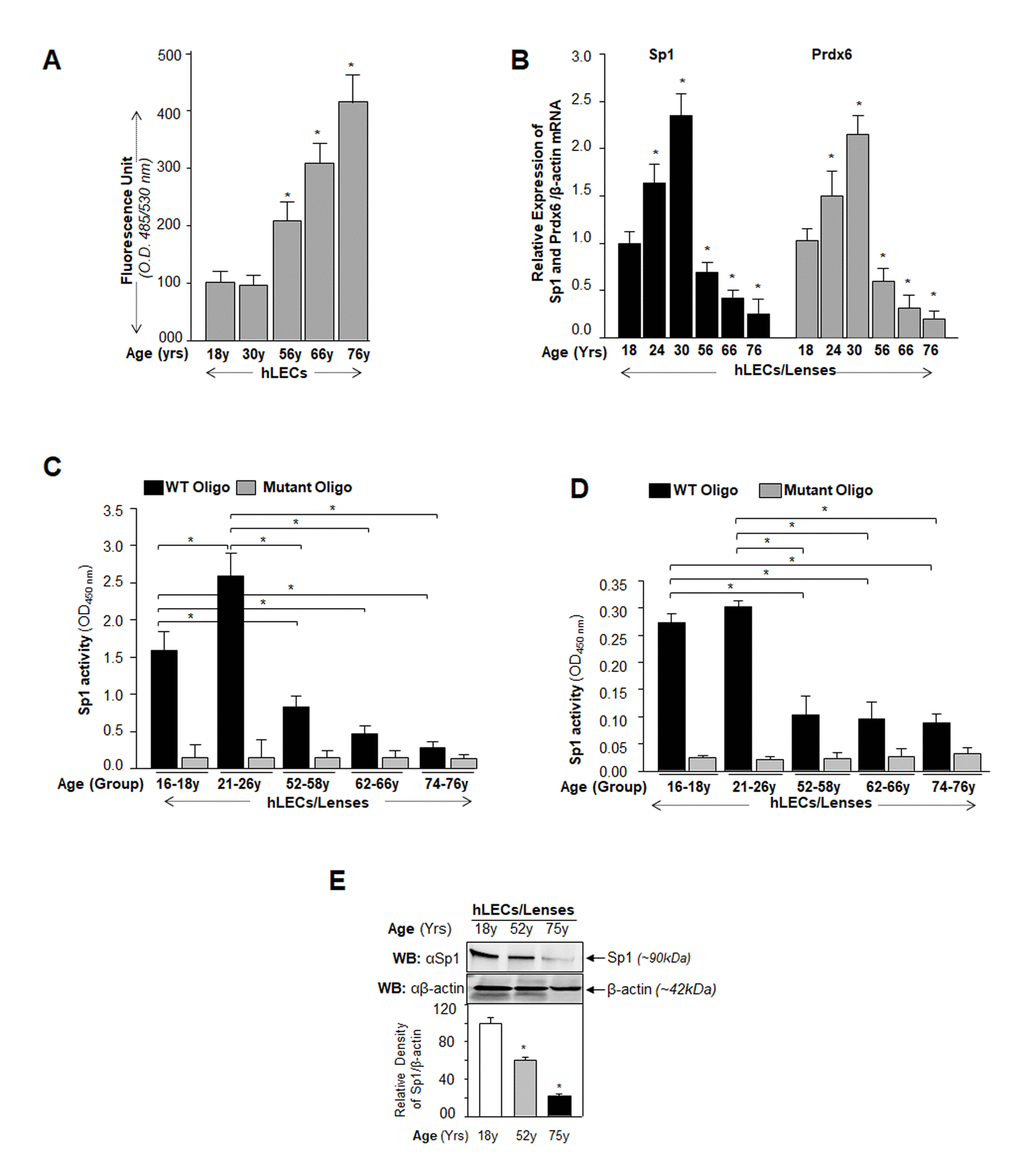
Figure 1.Aging human LECs/lenses showing elevated levels of ROS and progressive decline in Sp1 and Prdx6 expression connected to reduction in Sp1 activity. (A) ROS levels increased progressively in aging hLECs. Cells were cultured in 96 well plate (5000/well), and ROS were quantified using H2-DCF-DA dye assay as shown. Data represent the mean ± S.D. of two independent experiments. Younger (18y) vs aging samples; *p < 0.001. (B) Aging hLECs displayed progressive decline in levels of Sp1 and its target gene, Prdx6 mRNA. Total RNA was isolated from human LECs/ lenses of different ages as indicated and was processed for real-time PCR analysis with specific primers. The data represent the mean ± S.D. from three independent experiments. p values were determined for younger vs aging samples. *p < 0.001. (C) Aging/aged human lenses/LECs displayed significant loss of Sp1 activity. Nuclear extracts prepared from aging/aged hLECs/lenses were used for assay. LECs/lenses were divided into five age groups: 16-18y (n=6); 21-26y (n=6); 52-58y (n=8); 62-66y (n=8); 74-76y (n=8). Nuclear extracts containing equal amounts of protein were processed and assayed for Sp1 activity using a commercially available kit (Active motif) as described in Materials and Methods. The data represent the mean ± S.D. from three independent experiments. p values were determined for younger vs aging samples. *p < 0.001. (D) Nuclear extracts containing equal amounts of proteins were processed for Sandwich ELISA to measure the total Sp1 protein. Total Sp1 proteins were equalized with the O.D. of Sandwich ELISA and processed for Sp1 transactivation assay using a commercially available kit (Active motif) as ascribed in Materials and Methods. P value were determined for younger vs aging samples. *p < 0.001. (E) Aging hLECs showing significant loss of Sp1 protein. Cellular proteins were isolated from hLECs and human lenses of different ages as described in Materials and Methods section, and as indicated. An equal amount of protein was loaded onto SDS-PAGE, and immunoblotted using Sp1 antibody. Upper panel; expression levels of Sp1, Lower panel; membrane probed with β-actin antibody as loading/internal control. Each band of blot was quantified using densitometer shown below. Images are representatives from three independent experiments. P value were determined for younger vs aging samples. *p < 0.001.