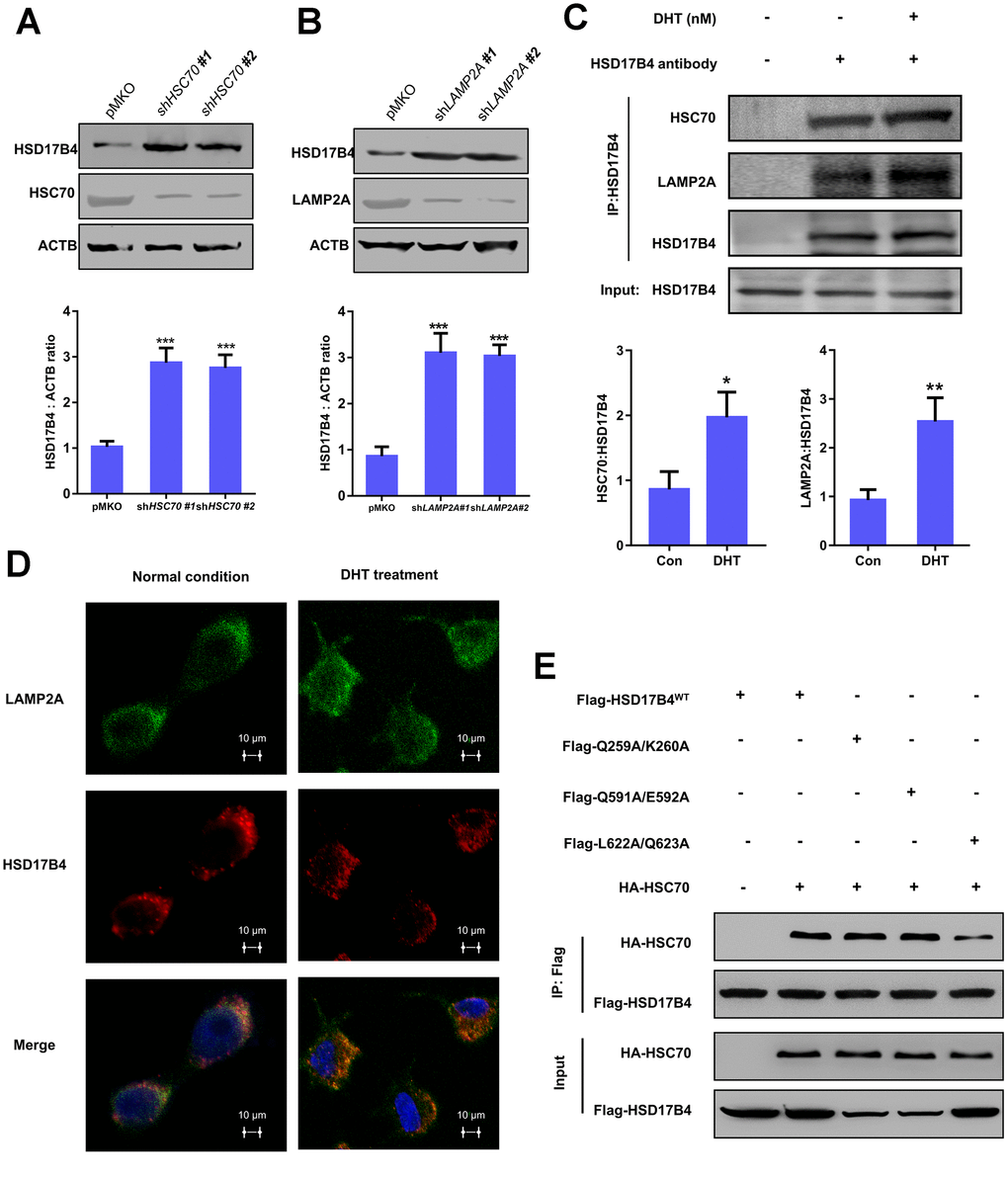
Figure 5.The degradation of HSD17B4 requires CMA. (A) HSC70 knockdown leads to accumulation of HSD17B4. HSC70 was transiently knocked down in LNCaP cells by shRNA. The knockdown efficiency and HSD17B4 protein level were determined by western blotting (upper panel). The HSD17B4 level was normalized against ACTB. **denotes P < 0.01. Error bars represent ±SD of triplicate experiments (lower panel). (B) LAMP2A knockdown leads to accumulation of HSD17B4. LNCaP cells stably expressed LAMP2A shRNA. The levels of HSD17B4 and LAMP2A proteins were determined by western blotting (upper panel). The relative HSD17B4 protein compared with ACTB was quantified. **denotes P < 0.01. Error bars represent ±SD of triplicate experiments (lower panel). (C) DHT promotes endogenous HSD17B4 binding with HSC70 and LAMP2A in LNCaP cells. LNCaP cells were cultured with or without DHT for 24 h before harvest. The interaction between endogenous HSD17B4 and HSC70 or LAMP2A was determined by coimmunoprecipitation and western blotting. (D) DHT enhances the interaction between HSD17B4 and LAMP2A. LNCaP cells were cultured with or without DHT for 24h as indicated and then paraformaldehyde fixed, blocked, and processed for double immunofluorescence with antibodies against LAMP2A (green) and HSD17B4 (red). Merged images of both channels are shown in the lower panel. Bar: 10 μm. (E) Identification of an HSC70 binding motif in HSD17B4. The binding between HSD17B4 mutants and HSC70 was analyzed by IP-western. Data are shown as the mean ± SD (n = 3) or typical photographs of one representative experiment. Similar results were obtained in three independent experiments. *p < 0.05, **p < 0.01, ***p <0.001.