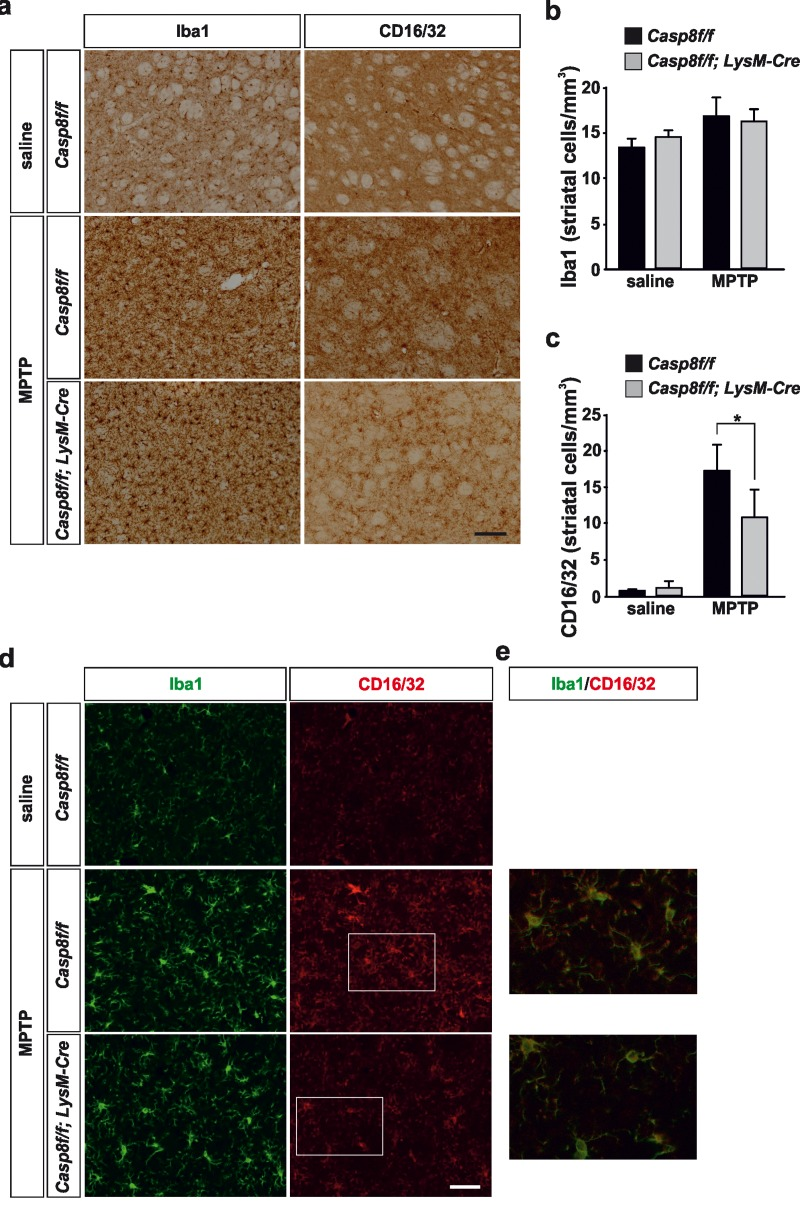
Figure 5.Microglial caspase-8 deficiency ameliorates MPTP-induced proinflammatory microglia activation in the striatumPanel (a) shows illustration of Iba1 and CD16/32-labeled microglia in the striatum in response to either saline or MPTP in Casp8fl/fl mice and CreLysMCasp8fl/fl mice. Injection of saline in Casp8fl/fl mice was not different from CreLysMCasp8fl/fl mice and hence only Casp8fl/fl mice saline is shown. Panels (b) and (c) show the stereological analysis of Iba1 (b) and CD16/32 (c) in the striatum in response to MPTP. Results are the mean ± SD of a minimum of four independent experiments and are expressed as number of cells per mm3. Statistical significance was calculated by analysis of variance followed by the least significant difference post hoc test for multiple range comparisons (p <0.05). Panel (d) shows illustration of dual immunofluorescence of Iba1 and CD16/32-labeled microglia in the striatum in response to either saline or MPTP in Casp8fl/fl mice and CreLysMCasp8fl/fl mice. Panel (e) show higher magnification photographs of dot boxes depicted in panel (d). Note the drastic changes in the microglia population in terms of Iba1-labeling in response to MPTP in both Casp8fl/fl mice and CreLysMCasp8fl/fl mice (a,d). Note the low levels of CD16/32 labeling in the unlesioned striatum (a, c, d) and how this marker is robustly up-regulated in response to MPTP in Casp8fl/fl mice (a, c, d). Also note how the MPTP-induced up-regulation of proinflammatory microglia in tems of CD16/32 is hindered in CreLysMCasp8fl/fl mice. Scale bar: a: 125 μm; d: Iba1 and CD16/32 staining: 50 μm; merge: 20 μm.