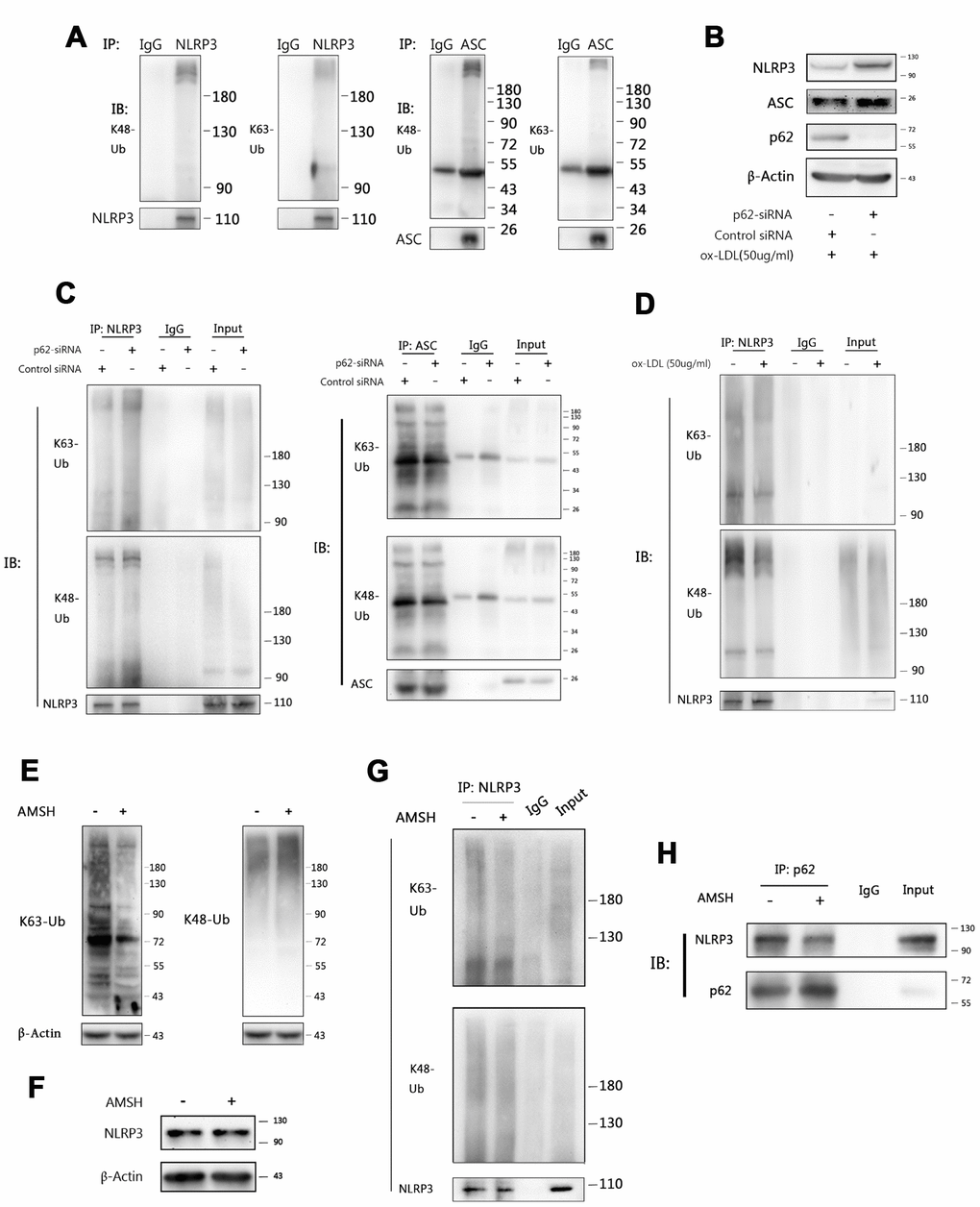
Figure 6.P62 binds to NLRP3 via the K63 polyubiquitin chains. (A) The immunoblot analysis of NLRP3 and ASC immunoprecipitates of Mφ stimulated with ox-LDLs (50 ug/ml) for 24 hours. (B) The immunoblot analysis of the total lysates of Mφ transfected with control siRNA or p62-siRNA for 24 hours, and subsequently stimulated with ox-LDLs (50 ug/ml) for another 24 hours. (C) The immunoblot analysis of NLRP3 (left) and ASC (right) immunoprecipitates of Mφ treated as described in (B). (D) The immunoblot analysis of NLRP3 immunoprecipitates of Mφ treated with or without ox-LDL (50 ug/ml) for 24 hours. (E) The detection for K63 (left) and K48 (right) polyubiquitin chains in the total foam-cell lysates pretreated with or without AMSH by immunoblotting. Prior to immunoblotting, 50 ul of ox-LDL stimulated Mφ lysates were administered with or without 1 ul of AMSH (AMSH concentration: 500 nM), and incubated at 37°C for 30 minutes. (F) The immunoblot analysis of NLRP3 and β-Actin in the total foam-cell lysates pretreated with or without AMSH (500 nM), as described in (E). (G) The NLRP3 immunoprecipitates obtained from ox-LDL stimulated Mφ lysates were treated with or without AMSH (500 nM), as described in (E), and subsequently subjected to immunoblotting. (H) The p62 immunoprecipitates obtained from foam-cell lysates were treated with or without AMSH (500 nM), as described in (E), and subsequently subjected to immunoblotting. All experiments were independently repeated for at least three times.