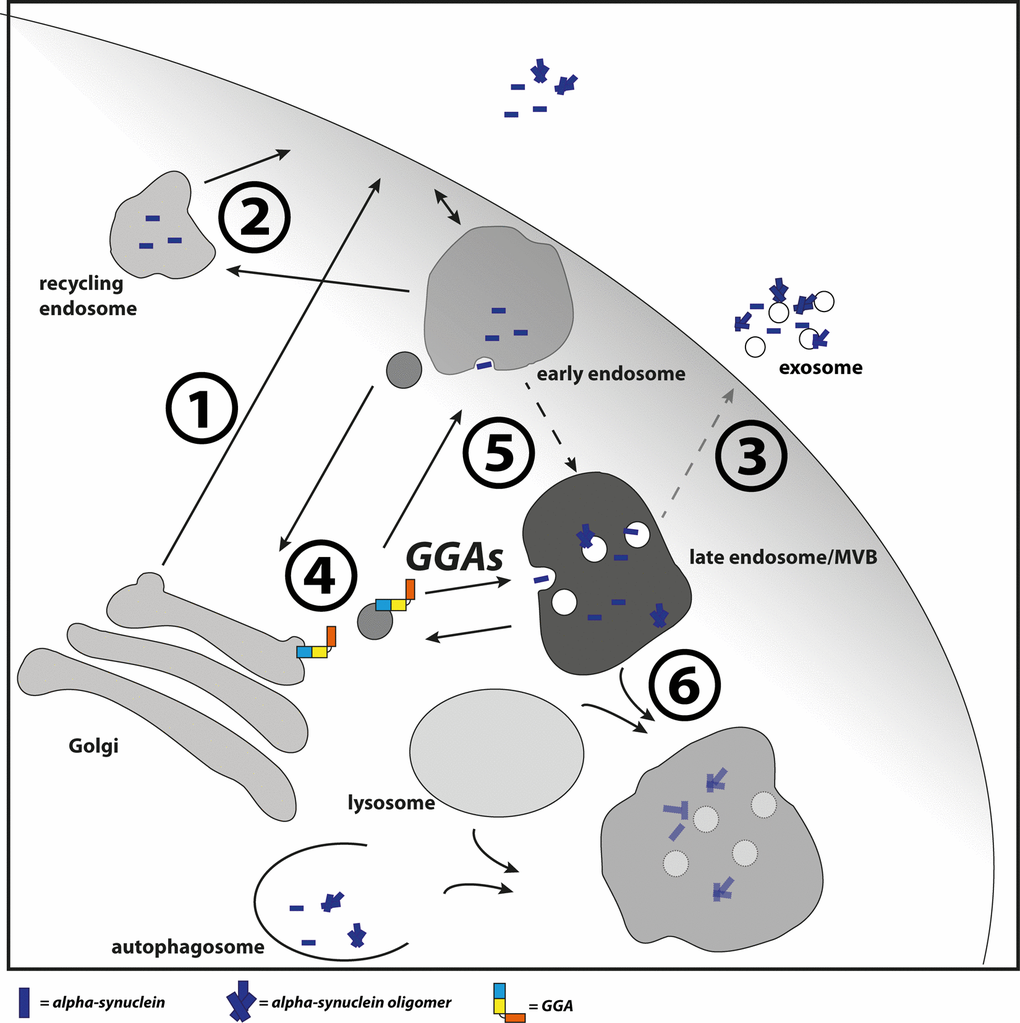
Figure 6.GGAs contribute to α-syn transport into the endolysosomal pathway. Different pathways have been described which contribute to α-syn secretion. This includes the secretory pathway (1), recycling pathway via early endosomes and recycling endosomes (2) and upon disturbed lysosomal degradation the release of MVB content at the plasma membrane (3). GGAs contribute to α-syn transport or occurrence of α-syn in the endolysosomal pathway. Intracellular α-syn aggregation and secretion can be triggered by different mechanisms. The D620N mutation in VPS35 identified in familial PD cases lead to VPS35 loss of function and mis-sorting of cathepsin D (4) and thereby to reduced lysosomal degradation of α-syn. GGAs might increase α-syn aggregation directly through enhanced transport into acidic environment or indirectly through enhanced residence in the acidic pH (5). In case of disturbed lysosomal function (6), α-syn oligomer loaded MVBs will fuse with the plasma membrane and release their content into the extracellular space (3).