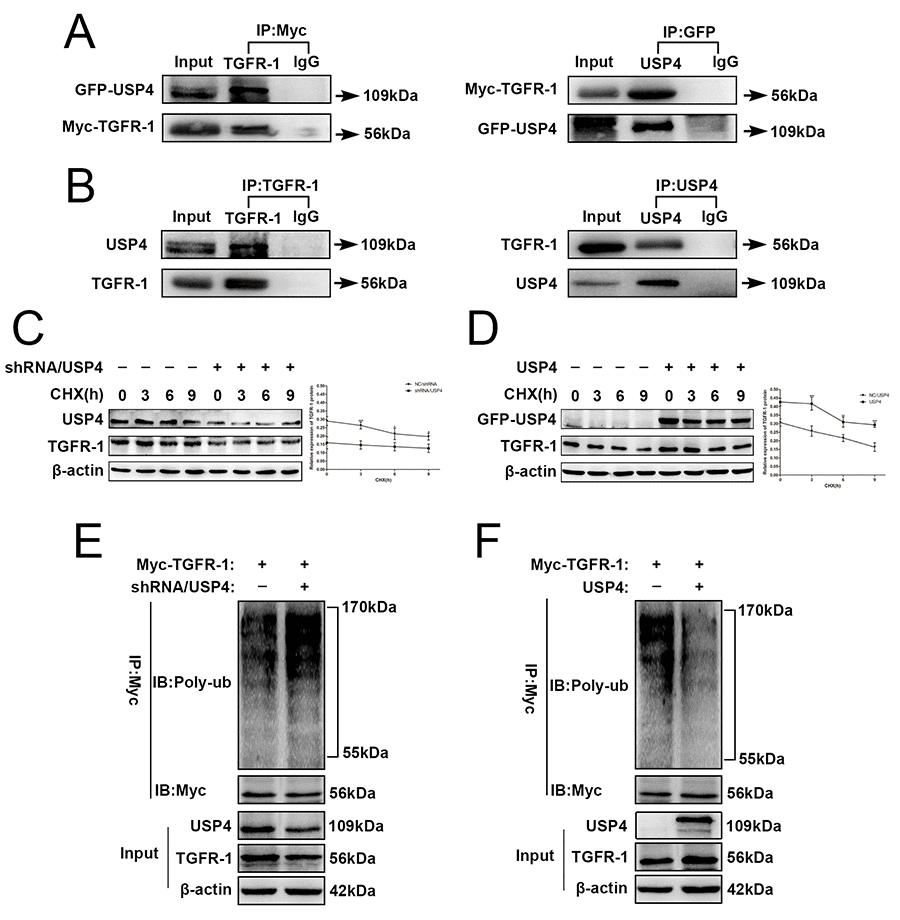
Figure 6.USP4 directly interacted with and deubiquitinated TGFR-1 in HCC cell. (A) USP4 interacted with TGFR-1 at exogenous levels. Immunoblotting analysis of lysates after immunoprecipitation from SK-Hep1 cells co-transfected with GFP-USP4 and Myc-TGFR-1. IgG was used as a negative control. (B) USP4 interacted with TGFR-1 at endogenous levels. Cell lysates from wild type SK-Hep1cells were immunoprecipitated with anti-USP4 or anti- TGFR-1 antibody, followed by immunoblotting with anti- TGFR-1 or anti-USP4 antibody, respectively. IgG was used as a negative control. (C) USP4 knockdown reduced the stability of TGFR-1 protein (* P < 0.05, *** P < 0.001). (D) USP4 overexpression elevated the stability of TGFR-1 protein (** P < 0.01, *** P < 0.001). SK-Hep1-shRNA/USP4 cells or SK-Hep1-USP4 cells were treated with protein synthesis inhibitor CHX (100 µg/mL) for the indicated times. Then, proteins were extracted and Western blotting was performed. (E) Effect of USP4 knockdown on the polyubiquitin levels of TGFR-1. (F) Effect of USP4 overexpression on the polyubiquitin levels of TGFR-1. SK-Hep1-TGFR-1 cells were co-infected with shRNA/USP4 or USP4. 48h later, cells were treated with proteasome inhibitor MG132 for 6 hours. Then, TGFR-1 was immunoprecipitated with anti-Myc antibody, and the polyubiquitination of TGFR-1 was detected by immunoblotting.