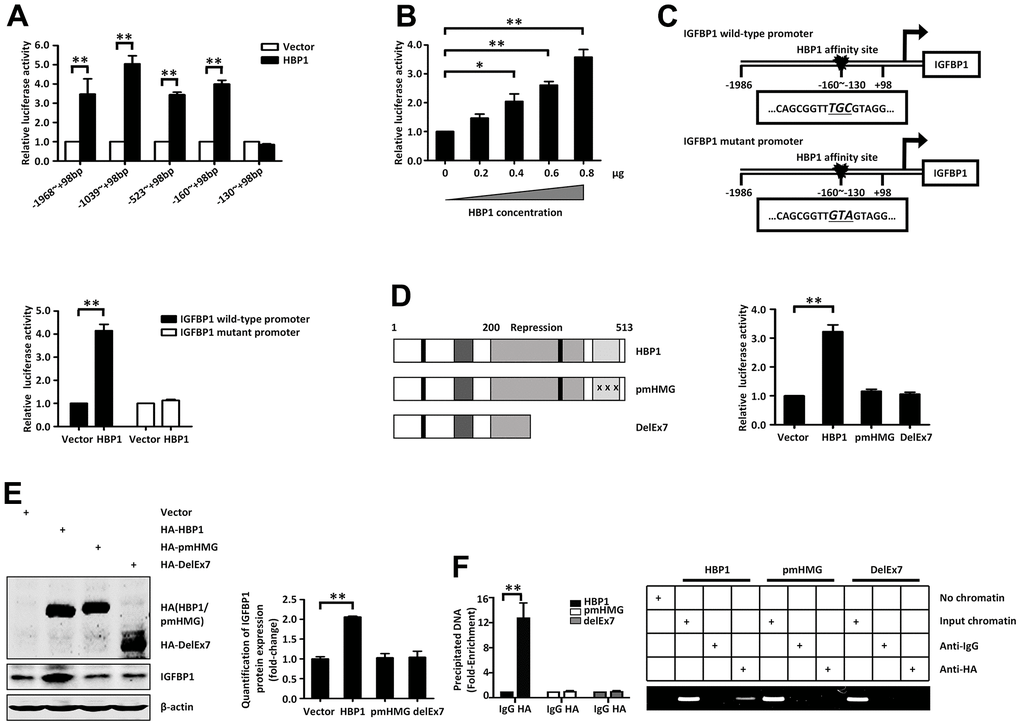
Figure 4.Insulin and palmitic acid reduce the protein expression of HBP1 and the binding of HBP1 to the IGFBP1 promoter. (A) The relative luciferase activities of HBP1 on IGFBP1 promoters of different lengths. 293T cells were co-transfected with the indicated length of IGFBP1 promoter and HA-HBP1 plasmid. Luciferase activities were determined 24 hours after transfection and were analyzed from four separate experiments. (B) HBP1 enhances IGFBP1 promoter activity in a dose-dependent manner. 293T cells were co-transfected with full-length IGFBP1 promoter and different doses (0, 0.2, 0.4, 0.6, 0.8 μg) of HA-HBP1 plasmid. The luciferase activities were analyzed as the means ± S.D. from four separate experiments. (C) The integrity of affinity sites is indispensable for HBP1 up-regulating IGFBP1 promoter in vivo. The schematic diagrams of the wild-type IGFBP1 promoter and its mutant promoter are shown in top panel. The mutant promoter of IGFBP1 contains four point mutations at the HBP1 affinity site (TCAA to CTGG) in comparison with wild-type IGFBP1 promoter. The relative luciferase activities of HBP1 on wild-type and mutant IGFBP1 promoter were analyzed from four separate experiments and shown in bottom panel. (D) Wild-type HBP1 rather than its mutants activates IGFBP1 promoter activities. Schematic diagrams of wild-type HBP1 and its associated mutants are shown in left panel. 293T cells were co-transfected with wild type IGFBP1 promoter and HBP1 or its mutant plasmids. The luciferase activities were analyzed from four separate experiments (right panel). (E) Expression of exogenous HBP1 increases IGFBP1 protein level. 293T cells were transfected with HBP1 and its associated mutants. The protein level of HA (HA-HBP1, HA-pmHMG and HA-delEx7) and IGFBP1 was measured by Western blotting. β-actin was used as a loading control. Quantification was normalized to β-actin. (F) The combination of HBP1 and endogenous IGFBP1 promoter depends on the HMG domain of HBP1. ChIP assays were performed to verify the binding of exogenous HBP1 to the endogenous IGFBP1. 293T cells were transfected with HA-HBP1 or HA-pmHMG or HA-DelEx7. The region from position -224 to -6 contains the HBP1 affinity site and was analyzed by specific PCR (right panel) and Realtime-PCR (left panel). Anti-HA antibody was used in the indicated lanes. Data were the mean ± SD by a two-tail, unpaired Student’s t-test. *, p<0.05. **, p<0.01.