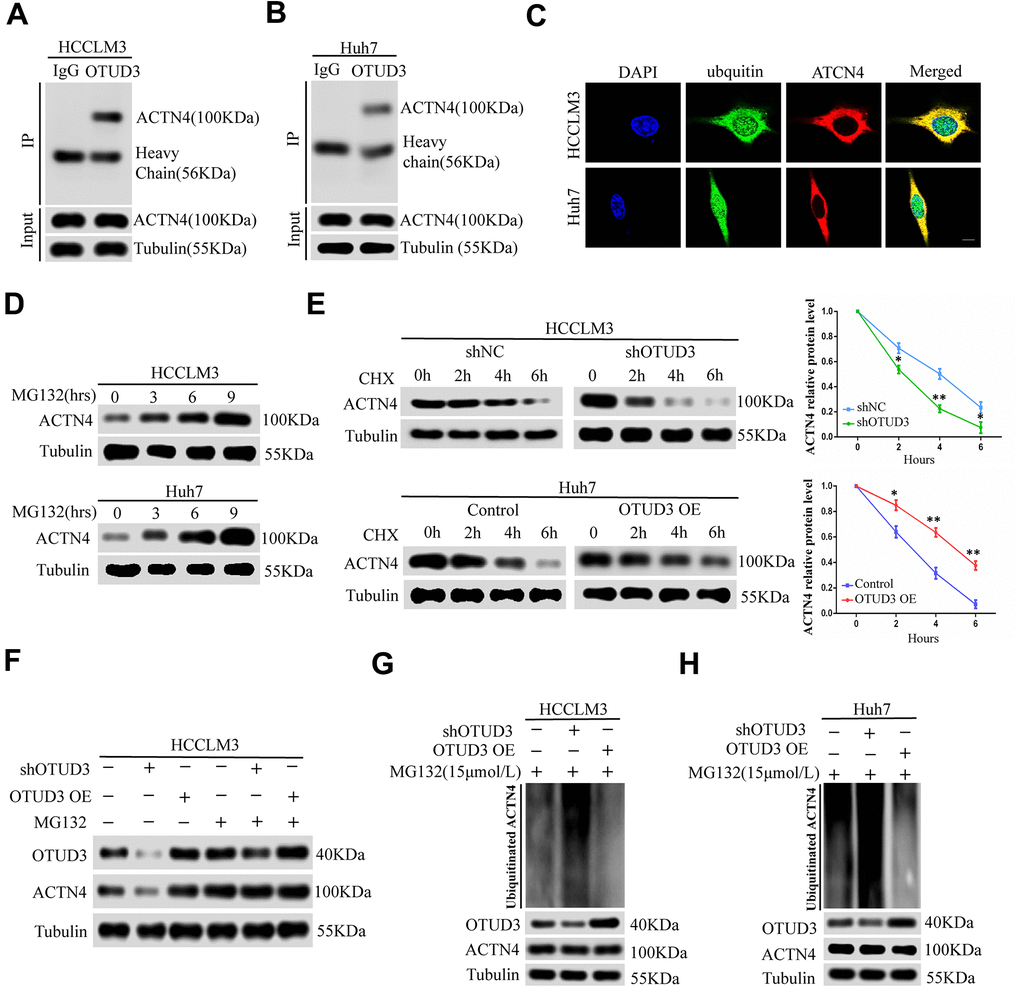
Figure 5.OTUD3 enhances the stability of ACTN4 through deubiquitylation. (A, B) co-IP experiments between endogenous OTUD3 and ACTN4 in HCCLM3 and Huh7 cells. ACTN4 was detected in the immunoprecipitation when the anti-OTUD3 antibody was used as bait. (C) Colocalization of OTUD3 and ACTN4 in HCCLM3 and Huh7 cells (Scale bar: 14μm). (D) HCCLM3 and Huh7 cells were treated with 15μM proteasomal inhibitor MG132 for the indicated time, and the levels of ACTN4 were then detected. (E) HCCLM3 cell transfected with OTUD3 shRNA or shNC together with stably OTUD3 overexpressing Huh7 cells and negative control were treated with 20μM cycloheximide (CHX). Cells were collected at different time points and were detected ACTN4 protein expression. (F) HCCLM3 cells with OTUD3 knockdown or OTUD3 overexpression were treated with MG132 (15μM). Cells were collected at 6 h and immunoblotted with the antibodies indicated. (G, H) the knockdown or upregulation of OTUD3 altered the ubiquitination of ACTN4 in both HCCLM3 and Huh7 cells. The cells in each group were treated with MG132 (15μM). The levels of ubiquitin-attached ACTN4 were detected by western blot analysis with ubiquitin (Ub) antibody.