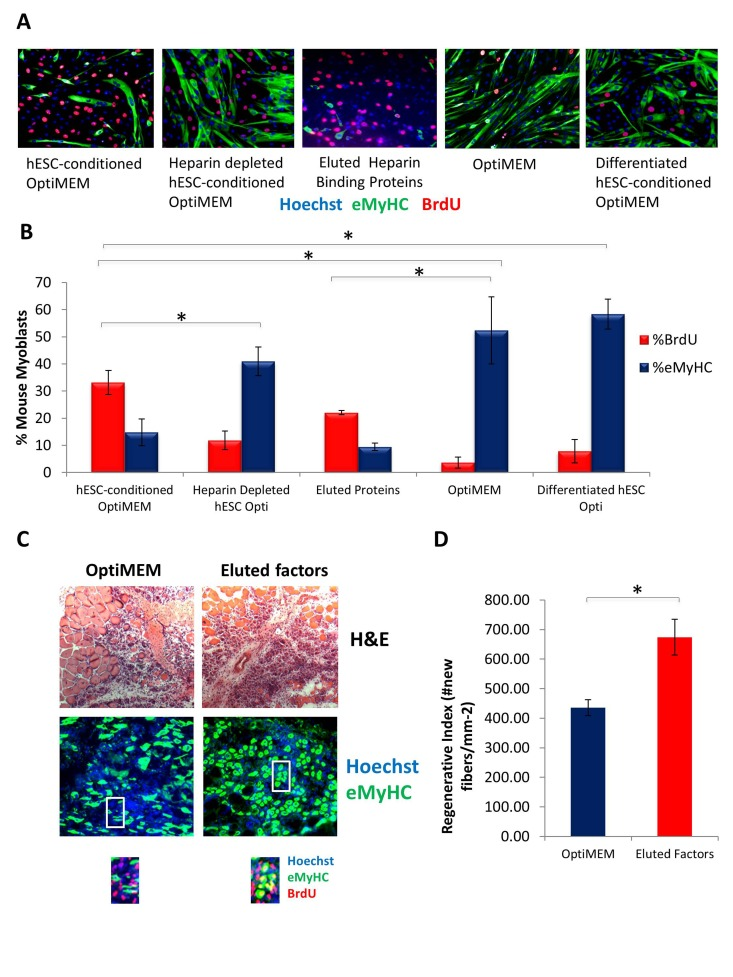
Figure 5.Pro-regenerative Embryonic Factors Contain Heparin Binding Domains(A) Primary Mouse Myoblasts were cultured for 24 hours in 50% fusion/differentiation medium + 50% of the specified medium. A 2 hour BrdU pulse was performed before cell fixation to label proliferating cells. Immuno-fluorescence was performed for eMyHC (green) and BrdU (red), with Hoechst (blue) labeling all nuclei. Representative images are shown. (B) Proliferation and differentiation of fusion-competent myoblasts were quantified by cell scoring in 25-100 random fields of each condition using an automated imager and MetaXpress cell scoring software. Results are displayed as the mean percent of BrdU+ or eMyHC+ proliferating or differentiating cells +/−SD, respectively. N=6 *P< 3×10^−45 for hESC-conditioned Opti-MEM compared to differentiated hESC-conditioned Opti-MEM, and hESC-conditioned Opti-MEM compared to Opti-MEM. *P<0.005 for hESC-conditioned Opti-MEM compared to heparin depleted hESC-conditioned Opti-MEM, and hESC-conditioned Opti-MEM compared to differentiated hESC-conditioned OptiMEM. *P< 5×10^−7 for hESC-conditioned Opti-MEM compared to Opti-MEM, and Eluted Proteins compared to Opti-MEM. (C) Old Tibialis Anterior muscles were injured with cardiotoxin (see Methods). Heparin bound and eluted protein or vehicle control (Opti-MEM) were injected into sites of injury on Day 0 and Day 2. BrdU was injected (intraperitoneal) at 3 days post injury to label proliferating, fusion-competent myoblasts. Animals were sacrificed and muscle was collected 5 days post injury. Cryosections (10 ?m) were analyzed by hematoxylin/eosin (H&E) staining and immunostaining for embryonic myosin heavy chain (eMyHC, shown in green) and BrdU incorporation (shown in red). Hoechst stains nuclei (blue). As shown by representative images, the regenerative outcome of old muscle given eluted factors was significantly improved as compared to old muscle given Opti-MEM vehicle control, based on significantly diminished scar tissue formation, larger and more dense de novo myofibers and an increase in the numbers of eMyHC+ myofibers with centrally-located BrdU+ nuclei that replaced the damaged tissue. (D) Regeneration of old mouse Tibialis Anterior 5 days post injury, that received eluted factors or vehicle, was quantified from muscle sections, and is presented as the number of newly regenerated myofibers per square millimeter of injury site. Error bars indicate SD, n=3 mice per group. *P <0.02 between old given eluted factors and old given vehicle control.