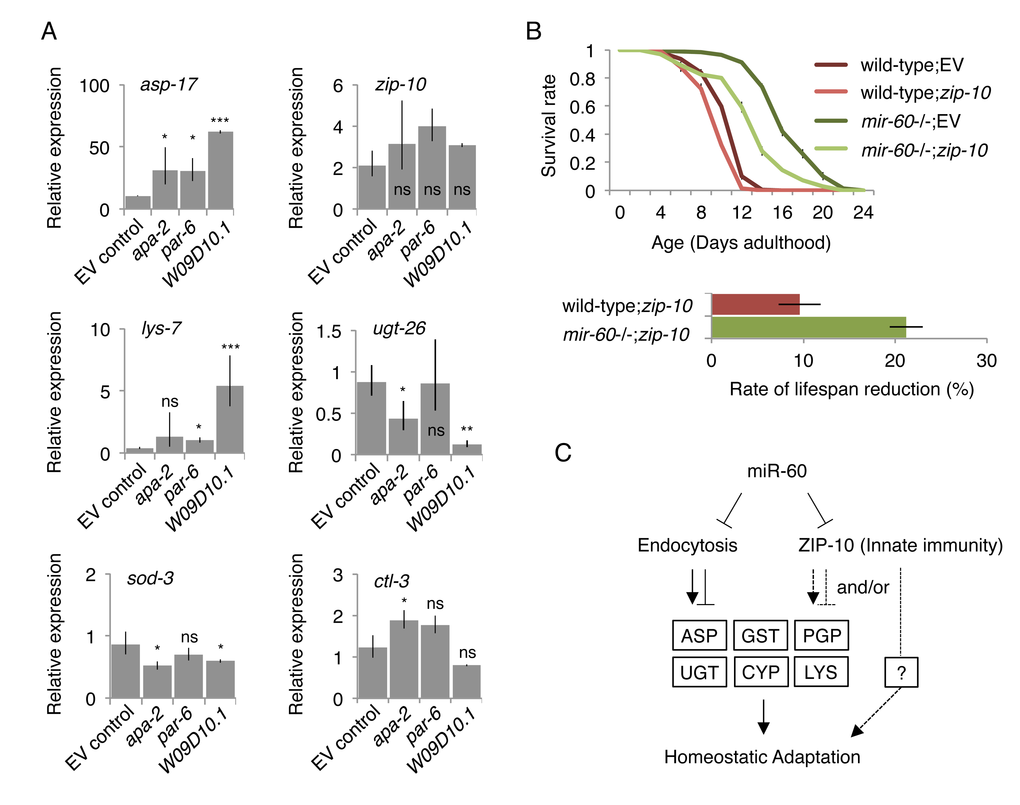
Figure 7.Inhibiting miR-60 target candidates abolish the mir-60 loss-induced gene expression changes and adaptive response against oxidative stress. (A) Expression was examined by qRT-PCR in the mir-60;spe-9 double mutant background, and the results were standardized by the expression level in the control spe-9 animals exposed to the empty vector control in RNAi. Error bars represent SE and p-values were calculated by paired t-test based on 3 independent trials of sample preparation: *p<0.1; **p<0.01; ***p<0.001. (B) Survival curves of wild-type and mir-60 mutant animals exposed to zip-10 RNAi are shown. A small bar graph below represents the rate of lifespan reduction of zip-10 RNAi-treated animals compared to those treated with the control RNAi. Error bars represent SE calculated from 3 replicates. The detailed results are available in Supplemental Fig 6. (C) A model illustrates the mechanism underlying mir-60 loss-induced adaptive response against chronic oxidative stress. Arrows and blunt arrows denote positive and negative interactions, respectively. miR-60 appears to directly modulate the activity of endocytosis machinery, which regulates downstream expression of genes, such as those encoding Aspartyl protease (ASP), P-glycoprotein (PGP), glutathione S-transferase (GST), UDP glucuronosyltransferase (UGT), cytochrome P450 (CYP) and lysozyme (LYS). ZIP-10, which also seems to be directly controlled by miR-60, promotes homeostatic adaptation possibly through mir-60 loss-responsive factors such as PGP, and/or unidentified factor(s) (shown by broken lines).