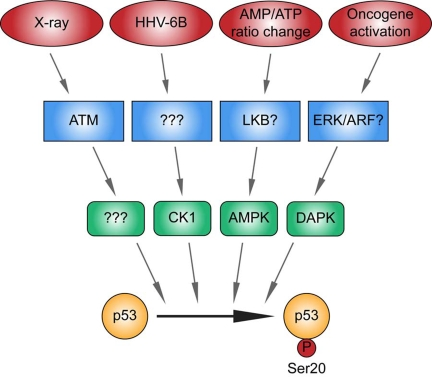
Figure 3.Different kinase signaling pathways link distinct stress signals to catalyze p53 phosphorylation at Ser20 in the TAD1 transactivation domain.
p53 is activated by distinct stresses, some of which include as indicated,
ionising radiation, viral infection, metabolic stress induced by an altered
AMP/ATP ratio, and oncogene activation. The X-ray-induced Ser20 site kinase
is ATM-dependent, but its identity is unknown (highlighted by "?"). CK1 is
the DNA virus HHV-6B-induced p53 Ser20 kinase, but the upstream sensor is
currently undefined (highlighted by "?"). The Ser20 site kinase induced by
an elevated AMP/ATP ratio is AMPK, and LKB is the likely upstream sensor.
DAPK-1 is the p53 Ser20 kinase induced by inappropriate oncogene
activation, and ERK or ARF are the likely upstream sensors. These data
support the formation of a model suggesting that the phosphorylation of p53
at Ser20 is triggered by distinct stress-responsive signaling cascades.
Future analysis will be required to determine the identity of all the
enzymes that mediate stress-induced phosphorylation at this site and
"integrate" the p53 response and developing disease models that deregulate
these signaling cascades.