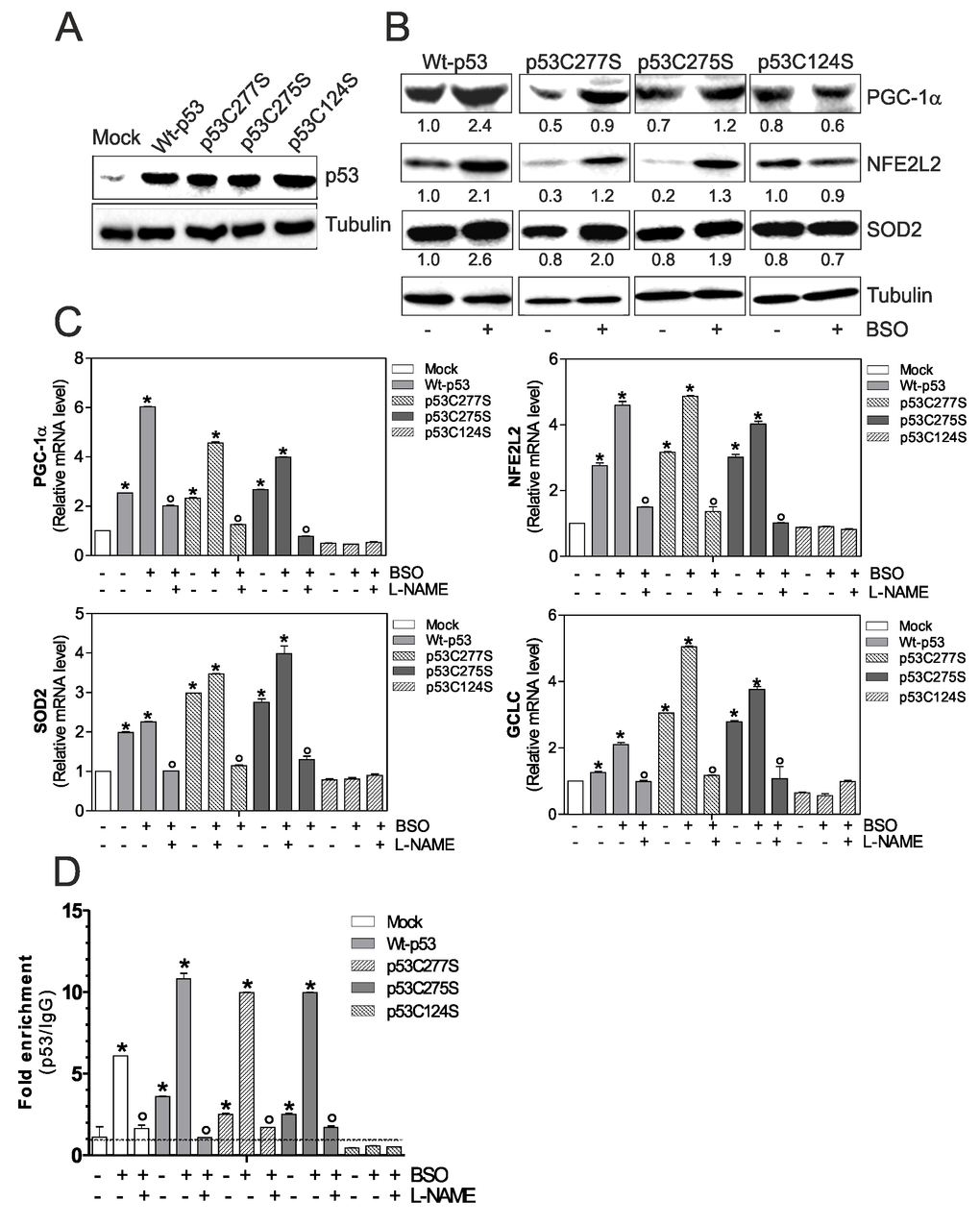
Figure 1.p53C124S mutant fails to induce NO/PGC-1α-mediated antioxidant pathway in C2C12 myoblasts. (A, B) C2C12 myoblasts were transfected with pcDNA3.1 vector containing cDNA for wild type p53 (Wt-p53), three single p53 mutants in DBD (p53C277S, p53C275S, p53C124S) or with empty vector (Mock). After 15 h from transfection, myoblasts were treated with 1 mM BSO for 24 h. Cells were lysed and 20 μg of proteins were loaded for Western blot analysis of p53, PGC-1α, NFE2L2 and SOD2. Tubulin was used as loading control. Numbers indicate the density of immunoreactive bands calculated using the Software Quantity one (Bio-Rad) and reported as the ratio of PGC-1α, NFE2L2 and SOD2/Tubulin. (C) L-NAME (100 μM) was added 1 h before BSO treatment (15 h) and maintained throughout the experiment. Total RNA was isolated and relative mRNA levels of PGC-1α, NFE2L2, SOD2 and GCLC were analyzed by RT-qPCR. Data are expressed as means ± S.D. (n=4; *p<0.001 vs Mock; °p<0.001 vs BSO-treated cells). (D) ChIP assay was carried out on cross-linked nuclei from Mock, Wt-p53, p53C277S, p53C275S and p53C124S cells using p53 antibody followed by qPCR analysis of p53RE. Dashed line indicates the value of IgG control. Data are expressed as means ± S.D. (n=3; *p<0.001 vs Mock; °p<0.05 vs BSO-treated cells). All the immunoblots reported are from one experiment representative of four that gave similar results.