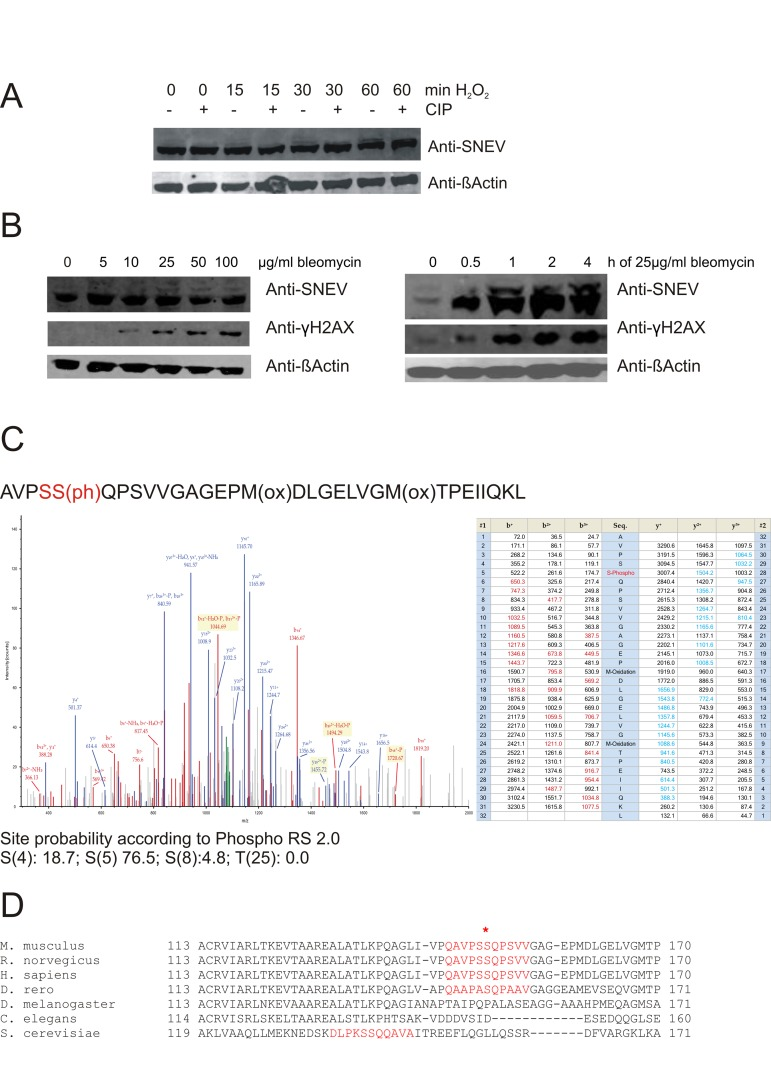
Figure 1.Upon oxidative stress, SNEV is detected as double band, probably representing a phosphorylated species(A) Upon treatment with hydrogen peroxide, additional bands of higher molecular weight were detected with anti-SNEV antibody. These bands disappear upon incubation with phosphatase. (B) Similarly, upon bleomycin treatment of fibroblasts, we detected an additional band with anti-SNEV antibody, which increased in a dose-and time-dpendent manner. Fibrolblasts were incubated with 0, 5, 10, 25, 50 or 100μg/ml bleomycin for 1hour (left panels) or with 25μg/ml bleomycin for 0, 0.5, 1, 2, 4 hours (right panels), scraped on ice in 2x SDS loading dye and subjected to Western Blotting with anti-SNEV antibody. Anti-β-actin was used to ensure equal loading. Anti-γH2AX antibody was used to confirm that the treatment induces DNA damage. (C)Collision-induced dissociation spectrum of the SNEV peptide AVPSS(ph)QPSVVGAGEPM(ox)DLG Indeed, a phosphorylation was detected and assigned to S149 with a probability of 76.5% (Fig. 1C). While the consistence of three different spectra with different m/z ratios underscores the correct assignment of the phosphorylation to this site, the remaining uncertainty comes from the presence of 3 serine residues within a quite long peptide. Together with the problem of lack of trypsin or chymotrypsin proteolytic sites near these serines, a better proof of S149 as the really phosphorylated serine by mass spectrometry is hampered. (D) Sequence comparison of SNEV homologues in the putative ATM target site. The consensus sequence surrounding the phospho-SQ site on ATM substrates that are regulated by DNA damage is conserved in the SNEVamino acid sequences across different vertebrates, but not in non-vertebrates.