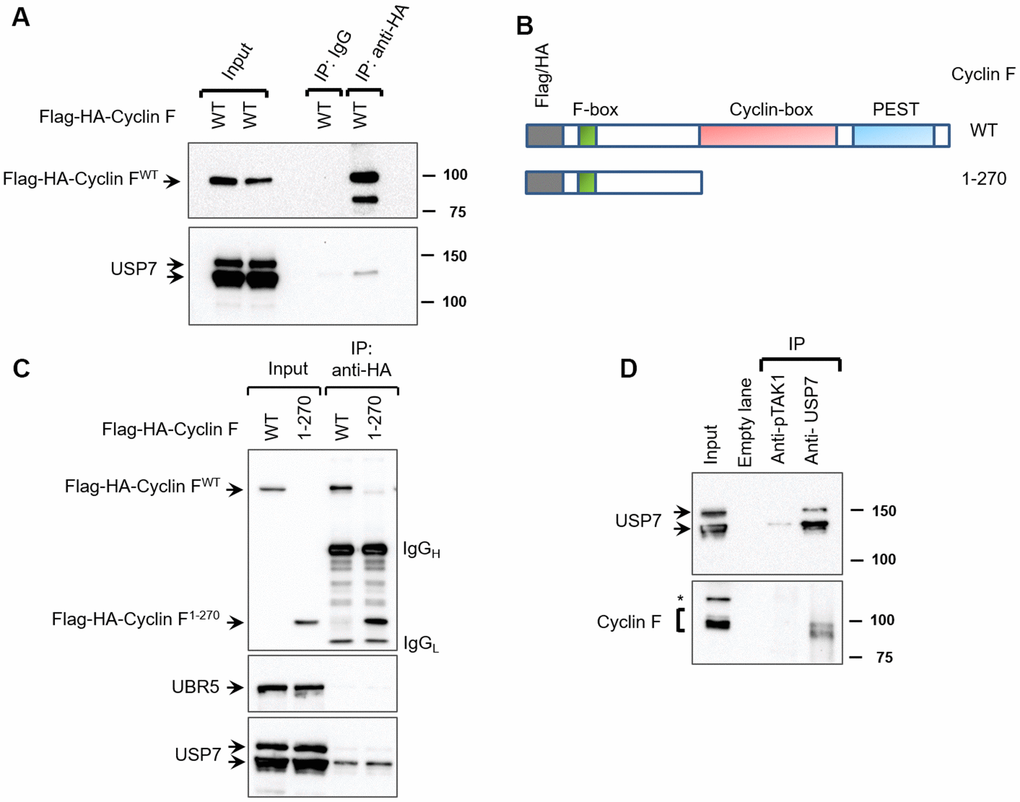
Figure 1.Cyclin F interacts with USP7. (A) HEK-293T cells transfected with Flag-HA-Cyclin F were lysed and immunoprecipitated with anti-HA antibody or non-specific mouse immunoglobulin (IgG) as loading control. Immunocomplexes were immunoblotted as indicated. (B) Schematic representation of cyclin F WT and cyclin F1-270 truncated mutant, highlighting the F-box, cyclin-box and PEST regions. (C) HEK-293T cells transfected with Flag-HA-Cyclin F WT or Flag-HA-Cyclin F1-270 were immunoprecipitated and immunoblotted as indicated. (D) Endogenous USP7 was immunoprecipitated from HEK-293T cell extracts, with anti-USP7 antibody or an unrelated, anti-p-TAK1 antibody as a loading control (denoted as control). Immunocomplexes were immunoblotted as indicated. Asterisk denotes non-specific band.