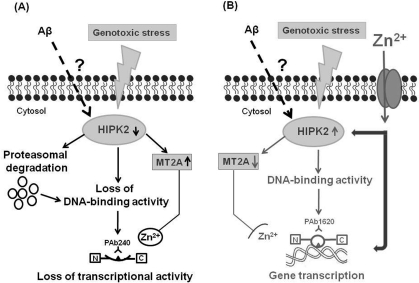
Figure 2.Working hypothesis for a putative link among p53 protein, soluble Aβ and HIPK2.The figure shows a novel mechanism of HIPK2 deregulation mediated by Aβ. HIPK2, when activated in response to DNA damaging agents, is able to interact physically and functionally with p53 and phosphorylate p53 at serine 46, thus regulating p53-induced apoptosis. HIPK2 also acts as transcriptional corepressor and deregulates the promoter metallothionein 2A (MT2A). MT2A may regulate p53 activity inducing protein folding through zinc modulation. In the presence of soluble Aβ, HIPK2 expression and activity are inhibited through Aβ-induced degradation via the proteasome system (panel A). HIPK2 deregulation results in the induction of MT2A (panel A), that exerts its Zn2+ chelator function. As a consequence, p53 protein misfolding (changing the wild-type conformation to a conformationally altered status) with subsequent abolishment of wild type p53 DNA binding and transcriptional activity occurs (panel A). Zinc supplementation counteracts Aβ effects on HIPK2 regulation (panel B). Zinc enters into cells through specific zinc transporters, that are required to convey this ion across cellular membranes, since zinc is unable to passively diffuse across cell membranes. Zinc can directly restore p53 function (panel B). In addition, zinc can also affect HIPK2 function, thus resulting in HIPK2 reactivation (panel B). As consequence, MT2A is deregulated and p53 conformational can switch to the wild-type and transcriptional active form (panel B).