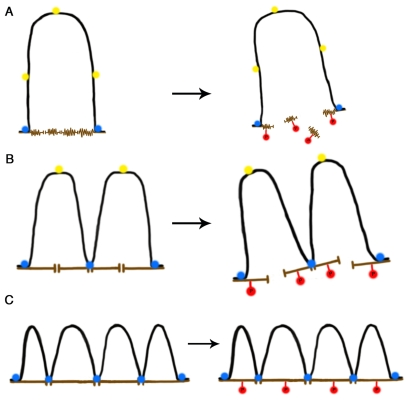
Figure 1.A self-stabilizing tensegrity model for DNA-NM interactions in the cell nucleus as a function of age. (A) In a newborn cell NM
proteins are in a compacted immature state (brown), thus the NM contact
surface is reduced and so a large DNA loop (black) is anchored to two NM
segments by means of two MARs that became actual LARs (blue circles) while
three potential MARs (yellow circles) cannot attach to the NM due to steric
hindrance and lack of enough contact surface. During mitosis biochemical
modification of NM proteins (e.g., phosphorylation, red circles) cause
disassembly of the NM network leading to disappearance of the cell nucleus.
(B) In an adult cell the NM proteins are in a more extended state
offering a larger contact surface, thus further potential MARs become
actualized as LARs reducing the average DNA loop size and increasing the
DNA-NM interactions. Yet phosphorylation of NM proteins leads to nuclear
disassembly during mitosis. (C) In a senescent cell the NM proteins
are fully extended thus offering enough contact surface for several
potential MARs to become actualized as LARs since steric hindrance is
further reduced. DNA loops become shorter on average and DNA-NM
interactions are significantly more numerous. Phosphorylation of NM
proteins during mitosis cannot lead to nuclear disassembly since the
DNA-loops keep separate NM segments bound together and stabilized by means
of the LARs attached to the NM. Thus the available energy becomes limiting
for disassembling the nucleus and the cell cannot enter or perform mitosis.