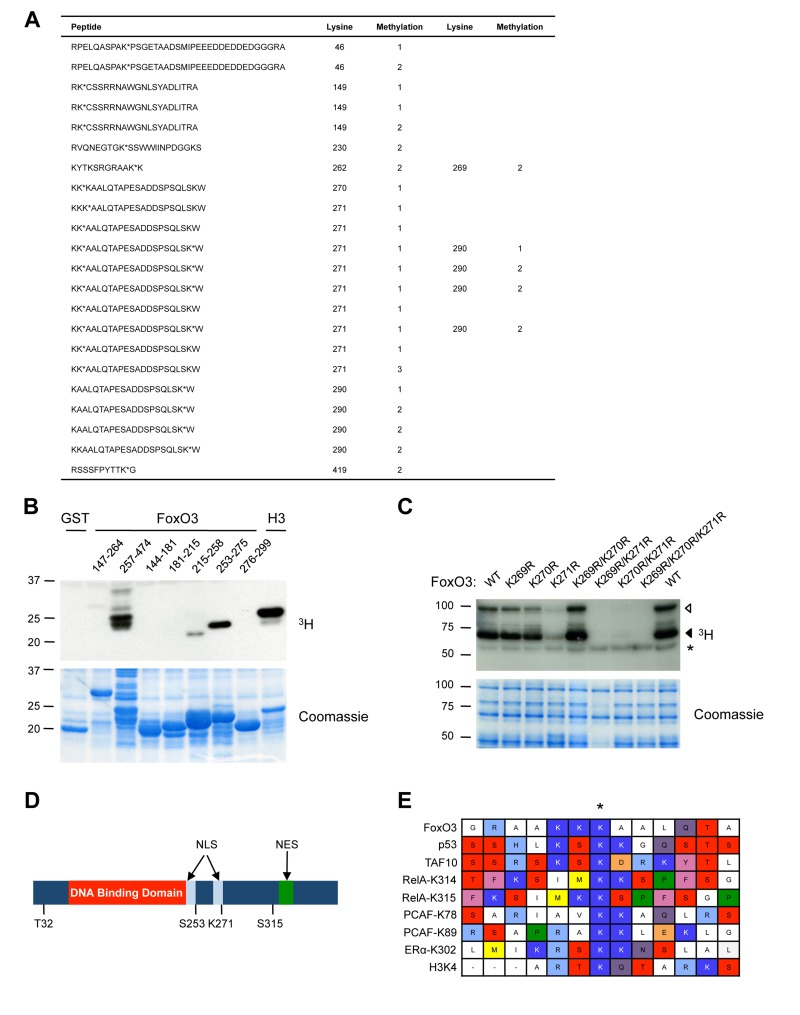
Figure 2.Set9 mono-methylates FoxO3 at K271 in vitro(A) Tandem mass spectrometry on in vitro methylated full-length FoxO3. Peptides containing methylated lysines are shown and the number of the methylated lysine in the human FoxO3 amino acid sequence is indicated. Methylated lysines are followed by an *. The type of methylation (mono-, di-, or tri-) is indicated by 1, 2, or 3, respectively. (B) Deletion analysis to map the region of FoxO3 containing the methylation site. In vitro methylation of overlapping fragments spanning the N-terminal domain of FoxO3 by Set9. (C) Methylation of FoxO3 WT or mutants of specific lysine residues. Each mutant was made in the context of the GST-FoxO3 protein (amino acid 1-525). ◁: FoxO3, ◀: FoxO3 degradation product, *: Set9 auto-methylation. (D) Location of K271 compared to other domains and PTMs of FoxO3. Listed are Akt phosphorylation sites (T32, S253, and S315), DNA binding domain, and NLS and NES (nuclear export sequence). K271 is the final amino acid in the second part of the bipartite FoxO3 NLS. (E) Alignment of the region surrounding the residues methylated by Set9 in a series of known Set9 substrates.