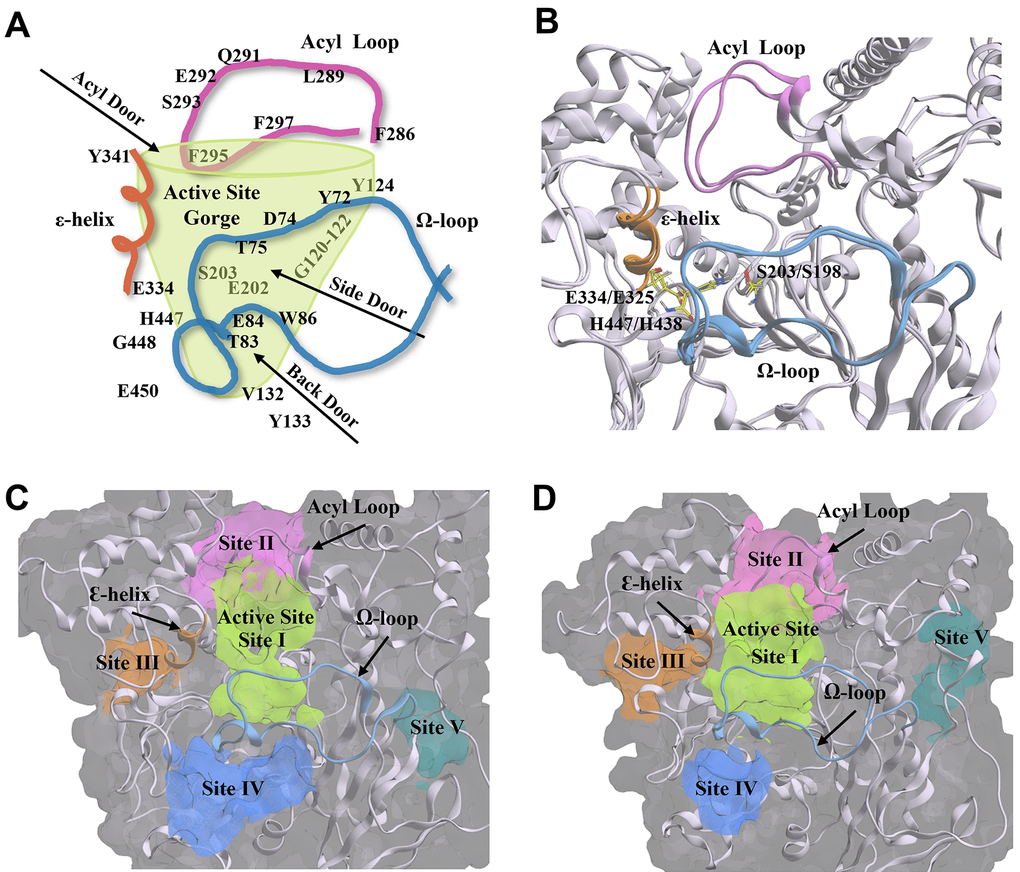
Figure 10.Structural components and amino acid residues that are essential for the catalytic activity of acetylcholinesterase (AChE) and butyrylcholinesterse (BChE). (A) 2D depiction of the entry/exit routes (“doors”) to the active site gorge of AChE. Amino acids are labelled and arranged according to their structural placement around the gorge, with those located on the opposite side to the viewing plane shown through the transparent green gorge wall. (B) AChE (PDB: 4M0E, 2.00 Å) [91] and BChE (BChE; PDB: 4TPK, 2.70 Å) [92] enzyme ribbon structures are overlayed to show structural conservation between the two enzymes. The catalytic triad residues are shown in yellow. (C, D) The five main compound binding site pockets (I-V) of AChE (C) and BChE (D) are identified as the enzyme active site gorge (lime green, I), pocket behind ChE acyl loop (pink, II), pocket behind ChE catalytic glutamate (orange, III), pocket behind ChE active site tryptophan (Back Door, blue, IV), and other binding pocket (teal, V). Structural features of AChE and BChE including the acyl loop (pink), Ω-loop (blue), and ε-helix (orange) are shown throughout each panel. Figures were generated using Microsoft PowerPoint for Microsoft 365 MSO (version 2409 build 18025.20104; Microsoft Corporation, Redmond, WA, USA) and Molecular Operating Environment 2022.02 (Chemical Computing Group ULC, Montreal, Quebec, Canada).