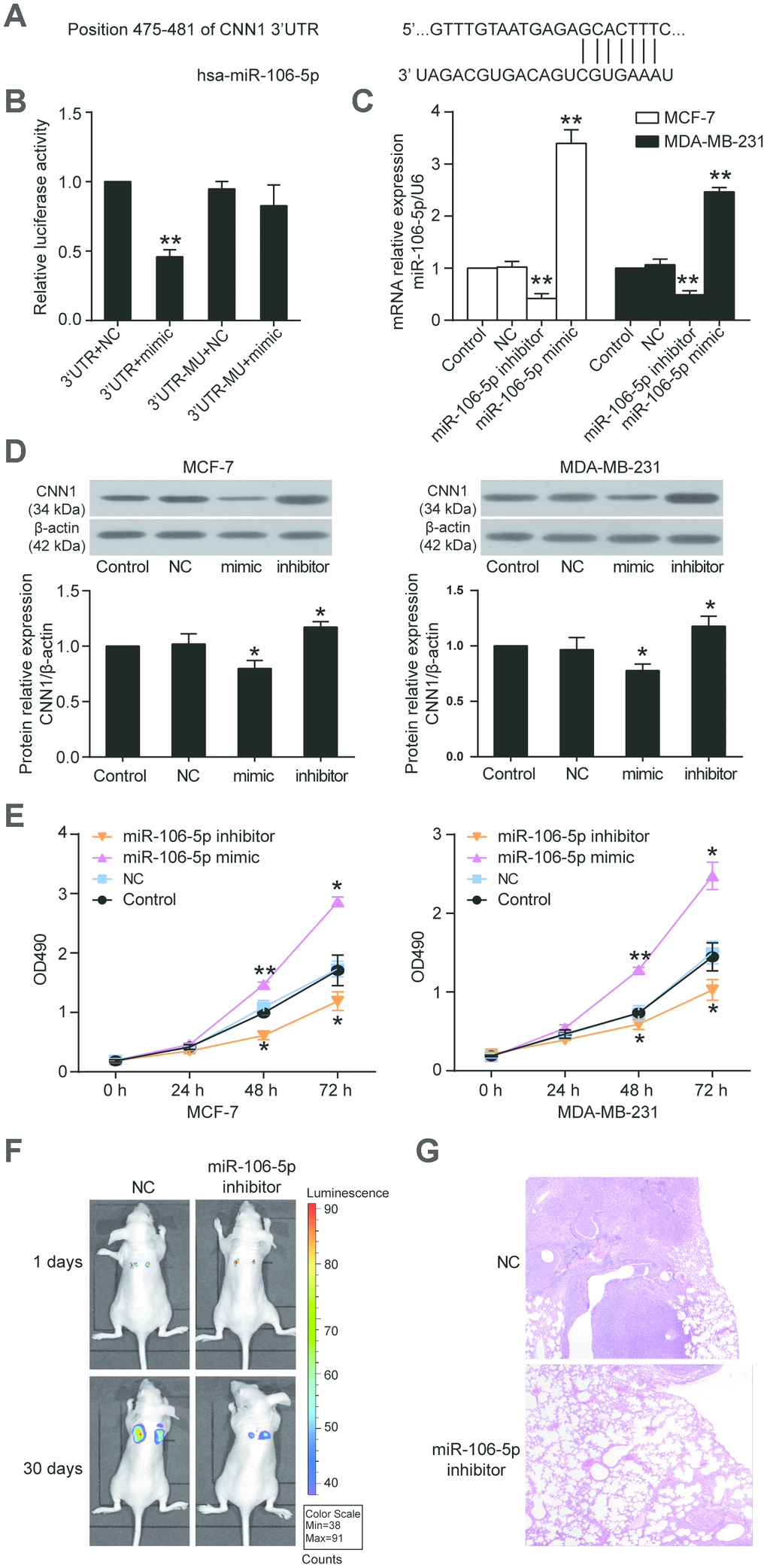
Figure 7.CNN1 was the target gene of miR-106b-5p, and the miR-106b-5p promoted cell proliferation. (A) The CNN1 3’UTR contained a binding site of miR-106b-5p by miRDB prediction. (B) The dual-luciferase reporter assay revealed the interaction between CNN1 3’UTR and miR-106b-5p. The HEK293 cells were co-transfected with the miR-106b-5p mimic and CNN1, or miR-106b-5p mimic and mutated CNN1. 3’UTR, wild-type CNN1 containing 3’UTR binding site. mimic, miR-106b-5p mimic. MU, mutated CNN1 without the 3’UTR binding site. NC, negative control. **P<0.001 vs. 3’UTR+NC. (C) The qRT-PCR was performed to detect the transfection efficiency of miR-106b-5p mimic and inhibitor in MCF-7 and MDA-MB-231 cells. (D) The miR-106b-5p mimic successfully inhibited CNN1 expression, while the miR-106b-5p inhibitor successfully upregulated CNN1 expression. The CNN1 protein expression was examined by immunoblotting assay after upregulation or downregulation of miR-106b-5p. mimic, the cells were transfected with miR-106b-5p mimic. inhibitor, the cells were transfected with miR-106b-5p inhibitor. (E) The miR-106b-5p mimic enhanced cell proliferation, and the miR-106b-5p inhibitor suppressed cell proliferation. The CCK8 assay was performed to detect the cell proliferation after transfection of miR-106b-5p mimic or inhibitor for 0 h, 24 h, 48 h, and 72 h. *P<0.05 vs. Control and **P<0.001 vs. Control. (F) Representative live bioluminescence images from mice treated with MCF-7 cells transfected with negative control or miR-106b-5p inhibitor. (G) Representative hematoxylin and eosin (H&E)-stained lung sections from mice treated with MCF-7 cells transfected with negative control or miR-106b-5p inhibitor.